Abstract
This study evaluated bacterial skeletal disease in conjunction with the major histocompatibility complex (MHC) in a genetically pure line of broiler breeder chickens. Chickens from six broiler breeder flocks were examined for skeletal lesions, bacterial pathogens, and MHC genotype. During a 10-week period, eighty-eight, 9- to 21-week-old lame chickens and 34 normal, age-matched controls were selected. Tenosynovitis, arthritis, and femoral or tibiotarsal (or both) osteomyelitis occurred in 86 of 88 (97.7%) lame chickens. Ninety-five bacterial isolates were obtained from 83 of 88 (94.3%) lame birds and 4 of 34 (11.8%) controls. Staphylococcus spp. was isolated from 72.6% of the skeletal lesions, predominantly Staphylococcus aureus (38.9%). MHC B complex genotypes were determined by hemagglutination for 88 lame birds, 34 controls, and 200 randomly selected birds from each of the six flocks (1,200 total). Combined chi-square analysis revealed that the homozygous MHC genotypes BA4/A4 (x2 = 14.54, P = 0.0063) and BA12/A12 (x2 = 42.77, P = 0.0001) were overrepresented in the sample of symptomatic birds compared with random samples from the same flocks. The homozygous A4 and A12 MHC genotypes influenced flock chi-square values more than the corresponding heterozygotes. An MHC B complex influence on bacterial skeletal disease was apparent in this line of broiler breeders.
Generalized leg weakness and lameness are predominant economic and welfare concerns among poultry producers. 22 Significant lameness has been historically attributed to various noninfectious dysplastic and metaplastic abnormalities, with less emphasis placed on the infectious counterparts. 22 Recent commercial and experimental flock analyses, however, revealed an increasing incidence of staphylococcal chondronecrosis with osteomyelitis in broilers. 22 These analyses revealed that 20.4% of lame individuals in two commercial flocks had typical bacterial skeletal lesions that were primarily associated with Staphylococcus aureus. Originally described in pheasants and turkeys during the 1930s, staphylococcosis is now considered a major contributing factor in the development of crippling mortality and reduced production among commercial broiler chickens. 2
Despite the prevalence of the natural disease, staphylococcosis is difficult to reproduce experimentally. A recent successful model used ad libitum feeding protocols and immunosuppression induced by chicken infectious anemia virus (CAV) and infectious bursal disease virus. 2, 22 This further supports the mechanism that multiple stresses and immunologic compromise are important pathogenic components for disease development. 22 These immunologic responses are dependent on both humoral and cell-mediated immunity, and the major histocompatibility complex (MHC) is central to this regulation.
Genetic control of immunoresponsiveness has been confirmed with S. aureus polyclonal activators of T lymphocytes. 8 Antigen recognition may be dependent on a single class-I or class-II MHC genotype, and specific genotypes are linked to the immunoregulation of various pathogens. 14 Increased resistance or susceptibility to several common poultry diseases, including tibial dyschondroplasia, Marek's disease, Rous sarcoma, and cecal coccidiosis, has been unequivocally linked to certain MHC genotypes. 9, 12, 16, 23, 25 In addition, MHC associations with moderately and highly pathogenic strains of S. aureus have been described in White Leghorn chickens. 8 Albeit, staphylococcosis and MHC associations have not been established in broiler breeder chickens.
The objectives of this study were to evaluate lameness and skeletal lesions in a genetically pure line of broiler breeder chickens, identify bacterial pathogens from the lesions, and evaluate the association of MHC influence on the development of bacterial skeletal disease.
Materials and Methods
Broiler breeder chickens
Six 9- to 21-week-old broiler breeder flocks (A through F) in a genetically pure line designated line A were studied. Flocks A through D, and E and F were located on farms 1 and 2, respectively, in a commercial breeder facility in the United States. All chickens were identified individually by wing band applied at hatch. The chickens were reared in solid-wall biosecure houses with cross-ventilation and evaporative cooling. Male and female broiler breeders were kept in adjacent floor pens separated by mesh fencing with a population of approximately 2,000 females and 500 males per flock. They were fed standard, nonmedicated commercial starter and grower rations in conservative amounts to control body weight gain, and water was supplied ad libitum. The chickens were vaccinated for infectious bursal disease and Marek's disease at 1 day of age and for chicken infectious anemia at age 13 weeks. In addition, CAV-specific antibodies were not detected in flocks F and E at 9- and 11 weeks of age, respectively, using a commercial enzyme-linked immunosorbent assay (Chicken anemia virus antibody test, IDEXX Laboratories, Westbrook, ME). Flocks A through D were immunized with live CAV vaccine before the initiation of the study.
Clinical examination
During a 10-week period (mid-June through August), chickens were observed daily for evidence of gait abnormalities. Lameness was evaluated and semiquantitatively categorized on a five-point scale, similar to a previously described gait analysis protocol, as summarized in Table 1. 15 Chickens with lameness, reluctance to move, or recumbency, and age-matched clinically normal control birds were selected from the general population, averaging four lame birds per day. Thirty-four control birds were selected from the general population. Numbers of lame and control birds were evenly distributed over all flocks.
Categorization and lameness assessment among 88 lame broiler breeders.

∗ Methodology for lameness categorization and semiquantitative assessment is modified from Kestin et al. 15
Necropsy
The chickens were euthanatized by cervical dislocation, and a necropsy was performed on each carcass. The bursa of Fabricius, spleen, and thymus were examined for gross lesions and fixed by immersion in 10% neutral-buffered formalin (NBF).
Before opening each carcass, the legs were removed to eliminate traumatic damage and reduce potential contamination of the femoral head. The coxofemoral joints were disarticulated by incising the skin over the greater trochanter, joint capsule, and round ligament of the head of the femur. Surrounding musculature and tendinous attachments were bluntly dissected, and the pelvic limbs were transported to a central laboratory for further dissection and subsequent bacterial culture in a clean environment. The pelvic limbs were inspected for gross lesions consisting of arthritis, tenosynovitis, and osteomyelitis. If no gross lesions were found, one half of both the left and right proximal femur, tibiotarsus, and gastrocnemius and surrounding tendon sheaths were fixed by immersion in 10% NBF. The remaining skeletal tissues were collected for bacterial culture.
Histopathology
Formalin-fixed tissues from six lame birds that had either negative bacterial cultures or no significant gross lesions and two Staphylococcus spp.–positive birds were histologically examined. Each femur and tibiotarsus was decalcified in 10% buffered formic acid for 36 hours. The decalcified bones, gastrocnemius and surrounding tendon sheaths, bursa of Fabricius, spleen, and thymus were routinely processed, embedded in paraffin, and sectioned at 4–6 μm. All sections were stained with hematoxylin and eosin (HE) (Gill's No. 2) using eosin phloxine counterstain. Definitive histomorphologic diagnosis of bacterial skeletal disease required coccoid bacterial colonies consistent with Staphylococcus spp. in the presence of inflammation in HE-stained sections.
Bacteriology
The cut surfaces of the proximal femur, tibiotarsus and gastrocnemius and surrounding tendons were dipped in 95% ethanol, flame sterilized, and sampled with sterile Culturette™ swabs (Becton Dickinson Microbiology Systems, BD Diagnostic Systems, Sparks, MD). The swabs were streaked onto blood (BBL™ Prepared Media, BD Diagnostic Systems, Sparks, MD), mannitol salt (Difco Catapult, BD Diagnostic Systems, Sparks, MD), and MacConkey agar plates (BBL™ Prepared Media, BD Diagnostic Systems, Cockeysville, MD) and incubated aerobically in a CO2 incubator for 24 hours at 37° C. Subsequently, a BBL™ Staphyloslide Latex Test (BD Diagnostic Systems, Sparks, MD) for S. aureus was performed on all colonies resembling Staphylococcus spp. Individual bacterial isolates were then placed on tryptic soy agar slants (Difco Catapult, BD Diagnostic Systems, Sparks, MD) and incubated for additional growth to produce adequate amounts of pure isolates. The purified isolates were maintained in tryptic soy broth containing 20% glycerol (Difco Catapult, BD Diagnostic Systems, Sparks, MD) and stored at −70° C. Gas chromatographic analysis was performed on all bacterial isolates using standard extraction and characterization techniques outlined in the manufacturers' instructions (Hewlett-Packard Midi-system, Hewlett-Packard Company, Palo Alto, CA).
MHC B complex immunotyping by hemagglutination
Initially, a 2.0-ml sample of whole blood was obtained from each bird (n = 122) via brachial venipuncture, diluted 1 : 10 (vol/vol) in 10% sodium citrate, and stored at 4° C for subsequent MHC B complex hemagglutination immunotyping. Broiler B complex MHC genotypes were characterized by hemagglutination using antisera and methods established in the laboratory of Drs. Warren Johnson and S. J. Ewald. 20 In addition to the selected population of 122 diseased and control birds, whole blood was collected from 200 randomly selected, 20-week-old birds from each of the six flocks (n = 1,200) for flock MHC B complex genotype distribution analysis.
Statistical analysis
The genotype distribution among lame individuals was statistically compared with the normal flock genotype distribution using chi-squared analysis.
Results
Clinical signs and gross pathology
The mean age of all chickens (n = 122; 88 clinically lame and 34 age-matched control birds) was 12.8 weeks (range 9–21 weeks), and the majority (57 of 88; 64.7%) of lame individuals were male. The average body weight was 4.48 and 3.5 kg for males and females, respectively. Significant positive correlations between lameness and body weight or sex were not observed in this line of broiler breeders. The onset and progression of overt gait abnormalities, ranging from slight ambulation deficits to recumbency, were insidious but rapid, often developing within 12–24 hours. All age-matched control birds were female (30 of 34; 88.2%) or missexed males (4 of 34; 11.8%). No lameness was detected in the 34 controls. Clinical assessment revealed increased expression of moderate (lameness score = 3) to severe (lameness score = 5) lameness among the selected population (Table 1).
In the lame chickens, gross lesions included osteomyelitis, arthritis, and tenosynovitis or combinations thereof (Table 2). Osteomyelitis, characterized by focal to locally extensive accumulations of fibrinonecrotic exudate with variable bone lysis, occurred most commonly in the proximal femoral physis (Fig. 1). Similar osteolytic lesions were present in the tibiotarsus. Tibiotarsal osteomyelitis was often accompanied by separation of the articular cartilage, exposing the sub-chondral bone. Arthritic lesions in the coxofemoral, femorotibial, and tibiotarsal joints consisted of distention of the respective joint capsule with purulent to caseous exudate and multifocal lysis of the articular cartilage. Similar caseous lesions occurred in the gastrocnemius and surrounding tendon sheaths. Nodular collections of purulent exudate filled the affected tendon sheath, and the respective tendon was often hyperemic to hemorrhagic. The adjacent fascia was edematous, and the gastrocnemius muscles had variable atrophy (Fig. 2). Other gross findings included a fractured wing, valgal angular limb deformities, and costochondral widening. Four of the lame individuals and all controls had no gross lesions.
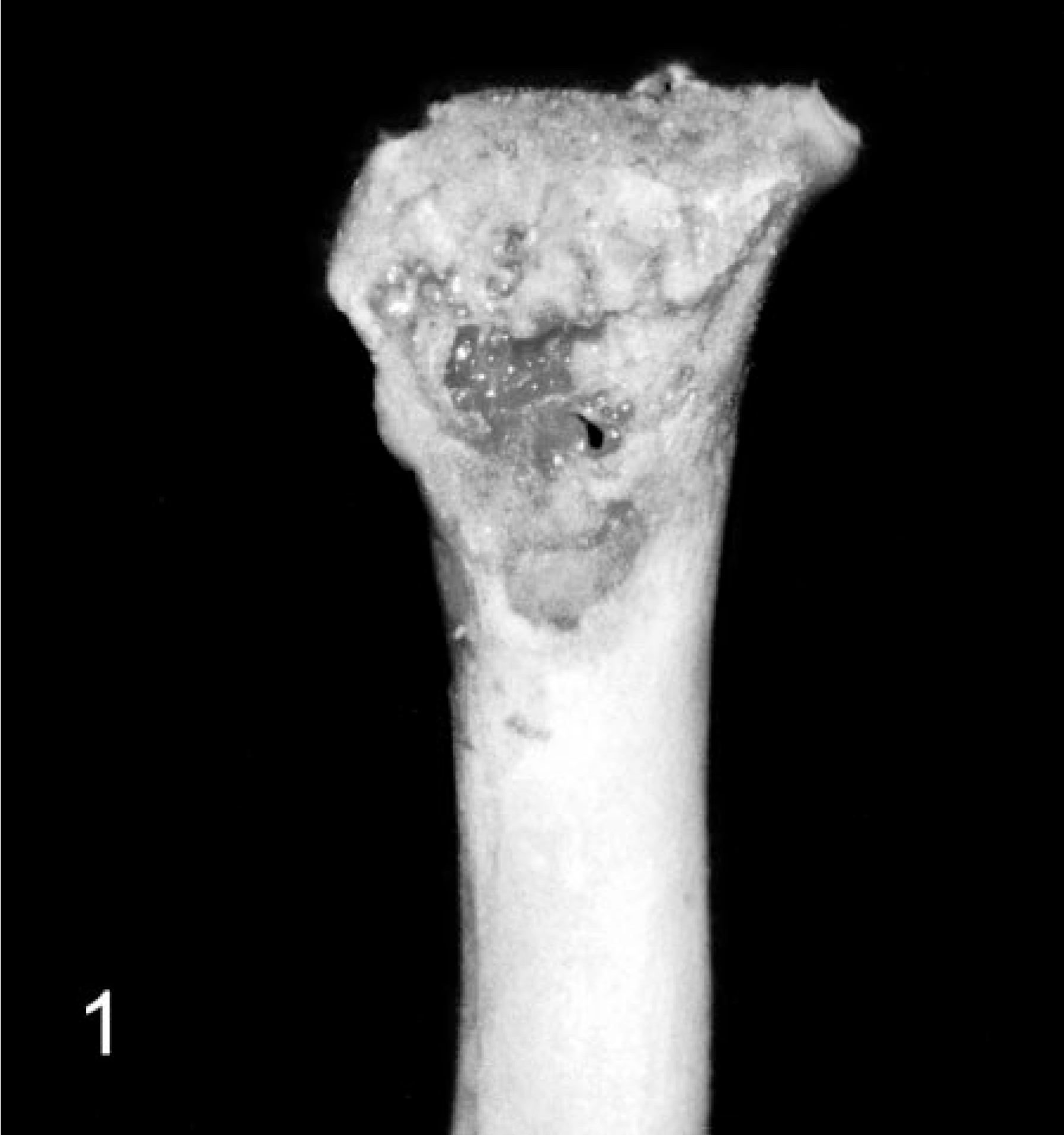
Femur; broiler breeder chicken with femoral head and neck necrosis. Note the loss of normal femoral head and neck architecture, accumulation of fibrinonecrotic exudate along the proximal femoral epiphysis, absence of the articular cartilage, and exposure of necrotic subchondral bone.
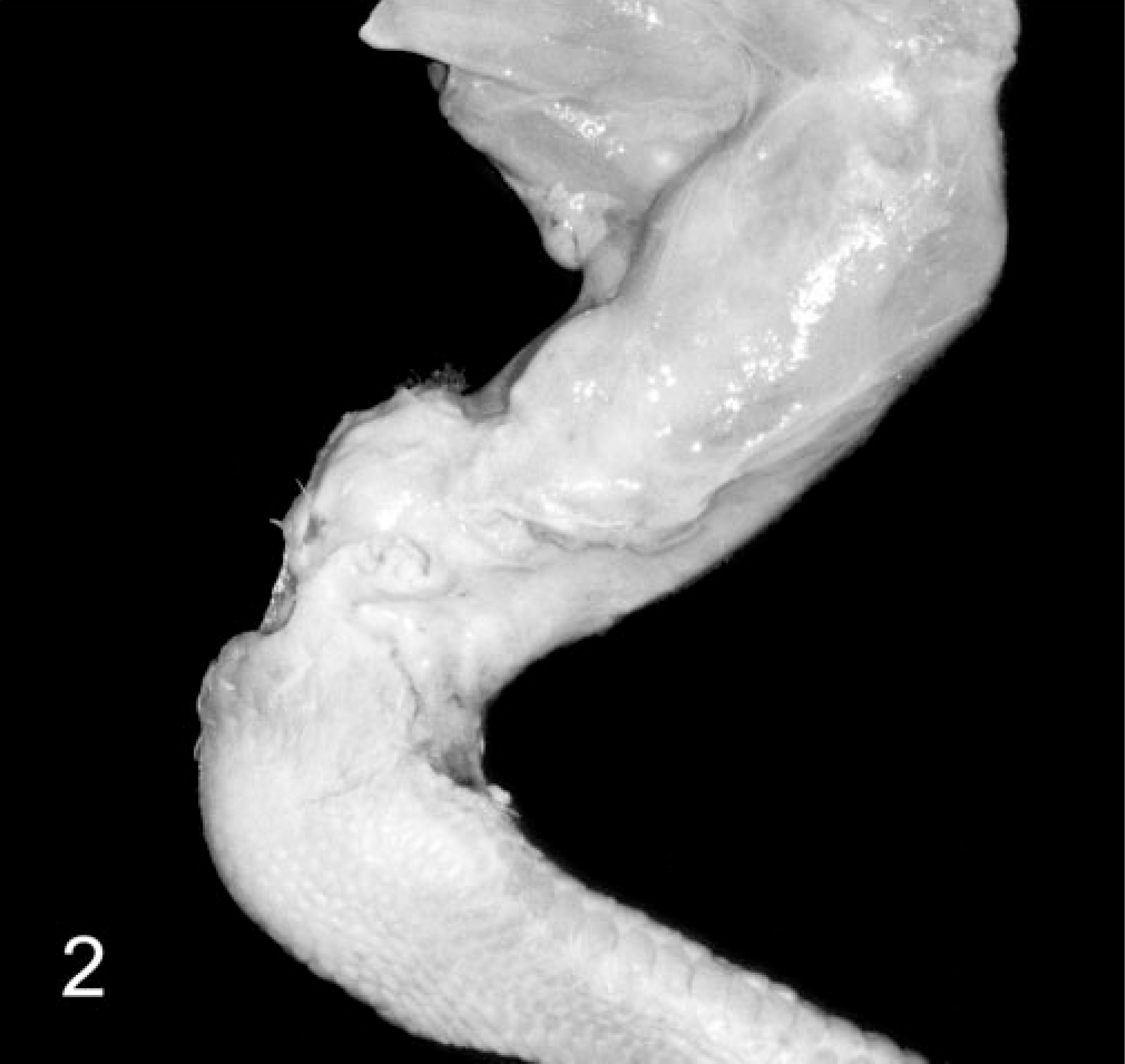
Pelvic limb; broiler breeder chicken. Multinodular distention of the gastrocnemius and surrounding tendon sheaths with turbid exudate. There is marked atrophy of the gastrocnemius muscle.
Summary and distribution of gross lesions among 88 lame and 34 clinically normal, age-matched control broiler breeders.
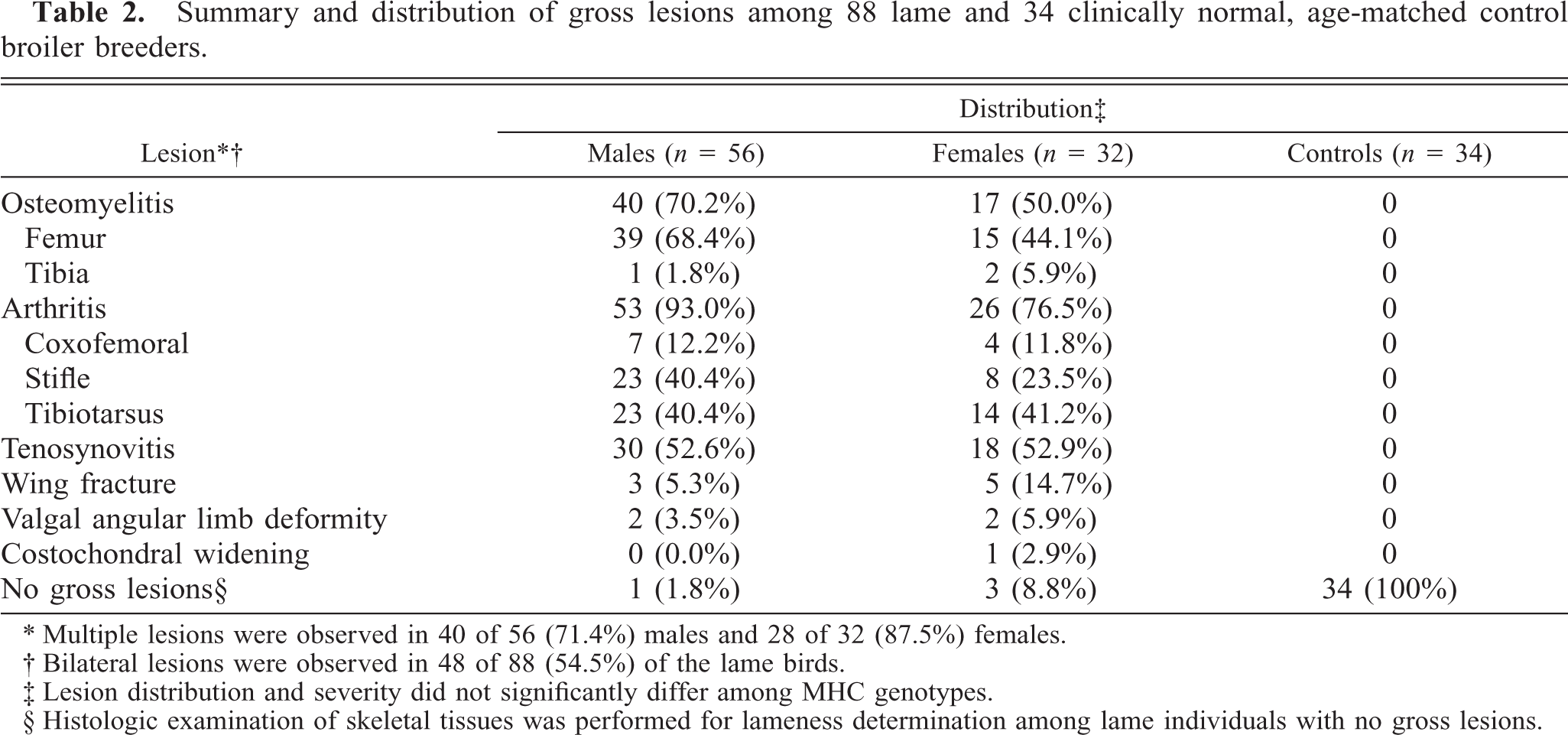
∗ Multiple lesions were observed in 40 of 56 (71.4%) males and 28 of 32 (87.5%) females.
† Bilateral lesions were observed in 48 of 88 (54.5%) of the lame birds.
‡ Lesion distribution and severity did not significantly differ among MHC genotypes.
§ Histologic examination of skeletal tissues was performed for lameness determination among lame individuals with no gross lesions.
Bacteriology
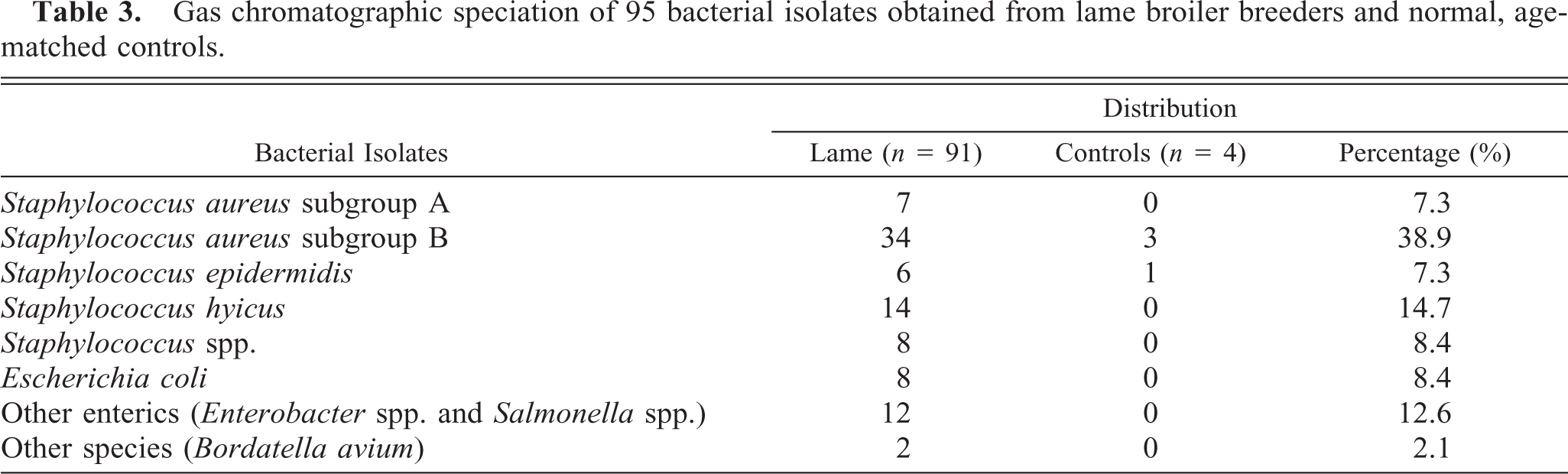
Culture of the proximal femoral physis, tibiotarsus, and gastrocnemius and surrounding tendon sheaths from each chicken (n = 122) yielded 95 bacterial isolates including Staphylococcus spp., Escherichia coli, and several other species in 83 of 88 (94.3%) lame chickens and 4 of 34 (11.8%) controls (Table 3). A preliminary identification for S. aureus was confirmed in 43 of 95 (45.3%) of the bacterial isolates with BBL™ Staphyloslide Latex agglutination. Gas chromatographic speciation confirmed the predominance of Staphylococcus spp. with S. aureus subgroup B being the most prevalent pathogen.
Gas chromatographic speciation of 95 bacterial isolates obtained from lame broiler breeders and normal, age-matched controls.
Histopathology
Eighty-three of 88 chickens were positive for bacterial skeletal disease based on criteria established at gross necropsy and bacteriology. Formalin-fixed tissues from six culture-negative birds were histologically examined. Histomorphologic criteria confirmed staphylococcal osteomyelitis and tenosynovitis in two of six (33%) of the lame chickens that had either negative bacterial cultures and/or no significant gross lesions. Multiple physeal vessels were occluded by coccoid bacterial colonies and were surrounded by heterophils, giant cell macrophages, and cellular debris, which replaced the normal physeal architecture. In one bird, the gastrocnemius tendon and tendon sheath were expanded by a diffuse heterophilic infiltrate and cellular debris. Fibrosis occurred with secondary synovial cell hyperplasia and synechia formation. The spleen, thymus, and bursa of Fabricius were histologically normal.
MHC association
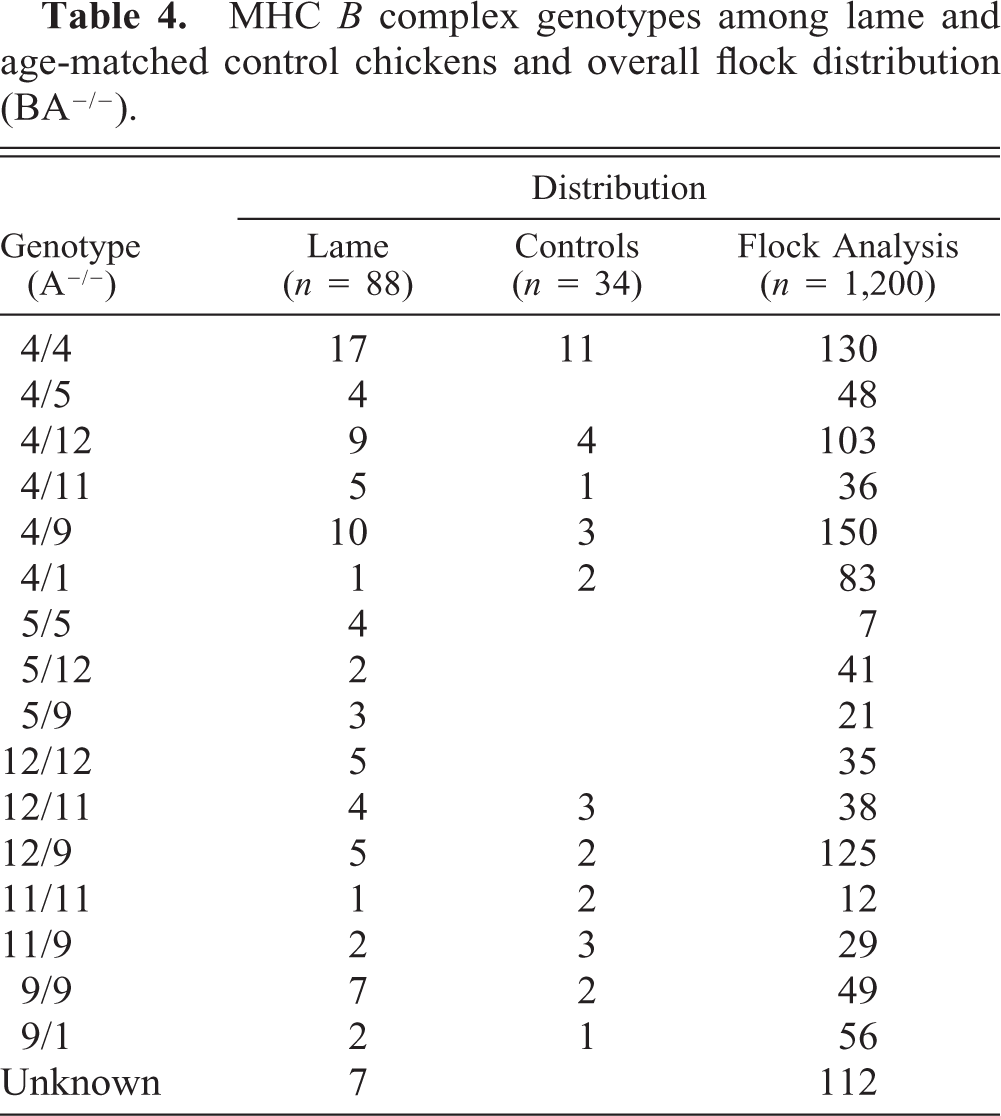
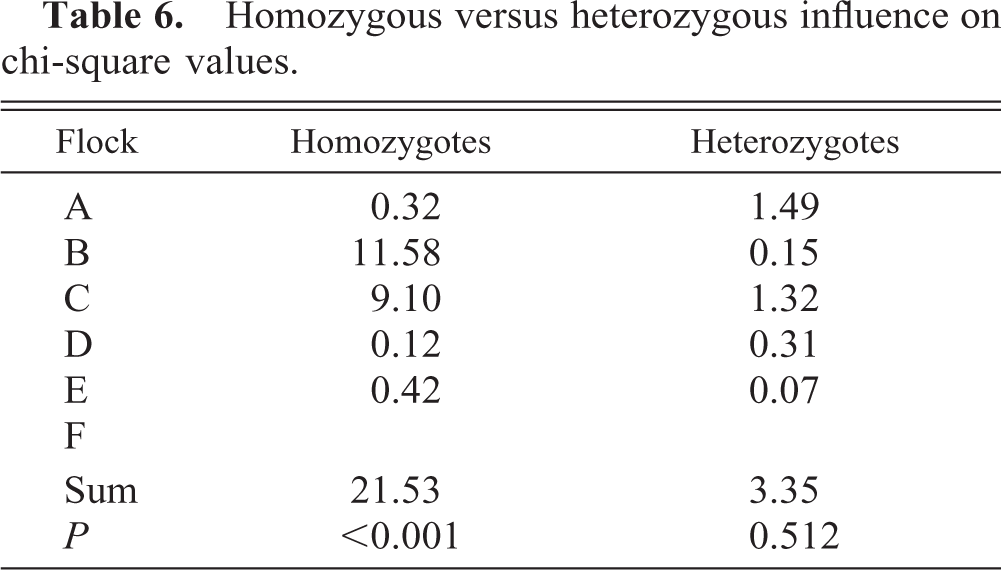
At least 16 genotypes were represented in the lame and control individuals (Table 4). A total of 28 different genotypes, resulting from hemagglutination using seven different haplotype-specific antisera, were represented and evenly distributed among the six flocks (data not shown). Two MHC B complex genotypes, A4 and A12, were associated with a significantly higher prevalence of lameness than were 14 other genotypes (Table 5). Frequency distribution and chi-square analysis revealed that the homozygous genotypes BA4/A4 (χ2 = 14.54, P = 0.0063) and BA12/A12 (χ2 = 42.77, P = 0.0001) were overrepresented in the sample of lame birds compared with the random sample from the same flocks. The combined chi-square values from each flock were significant, with A4 and A12 being prevalent in the most influential genotypes (Table 5). The homozygous individuals contributed greater to the respective chi-square values than the corresponding heterozygotes (Table 6).
MHC B complex genotypes among lame and age-matched control chickens and overall flock distribution (BA−/−).
Combined flock chi-square analysis and the most influential MHC B complex genotypes in each flock.
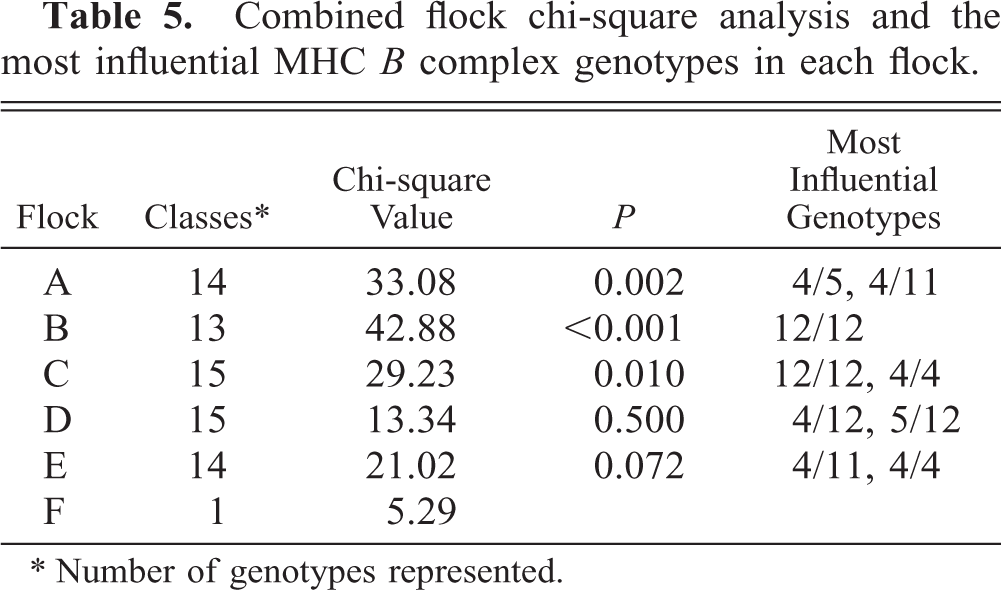
∗ Number of genotypes represented.
Homozygous versus heterozygous influence on chi-square values.
Discussion
The avian MHC gene family is unequivocally associated with increased resistance or susceptibility to several common poultry pathogens, most notably Marek's disease virus. 3, 10 There is a general paucity of information, however, regarding both MHC-associated differential immunity to bacterial infections and genetic control of immunoresponsiveness during natural disease outbreaks. 18 This is the first report describing an MHC B complex influence on the development of bacterial skeletal disease in broiler breeder chickens, where Staphylococcus spp. was the predominant pathogen. Clinical signs and lesions were similar and consistent with natural and experimental staphylococcal osteomyelitis and tenosynovitis. 2, 11, 27 Moreover, this study parallels MHC-mediated resistance after S. aureus challenge in inbred congenic lines of White Leghorn chickens. 6
Several models for staphylococcosis use either direct intracardiac or intravenous inoculation for experimental disease development. 2, 11, 27 The respiratory tract or mucous membrane colonization likely serves as a common portal of entry during natural disease. 2, 22 Experimental models for aerosol-mediated staphylococcal osteomyelitis require repeated high-dose challenge, but often with inconsistent results. 2, 22 Aerosol exposure in conjunction with virally induced immunosuppression and ad libitum feeding protocols enhanced the development of bacterial chondronecrosis and osteomyelitis. 22 These results imply that the pathogenesis is multifactorial, involving interactions between the host immune system, environmental influences, and bacterial virulence. Consequently, the aim of this study was to investigate the epidemiology of a natural skeletal disease with emphasis on the immunogenetic factors associated with disease development.
Two MHC B complex genotypes, A4 and A12, were associated with increased susceptibility to bacterial skeletal disease within the examined flocks. This over-representation of select genotypes among affected birds is suggestive of a potential MHC influence on the outcome of disease. The avian MHC, originally identified serologically as an extensively polymorphic erythrocyte antigen or blood group system, has three closely linked major loci, B-F, B-L, and B-G. 4, 13, 17, 19 The B-F determinants and B-L immune response genes are equivalent to the mammalian class-I and class-II genes, respectively. However, the B-G erythrocyte-specific class IV alloantigens have not been identified in human or murine counterparts. 18 Additional genes in the chicken MHC possess structural and functional homology with mammalian genes involved in lymphocyte activation and transporter associated with antigen-processing proteins. 18 As such, the MHC genes are a crucial component in the initial antigen recognition and perpetuation of immune regulation.
Molecular surveys have mapped disease resistance to the B-F/B-L region. 3, 10 The B-F and B-L MHC regions encode for cell surface molecules that act as restriction elements for host cellular interactions, thus affecting immunoresponsiveness. 28, 30 These associations have been studied with inbred congenic chicken lines that principally differ in MHC haplotype. 1 Moreover, MHC-mediated resistance to Salmonella spp. occurred in inbred congenic lines homozygous for the B4 haplotype. 5 Age-dependent roles for chicken heterophil function are correlated with MHC enhancement of antibody production, complement activation, and the production of acute phase proteins. 7, 26 Thus, MHC-induced differential immunity to bacterial pathogens may be age related and involve genetic interactions between MHC and non-MHC loci.
The anatomic distribution of gross lesions among these lame broiler breeders was similar to previous reports. 2, 11, 22, 27 The diagnosis of bacterial skeletal disease was established by consistent gross lesions, bacterial isolation, or histologic lesions (or all). Osteomyelitis occurred most frequently in the proximal femoral physis and metaphysis, consistent with bacterial deposition in the terminal metaphyseal capillary network, where laminar blood velocity is decreased. 22 Localized extension into the surrounding joints, tendon sheaths, and soft tissues commonly occurs with severe osteomyelitis. For example, multiple lesions observed in the lame birds often consisted of osteomyelitis with arthritis of the corresponding joint or localized tenosynovitis (or both). Although gross lesions occurred in most lame chickens, histologic examination identified bacterial skeletal lesions in two of six lame birds with neither gross lesions nor bacterial isolates.
In addition to alterations in the host immune response, staphylococcal virulence factors are also related to septicemia and skeletal disease. 2 Poultry isolates have avian-specific phage groups and ecovars containing the ermC gene determinant. 2, 21, 24 Most strains possess multiple virulence factors, including protein A, β-hemolysin, fibrinolysin, and dermonecrotic toxin, which may act synergistically to elude the immune response.
In this study, spontaneously lame broiler breeder chickens with bacterial osteomyelitis, arthritis and tenosynovitis were identified as a natural disease. The clinical and pathologic presentations were consistent with previously reported staphylococcosis in immature broiler breeders in commercial broiler production. The corresponding high incidence of bacterial infection in two significantly overrepresented genotypes indicates an MHC B complex influence on the expression of bacterial skeletal disease, with Staphylococcus spp. predominant. The current demonstration of differential immunity linked to the MHC B complex may provide insights into preventative measures for bacterial skeletal disease. Characterization of the mechanism of the MHC and host immunoresponsive influences on bacterial skeletal disease will require experimental disease reproduction and analysis within a larger, controlled population.
Footnotes
Acknowledgements
We thank Dr. Robert Norton, Kenneth Macklin, Stephen Martin, and Beth Landreth for their technical assistance.
