Abstract
Avian leukosis virus subgroup J has a high tropism for myeloid lineage cells and frequently induces neoplastic transformation of myelocytes. The impact of congenital avian leukosis virus subgroup J infection on the function of circulating heterophils and susceptibility to staphylococcal infection was investigated. Six-week-old broiler chickens negative for exogenous avian leukosis viruses or congenitally infected with avian leukosis virus subgroup J were inoculated intravenously with 106 colony-forming units of Staphylococcus aureus, and pre- and postinoculation heterophil function was assessed. All chickens developed a leukocytosis with heterophilia after inoculation, but total leukocyte and heterophil counts were significantly higher in leukosis-negative chickens than in virus-infected chickens. Tenosynovitis was more severe in leukosisnegative chickens, and 2/10 (20%) of the virus-infected chickens had no histologic evidence of tenosynovitis. Osteomyelitis in the tibiotarsus or tarsometatarsus developed in 5/10 (50%) of the chickens in each group. S. aureus was recovered from the hock joint of 6/10 (60%) of the chickens in each group. Heterophils from all chickens exhibited similar phagocytic ability pre- and postinoculation. Heterophils from virus-infected chickens exhibited less bactericidal ability preinoculation than did heterophils from leukosis-negative chickens. However, postinoculation bactericidal ability was similar in both groups. Avian leukosis virus subgroup J provirus was present in heterophils isolated from congenitally infected chickens. Heterophils isolated from broiler chickens congenitally infected with avian leukosis virus subgroup J exhibit no significant functional deficits, and infected and uninfected chickens exhibit similar susceptibility to staphylococcal infection.
Keywords
Avian heterophils are equivalent to mammalian neutrophils and are the predominant leukocyte in acute hematologic and tissue exudative responses to inflammatory stimuli. 15 Heterophils are also important tissue phagocytes for host defense against microbial pathogens. Heterophils primarily use oxygen-independent mechanisms for microbicidal activity by employing granule contents such as cationic peptides and lysozyme. Both intact and lysed heterophils exhibit microbicidal activity, indicating these cells most likely can perform this function by either phagocytosis of organisms or extracellular release of granule contents. 16
Staphylococcal tenosynovitis and osteomyelitis are economically important diseases of chickens and turkeys. Experimental staphylococcal osteomyelitis in the chicken has also been established as a model for human acute hematogenous osteomyelitis. 9,11,12 In this model, 104–108 Staphylococcus aureus are injected intravenously, and osteomyelitis and tenosynovitis develop as soon as 24 hours postinoculation. Bacteria are deposited within metaphyseal vessels terminating in the physeal zone of hypertrophy, and osteomyelitis develops within the adjacent metaphysis, frequently in long bones. 11 Inflammatory cell infiltrates in experimental staphylococcal osteomyelitis and tenosynovitis are primarily heterophils, consistent with their role as the main leukocyte in tissue exudative responses. 9 Leukocytosis with heterophilia is also a consistent finding in natural cases of staphylococcal tenosynovitis and osteomyelitis. 3,5 Heterophils collected from chickens with experimental staphylococcal tenosynovitis and osteomyelitis exhibited increased adherence, chemotaxis, phagocytosis, and killing of bacteria. 2 These predictable heterophil responses to reproducible stimuli make experimental staphylococcal tenosynovitis and osteomyelitis in chickens an appropriate model to investigate viral effects on heterophil function.
Avian leukosis viruses (ALVs) are oncogenic retroviruses that typically induce B-cell lymphosarcoma in commercial poultry. In 1991, a novel subgroup of ALV designated J (ALV-J) was described in meat-type chickens. 21 In contrast to previously described ALVs, ALV-J isolates have a low lymphoid cell and a high myeloid cell tropism in vitro. 4 Consistent with this tropism, infected chickens frequently develop myelocytomas, histiocytic sarcomas, or erythroblastomas, but lymphosarcomas have not been documented. 22,31 Some ALVs suppress immune function in the host by infecting leukocytes. Subgroup B ALVs include dramatic immunosuppression by interfering with macrophage-derived, antigen-presenting cell function. 23–25 Persistent synthesis and accumulation of integrated and unintegrated proviral DNA in infected antigen-presenting cells have been proposed as mechanisms for this functional deficit. 8 In our preliminary investigations of congenitally ALV-J–infected broiler chickens, ALV-J provirus was frequently detected in DNA isolated from peripheral blood heterophils by polymerase chain reaction (PCR). The impact of ALV-J infection on heterophil function has not been investigated. In the following study, the staphylococcal tenosynovitis and osteomyelitis model was used to investigate heterophil function in broiler chickens with and without congenital ALV-J infection.
Materials and Methods
Production of broiler chickens
Broiler chickens congenitally infected with ALV-J (four males, six females) and broiler chickens with no detectable exogenous ALVs (five males, five females) were produced from established ALV-J–positive and ALV-negative breeder flocks as described previously. 29 Their ALV and ALV-J status was verified at hatch by ALV p27 enzyme-linked immunosorbent assay (ELISA) IDEXX Laboratories, Westbrook, ME) and reverse transcription PCR amplification of viral genomic RNA extracted from plasma using previously described methods. 28,29 All broiler chickens were identified individually by wing banding at hatch. ALV-J–infected chickens were raised separately from ALV-negative chickens in Horsfall-Bauer isolation units to 4 weeks of age. Chickens were then transferred to separate floor pens for the remainder of the study. Chickens received an unmedicated growth ration (University of Georgia, Athens, GA) and water ad libitum. Chickens received no vaccinations and were weighed weekly.
Experimental model
S. aureus strain 6-921, a pathogenic capsular antigen type 8 strain isolated from a chicken, 7 was maintained in brain-heart infusion (BHI) broth. The number of colony-forming units per microliter was determined by obtaining optical density readings at 595 nm and was verified by plating serial dilutions on blood agar. Bacterial subcultures in log phase growth were used for inoculation and bactericidal assays.
Ten 6-week-old broiler chickens that were congenitally infected with ALV-J and 10 that were ALV negative were each inoculated intravenously with 106 S. aureus suspended in 0.1 ml of Hanks' balanced salt solution (HBSS; Sigma Chemical Co., St. Louis, MO). A 4-ml sample of ethylenediaminetetraacetic acid (EDTA)-anticoagulated whole blood was obtained from the brachial vein of all chickens 24 hours prior to inoculation and 5 days postinoculation for heterophil isolation and function assays. A 0.2-ml sample of EDTA-anticoagulated whole blood was obtained from the brachial vein of five randomly selected congenitally infected chickens and five randomly selected ALV-negative chickens 6 days postinoculation for leukocyte counts. All chickens were necropsied 6 days postinoculation and examined for gross lesions. A serum sample was obtained from each chicken immediately before necropsy. Samples were tested for seroconversion to Newcastle disease virus, infectious bronchitis virus, infectious bursal disease virus, chicken anemia virus, avian reovirus, avian encephalomyelitis virus, Mycoplasma gallisepticum, Mycoplasma synoviae (IDEXX Laboratories), and infectious laryngotracheitis virus (Kirkegaard and Perry Laboratories, Gaithersburg, MD) by ELISA following the manufacturers' instructions.
Histopathology
At necropsy, the distal left tibiotarsus, left tibiotarsal–tarsometatarsal (hock) joint, proximal left tarsometatarsus, and attached digital flexor tendons were collected from each chicken and fixed by immersion in 10% neutral buffered formalin. The distal tibiotarsus and proximal tarsometatarsus were decalcified in a solution of equal parts of 8 N formic acid and 1 N sodium formate. Sagittal slices through each decalcified bone and the digital flexor tendons were processed, embedded, sectioned, and stained using routine methods. One hematoxylin and eosin (HE)-stained section from each bone and one HE-stained section of the flexor tendons were examined from each chickens. Osteomyelitis was recorded as present in a chicken if either bone had lesions. Tenosynovitis was recorded as present in a chicken if the flexor tendon section showed lesions in the tendon or sheath. Additionally, the severity of any tenosynovitis was assessed for each chicken based on the most severe histologic lesions within the section. Tenosynovitis was defined as mild if lesions consisted of minimal synovial hyperplasia with rare perivascular heterophils. Moderate tenosynovitis was defined as moderate synovial hyperplasia with more prominent subsynovial heterophils and infrequent heterophil exocytosis but without exudation into the synovial space. Severe tenosynovitis was defined as prominent synovial hyperplasia with abundant heterophil infiltrates, heterophil exocytosis, and the accumulation of exudate within the synovial space.
Microbiology
At necropsy, the right hock joint of each chicken was aseptically incised and swabbed using a single swab (Culturette, BD Biosciences, Franklin Lakes, NJ). Swabs were streaked onto blood agar plates and incubated aerobically for a minimum of 3 days at 37 C. Colonies on plates were examined and confirmed as S. aureus using a commercial kit (BBL Staphyloslide Latex test kit; BD Biosciences).
Leukocyte counts
Total leukocyte counts were performed manually (Eosinophil Unopette system; BD Biosciences) as described for avian species with minor modifications. 6 Granulocytes in all squares in the hemocytometer could not be easily counted manually because of extremely high granulocyte counts in the samples. Therefore, only granulocytes in the upper right, middle, and lower left squares of both sides of the hemocytometer were counted. The sum for each side was multiplied by 3 to obtain a total count for each side, and the total for both sides was used to calculate the total leukocyte count according to a published formula. 6 Differential leukocyte counts were performed manually for 200 leukocytes in blood smears stained with Wright's stain.
Heterophil isolation
Heterophils were isolated from peripheral blood samples following an established protocol with some modifications. 2,7 For each chicken, the 4-ml sample of EDTA-anticoagulated whole blood was mixed with 1% methylcellulose in calcium- and magnesium-free (CMF) HBSS with 1% fetal bovine serum (FBS; Mediatech, Herndon, VA) at a ratio of 1.5:1 and centrifuged at 25 × g for 15 minutes. The plasma and buffy coat were harvested, washed once in CMF HBSS 1% FBS, and suspended in 3 ml of the same solution. The cells were centrifuged for 20 minutes at 225 × g over 3 ml of Histopaque 1077 (Sigma). The resultant pellets of heterophils and erythrocytes were washed once in CMF HBSS 1% FBS and then suspended for 10 minutes in 10 ml of 0.87% NH4Cl (Sigma) and 0.1% KHCO3 (Sigma) in sterile water to lyse erythrocytes. The remaining heterophils were washed in CMF HBSS 1% FBS and quantified using an automated cell counter (Coulter Counter® Model D2N; Coulter Corp., Hialeah, FL). Cytocentrifuge preparations of each heterophil suspension were made (Cytospin® cytocentrifuge; Shandon, Pittsburgh, PA). Each preparation was stained with Wright's stain and examined to confirm the purity of the isolation.
Detection of provirus
An aliquot of 3 × 106 heterophils in HBSS from each chicken was stored at −20 C until nucleic acid extraction. Total DNA was extracted with a kit (Wizard® Genomic DNA Purification Kit; Promega, Madison, WI) following the manufacturer's instructions. Extracted DNA from each chicken was suspended according to the manufacturer's recommendations. PCR was performed as previously described with an ALV-J–specific primer set. 28,29 DNA was amplified in a 50-µl reaction volume for 36 cycles at an annealing temperature of 55 C (PTC-200 DNA Engine thermal cycler; MJ Research, Waltham, MA). Products were electrophoresed on 1% agarose gels, stained with ethidium bromide, and visualized under ultraviolet light.
Heterophil phagocytosis
Heterophil phagocytic ability was assessed using fluorescent latex particles and a modification of a previously described protocol. 2,7 The assay was performed in triplicate for each chicken. A suspension of 1 × 106 heterophils in RPMI (Mediatech) with 1% FBS was placed in a 5-ml tube with no additive (Vacutainer; Fisher Scientific, Norcross, GA). Heterophils fixed in 3% paraformaldehyde were used as a negative function control for the assay. An aliquot of 2.44-µm fluorescent latex particles (Polysciences Corp., Warrington, PA) opsonized with serum obtained from specific-pathogen-free (SPF) chickens (Charles River SPAFAS, North Franklin, CT) was added to each tube for a total heterophil:particle ratio of 1:20. Latex particles were sonicated for 90 seconds to ensure most existed as single particles before adding them to heterophils. Heterophils and particles were centrifuged for 5 minutes at 25 × g to increase contact between particles and cells. Tubes were incubated in a shaking water bath for 45 minutes at 41 C in the dark. After incubation, heterophils were washed with CMF HBSS 1% FBS with 0.02% EDTA and resuspended in HBSS. Phagocytosis was assessed by flow cytometric counting of 1 × 105 heterophils from each tube (Epics XL-MCL Elite flow cytometer; Coulter). The analyses were standardized by creating bit map gates around paraformaldehyde-fixed control heterophil scatter plots using both forward angle and 90° light scatter as described previously. 2 Results for each sample of heterophils were expressed as the percentage of heterophils containing fluorescent particles. The percentage of phagocytosis for each bird was calculated as described previously using the following formula: % phagocytic heterophils = % phagocytic test heterophils − % phagocytic fixed heterophils. 7
Heterophil bactericidal ability
Bactericidal ability in vitro was assessed by a colorimetric killing assay using the tetrazolium compound 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT; Sigma) as described for neutrophils and modified for avian heterophils. 7,30 Bactericidal ability was assessed against both the challenge strain of S. aureus, 6-921, and a pathogenic capsular antigen type 5 strain of S. aureus, 8-5658A, isolated from a chicken. 7 Strain 8-5658A was maintained in BHI as was strain 6-921. Log phase growth cultures of each bacterium were opsonized with serum from specific-pathogen chickens (Charles River SPAFAS) and then suspended at 5 × 107 cells/ml in HBSS as determined by optical density measurements for all assays. Counts were verified by plating serial dilutions on blood agar. Assays for each chicken and all controls were run in quadruplicate. Three × 105 heterophils in HBSS 1% FBS and 2.5 × 106 bacteria in HBSS were added to each test well in a 96-well flat-bottom cell culture plate (Nalge Nunc International, Naperville, IL) for a total heterophil:bacteria ratio of 3:25. For bacterial control wells, HBSS 1% FBS was added instead of heterophils. For cell control wells, HBSS was added instead of bacteria. Plates were centrifuged at 400 × g for 5 minutes to increase contact between heterophils and bacteria and then incubated for 1 hour at 37 C. After incubation, plates were centrifuged at 600 × g for 10 minutes, and the medium was carefully removed by pipetting. Heterophils were then lysed by the addition of 80 µl of sterile water for 5 minutes, followed by 150 µl of BHI. To cultivate remaining viable bacteria, plates were incubated for 3 hours at 37 C. To quantify cultivated bacteria, 50 µg of MTT was added to each well, incubation was continued for an additional 10 minutes, and 10 µl of 0.04 N HCl was added to each well. Plates were immediately cooled on ice, and optical density was determined at 570 nm using an automated microplate reader (MR-700; Dynatech Laboratories, Chantilly, VA). The percentage of bacteria killed was determined for each chicken using the following formula: % killing = 100 × [optical density of bacterial control well − (optical density of test well − optical density of cell control well)]/optical density of bacterial control well. 7
Statistical analysis
All numeric data were analyzed using a one-way analysis of variance. For heterophil phagocytosis data, percentages were transformed to arcsines for analysis. For bactericidal ability data, analyses were performed on optical density values. A significance level of 0.05 was used for all statistical analyses.
Results
Immediately before staphylococcal inoculation, mean body weights for 6-week-old ALV-negative chickens were 2,341 g for males and 1,864 g for females. Mean body weights for 6-week-old ALV-J–infected chickens were 1,218 g for males and 1,404 g for females, corresponding to 52% and 75% of the body weights for ALV-negative male and female chickens, respectively. This severe body weight suppression is similar to previously published body weight suppression associated with ALV-J infection. 29 Chickens in both groups developed clinical signs of lameness and reluctance to move, consistent with staphylococcal tenosynovitis and osteomyelitis. All chickens were clinically affected by 5 days postinoculation. At necropsy, grossly affect hock joints and tendon sheaths contained mild to copious amounts of thick, tan exudate. Gross joint or tendon involvement was apparent unilaterally or bilaterally in 3/10 (30%) of the ALV-negative chickens and 4/10 (40%) of the ALV-J–infected chickens. Visceral lesions and neoplasia were not observed in any chickens.
For all chickens, tendon sections typically contained three to five tendons and sheaths. All chickens in the ALV-negative group had inflammatory lesions consistent with tenosynovitis in at least one tendon sheath, but two ALV-J–infected chickens had no histologic evidence of tenosynovitis, and lesions were less severe in these chickens (Fig. 1). In cases of tenosynovitis, inflammatory lesions consisted of synovial hyperplasia, heterophil infiltrates, and exudation into the synovial space (Fig. 2). Multifocal synovial cell necrosis was present in some sections. Subsynovial vessels were frequently dilated and had heterophil margination. Exudate in the synovial space consisted of heterophils, sloughed synovial cells, and necrotic cell debris. Lesions were confined to the synovial membrane and synovial space without involvement of the tendon proper. Bacteria were not observed in any lesions. In two ALV-negative chickens and one ALV-J–infected chicken, subsynovial lymphoid infiltrates with follicle formation were rarely present in both tendon sheaths with a heterophilic tenosynovitis and sheaths with no acute inflammatory lesions. All chickens had low serologic titers to avian reovirus (geometric mean titer = 3,410) at the termination of the study, and the lymphoid infiltrates were morphologically consistent with chronic reovirus infection. 17 Seroconversion to other avian pathogens was not detected. In both ALV-negative and ALV-J–infected groups, 5/10 (50%) of the chickens developed osteomyelitis. Overall, 6/20 (30%) of the tibiotarsi and 5/20 (25%) of the tarsometatarsi examined had lesions consistent with osteomyelitis. One ALV-negative chicken had lesions in both bones. Lesions of osteomyelitis consisted of single or multiple colonies of bacterial cocci, bone marrow necrosis, and infiltrates of necrotic heterophils with fewer macrophages and giant cells (Fig. 3). Lesions were typically located within the zone of ossification or zone of hypertrophy of the growth plate, frequently at the termination of metaphyseal vessels (Fig. 4). Osteomyelitis was also occasionally present within the epiphysis.
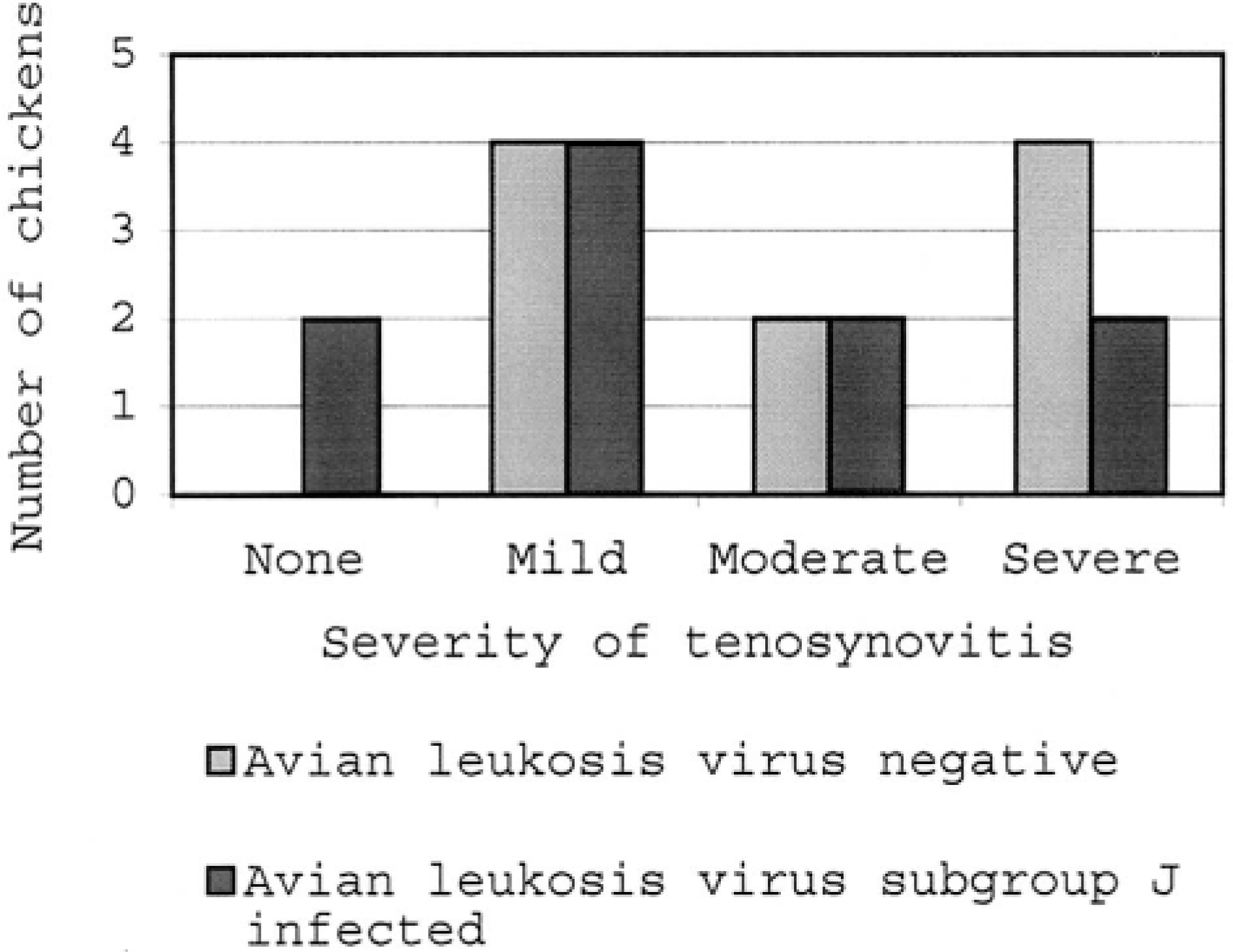
Frequency distribution of tenosynovitis lesions in avian leukosis virus–negative and avian leukosis virus subgroup J–infected chickens. The numbers of chickens in each group are indicated for each category of lesion severity.
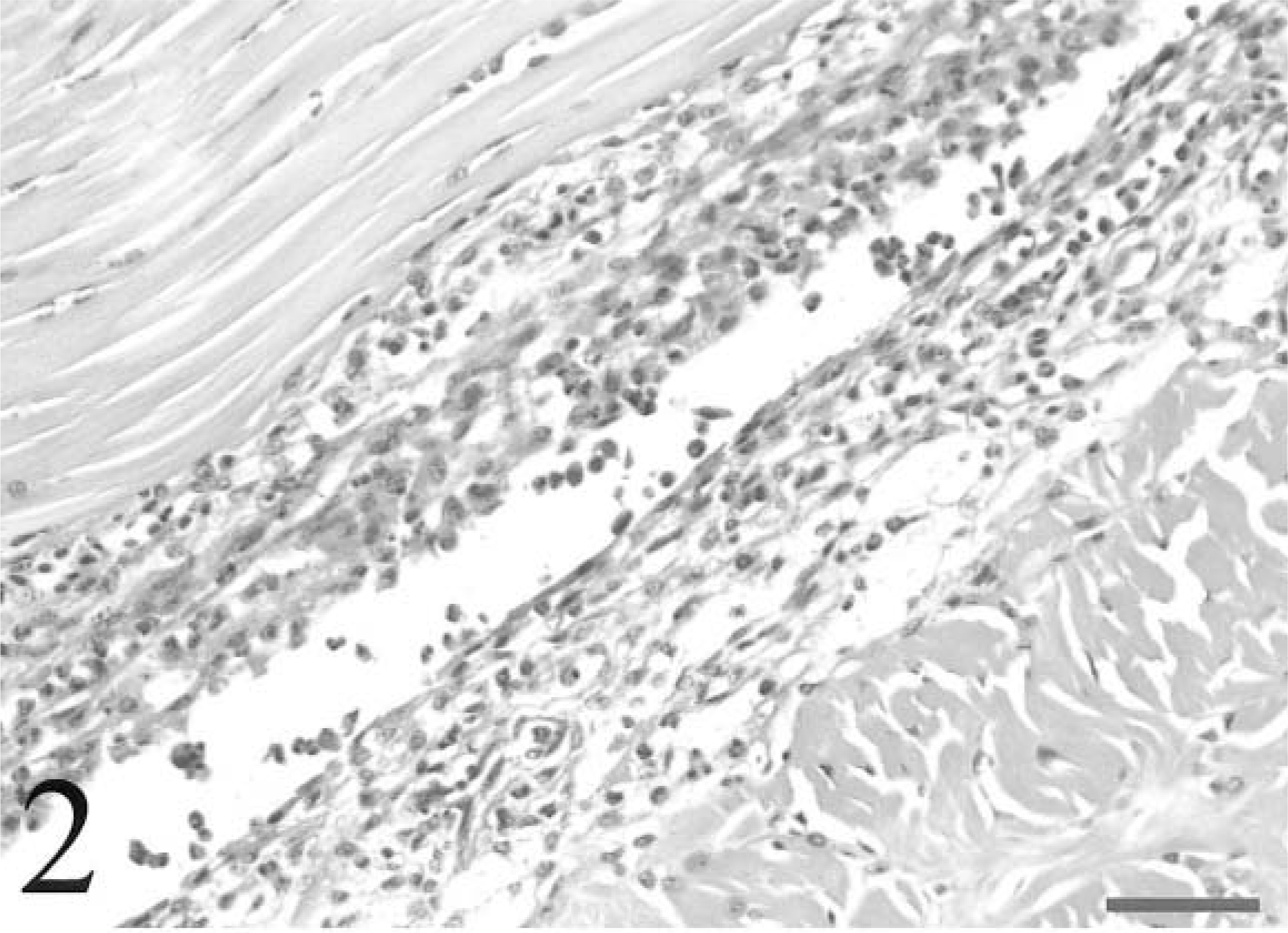
Digital flexor tendon and sheath; avian leukosis virus–negative chicken inoculated with Staphylococcus aureus. The synovium exhibits multifocal hyperplasia and necrosis with heterophil exocytosis. Heterophils are also prominent in subsynovial vessels. The synovial space contains necrotic cells and heterophils. HE. Bar = 50 µm.
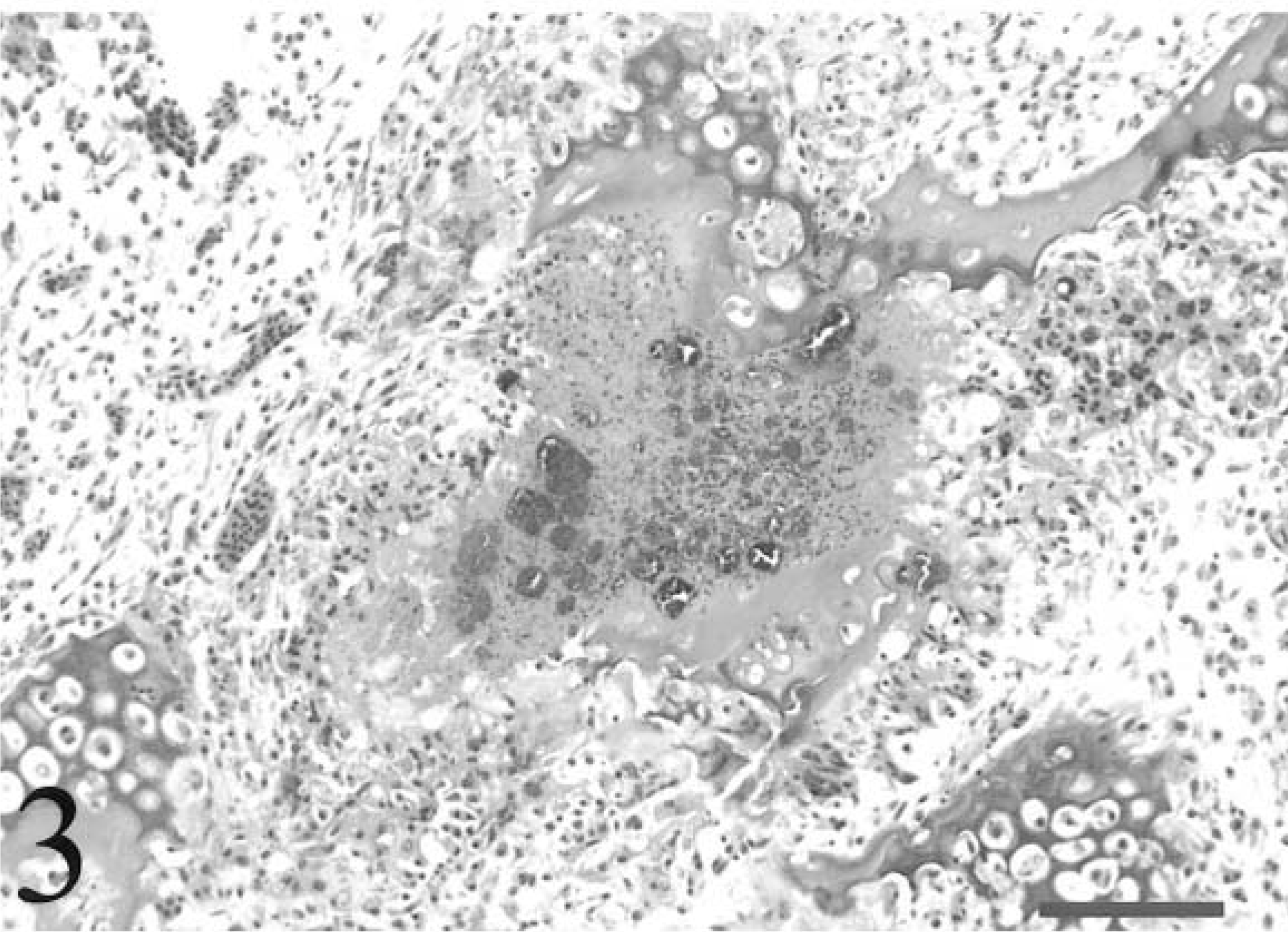
Tibiotarsus; avian leukosis virus–negative chicken inoculated with Staphylococcus aureus. Osteomyelitis disrupts a cartilage column in the zone of ossification of the growth plate. Multiple colonies of bacteria are surrounded by proteinaceous material, necrotic heterophils, and necrotic bone marrow. Fragments of the cartilage column also exhibit chondrocyte necrosis. HE. Bar = 80 µm.
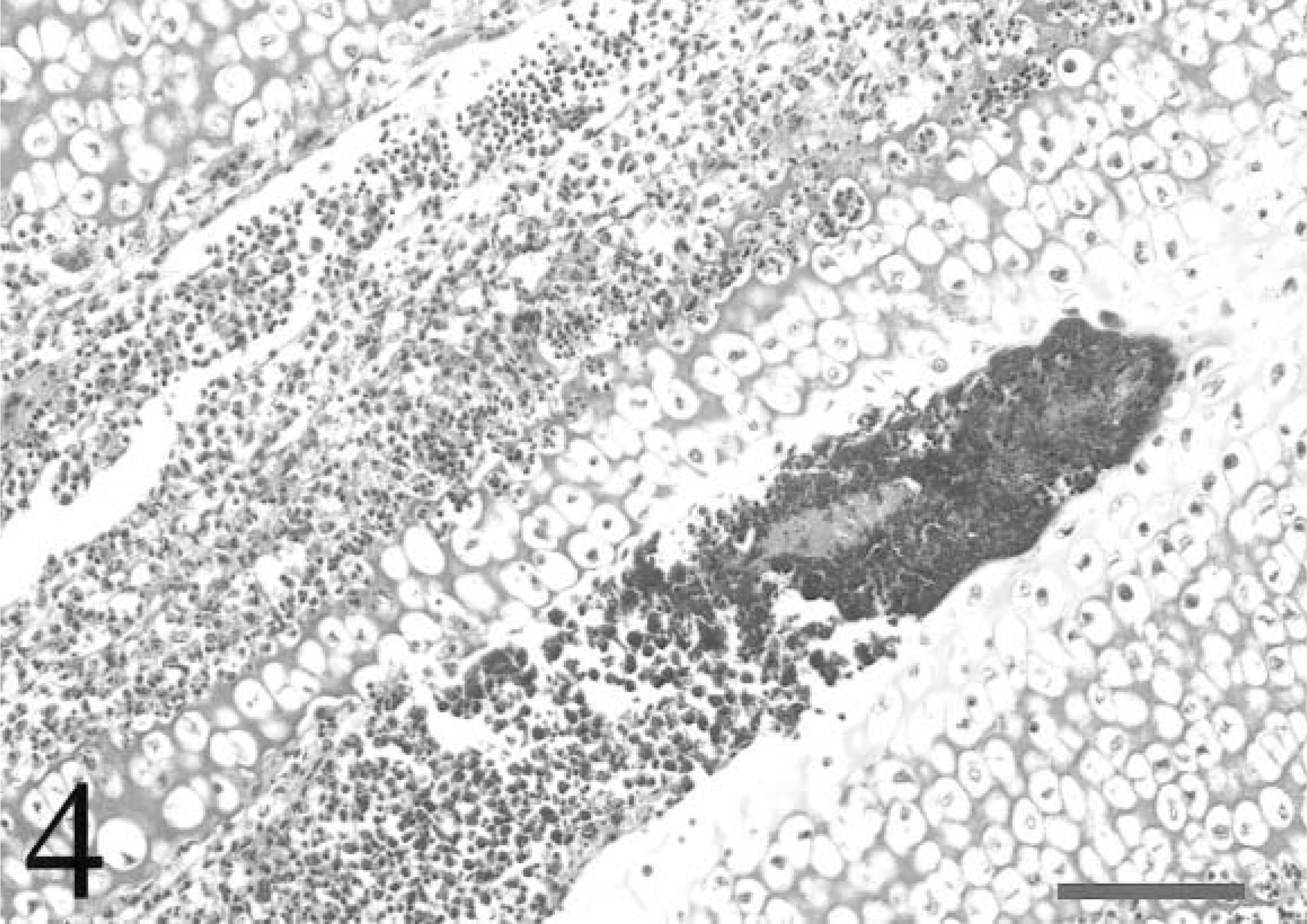
Tibiotarsus; avian leukosis virus–negative chicken inoculated with Staphylococcus aureus. Bacteria completely fill the termination of a metaphyseal vessel penetrating the zone of hypertrophy of the growth plate. Necrotic cell debris is present adjacent to the bacteria and within an adjacent vessel. Cartilage adjacent to the bacteria exhibits matrix eosinophilia and chondrocyte necrosis. HE. Bar = 80 µm.
S. aureus was recovered aerobically from the right hock joint of 6/10 (60%) of the chickens in each group. No other bacteria were recovered.
Leukocyte parameters are summarized for each group in Table 1. All chickens developed a severe leukocytosis characterized by a heterophilia and monocytosis, but total leukocyte and heterophil numbers were significantly higher postinoculation in ALV-negative chickens than in ALV-J–infected chickens. The percentages of heterophils, lymphocytes, and monocytes were very similar between the groups. Band heterophils were slightly more frequent in ALV-negative chickens, but this difference was not significant. Rare minimal heterophil cytoplasmic vacuolation was seen in 3/5 (60%) of the ALV-negative and 4/5 (80%) of the ALV-J–infected chickens, but overt toxic changes were not apparent in heterophils from any of the chickens in either group.
Total and absolute leukocyte counts for avian leukosis virus (ALV)-negative and ALV subgroup J–infected chickens inoculated with Staphylococcus aureus (○ ± SD). Counts are given as the number of leukocytes per microliter whole blood.

∗ Significant difference between the groups, one-way analysis of variance (P < 0.05).
Heterophil isolation and subsequent phagocytosis and bactericidal assays were performed successfully in six ALV-J–infected and five ALV-negative chickens. For the other chickens, clot formation in whole blood samples interfered with separation of heterophils and erythrocytes from mononuclear cell fractions, and insufficient numbers of heterophils were recovered. Isolated heterophil suspensions obtained successfully from the six ALV-J–infected and five ALV-negative chickens contained 95–99% heterophils with minimal erythrocyte contamination. Other leukocytes and thrombocytes were not observed. A 545-base pair PCR product consistent with ALV-J provirus was amplified from all pre- and postinoculation heterophil DNA from all ALV-J–infected chickens (Fig. 5). No PCR product was amplified from heterophil DNA from any of the ALV-negative chickens.
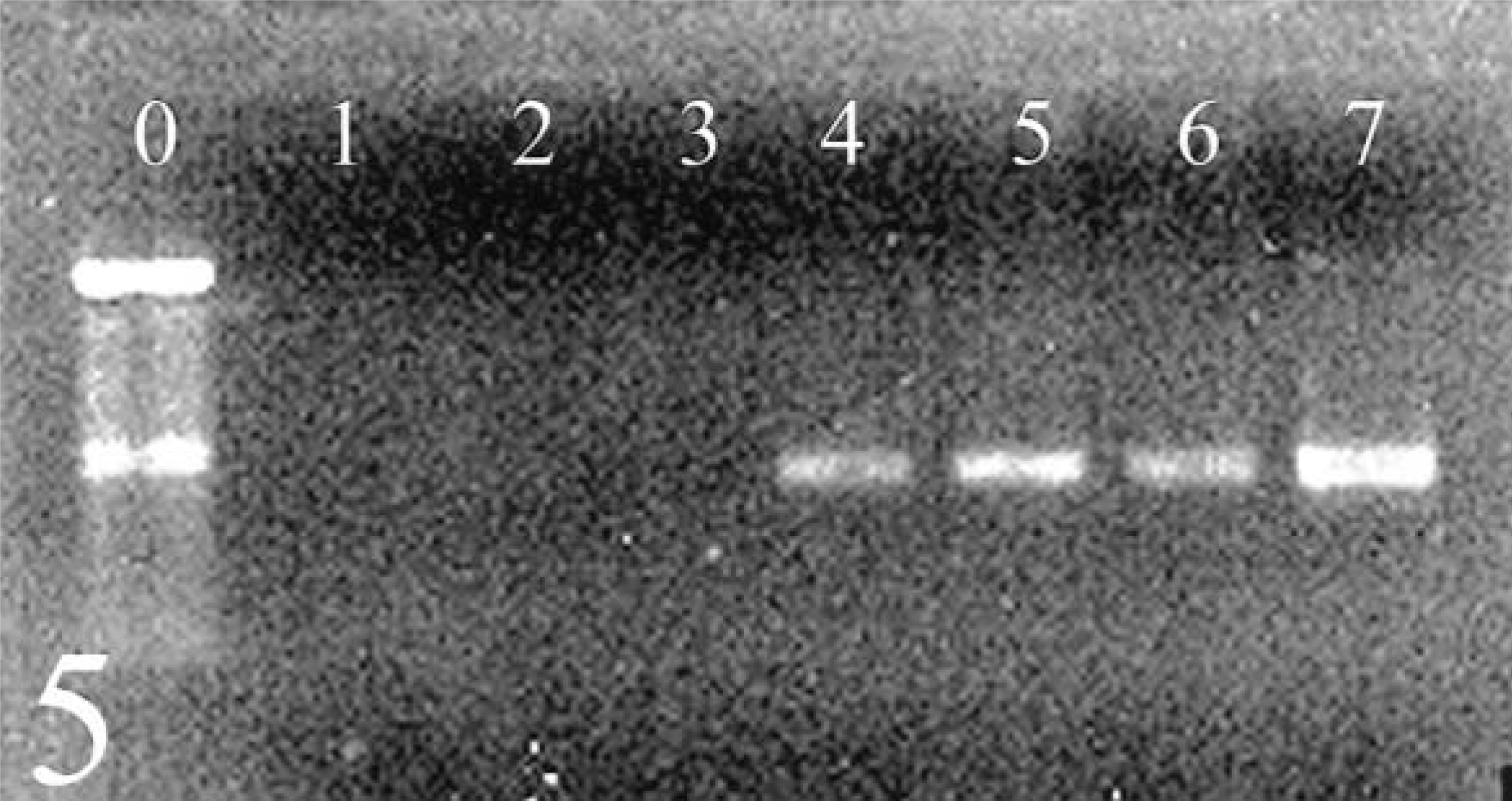
Agarose gel electrophoresis of PCR products obtained from the amplification of heterophil DNA from avian leukosis virus (ALV)-negative (lanes 2, 3) and ALV subgroup J–infected (lanes 4–6) chickens. The expected 545-base pair product indicating the presence of ALV subgroup J provirus was obtained from all infected chickens. Lane 0 = 100-base pair marker; lane 1 = water control; lane 7 = positive control plasmid with provirus insert.
Results for the phagocytosis and bactericidal assays are summarized in Table 2. The percentage of heterophils with phagocytosed latex particles did not differ significantly between ALV-negative and ALV-J–infected chickens either pre- or postinoculation. The percentage of phagocytosis did not increase significantly in either group postinoculation. Although ALV-negative chickens exhibited a greater increase than did ALV-J–infected chickens, this difference was not significant. Both groups exhibited significant increases in heterophil bactericidal ability against the capsular antigen type 8 challenge S. aureus strain, 6-921, and the type 5 capsular strain, 8-5658A, post inoculation. Heterophils from the ALV-negative group killed more of the type 5 capsular strain preinoculation than did heterophils from the ALV-J–infected group. However, bactericidal ability was not significantly different between the groups against either strain of S. aureus either pre- or postinoculation. Bactericidal ability against strain 6-921 was significantly greater than that against strain 8-5658A for both groups of chickens both pre- and postinoculation.
Results of latex particle phagocytosis and bactericidal assays for avian leukosis virus (ALV)-negative and ALV subgroup J–infected chickens inoculated with Staphylococcus aureus. Results are given for assays performed pre- and postinoculation and are expressed as percentages (○ ± SD). Bacterial killing results are given for both the S. aureus capsular antigen type 8 challenge strain (6-921) and the type 5 strain (8-5658A).

∗ Significant increase in postinoculation compared with preinoculation killing, one-way analysis of variance (P < 0.05).
Discussion
Of the eight capsular polysaccharide types described for S. aureus, capsular antigen type 5 and type 8 are most frequently isolated from staphylococcal lesions in poultry, implying increased pathogenicity of these capsular types for poultry. 10 Capsular antigen types 5 and 8 are composed of repeating polymers, similar to other capsular antigen types and teichoic acids, but differ in substitutions that may confer differences in antigenicity and pathogenicity. 13,20,21 Although type 5 strains of S. aureus are more frequently isolated from staphylococcal lesions in poultry than are type 8 strains, both strains will induce osteomyelitis and tenosynovitis, and a type 8 strain was selected for this study, similar to the model as previously described. 9,10
Intravenous inoculation of S. aureus produced tenosynovitis and osteomyelitis in ALV-negative chickens and in chickens congenitally infected with ALV-J. The clinical signs and gross and histologic lesions were similar to those previously described for both natural and experimental staphylococcal tenosynovitis and osteomyelitis in chickens. 2,9,11,12,17 Because a uniform challenge of the same number of organisms was used for all chickens, the severely stunted ALV-J–infected chickens received more organisms per kilogram of body weight. The staphylococcal osteomyelitis model has been described using both a uniform challenge and a challenge based on body weight. 9,12 However, the development of tenosynovitis and osteomyelitis is dependent on the establishment of bacteremia, and log differences in the numbers of organisms are required to produce differences in the establishment of bacteremia and frequency of lesions. 2,9 An intravenous challenge based on body weight would not have resulted in log differences in the numbers of organisms administered to any chickens in this study. Therefore, a uniform challenge similar to that previously described to investigate heterophil function was used. In this study, a challenge of 106 organisms was sufficient to produce both osteomyelitis and tenosynovitis, whereas others have reported that a challenge of 107 organisms is necessary to produce osteomyelitis. 2
The recovery of S. aureus from the hock joint and the development of osteomyelitis were remarkably similar between the groups. The development of osteomyelitis is dependent on a continuous bacteremia. 9 Therefore, the similar development of osteomyelitis in both groups implies that the resistance to the establishment and maintenance of bacteremia was also similar in both groups. Because the heterophil is important for host defense against microbial pathogens, this finding provides indirect evidence for similar heterophil function in both groups. Colonies of bacteria, necrosis, and inflammation were frequently present at the termination of metaphyseal vessels in the zone of hypertrophy in the growth plate, consistent with the hypothesis that frequent deposition of bacteria at termination of these vessels induces metaphyseal osteomyelitis in chickens. 11 In contrast, the development of tenosynovitis was more frequent and more severe in the ALV-negative group than in the ALV-J–infected group. Because bacteremia is also required for the development of tenosynovitis in the avian model, the milder and less frequent tenosynovitis in the ALV-J–infected chickens implies that they may have been more effective at limiting the establishment and maintenance of bacteremia. Factors contributing to the severity of staphylococcal tenosynovitis are not well defined. Heterophil infiltrates can directly damage tissues by degranulation, and the more severe heterophilia in the ALV-negative chickens may have allowed greater heterophil infiltration of the synovium and therefore more tissue damage. 15 The reasons for the higher total leukocyte and heterophil counts in the ALV-negative chickens than in the ALV-J–infected chickens are unknown. Possible factors contributing to this finding could include impaired leukocyte production, maturation, or release or shortened leukocyte lifespan in ALV-J–infected chickens. Alternatively, the more severe tenosynovitis may have induced the higher total leukocyte and heterophil counts in the ALV-negative chickens. Both groups of birds became infected with avian reovirus during the course of the study. However, the reovirus infection appeared chronic and probably did not significantly impact the severity of the acute staphylococcal lesions.
Similar heterophil bactericidal ability was present in both groups of chickens pre- and postinoculation with no significant differences between the groups. This observation indicates that heterophils from ALV-J–infected chickens are capable of similar function and functional enhancement after bacterial challenge. Heterophils from both groups of chickens killed more of the capsular antigen type 8 strain of S. aureus relative to the capsular antigen type 5 strain both pre- and postinoculation, as in a previous investigation. 7 A greater innate ability of heterophils to kill the type 8 strain may explain why this capsular antigen strain is not isolated from poultry lesions as frequently as capsular antigen type 5 S. aureus. Heterophils will also kill significantly more nonpathogenic S. aureus than pathogenic strains in vitro, and the increased killing of the type 8 strain also may reflect a lower pathogenicity of this organism in chickens. 19 In this study, killing of both capsular antigen types was achieved using nonspecific sera for opsonization, similar to previous findings. 1 No functional enhancement of latex particle phagocytosis was observed postinoculation in either group of chickens, although the percentages of heterophils exhibiting phagocytosis were similar to or even slightly greater than those in previously published studies. 2,7 The reason for the lack of phagocytic enhancement is unknown. In natural cases of staphylococcal osteomyelitis and tenosynovitis, heterophils exhibit no enhancement of phagocytic and bactericidal ability or may even exhibit reduced chemotactic function, possibly caused by the prolonged nature of the disease and subsequent heterophil fatigue. 3,5 However, chickens were acutely infected in this study, and enhancement of both bactericidal and phagocytic ability would be expected.
The consistent detection of provirus in DNA isolated from heterophil suspensions from ALV-J–infected chickens is consistent with the described tropism of ALV-J. The lack of any apparent impact on heterophil function suggests that ALV-J may differ in its effects on the host from the subgroup B ALVs that impact antigen-presenting cell function by proviral synthesis. Alternatively, the provirus amplified by PCR may have originated from contaminating erythrocytes. However, considering the selective transformation of immature granulocytes during ALV-J infection, the frequent presence of provirus in heterophils would be expected. Impaired leukocyte function and increased susceptibility to bacterial infection have been described for other retroviral infections. For example, human immunodeficiency virus (HIV)-infected patients with low CD4 lymphocyte counts have reduced neutrophil phagocytosis of bacteria, and HIV infection is considered a risk factor for septic arthritis in hospitalized patients. 26,27 We were unable to document a similar phenomenon for ALV-J infection. Staphylococcal antigens can also enhance HIV replication, implying that concurrent bacterial infections may also be an important factor in the pathogenesis of HIV infection. 14,18 The role of staphylococcal antigens in the enhancement of ALV infection is unknown and warrants further investigation, because staphylococcal infections are relatively common in poultry.
Heterophils from ALV-J–infected chickens exhibit no functional deficits and adequate functional enhancement after staphylococcal infection, and ALV-J–infected chickens do not exhibit increased susceptibility to staphylococcal infection.
Footnotes
Acknowledgements
This research project was supported by a grant from the US Poultry and Egg Association.
