Abstract
Inducible nitric oxide synthase (iNOS) is important in the control of a number of intracellular pathogens, including mycobacteria, and is a marker of classic macrophage activation. In human granulomatous diseases such as leprosy, a spectrum of granulomatous lesions is described, ranging from the tuberculoid to lepromatous types. Tuberculoid granulomas are associated with enhanced iNOS production and improved clinical outcomes over the lepromatous types. The aim of this study is to determine whether an association exists between morphology of bovine Johne's disease granulomas and lesion macrophage effector functions. To accomplish this, we retrospectively evaluated 24 cases of bovine Johne's disease. In each case, we recorded the predominant granuloma morphology and evaluated iNOS immunoreactivity and bacterial burden by acid-fast stains and mycobacterial immunolabeling. The results of this study demonstrate that all cases had granulomas with features most similar to the lepromatous type. This morphology correlated with heavy bacterial burdens demonstrated by acid-fast staining and mycobacterial immunoreactivity. None of the cases had high expression of iNOS in mycobacterial-positive granulomas. When iNOS immunoreactivity was identified, it was usually located near the crypts and was distinct from the granulomatous foci.
Keywords
Inducible nitric oxide synthase (iNOS) plays a crucial role in host defense and is a major killing mechanism within macrophages. iNOS is one of three isoforms of nitric oxide synthase that generates nitric oxide (NO) from
iNOS expression is induced by several types of signals. Environmental factors such as hypoxia and bacterial components including lipopolysaccharide and mycobacterial lipoarabinomanins will lead to iNOS expression in macrophages. 23, 37 In addition, iNOS is strongly induced by T-helper 1 (Th1)–type cytokines including interferon-γ (IFNγ), interleukin 2, and tumor necrosis factor–α; therefore, iNOS expression is a marker of Th1-mediated macrophage activation. 15, 20
Similar to other species of pathogenic mycobacteria, Mycobacterium avium subspecies paratuberculosis (M. a. ptb) is an intracellular pathogen of macrophages and monocytes. 46 The pathogenesis of M. a. ptb infection at the macrophage level is incompletely understood, with much of our current understanding derived from the M. avium and M. tuberculosis literature and from in vitro studies on M. a. ptb infection in bovine and murine macrophages. Collectively, these data suggest that if macrophages can be appropriately activated, they will kill intracellular M. a. ptb. 22, 43, 47
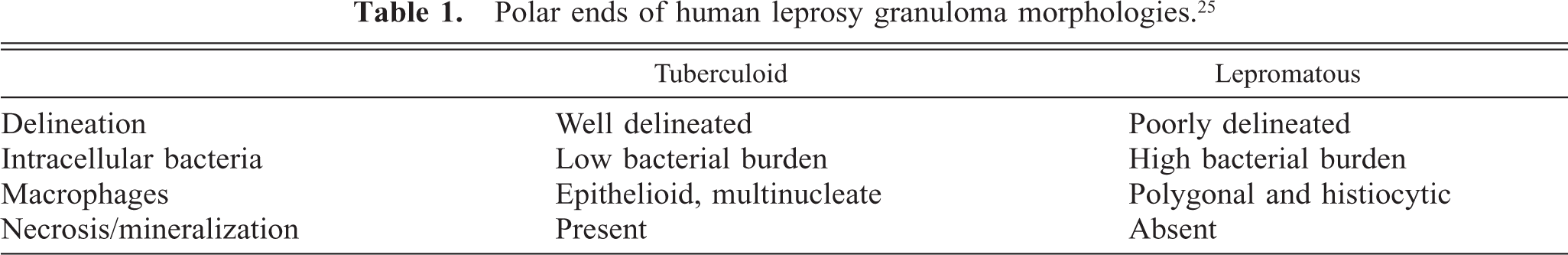
The characteristic intestinal lesion of clinical Johne's disease is granulomatous enteritis; however, there are variations in the morphology of granulomatous infiltrates encountered. Johne's lesions in sheep have been best classified using several different systems, one of which describes a spectrum of granuloma types that have features in common with the lepromatous, borderline, and tuberculoid granulomas of leprosy. 35, 39 Differences in granuloma types in bovine Johne's disease have been less thoroughly evaluated, and classification schemes have used both severity and distribution to define lesion categories. 5, 8 Although not as distinct as sheep, tuberculoid-like and lepromatous-like granulomas lesions have been reported in cattle. 11, 26 In humans, control of mycobacterial infection correlates with granuloma morphology, with tuberculoid granulomas imparting a more favorable outcome than lepromatous types. 14, 27, 29 iNOS expression is high in human tuberculoid granulomas resulting from effective macrophage activation, which likely contributes to control of mycobacterial proliferation. 15, 25
In bovine Johne's disease, the relationship between granuloma morphology and mycobactericidal activity is not as well defined as it is for human granulomas. The goal of this study is to determine whether an association exists between bovine M. a. ptb granuloma morphology and antimicrobial effector function, using expression of iNOS as a marker. Our hypothesis is that granulomas of clinically infected animals are most similar to the lepromatous type that lack iNOS expression. We tested this hypothesis with a retrospective study evaluating bovine intestinal lesions from 24 cases diagnosed with Johne's disease through the Iowa State University's Veterinary Pathology and Veterinary Diagnostic Laboratory necropsy services. We evaluated intestinal granulomas for severity and distribution and for lepromatous and tuberculoid features (Table 1). We then evaluated iNOS, mycobacterial antigen, and the macrophage marker CD68 (to determine origin of iNOS+ cell types) with an immunohistochemical staining technique. We present evidence in this study that granulomas in the intestinal tract of clinical bovine Johne's disease have features similar to lepromatous granulomas, which lack significant iNOS expression.
Polar ends of human leprosy granuloma morphologies. 25
Materials and Methods
Reagents
Antibodies used are as follows (see Table 2). Protease, Trizma base, normal goat serum (Sigma, St. Louis, MO); hydrogen peroxide (Fisher, Hanover Park, IL); horseradish peroxidase (HRP)–streptavidin, alkaline phosphatase–streptavidin (Zymed, San Francisco, CA); diaminobenzidine (DAB) substrate solution (Dako, Carpinteria, CA); Vector Red alkaline phosphatase substrate (Vector, Burlingame, CA).
Primary/secondary antibodies, suppliers and conditions.

Case selection
Formalin-fixed and paraffin-embedded bovine intestinal tissues with a diagnosis of Johne's disease were selected from the archives of the Iowa State University Veterinary Pathology and Veterinary Diagnostic Laboratory necropsy services. Bovine submissions with this diagnosis were reviewed from 1993 to 2002. To be included, sections of intestine had to have limited postmortem autolysis, with clearly retained mucosal architecture and granulomatous lesions of M. a. ptb infection present. Twenty-four cases were deemed suitable to include in the study. All cases had typical presenting histories of chronic Johne's disease, eight cases were confirmed by culture, two further cases had positive serology. A lymph node with typical tubercle granulomas taken from a bovine infected with M. bovis (Dr. Y. Niyo at the National Veterinary Services Laboratories, Ames, IA) was used as a positive control for iNOS expression in bovine granulomatous inflammation.
Histopathology
The entire length of the intestinal section was examined for each case. Cases were classified based on a score for severity of granulomatous infiltrates using 1 for mild, 2 for moderate, and 3 for marked and by distribution using focal, multifocal, and diffuse categories. Macrophage morphology in each case was evaluated using polygonal, epithelioid, histiocytic, and multinucleate as categories. Using distribution, bacterial burden, and macrophage morphology as parameters, we then compared each case to tuberculoid and lepromatous granulomas.
Immunohistochemistry
Polyclonal rabbit anti–M. bovis has extensive cross-reactivity with M. a. ptb and is a useful tool for the identification of M. a. ptb in bovine granulomas. 7 The intestinal lesions of M. a. ptb infection were readily differentiated from those of intestinal tuberculosis caused by M. bovis by lack of caseous necrosis and mineralization.
Before staining, slides were deparaffinized, rehydrated, and processed for immunohistochemistry (IHC).
iNOS/M. a. ptb combined staining
Antigen retrieval was accomplished by microwaving slides in a pH 10 buffer for 5 minutes. Primary (α-iNOS) and appropriate secondary antibody were added to slides under the conditions described in Table 2. HRP-streptavidin diluted 1 : 200 was added to the tissue for 15 minutes. DAB was added to the slides until optimal staining developed (12 minutes). Primary (α-M. bovis) and appropriate secondary antibodies were added to the slides under the conditions described in Table 2. Alkaline phosphatase–streptavidin diluted 1 : 200 was added to the slide (15 minutes). Vector Red Substrate was added until optimal staining developed (20 minutes).
CD68 stain
Antigen retrieval for the CD68 stain was accomplished with 2% protease for 15 minutes. Primary and appropriate secondary antibody were added under the conditions described in Table 2. Cross-reactivity of mouse anti-human CD68 with bovine macrophages has been reported previously. 1 The following controls were included in the study: stains with the primary antibody omitted and stains where an irrelevant (isotype control) antibody replaced the specific primary antibody.
Acid-fast staining (Ziehl-Neelsen method)
Slides were deparaffinized and rehydrated in distilled water. Carbol fuchsin solution was added for 45 seconds while microwaving at 450 W. Slides were rinsed in 0.5% acid alcohol for decolorization. Slides were rinsed for 1 minute in tap water then counterstained in methylene blue for 1 minute. Acid-fast stains were quantified by the following scoring system: 0 = no acid-fast bacteria,
Evaluation of stained slides
A previously described method used for evaluation of iNOS in bovine tissues was adapted for use in this study. 17 In brief, immunoreactivity of iNOS, M. a. ptb, and CD68 was evaluated for relative frequency and anatomic location within each lesion. Overlap of markers in a given focus was evaluated by examination of serial sections. A semiquantitative approach was used to score the frequency of positive cells by location. The immunoreactivity of the stains was scored by the following scale 0 = no immunoreactive cells, 1 = rare immunoreactive cells, 2 = scattered immunoreactive cells, 3 = clusters of immunoreactive cells, 4 = nearly diffuse staining. Slides were initially examined independently by two investigators and differences in scoring resolved during a combined session.
Results
Histopathology of M. a. ptb granulomas
Severity and distribution
Twenty-two (92%) cases had granulomatous inflammation in the 3 (marked) category. Two cases (cases 20, 21) had a severity score of 2 (moderate) (8%). There were equal numbers of cases in the diffuse (cases 1–12) and multifocal (cases 13–24) distribution categories (Table 3).
Case signalments and granuloma morphologies by HE staining.∗
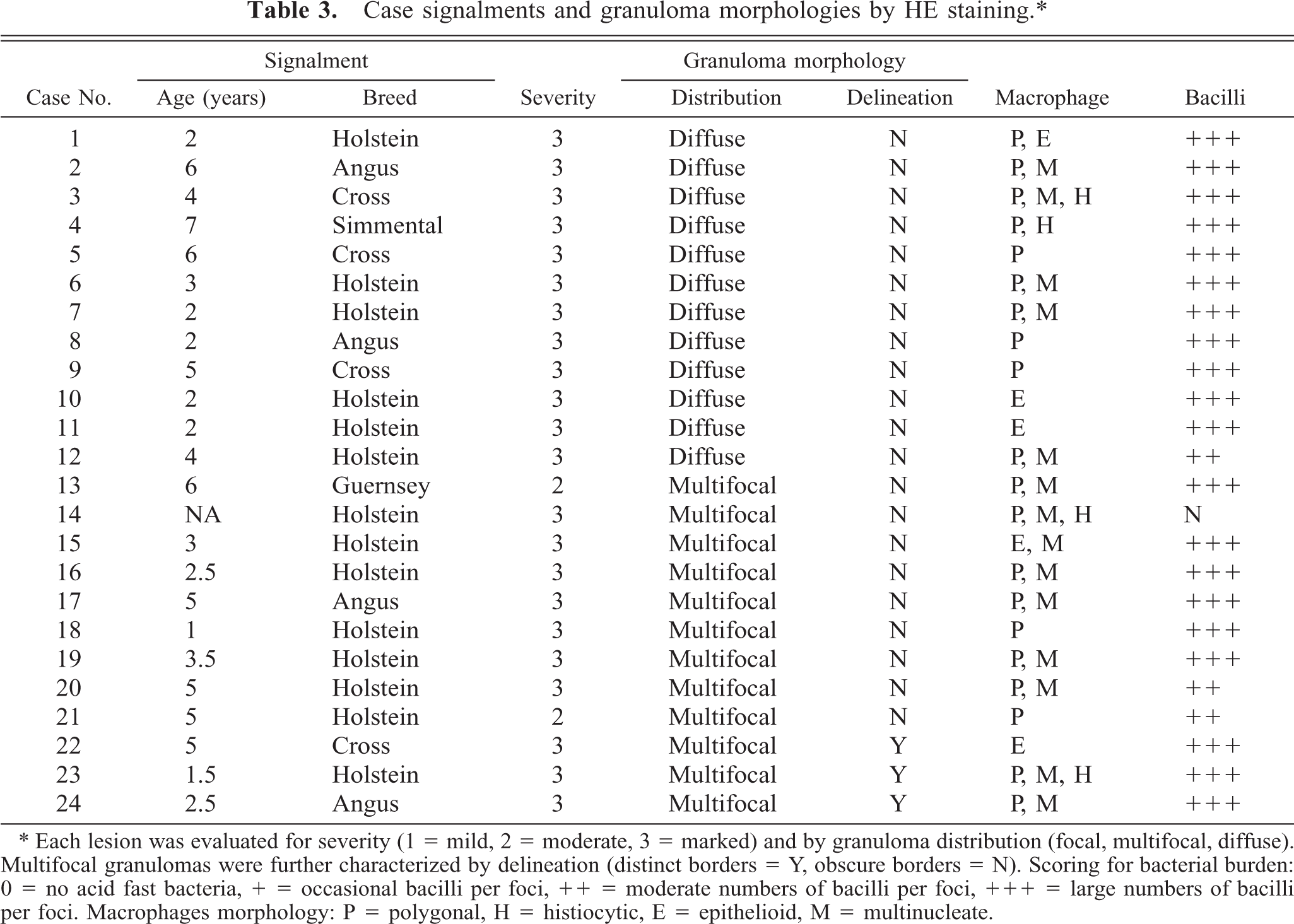
∗ Each lesion was evaluated for severity (1 = mild, 2 = moderate, 3 = marked) and by granuloma distribution (focal, multifocal, diffuse). Multifocal granulomas were further characterized by delineation (distinct borders = Y, obscure borders = N). Scoring for bacterial burden: 0 = no acid fast bacteria, + = occasional bacilli per foci, ++ = moderate numbers of bacilli per foci, +++ = large numbers of bacilli per foci. Macrophages morphology: P = polygonal, H = histiocytic, E = epithelioid, M = multinucleate.
Granuloma morphology, macrophage morphology, and intracellular bacterial burden
We identified three granuloma morphologies from M. a. ptb. infected animals in this study. Cases 1–12 (50%) granulomas were in the first category, which had diffuse distribution. These granulomas were widespread and tended to expand the mucosal surface and extend into the sub-mucosa of the section (Fig. 1). Cases 13–21 (38%) granulomas fell into the second category, with multi-focal distribution without clear delineation of granuloma boundaries. These lesions consisted of variable-sized and poorly delineated macrophage infiltrates along with scattered individual macrophages and multinucleate giant cells throughout the lamina propria. Cases 22–24 (12.5%) granulomas fell into the third category, with multifocal distribution and well-delineated boundaries (Fig. 2). Granuloma borders were distinct because of an abrupt border with normal adjacent lamina propria. Macrophage morphology in all cases was variable, and more than one morphology was often present within a given lesion (Table 3). The majority (20) of cases had lesions that consisted mainly of polygonal macrophages with abundant and often vacuolated cytoplasm. Multinucleate cells were also common. Three cases had histiocytic macrophages, and four cases had lesions that consisted mainly of epithelioid macrophages (Table 3). Acid-fast bacilli were present within macrophages in all cases. Twenty-one cases (88%) had heavy (+++) bacterial burdens. Cases 12, 20, and 21 (12.5%) had moderate (++) numbers of bacilli within macrophages (Table 3). These three cases were in either the diffuse or in the multifocal, poorly delineated granuloma categories (Table 3). The histopathology of the lymph node lesion from the M. bovis infected animal was consistent with the tubercle type granuloma described for M. bovis infection in cattle (Fig. 3). 38
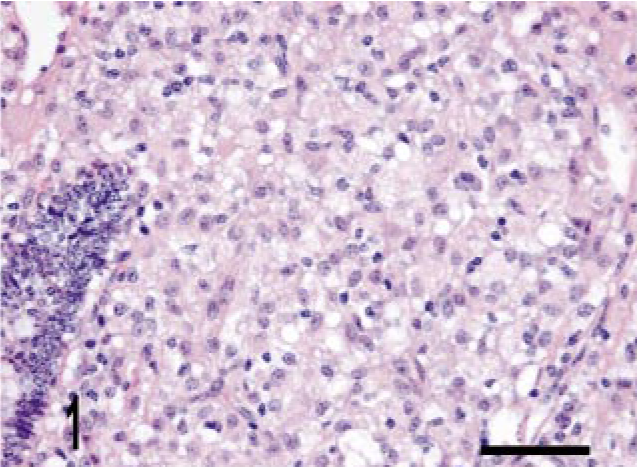
Intestine; bovine, infected M. a. ptb, case No. 5. Diffuse granuloma within the lamina propria. HE stain. Bar = 38 μm.
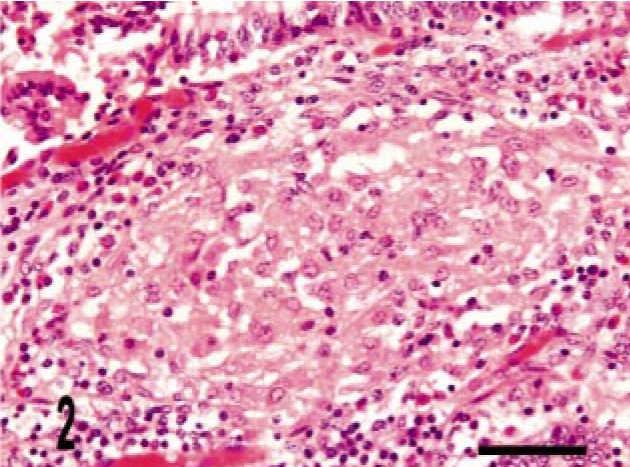
Intestine; bovine, infected M. a. ptb, case No. 24. Well-delineated granuloma within the lamina propria. HE stain. Bar = 38 μm.
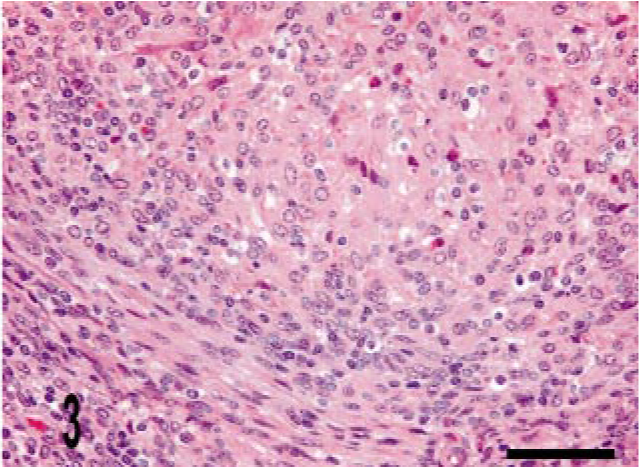
Lymph node; bovine, infected M. bovis. Tubercle granuloma with border of fibrous tissue. HE stain. Bar = 38 μm.
IHC of M. a. ptb granulomas
Mycobacterial staining
Granulomas in all 24 M. a. ptb. cases had diffuse mycobacterial immunoreactivity (score of 4), which tended to intensely label lesion macrophages (Figs. 4, 5). The M. bovis granuloma had little to no staining for mycobacterial antigen (score of 0–1) (Fig. 6). Mycobacterial immunoreactivity strongly correlated with heavy bacterial burdens demonstrated by acid-fast staining (Figs. 7–9).
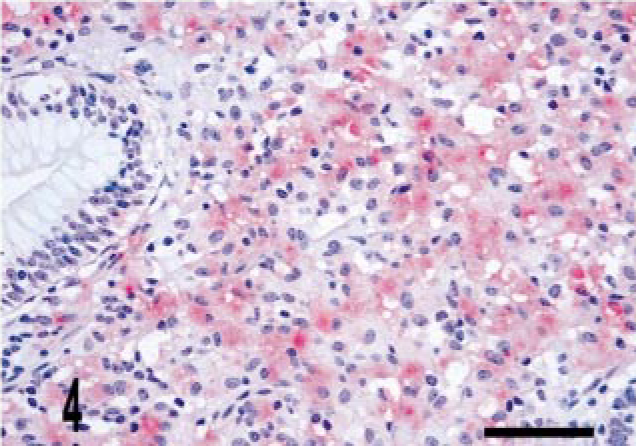
Intestine; bovine, infected M. a. ptb, case No. 5. The granuloma does not stain for iNOS but does stain diffusely for mycobacterial antigen. Dual M. bovis and iNOS immunolabeling. ABC method with Harris' hematoxylin counterstain. iNOS stain is brown, whereas M. bovis stain is red. Bar = 38 μm.
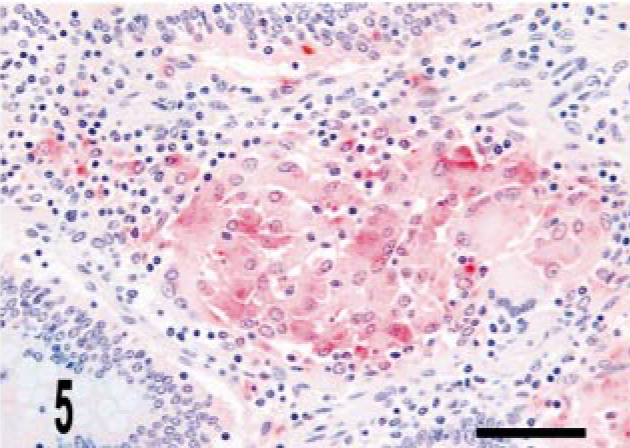
Intestine; bovine, infected M. a. ptb, case No. 24. The granuloma does not stain for iNOS but does stain diffusely for mycobacterial antigen. Dual M. bovis and iNOS immunolabeling. ABC method with Harris' hematoxylin counterstain. iNOS stain is brown, whereas M. bovis stain is red. Bar = 38 μm.
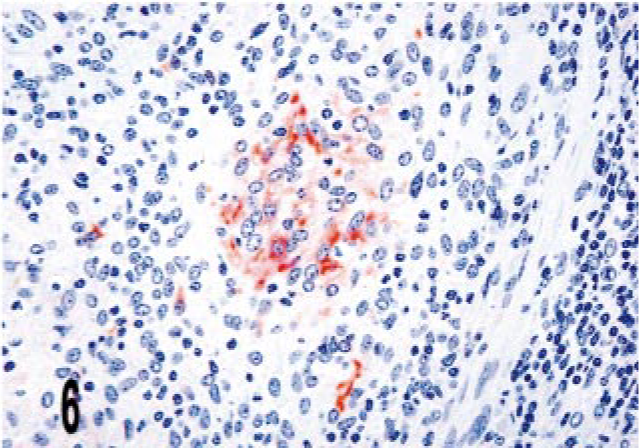
Lymph node; bovine, infected M. bovis. The center of the granuloma contains multiple iNOS immunoreactive cells (arrow). Dual M. bovis and iNOS immunolabeling. ABC method with Harris' hematoxylin counterstain. iNOS stain is brown, whereas M. bovis stain is red. Bar = 38 μm.
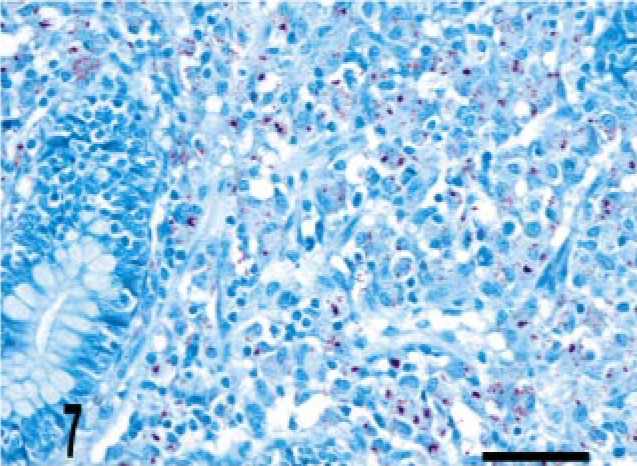
Intestine; bovine, infected M. a. ptb, case No. 5. Numerous acid-fast bacilli are present within the granuloma. Ziehl-Neelsen method. Bar = 38 μm.
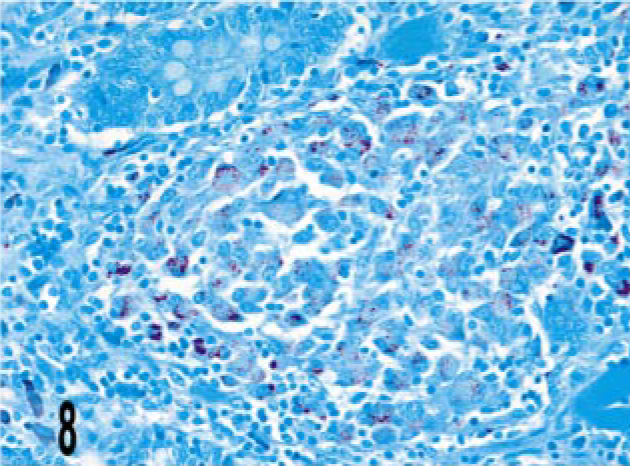
Intestine; bovine, infected M. a. ptb, case No. 24. Numerous acid-fast bacilli are present within the granuloma. Ziehl-Neelsen method. Bar = 38 μm.
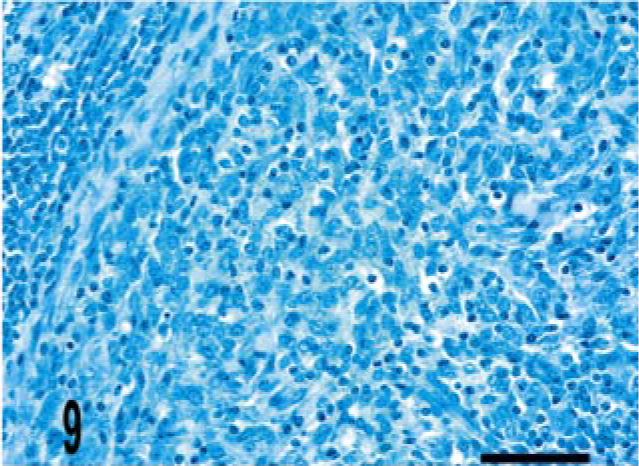
Lymph node; bovine, infected M. bovis. Few to no acid-fast bacilli are present within the granuloma. Ziehl-Neelsen method. Bar = 38 μm.
iNOS staining
The vast majority of granulomas from M. a. ptb. animals lacked iNOS immunoreactivity (scores of 0–1) (Figs. 4, 5). Variation in granuloma morphology did not significantly alter iNOS expression (Table 4, Figs. 1b, 2b) When overlap of iNOS and M. a. ptb occurred within granulomas, it was typically single macrophages within a 40× field. Overlap was, however, more frequent in dilated crypts, where clusters of mycobacteria and iNOS-positive cells intermixed with moderate numbers of neutrophils and cell debris were present (crypt abscesses). iNOS expression in areas of neutrophilic infiltrate has been reported previously in bovine inflammatory lesions. 17 In 19 cases, iNOS staining was identified in low frequency (score of 2) near the basal aspect of the crypts in the deep lamina propria. In the lymph node granuloma from the M. bovis infected animal there was multifocal iNOS immunoreactivity within the granulomas (score of 2–3), which was most near the center as well as areas bordering foci of necrosis and neutrophil infiltration (Fig. 6).
Immunoreactivity in M. a. ptb intestinal lesions.∗

∗ 0 = no immunoreactive cells. 1 = rare immunoreactive cells, 2 = scattered immunoreactive cells, 3 = clusters of immunoreactive cells, 4 = nearly diffuse staining. Note: granulomas have high immunoreactivity for mycobacteria and low immunoreactivity for iNOS. Variation in granuloma morphology does not affect this staining pattern.
CD68 staining
CD68 is a marker of histiocytic cells and macrophages that recognizes a cytoplasmic epitope also known as KP-1. 6 In addition to diffuse labeling (score of 4) of granuloma macrophages (Figs. 10–12), CD68 immunoreactive cells were also located in the deep aspect of the lamina propria adjacent to the crypts (score of 2) (Fig. 11). To better determine the origin of iNOS-expressing cells near the crypts, we evaluated serial sections for iNOS and CD68 expression. We observed that iNOS immunoreactivity associated with the crypts included, but was not limited to, CD68-positive cells (Figs. 13, 14).
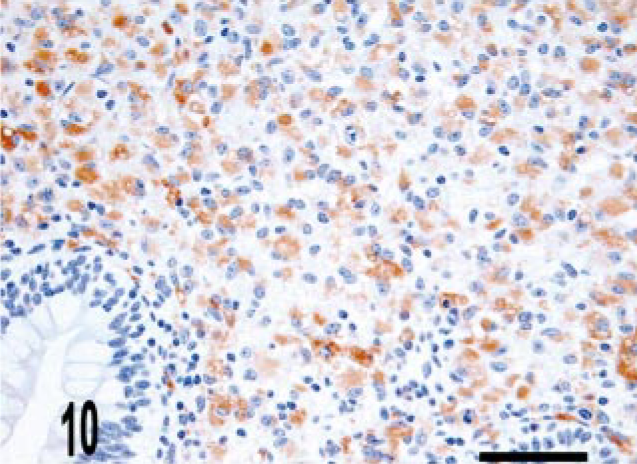
Intestine; bovine, infected M. a. ptb, case No. 5. Macrophages within the granuloma stain with CD68. CD68 immunolabeling. ABC method with Harris' hematoxylin counterstain. Bar = 38 μm.
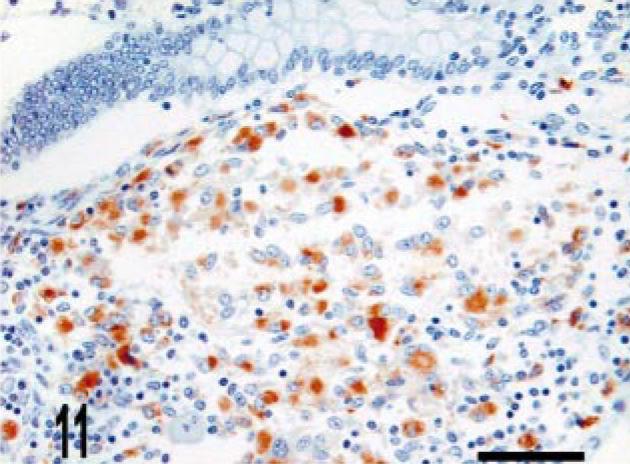
Intestine; bovine, infected M. a. ptb, case No. 24. Macrophages within the granuloma stain with CD68. CD68 immunolabeling. ABC method with Harris' hematoxylin counterstain. Bar = 38 μm.
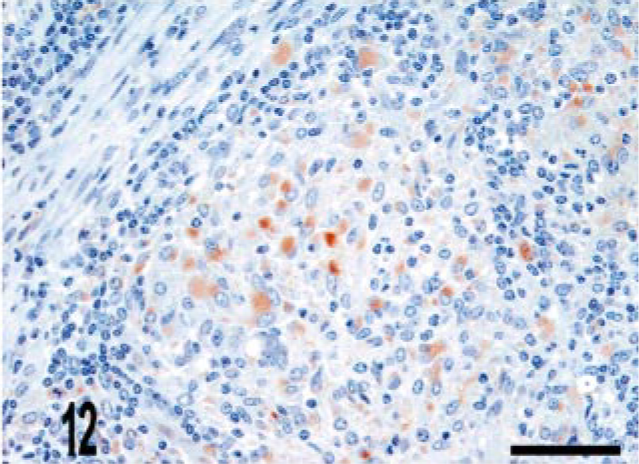
Lymph node; bovine, infected M. bovis. Macrophages within the granuloma stain with CD68. CD68 immunolabeling. ABC method with Harris' hematoxylin counterstain. Bar = 38 μm.
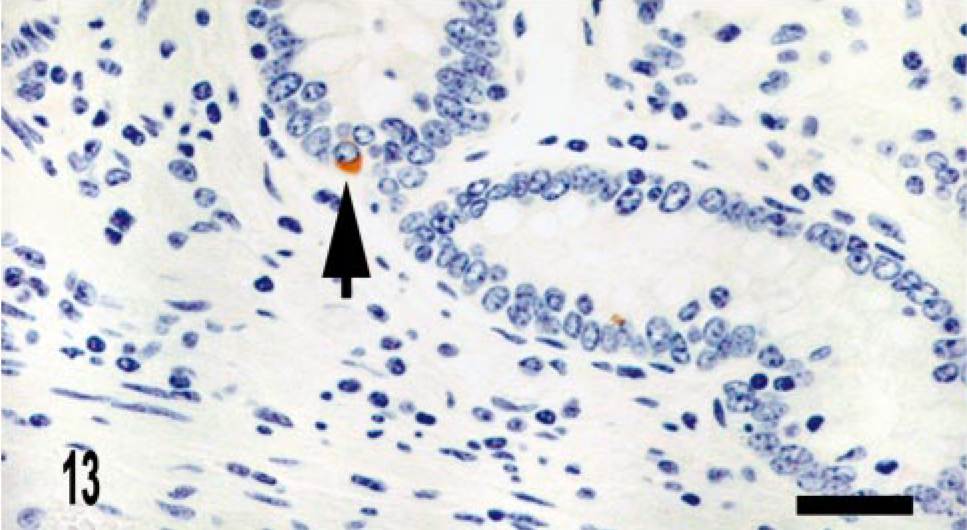
Intestine; bovine, infected M. a. ptb, case No. 13. iNOS immunoreactive cell associated with crypt (arrow). Dual M. bovis and iNOS immunolabeling. ABC method with Harris' hematoxylin counterstain. iNOS stain is brown, whereas M. bovis stain is red. Bar = 28 μm.
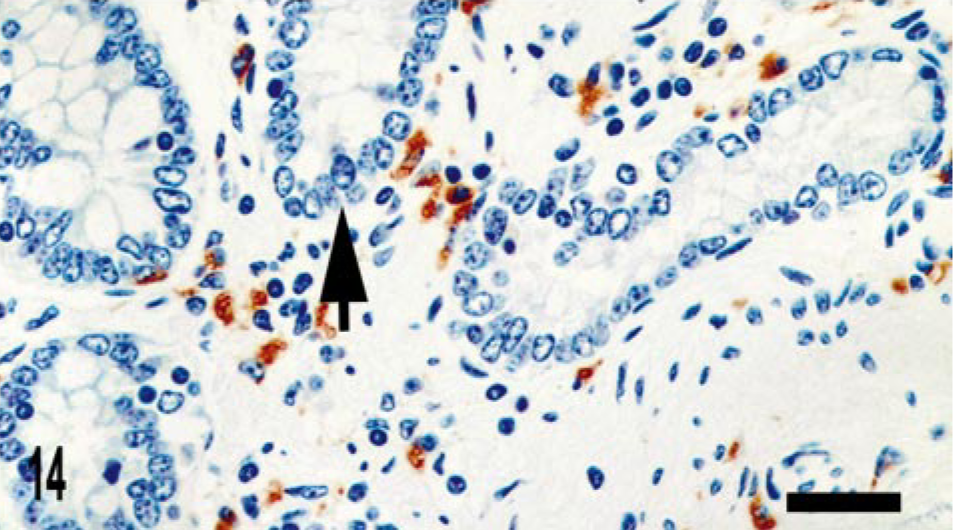
Intestine; bovine, infected M. a. ptb, case No. 13. CD68 staining cells are present adjacent to the crypts; however, iNOS-positive area of crypt is negative for CD68 staining. CD68 immunolabeling. ABC method with Harris' hematoxylin counterstain. Bar = 28 μm.
M. bovis granulomas
The histopathology of the lymph node lesion was consistent with the tubercle-type granuloma described for M. bovis infection in cattle (Fig. 3a). 38 There was multifocal iNOS immunoreactivity within granulomas (score of 2–3), which was most intense near the center as well as areas bordering foci of necrosis and neutrophil infiltration (Fig. 3b). Few to no bacilli were detected by acid-fast staining, and mycobacterial IHC (score of 0–1) was also negative (Figs. 3b, c).
Discussion
Morphologic distinctions of granulomas are significant in that granuloma types can correlate with specific cytokine profiles: lepromatous granulomas with a T-helper 2 (Th2) profile and tuberculoid granulomas with a Th1 profile. 4 Th1 cytokine influence leads to functional differences in macrophages compared with Th2 profiles including classic activation, enhanced iNOS expression, and enhanced mycobacterial killing. 15, 25 In ovine Johne's disease, it is speculated that early control of infection or “reactive paratuberculosis” is due to containment of the infection by Th1-like–mediated (tuberculoid) granulomas. 11, 12, 26
The majority of granulomas in this study (cases 1–21) had similar morphologic features including widespread distribution, poor delineation, and heavy bacterial burden, which comparatively, are features characteristic of lepromatous-type granulomas. A small number of cases (cases 22–24) had more discrete lesions with well-delineated boundaries, features found in tuberculoid-type granulomas. These granulomas, however, lacked additional tuberculoid features including bordering fibrosis, associated lymphocytic infiltrate, and low bacterial burdens.
There is some disparity in reports concerning tuberculoid-type granulomas in bovine M. a. ptb infection. In 1991, Kreeger et al. described tuberculoid granulomas as a common feature in late-stage lesions of bovine Johne's disease, which may be the result of reversal reactions. 26 Reversal reactions are a rapid change from borderline lesions to tuberculoid-type granulomas. 3 In a previous descriptive report on bovine M. a. ptb infection, Buergelt et al. did not identify tuberculoid granulomas in 51 cases of naturally infected cattle. 8 Considering the heavy bacterial burden and lack of other tuberculoid features, the multifocal well-delineated granulomas in this study overall have features closest to lepromatous types. It is also possible that these granulomas represent borderline-type lesions and with time would have completed a reversal reaction into a more tuberculoid morphology.
The vast majority of granulomas had high bacterial burdens, yet were negative for iNOS expression, suggesting decreased macrophage effector function. Although the immune responses in these cattle are largely unknown, it is likely that they had strong humoral responses typical of clinical M. a. ptb infection. Interleukin 10 (IL-10) and interleukin 4 associated with humoral or Th2 responses are inhibitory to macrophage iNOS generation.
11,
36,
40
IL-10 production has been reported in peripheral blood mononuclear cells from cattle clinically infected with M. a. ptb, as well as in vitro M. a. ptb–infected bovine macrophages.
13,
45
In mouse models, iNOS inhibition by Th2 cytokines is mediated by induction of the alternative pathway for
The role of iNOS and NO in control of M. a. ptb infection is not well established, and to the best of the author's knowledge, this is the first report of iNOS immunoreactivity in natural bovine M. a. ptb lesions. In mouse models, NO production is essential for control of M. tuberculosis and M. bovis but apparently enhances M. avium infection. 2, 18, 21 Specifically concerning M. a. ptb, peripheral blood monocytes harvested from experimentally infected calves during the early subclinical period have augmented NO production with M. a. ptb antigen stimulation. 44 In this study, the general lack of iNOS immunoreactivity in the M. a. ptb granulomas corresponds to high bacterial loads, which is contrasted by low bacterial burdens in the M. bovis granulomas, which had higher iNOS expression. Taken together, these data suggest that iNOS and NO play a role in control of M. a. ptb infection, especially during the early period after infection.
The M. bovis lymph node lesion was included in this study as an example of a tubercle granuloma with potential for high iNOS production. Consistent with the Th1 influence well described for bovine tuberculosis, these granulomas had tubercule morphology, multifocal iNOS expression, and low bacterial burdens. 9, 24, 27 Because we examined only a single lesion from one animal with M. bovis infection, we cannot conclude from this study that all M. bovis granulomas in cattle are positive for iNOS expression, but again this example is consistent with the expected Th1 cytokine profile.
iNOS immunoreactivity in the lamina propria in M. a. ptb–infected cattle was mainly located near the crypts, distinct from the granulomatous lesions. We used the macrophage marker CD68 to better identify the cell type expressing iNOS in this location. Serial sections demonstrated a zone of CD68+ cells near the basal aspect of the crypts, similar to a region described in human intestine. 42 In this study, not all iNOS-positive cells in the crypts stained with CD68. The location of these iNOS-positive/CD68-negative cells suggests that they are crypt enterocytes. iNOS expression in enterocytes at a low constitutive level has been reported in multiple species including humans, mice, and pigs. 19, 28, 34, 42 It is uncertain whether this represents truly constitutive expression or whether iNOS is continuously induced by antigens in the luminal environment. With inflammation, however, iNOS expression is rapidly increased, and NO derived from crypt epithelia may be an important contributor to mucosal injury in intestinal inflammatory conditions. 34, 42 We examined sections of intestine from five healthy adult cattle and found iNOS immunoreactivity at a much lower intensity and frequency than in the M. a. ptb–infected cattle (data not shown). Future comparative and immunohistochemical studies evaluating normal and inflamed bovine intestine using epithelial cell markers and nitrotyrosine would be beneficial in establishing the iNOS source and its role in mucosal inflammation in bovine intestine.
Summary
In this study we identified mild variations in granuloma morphology in bovine Johne's disease, but all cases had granuloma features closest to the lepromatous type. These granulomas lacked significant iNOS expression and had high bacterial loads, which suggests diminished effector responses. We speculate that, similar to the lepromatous form of human leprosy, the granuloma morphology and function in these cattle is associated with strong humoral and weak cell-mediated immune responses typical of clinical stages of bovine Johne's disease. An additional finding was the consistent expression of iNOS associated with the basal aspect of the crypts. Not all iNOS-positive cells near crypts were CD68 positive, incriminating bovine crypt enterocytes as a source.
Footnotes
Acknowledgements
This study was funded by the United States Department of Agriculture Veterinary Services Notice for the Cooperative Study of Johne's Disease 02-09. We thank Drs. M. Ackermann, A. Fales-Williams, J. Haynes, and S. Jones for their editorial assistance and Dr. Y. Niyo of the National Veterinary Services Laboratory for providing tissue from an M. bovis–infected animal. Jim Fosse and Biomedical Communications were instrumental in preparing images. In addition, we thank the Merck-Merial Summer Research Program.
