Abstract
Listeria monocytogenes (LM) is a Gram-positive facultative intracellular bacterium that causes fatal meningoencephalitis in humans and ruminants. A current paradigm predicts that intracellular bacteria are controlled by nitric oxide (NO) whose synthesis is catalyzed by inducible nitric oxide synthase (iNOS). The ability of macrophages (MΦ) to express iNOS shows extreme interspecies variability. Here the expression of iNOS and synthesis of NO was studied in listeric encephalitis of cattle, sheep, and goats. iNOS was expressed by a subset of MΦ in cerebral microabscesses in all three species. The level of iNOS expression and the density of cells per lesion expressing iNOS was highest in cattle, intermediate in sheep, and lowest in goats. The accumulation of nitrotyrosine (NT), an indicator of local NO synthesis, was observed in lesions of cattle but not in those of small ruminants. The density of iNOS-expressing cells in lesions was inversely correlated with the number of bacteria. No species differences were observed in regard to reactive oxygen intermediate (ROI) production by stimulated granulocytes, using the flow cytometric dihydrorhodamine-123 (DHR) method indicating ROI generation. Thus, the marked species differences in iNOS expression, NT accumulation, and LM content in lesions of ruminants with listeric encephalitis are explained by different amounts of ROI produced. It suggests that variations in the ability of MΦ to synthesize NO are of pathophysiological significance in listeriosis.
Nitric oxide (NO) produced by inducible nitric oxide synthase (iNOS) of macrophages (Mφ) has an important role in antimicrobial host defense and immunosurveillance. NO contributes to resistance to or recovery from infections by a large array of microbial pathogens, including viruses, bacteria, fungi, and protozoan and metazoan parasites (reviewed by MacMicking et al.23). On the other hand, NO produced by inducible nitric oxide synthase (iNOS)-expressing cells contributes to a variety of disease symptoms, extending from immunosuppression to apoptosis and tissue damage.10,11,29,33,34,40 The knowledge on the conditions of iNOS induction, signal transduction, gene activation, enzyme production, and activity firmly rests on studies performed in the mouse.22,27 The question whether human macrophages (Mφ) produce similar amounts of NO as do murine counterparts has been controversial,8,9,20,21,24,25,28,35,43,45 and it is unclear whether antimicrobial activity of interferon-γ-activated human Mφ rests on NO synthesis or on other as yet undetected pathways. As regards other species, our group and others have established that bovine Mφ are also capable of producing high amounts of NO by expression of iNOS.2,3,16,42 In contrast, Mφ of small ruminants such as goat and sheep were found to synthesize far lower amounts of NO, and iNOS expression was much weaker than in bovine cells stimulated in a similar manner.1
Thus, the species difference with regard to the ability of Mφ to synthesize NO exists not only between distantly related species such as human and mouse but also within closely related ruminants. This prompted us to look for iNOS expression at sites of infection and inflammation. Listeric encephalitis was chosen as a model since (1) this is a natural infection in goats, sheep, and cattle and (2) a combination of heat-killed Listeria monocytogenes (LM) and interferon-γ proved to be one of the strongest stimuli for the induction of iNOS in ruminant macrophages.19 Our report shows that, in animals succumbing to listeric encephalitis, iNOS was expressed in a subset of Mφ in microabscesses, regardless of the species.
While this proved that in vivo Mφ of low responder species are also capable of expressing iNOS, the study did not reveal whether there are quantitative differences with regard to NO synthesis and how this relates to the efficiency of bacterial growth control. Furthermore, nitric oxide may react with superoxide and generate the highly toxic peroxynitrite,5 which in turn reacts with proteins, thereby forming nitrotyrosine (NT). In this study, brain sections of cattle, sheep, and goats with listeric encephalitis were assessed (1) with regard to the intensity of iNOS expression, (2) the extent of NT accumulation, and (3) the number of bacteria per infection site. Peripheral blood-derived granulocytes of the three species were compared with regard to their capacity to generate reactive oxygen intermediates (ROI) in response to heat-killed LM or phorbol 12-myristate 13-acetate (PMA). The study shows that the ability of Mφ to express iNOS and to produce NO in vitro is closely correlated with the degree of iNOS expression and NT accumulation in microabscesses and is inversely correlated with the number of LM organisms per infection site. In contrast, the ability of granulocytes to produce highly reactive ROI in vitro does not show species variation. This is consistent with an important contribution of NO synthesis to antilisterial defense in the ruminant brain and adds credence to the view that interspecies variation in the ability of Mφ to mount an iNOS response is of general pathophysiologic significance.
Materials and Methods
Source and preparation of tissue samples
Animals referred to the Institute of Animal Neurology were necropsied. The formalin-fixed brains were sliced, and coronal sections were embedded in paraffin, cut at 4 μm, and mounted on positively charged glass slides (SuperfrostPlus, Menzel, Braunschweig, Germany) as described.19 Specimens from ruminants with other diseases with extensive malacias were treated accordingly. These included cases of cerebrocortical necrosis (CCN), polioencephalomalacia (toxic; vitamin B deficiency), focal symmetrical encephalomalacia (clostridial enterotoxemia), and suspected caprine arthritis encephalitis syndrome. Brains from infant (day 11 to day 18) rats intracisternally infected with LM were fixed by perfusion with paraformaldehyde (4% in PBS), embedded in paraffin, and then processed for immunohistochemistry. The infection model is described elsewhere.30 These brain sections were found to express iNOS and were used as a quality control for evaluating NT-specific antibodies.
Immunohistochemistry
General
Immediately before staining, slides were deparaffinized and processed for immunohistochemistry (IHC). In some cases (NT, LM), antigens were retrieved by treatment with trypsin (2 mg/ml trypsin 250, DIFCO, in 50 mM Tris buffer, pH 7.5) for 15 minutes. Endogenous peroxidase was inactivated by treatment with H2O2 (1% in PBS, 15 minutes at room temperature). Fc receptors were blocked by treatment with human IgG (Globuman Berna, Swiss Serum and Vaccine Institute, Berne, Switzerland). After these preparatory treatments, slides were incubated with the specific antibodies (1 hour at 37 C), followed by treatment with an appropriate second antibody and a detection system.
Primary antibodies
iNOS-specific antibodies were polyclonal rabbit IgG raised against murine iNOS (cat. no. 06-295 for ruminants, cat. no. 06-573 for the rat, both purchased from Upstate Biotechnology Inc., Lake Placid, NY, and used at 1:200). Antibody 06-295 was shown to crossreact extensively with iNOS from other species, including cattle and small ruminants15,19 (and unpublished observations). Monoclonal antibodies specific for nitrotyrosine were purchased from Calbiochem-Novabiochem (San Diego, CA) or from Hycult Biotechnology (purchased through Bioreba, Basel, Switzerland) or were generously provided by Dr. M. Shigenaga (UC Berkeley; antibody 39B6). The properties of the latter antibody were reported in abstract form38 and will be published in detail elsewhere (Girault et al., unpublished manuscript). Polyclonal NT-specific antibodies were from Upstate Biotechnology Inc. (cat. no. 06-284). Polyclonal rabbit-anti-LM (specific for serotypes 1 and 4) was purchased from DIFCO (Detroit, MI; cat. no. 2302-50).
Secondary antibodies
In the case of anti-iNOS and polyclonal anti-NT, biotinylated anti-rabbit IgG (cat. no. 11-065-045; Jackson Immunoresearch Laboratories, West Grove, PA) was used at 1:200. In the case of monoclonal anti-NT, biotinylated goat anti-mouse IgG (human and rat serum absorbed, cat. no. 16-18-15; Kirkegaard and Perry, Gaitherburgh, MD) was applied at 1:200. In the case of anti-LM, slides were treated with monoclonal anti-rabbit IgG conjugated to alkaline phosphatase (cat. no. A-2556; Sigma, St. Louis, MO) at 1:200.
Detection system
Specimens treated with biotinylated antibodies were stained with the ABC system (cat. no. 85-99-43; Vector, Burlingame, CA) combined with diamino benzidine as a substrate for peroxidase. Alkaline phosphatase reactions were performed with the fast red substrate: 0.5 mg/ml Naphthol SX-MX (cat. no. N-5000; Sigma) was added to 1 mg/ml fast red (Chroma Gesellschaft, Schmid GmbH, Köngen, Germany) in 100 mM Tris-HCI, pH 8.
Controls
In each staining batch for iNOS and NT, the following controls were included. Negative controls consisted of slides on which the first antibody was omitted. When staining samples from small ruminants, a section from cattle known to show a strong iNOS response was also stained as a positive control.
Validation of NT staining
A variety of polyclonal and monoclonal anti-NT antibodies were tested for their specificity in brain sections from rats intracisternally infected with LM. Sections were made from brains collected between day 3 (d3) and d5 postinfection, at a time when a number of foci with iNOS-expressing cells were observed (Fig. 1). Positive NT staining was obtained around the same time (d3–d5) and in the same areas as iNOS, and the staining intensity was strongest for the polyclonal antibody and somewhat weaker for the monoclonals tested (Fig. 2–4 and data not shown). The cells expressing iNOS and NT were colocalized. At the light microscopic level, staining for NT was restricted to small blebs, in contrast to that for iNOS, which was distributed over the whole cytoplasm. In order to prove the specificity of the antibodies, the following control tests were carried out: 1) Staining was abrogated in the presence of free NT (1 mg/ml) (Fig. 5). When staining was performed in the presence of aminotyrosine, no significant reduction in intensity was observed (data not shown). 2) Prior to staining, tissue sections were exposed to dithionite (100 mM in 100 mM borate buffer, pH 9.0), thereby reducing nitrotyrosine to aminotyrosine, which no longer binds NT-specific antibodies. When using dithionite exposure, the staining intensity was largely abrogated (Fig. 6). Judged by these criteria, all antibodies tested were specific for NT and exhibited the same pattern of staining signal. Since the polyclonal antibody provided the strongest staining, it was used for the assessment of ruminant listeric encephalitis.
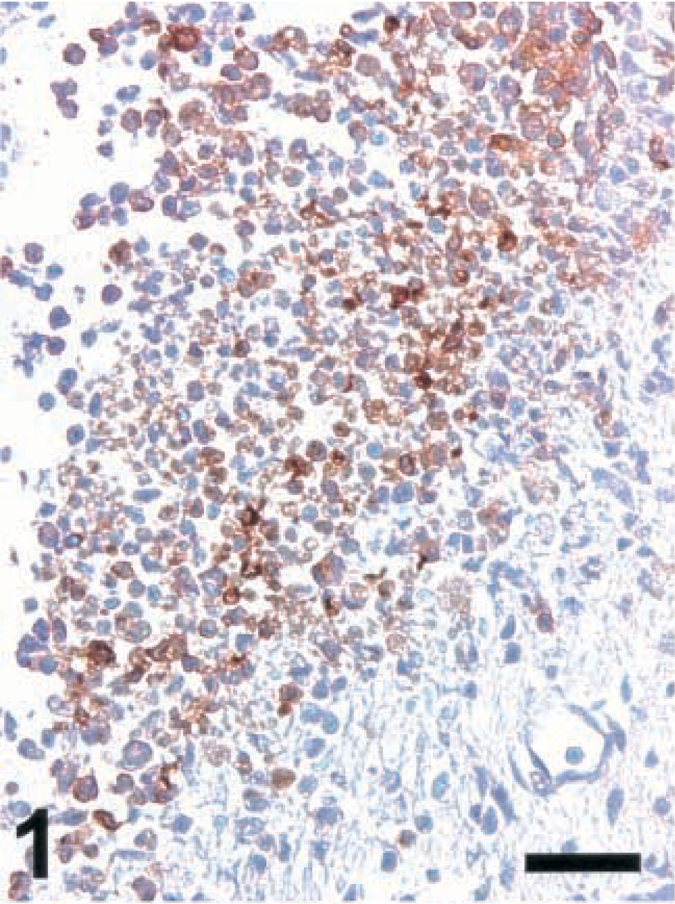
Brain; rat intracisternally infected with LM, rat no. 2. iNOS expression in an inflammatory lesion. IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
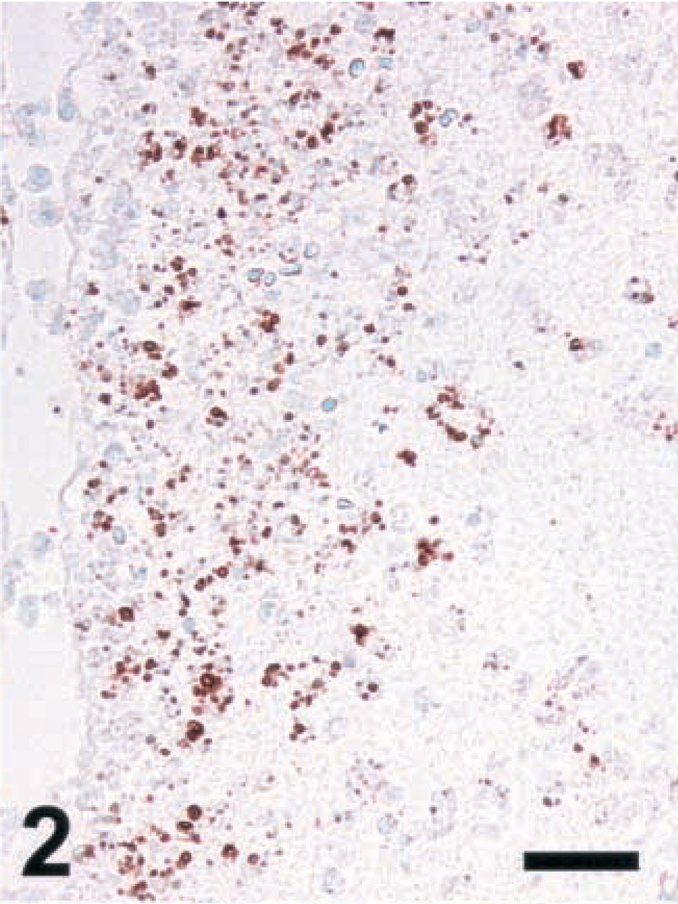
Brain; rat intracisternally infected with LM, rat No. 1. Staining for NT in an inflammatory lesion using a monoclonal antibody from Calbiochem. IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
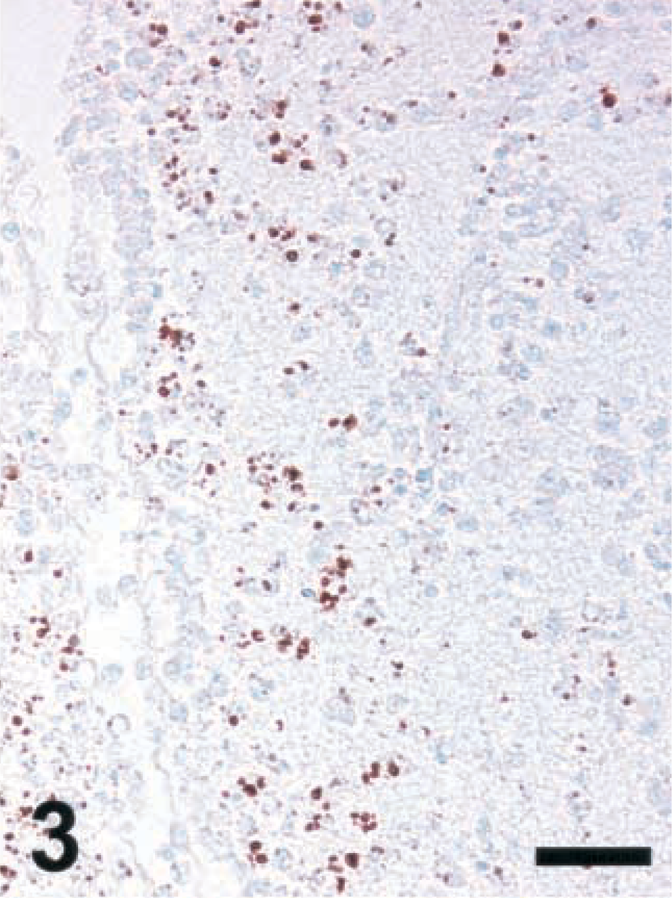
Brain; rat intracisternally infected with LM, rat No. 1. Staining for NT in an inflammatory lesion using a monoclonal antibody from Hycult Biotechnology. IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
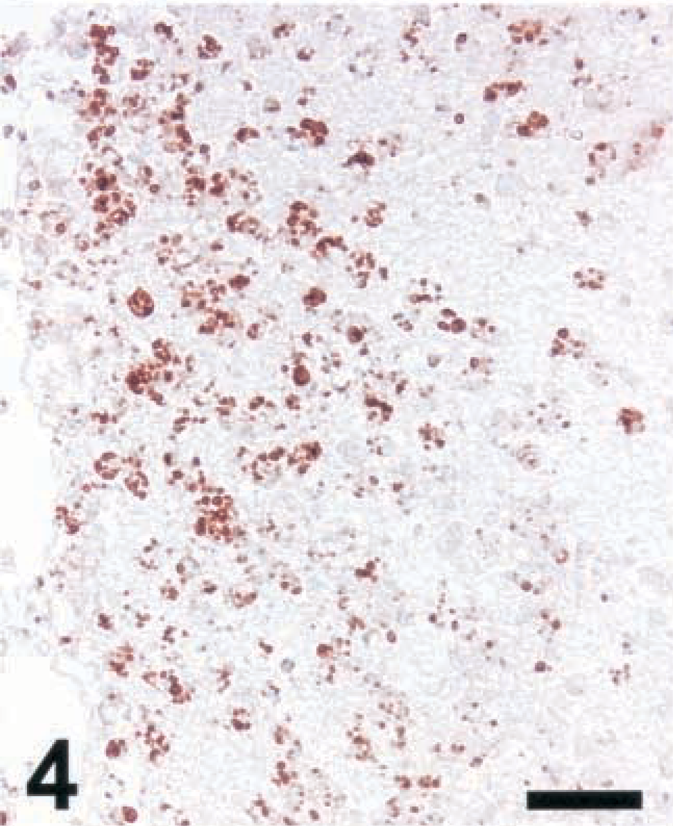
Brain; rat intracisternally infected with LM, rat No. 1. Staining for NT in an inflammatory lesion using a polyclonal antibody from Upstate Biotechnology Inc. IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
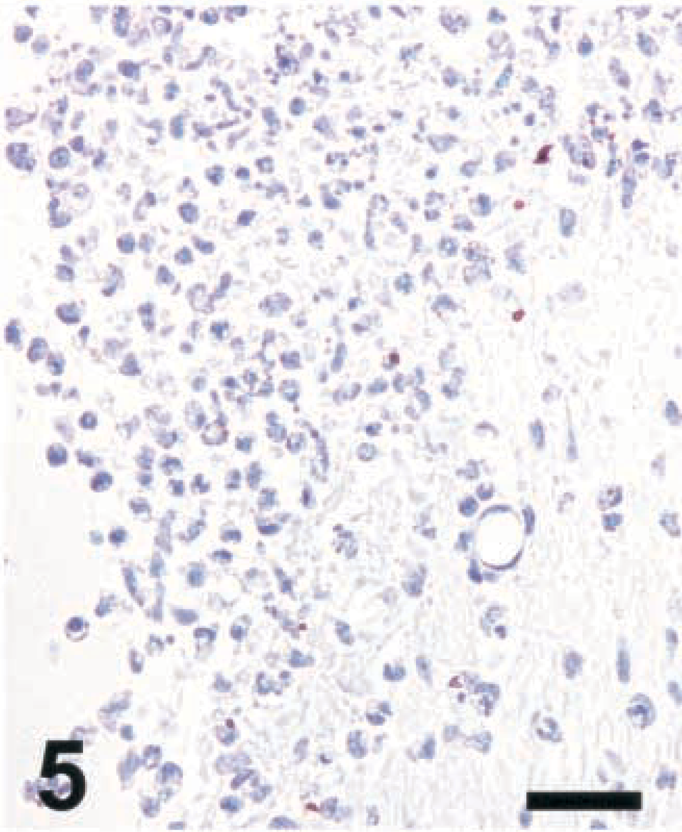
Brain; rat intracisternally infected with LM, rat No. 2. Staining for NT in an inflammatory lesion using a polyclonal antibody from Upstate Biotechnology Inc. Absence of NT staining when antibody reaction is performed in the presence of free NT (1 mg/ml). IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
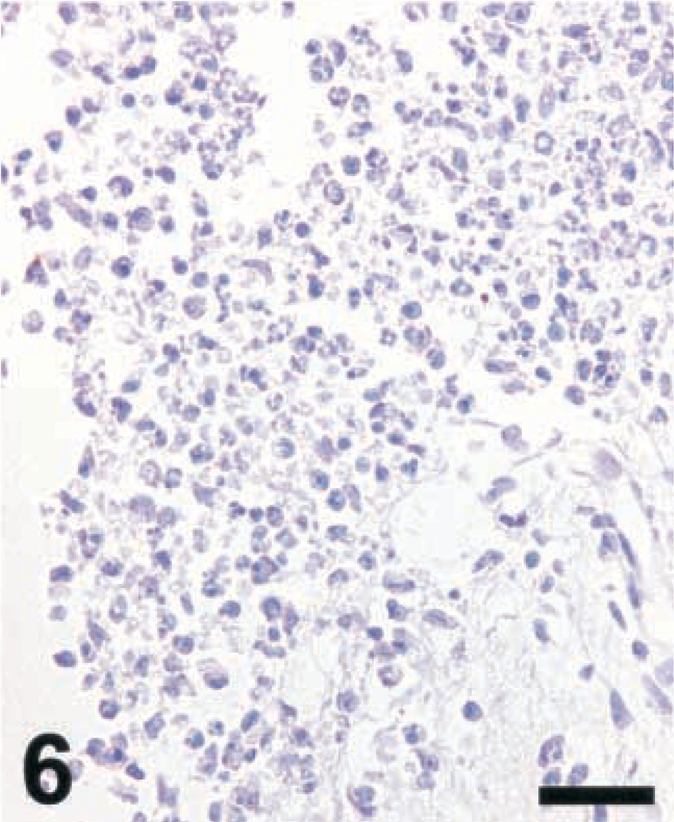
Brain; rat intracisternally infected with LM, rat No. 2. Staining for NT in an inflammatory lesion using a polyclonal antibody from Upstate Biotechnology Inc. Absence of NT staining when the section had been pretreated with dithionite (100 mM). IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
Evaluation of immunohistochemistry results
In order to objectively score the staining intensity of iNOS, pictures were taken from iNOS-positive microabscesses and were scored between 1 (absent) and 6 (strong) in a blinded fashion by another collaborator. Scores were recorded for size of microabscesses, density of iNOS-positive cells, and intensity of staining for iNOS. All microabscesses on one slide where evaluated for each parameter except when their number exceeded 10. In this case, a maximum of 10 microabscesses were randomly selected. One to four slides per animal were scored. Average scores were converted into new scores, which were taken as representative for a given animal. From these scores, medians and ranges were determined for each parameter and species.
The number of bacteria was determined in all microabscesses inspected for scoring. Microabscesses in which LM organisms were visible were photographed using a digital camera, and clearly visible organisms were counted on the screen. Microabscesses were usually smaller than the field of vision, but in some goat cases exceeded the latter in area. Particles having a larger size than a normal single bacterium were counted as two or three, depending on the area covered. The bacterial numbers were averaged for each slide, and the numbers determined for each slide were averaged for each animal. These averages were taken as representative for a given animal. Means and standard errors of the means were calculated for each species. Animals with a record for previous treatment with antibiotics were excluded.
Bacteria
LM isolated from cattle, sheep, and goats were obtained by the courtesy of Dr. E. Bannermann, Cantonal University Hospital, Lausanne, Switzerland. Isolates were picked at random and included biotypes 1/2a, 1/2b, 4b, and 4c. An individual colony from a tryptic soy agar plate was grown in tryptic soy broth overnight prior to washing and adding to RAW264.7 cell cultures.
Cell isolation
The mouse macrophage cell line RAW264.7, originally obtained from G. Werner-Felmayer, University of Innsbruck, was cultured in Dulbeccos's minimum essential medium (MEM) with penicillin (100 IU/ml), streptomcycin (100 µg/ml), amphotericin B (2.5 µg/ml), 2-mercaptoethanol (50 μm),
Peripheral blood leukocytes were isolated from heparinized venous blood collected aseptically from cattle, sheep, and goats. It was spun (10 minutes at 365 × g), washed in PBS, followed by two steps of erythrocyte lysis at room temperature using as a lysis buffer of NH4Cl 0.155 M, KHCO3 10 μm, EDTA 100 μm, pH 7.4. White blood cells were spun (10 minutes at 365 × g) and washed twice with PBS, followed by resuspension in PBS for use in flow cytometry.
Assessment of reactive oxygen intermediates
The production of ROI was assessed by a fluorescent method, using dihydrorhodamine-123 (DHR).13,31 Peripheral blood leukocytes were loaded with DHR (0.75 μm for 5 minutes at 37 C). Cells stimulated with either heat-killed LM or PMA (Sigma; 10−7 molar) were incubated at 37 C and were subjected to flow cytometry at 15 minutes after stimulation (PMA, LM) and every 15 minutes thereafter (LM) up to 60 minutes.
Statistical analysis
Ordinal data (scores) were expressed as medians and ranges. Bacterial numbers per lesion were expressed as averages and standard errors of the mean. Statistical significance (P < 0.05) of interspecies differences was assessed by Kruskall–Wallis test for multiple group comparisons and by the Mann–Whitney test for comparisons of two groups.
Results
Detection of iNOS in tissue sections of ruminants with listeric encephalitis
Brain sections from animals previously analyzed19 and more recently collected samples were analyzed according to Table 1. Ruminant listeriosis exhibits two types of characteristic lesions: microabscesses and perivascular cuffs. iNOS was never seen in perivascular cuffs. However, in 9 out of 9 cattle, 8 out of 10 goats, and 8 out of 8 sheep, iNOS was observed in microabscesses (Table 1), thus confirming our earlier data.19 In some sections, iNOS could not be detected, a phenomenon which was more frequent in goats than in sheep and cattle. The relative staining intensity was variable but was usually higher in cattle sections than in those of small ruminants (Figs. 7–9, Table 2). The density of iNOS-positive cells was significantly higher in cattle sections than in those of small ruminants (Figs. 7xs–9, Table 2).
Variability in iNOS expression in listeric encephalitis of ruminants.
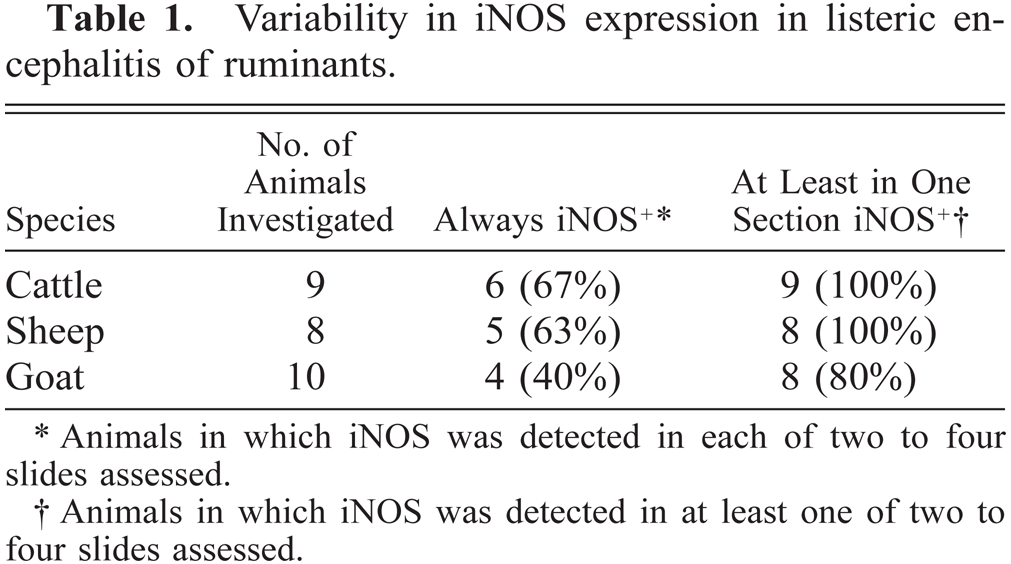
Animals in which iNOS was detected in each of two to four slides assessed.
Animals in which iNOS was detected in at least one of two to four slides assessed.
Size of microabscesses and level of iNOS expression in ruminant listeric encephalitis.
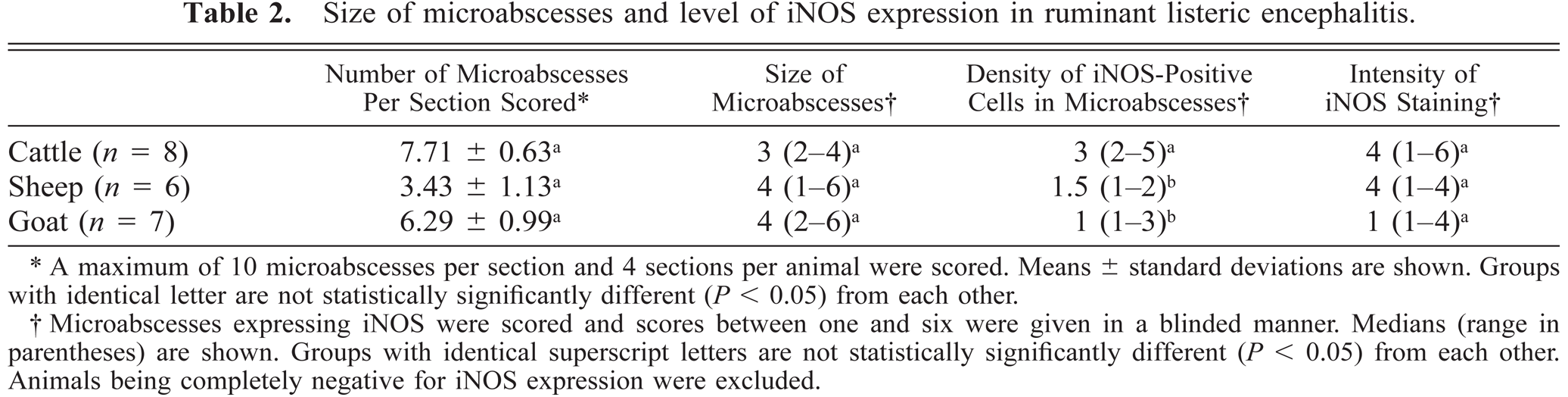
A maximum of 10 microabscesses per section and 4 sections per animal were scored. Means ± standard deviations are shown. Groups with identical letter are not statistically significantly different (P < 0.05) from each other.
Microabscesses expressing iNOS were scored and scores between one and six were given in a blinded manner. Medians (range in parentheses) are shown. Groups with identical superscript letters are not statistically significantly different (P < 0.05) from each other. Animals being completely negative for iNOS expression were excluded.
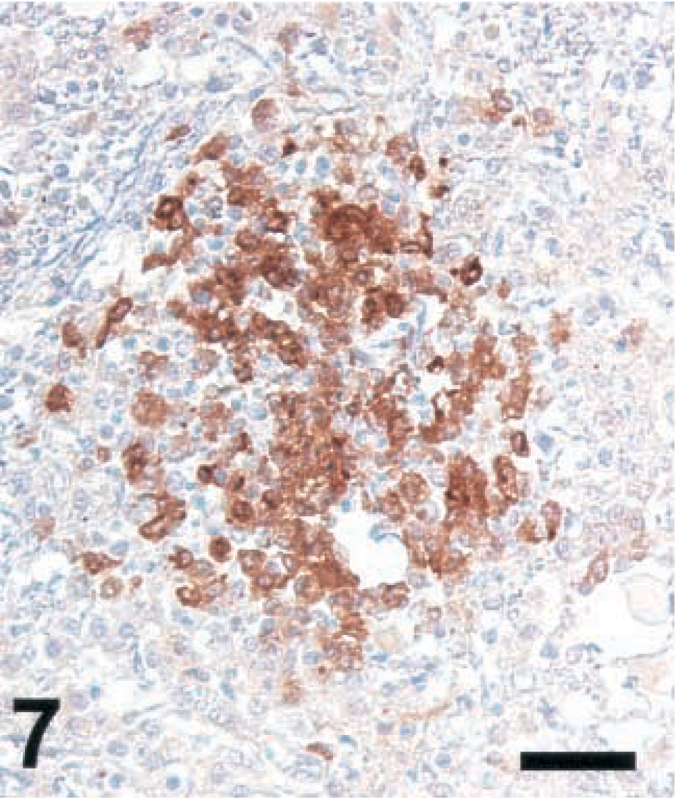
Brain; cow with listeric encephalitis, case No. 3. iNOS expression in a microabscess. IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
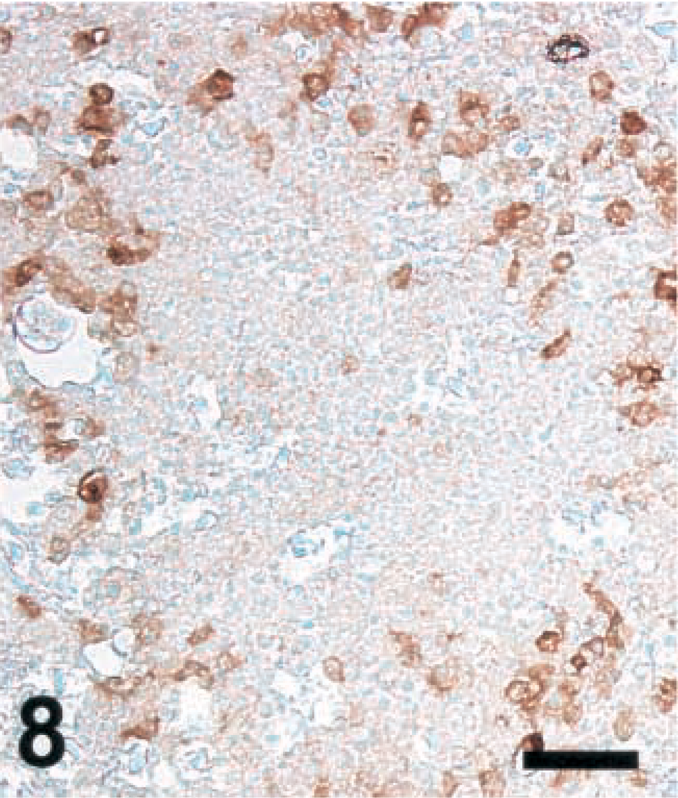
Brain; sheep with listeric encephalitis, case No. 19. iNOS expression in a microabscess. IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
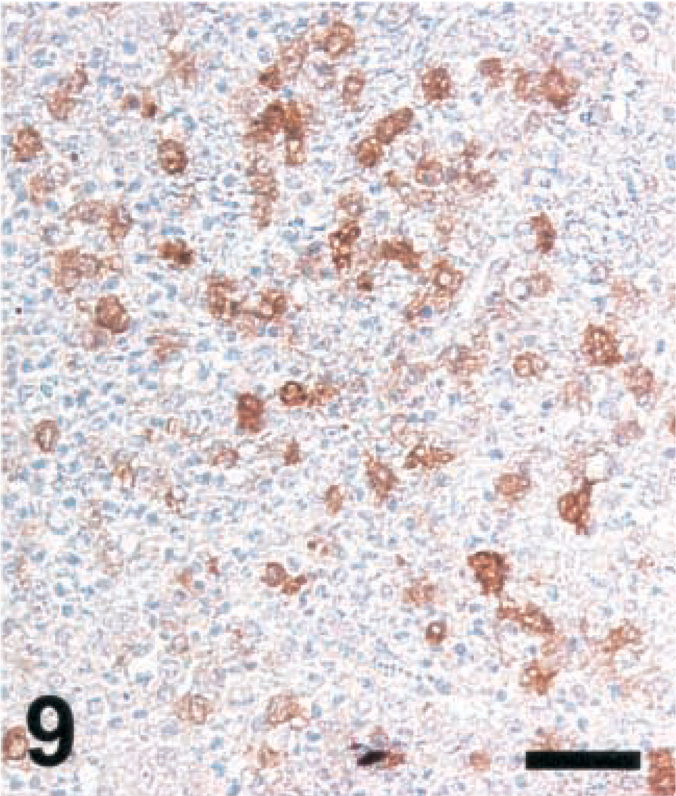
Brain; goat with listeric encephalitis, case No. 15. iNOS expression in a microabscess. IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
Detection of NT in tissue sections of ruminants with listeric encephalitis
Brain sections from ruminants with listeric encephalitis and adjacent to those stained for iNOS were stained for NT. This is an indicator of local generation of reactive nitrogen intermediates (RNI) such as peroxynitrite,5 NT was not detected in any of the sections remaining negative for iNOS (data not shown). In specimens from ruminants with other diseases with extensive malacias, including cases of cerebrocortical necrosis (CCN), polioencephalomalcia (toxic; vitamin B deficiency), focal symmetrical encephalomalacia (clostridial enterotoxemia), and suspected caprine arthritis encephalitis syndrome, no NT could be detected (data not shown). Six out of six cattle specimens showing a high level of iNOS expression were strongly positive for NT. (Two cattle brains were rated positive at an earlier occasion, but in the blocks presently evaluated, no iNOS expression could be detected; these specimens were negative for NT.) The morphology of the structures stained with NT-specific antibodies was similar to that seen in infected rats. In contrast, NT was detected in only one sheep and one goat, which showed positive staining for iNOS, and the NT signal was considerably weaker than in cattle (Figs. 10–12 and data not shown). In some cattle specimens that had a strong reaction for both iNOS and NT, double staining was performed. This showed that cells expressing both markers simultaneously were rare, although the areas in which NT was observed were colocalized with those staining for iNOS (data not shown). Thus, NT accumulation was observed only in microabscesses expressing high levels of iNOS.
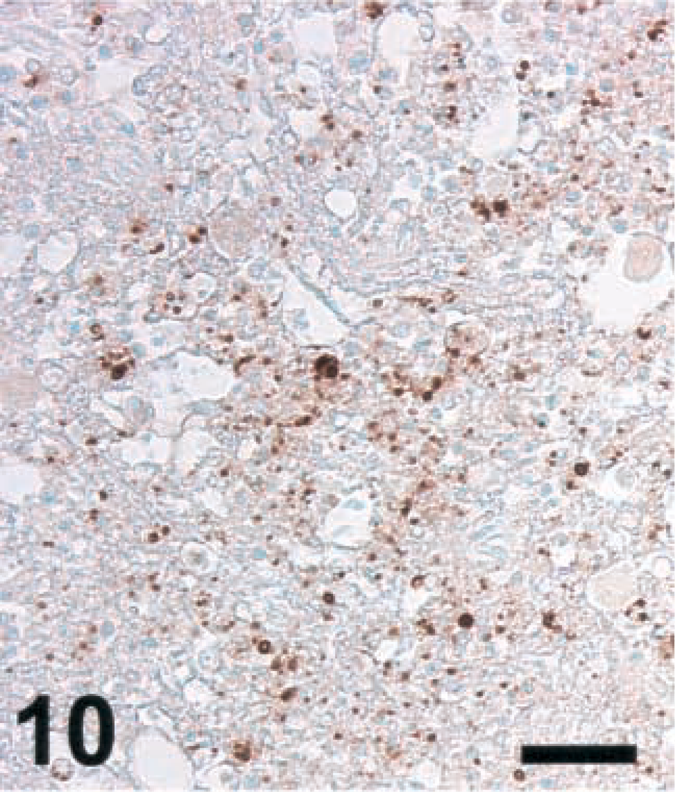
Brain; cow with listeric encephalitis, case No. 3. NT in a microabscess. IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
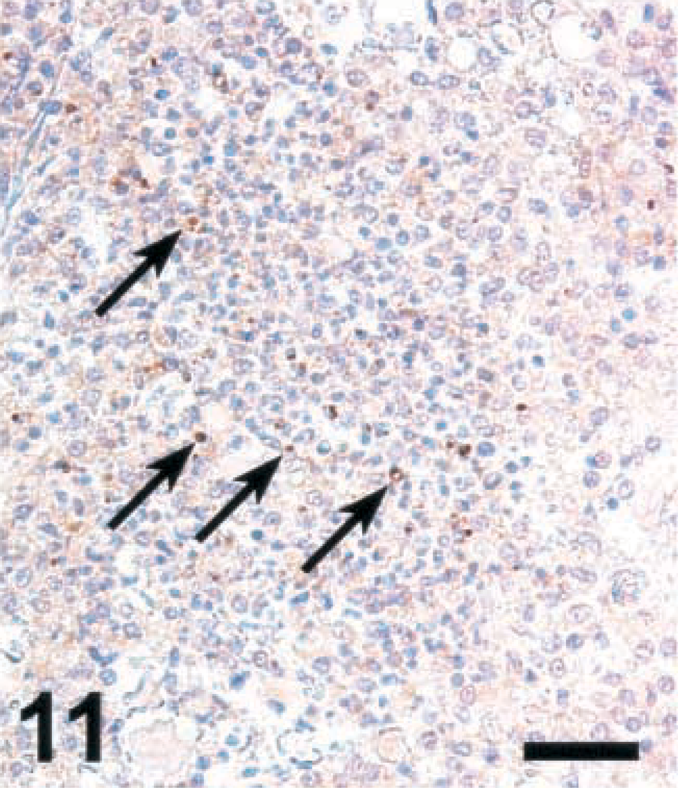
Brain; sheep with listeric encephalitis, case No. 18. Weak staining for NT in a microabscess (arrows). IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
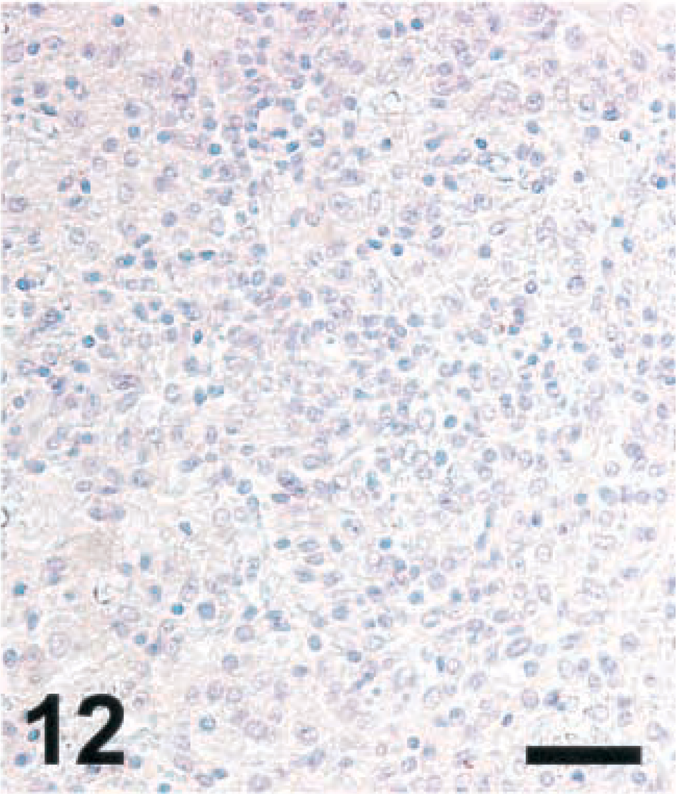
Brain; goat with listeric encephalitis, case No. 15. Absence of NT from the microabscess. IHC; peroxidase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
Bacterial counts are inversely correlated with iNOS expression
To determine whether the bacterial load in microabscesses is inversely correlated with the amount of NO and/or peroxynitrite produced, brain sections were stained for LM and photographed. LM organisms were counted as described in Materials and Methods. As the iNOS-expressing areas were often of a more diffuse type in small ruminants and a more compact type in cattle, the most compact type of lesions was selected in small ruminants. In lesions of cattle, there were significantly less bacteria than in those of small ruminants (Figs. 13–16). The counting method led to an underestimation of bacteria in goat lesions (see Materials and Methods). Thus, microabscesses of cattle contained two orders of magnitude less bacteria than those of goats, the sheep taking an intermediate position. Staining mouse RAW264.7 cells having ingested LM organisms from cattle, sheep, and goat isolates showed that bacteria isolated from all three species and picked at random were recognized by the monoclonal antibody used (data not shown). This rules out the possibility that bacteria found in cattle brains were not recognized by the antibody used, resulting in erroneously low numbers of bacteria per cattle lesion.
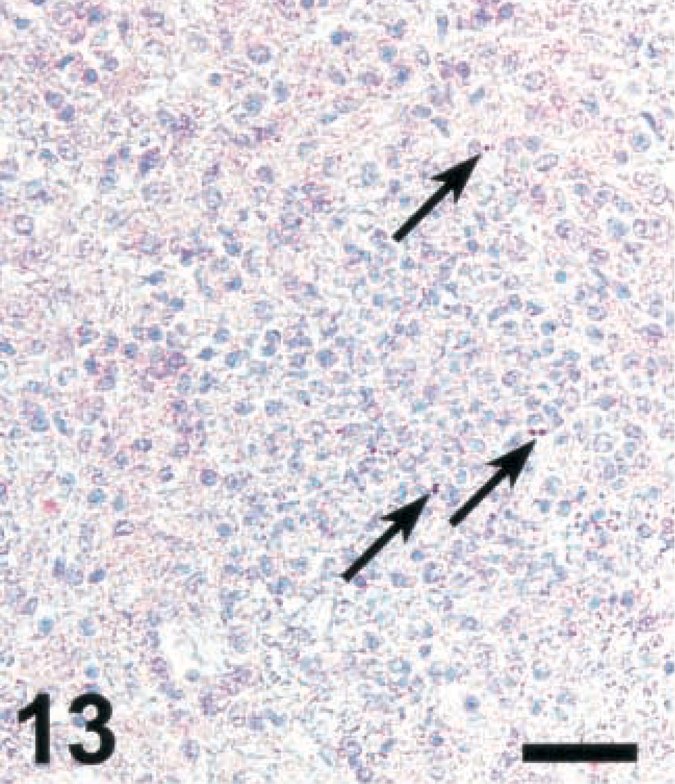
Brain; cow with listeric encephalitis, case No. 3. LM in a microabscess (arrows). IHC; alkaline phosphatase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
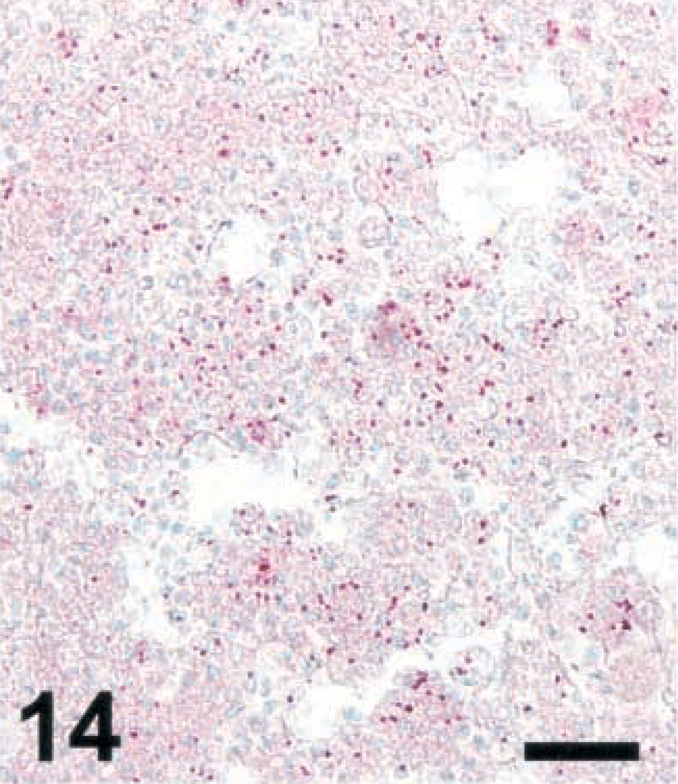
Brain; sheep with listeric encephalitis, case No. 22. LM in a microabscess. IHC; alkaline phosphatase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
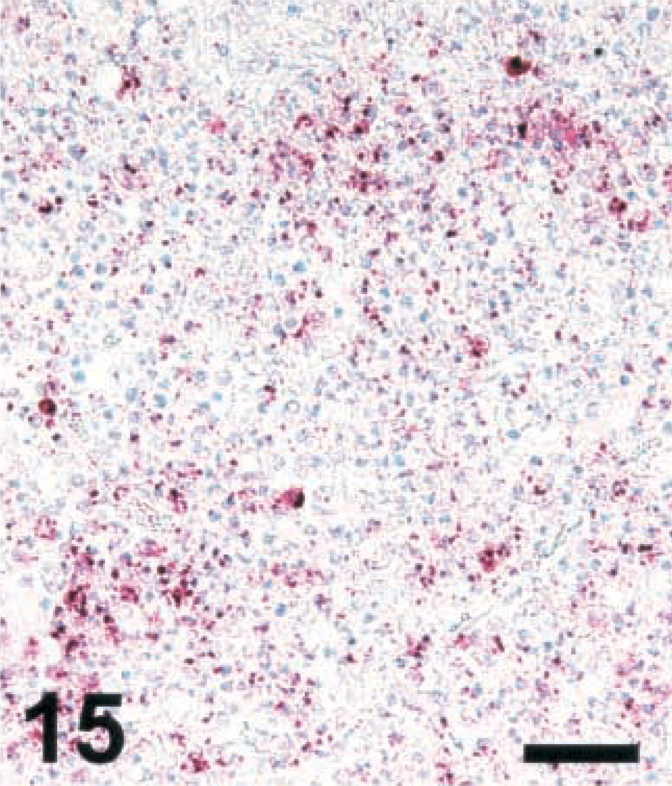
Brain; goat with listeric encephalitis, case No. 15. LM in a microabscess. IHC; alkaline phosphatase method, Mayer's hematoxylin counterstain. Bar = 67 μm.
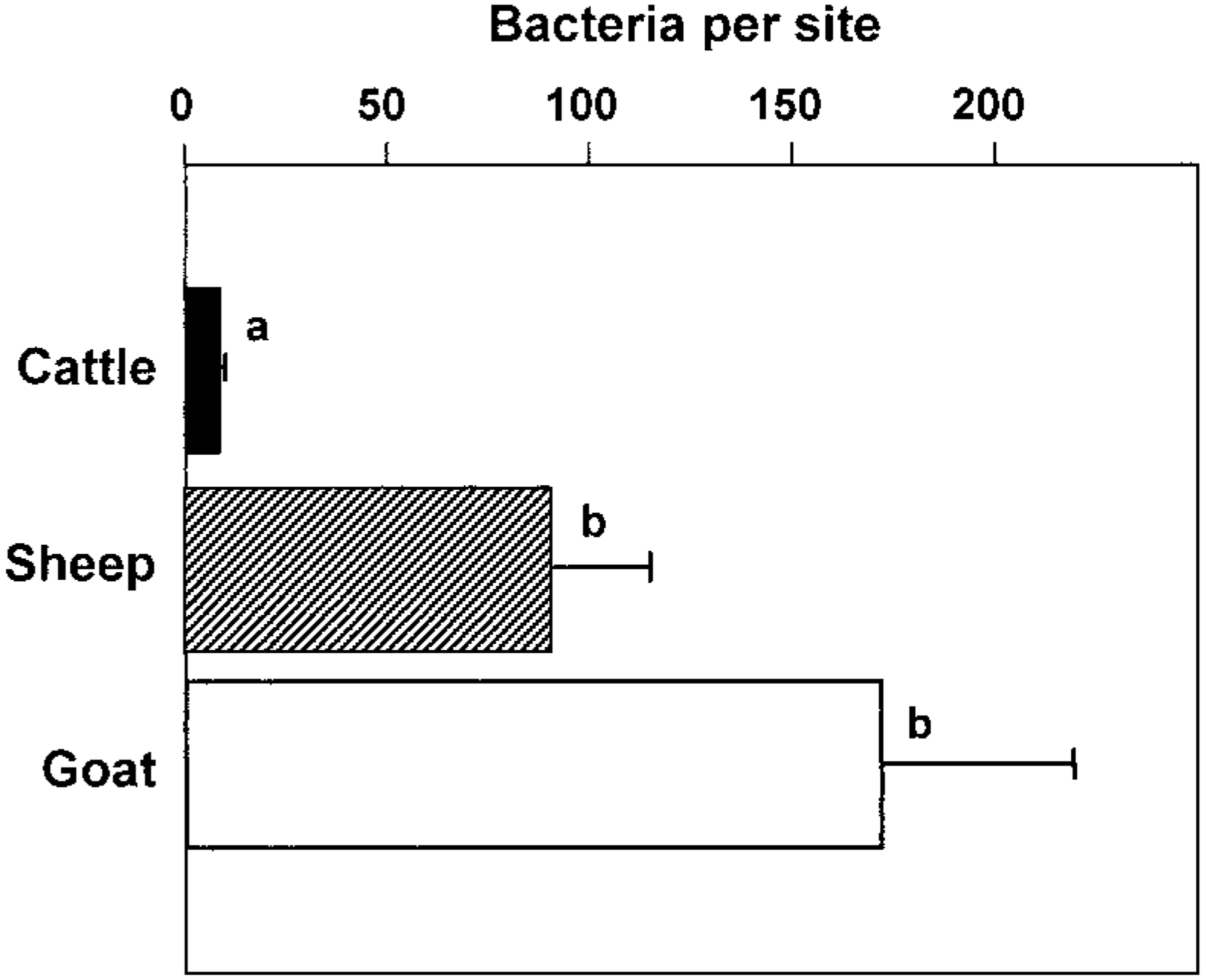
Numbers of LM within microabscesses in ruminant listeric encephalits, determined as described in Materials and Methods. Means and standard errors of the mean are shown. Groups with identical letters are not statistically significantly different (P < 0.05) from each other.
In Fig. 17, the correlation between the density of iNOS-expressing cells within lesions and the number of bacteria is shown for all animals. Small ruminants and cattle represented clearly separate populations. Overall, there was poor correlation between the number of bacteria on the one hand and the lesion size, the cellular iNOS staining intensity, or the number of sites detected on the other hand (data not shown).
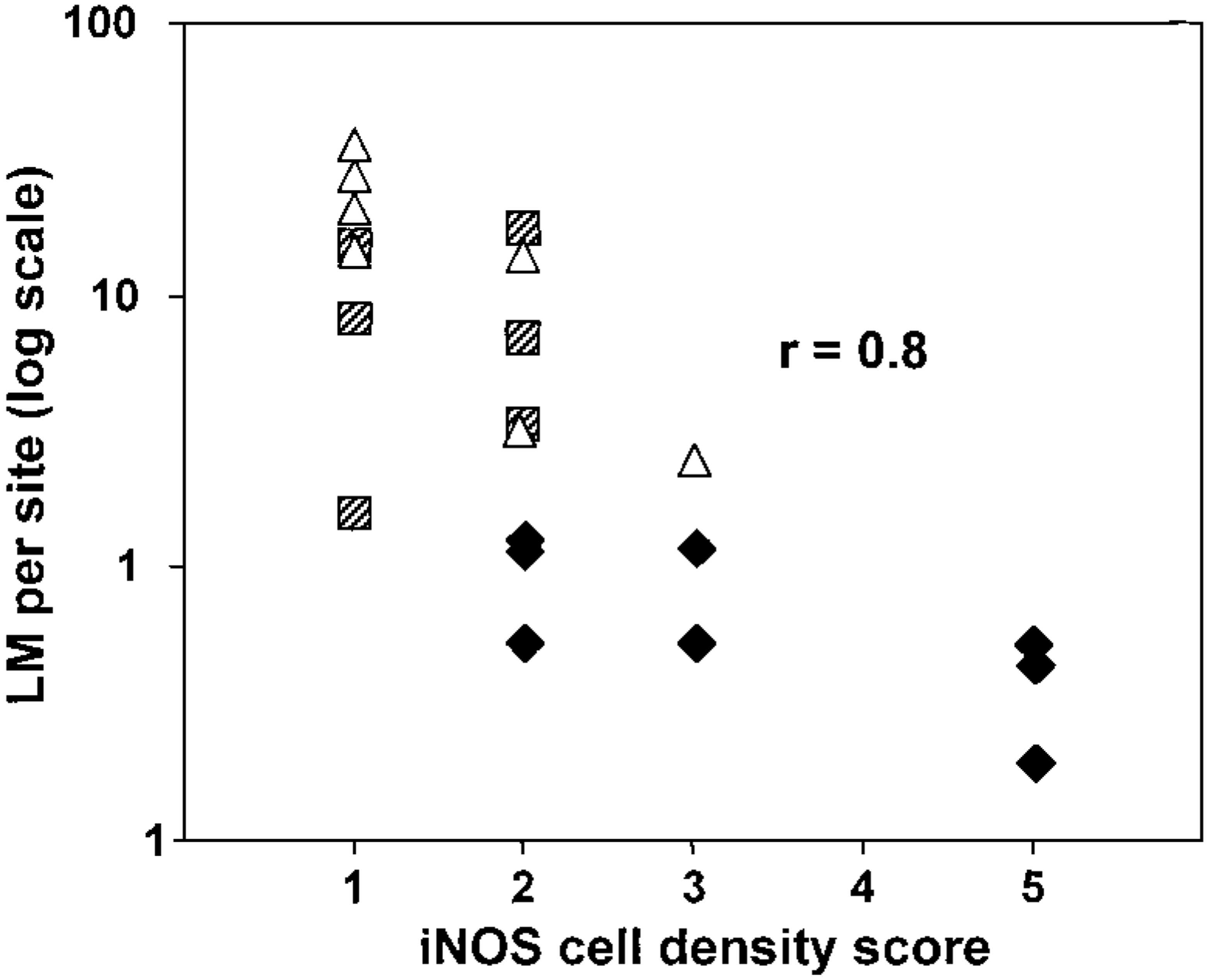
Correlation between the number of LM per animal per lesion and the density of iNOS-positive cells per lesion. Data summarized in Table 2 are plotted for each individual animal. Cattle, closed diamonds; sheep, hatched squares; goats, open triangles. The r value (correlation coefficient) refers to all animals combined.
ROI production
Evidence for the generation of reactive oxygen intermediates (ROI) was sought in various ways. Using antibodies recognizing Michaels adducts of hydroxynonenal (Calbiochem41), a positive signal was obtained neither in ruminant brains nor in the brains of rats experimentally infected with LM, suggesting that such antibodies are inappropriate indicators of ROI generation a posteriori in formalin-fixed paraffin-embedded tissue. To test whether a species difference may be noted in terms of ROI production, LM-stimulated or PMA-stimulated granulocytes of all three species were assesed for ROI production. In a flow cytometric assay, the compound DHR is internalized in a nonfluorescent form and oxidized to the fluorescent rhodamine upon reaction with ROI. Cells loaded with DHR were tested over time for an increase in fluorescence upon exposure to DHR. Granulocytes showed a strong signal upon exposure to PMA and only a weak response to heat-killed LM (Table 3). The response was completely resistant to NMMA (1 mM) and to azide (10 mM), ruling out an involvement of NO and HOCI in the DHR signal.17,39 No statistically significant difference was observed for the DHR signals of small ruminant and cattle granulocytes, suggesting that the interspecies variability with regard to ROI generation was low.
ROI in ruminant granulocytes stimulated with PMA or heat-killed LM, as determined by DHR flow cytometry.

Values represent mean relative fluorescence units (n = 3) ± standard deviation as determined in the presence of NMMA.
Values in parentheses denote mean stimulation indices.
Discussion
Host defense against intracellular pathogens rests largely on the ability of Mφ to synthesize NO. This has been exhaustively shown for a variety of organisms, extending from RNA and DNA viruses to Gram-positive, Gram-negative, and acid-fast bacteria to intracellular parasites and fungi.23 Large amounts of NO are generated by iNOS, which is induced in Mφ and other cells under the influence of cytokines and pathogen-derived constituents. This notion is true for laboratory rodents, but it is unclear whether this pathway is crucial for the elimination of intracellular pathogens in other species, including humans, in which the ability of Mφ to express iNOS in vitro is orders of magnitude lower than in rodents.8,9,20,21,24,25,28,35,43,45 Ruminants provide an opportunity to compare species whose Mφ show either a high (cattle) or a low (sheep, goats) iNOS reactivity in similar types of infections and/or inflammations. They all may develop an encephalitis upon infection with Listeria monocytogenes, a disease that is often fatal. In this study, the extent of iNOS expression in brain lesions of cattle, sheep, and goats with listeric encephalitis was assessed. To relate differences in iNOS expression to iNOS activity, NT accumulation, a hallmark of local NO production, was determined. To address whether high or low NO synthesis has an influence on bacterial growth control, lesions were assessed for the density of bacteria. The present study shows 1) that the rank order of iNOS expression in brain microabscesses is cattle > sheep > goat; 2) that this decreasing order is correlated with the level of NT accumulation in the lesions, which is confined to the areas of strong iNOS expression and is not found in areas free of iNOS or with a low or undetectable degree of iNOS expression; 3) that NT and iNOS expression are inversely correlated with the number of bacteria in microabscesses; and 4) that, in contrast with these species differences, there is little difference in the ability of peripheral blood granulocytes to generate ROI in vitro. These findings are consistent with the view that species differences in RNI production rather than ROI generation contribute to the species variation observed in the number of bacteria per lesion. They lend support to the hypothesis that RNI are of pathophysiologic significance in the control of bacterial growth in listeric encephalitis of ruminants.
In ruminant listeric encephalitis, LM organisms are never detected in perivascular cuffs, and these areas are also devoid of iNOS and NT despite the presence of CD68-positive, MHC-II-expressing cells.19 This is consistent with the view that special local conditions determine whether or not iNOS is expressed. It is tempting to speculate that one necessary element is the presence of bacteria. In vitro, Gram-positive organisms such as LM are iNOS inducers, and this can be strongly augmented by costimulation with interferon-γ,19 which, as a single agent, is only a poor iNOS inducer in ruminant Mφ.2 What promotes maintenance of iNOS expression in microabscesses of cattle, in which hardly any bacteria are detected, remains to be determined.
A pathophysiologically important congener of NO is peroxynitrite, a product of NO and superoxide.5 Formation of peroxynitrite is associated with the generation of NT, which can be stained for by immunocytochemistry.44 Tyrosine can also be nitrated by other reactive nitrogen species, e.g., in a complex reaction involving hypohalite.12 Several lines of evidence support the notion that the antibodies we used detected NT in the tissues investigated. NT was detected only in areas of high levels of iNOS expression, as usually occurring in microabscesses of cattle but not in those of small ruminants. No NT was detected in other areas of the brain from animals with listeric encephalitis nor was it seen in malacias of various other origins. Using the same antibodies, NT was also observed in infected lesions of rats inoculated intracisternally with LM and in the lungs of animals with bronchopneumonia.15 Thus, the presence of iNOS-expressing cells leads to NT accumulation only when their density exceeds a certain threshold. The possibility that insufficient amounts of ROI necessary for NT formation are locally generated is remote.
Basically, an infection with a moderate number of LM organisms is an acute, self-limiting process ending with the elimination of the organism. This is not only true for the classical rodent infection model in which a granulomatous infection of liver and spleen is the rule, it also applies to the brain, as we recently showed using an intracisternal infection of infant rats. Both innate and adaptive cellular immunities contribute to bacterial elimination. The ultimate mediator(s) contributing to the elimination of LM from the tissues is (are) not entirely clear, but a variety of studies point to an important role of ROI.18,37 Another series of reports suggests that reactive nitrogen intermediates are important,4,6,7,32 although this has been disputed by still other studies.14,32 According to one report, NO is essential for the elimination of LM in rodents but not in humans.6 A recent article provided in vitro evidence that peroxynitrite rather than NO itself is essential for the elimination of LM.26 The present study, in which natural rather than experimental listeriosis was assessed, is consistent with the view that iNOS-derived NO contributes to antilisterial activity in the brain.36
The variable number of bacteria in the lesions may be due to several mechanisms. It is consistent, however, with an important role of iNOS and NO in bacterial clearance from the brain. The fact that NT was observed in bovine and rat brain lesions suggests that, besides RNI, ROI is also locally generated. In this retrospective analysis, no evidence for ROI generation in vivo could be obtained. Therefore, peripheral blood-derived granulocytes were assayed for ROI generation using the fluorigenic probe DHR. This compound is a highly sensitive indicator of oxidant species. It was shown to indicate the presence of ONOOH when assayed in the absence of LMMA and of H2O2 in its presence.17,39 Regardless of the species, granulocytes showed a slight increase in the fluorescent signal when exposed to heat-killed LM and a strong signal when stimulated with PMA. This signal resisted a treatment with an NOS inhibitor, arguing for its dependence on ROI generation rather than RNI production. Monocytes of ruminant origin were found to induce low levels of ROI upon stimulation with heat-killed LM (K. A. Remer and T. W. Jungi, unpublished observations). Taken together, this is evidence against a species difference in the ability to mount an ROI response in listeriosis and strengthens our assumption that species differences in Mφ's ability to mount an iNOS response is causally related to the marked differences in bacterial numbers found in microabscesses.
In conclusion, this retrospective study reveals marked species variation in Mφ's ability to express iNOS in listeric encephalitis. It suggests that this difference is a likely explanation for the observed species variability in the effectiveness of antimicrobial mechanisms in the brain, a suggestion further corroborated by results from an experimental rat model of intracisternal inoculation with LM.30
Footnotes
Acknowledgements
This work was supported by the Swiss National Science Foundation (54041.98). The excellent technical assistance by Mrs. Carmen Cardoso is gratefully acknowledged. We also appreciate the generous gift of LM isolates from various ruminant species and the valuable comments and criticism by Drs. E. Peterhans and M. Schweizer, Institute of Veterinary Virology, and the helpful suggestions by Dr. Andreas Zurbriggen, Institute of Animal Neurology, University of Berne.
