Abstract
A clinically normal, 3-year-old female rhesus monkey (Macaca mulatta), which was part of a routine toxicology study, had a mass in the right ovary with metastases to the adjacent mesentery and lungs. The histologic features and immunohistochemistry results were consistent with the diagnosis of choriocarcinoma. Neoplastic cell types included cytotrophoblast (positive for cytokeratin), syncytiotrophoblast (positive for human chorionic gonadotropin), and extravillous trophoblast (positive for human placental lactogen). Because the neoplasm was present in the ovary, the uterus was normal, and the animal was not currently pregnant, this was considered a primary ovarian neoplasm of germ cell origin. The monkey had elevated serum levels of chorionic gonadotropin at the beginning of the study, indicating that, as in women, choriocarcinomas in monkeys can be associated with increased gonadotropin levels and that the tumor was preexisting at the start of the toxicology study.
Primary ovarian choriocarcinomas are malignant neoplasms that arise from germ cells of the ovary and comprise cells of trophoblastic lineage. They are exceedingly rare tumors, estimated to occur only in one of 369,000 human females. 5 Only one previous report of primary ovarian choriocarcinoma in a nonhuman primate exists, in a cynomolgus macaque (Macaca fascicularis). 13
A clinically normal, 3-year-old female rhesus monkey (Macaca mulatta) was sacrificed and a necropsy performed as part of a routine toxicology study. In the right ovary, there was a red and tan multinodular mass, approximately 3.0 cm in length and 1.5 cm in diameter (Fig. 1). Additional masses were present in the adjacent mesentery and lungs. The mesenteric mass was single, red, and multinodular, approximately 2.5 cm in length and 1.5 cm in diameter. There were two lung masses, in the left diaphragmatic and right apical lung lobes. Both were red and measured 2.0 cm and 3.0 mm in diameter, respectively. The uterus was normal, and the monkey was not pregnant. No other abnormalities were found.
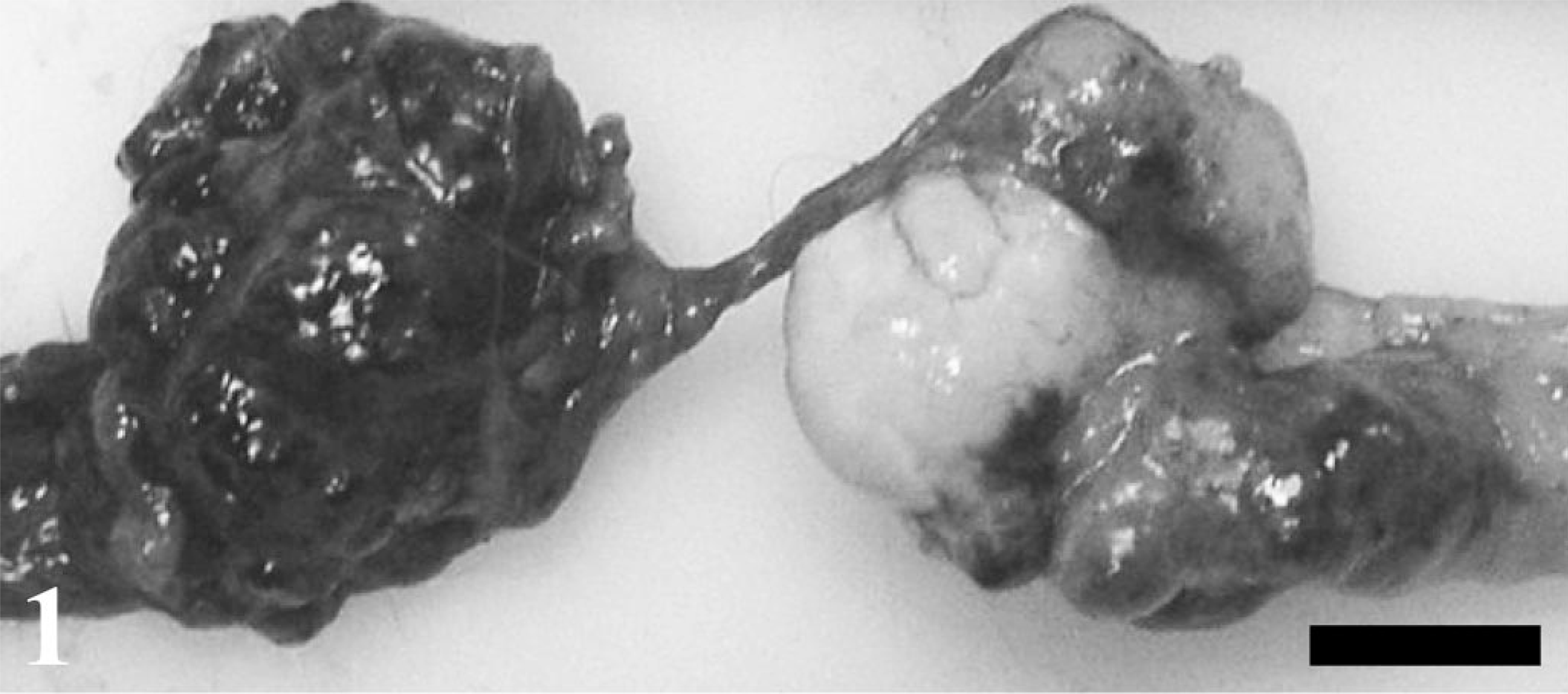
Ovary and mesentery; rhesus monkey. A large mass involves the entire right ovary (right). A metastatic mass is present in the adjacent mesentery (left). Bar = 1 cm.
Tissues were preserved in 10% neutral-buffered formalin and processed to hematoxylin and eosin–stained slides. Immunohistochemistry was performed on sections of the ovarian tumor, using a two-step peroxidase 3,3′-diaminobenzidine staining technique with the DAKO EnVision™ + System (DAKO Corp., Carpinteria, CA). Staining was performed for human chorionic gonadotropin (hCG) and human placental lactogen (hPL), using the respective polyclonal rabbit antibodies (DAKO Corp.). Staining for cytokeratin (CK) was performed using a cocktail composed of monoclonal antibody CAM 5.2 (Becton Dickinson, San Jose, CA) and monoclonal antibody AE1/AE3 (Zymed, San Francisco, CA).
Histologically, the primary mass of the right ovary was characterized by a pleomorphic cell population with numerous areas of necrosis, hemorrhage, and mineralization. The neoplasm comprised the entire right ovary, with no normal ovarian tissue present. The histologic features were consistent with a choriocarcinoma. Neoplastic cells were arranged both in large nodules as well as in more diffuse sheets and generally consisted of three distinct cell types. The majority of cells in the nodules were large, round to oval, with distinct cytoplasmic borders and foamy or vacuolated amphophilic cytoplasm. The nuclei were round to oval, with coarsely stippled chromatin and large, prominent nucleoli. These cells likely represented cytotrophoblast. Small numbers of large, multinucleated syncytial cells were observed in the inner layers of some nodules, particularly those filled with blood or necrotic debris. In some areas, the multinucleated or other neoplastic cells appeared to be in close contact with red blood cells (Fig. 2). The syncytial cells were consistent with syncytiotrophoblast, and the frequent arrangement of these cells in the inner layers and the cytotrophoblast in the outer layers of the nodules was similar to the arrangement of these cells in the normal placenta. The third type of neoplastic cells represented those that were more diffusely arranged. These cells were large, round to polygonal, and had amphophilic cytoplasm that frequently contained large, clear, and discrete vacuoles. The nuclei of these cells were large, variably round, folded or clefted, and often had intranuclear cytoplasmic herniations. These large cells were present individually or in small clusters and were usually surrounded by eosinophilic hyaline ground substance (Fig. 3). These cells likely represented extravillous trophoblast, and the eosinophilic ground substance was consistent with “fibrinoid,” a substance of uncertain structure that is produced by extravillous trophoblast in the normal placenta. Mitotic figures were numerous in all the cellular components, and occasional blood vessels had neoplastic cells within their walls.
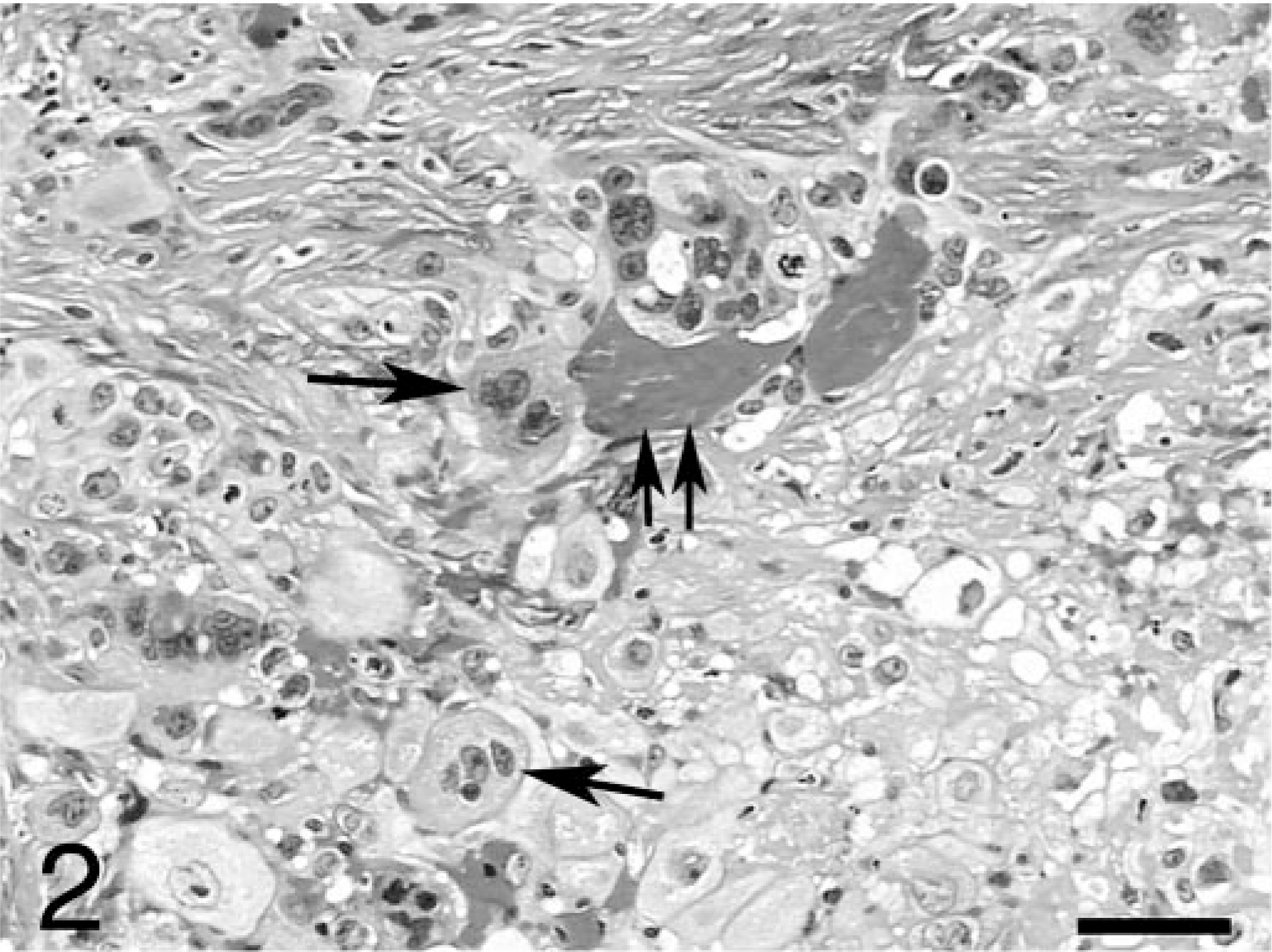
Ovary; rhesus monkey. Region of the ovarian tumor showing syncitiotrophoblast (arrows), some of which are directly adjacent to red blood cells (double arrow). HE. Bar = 50 μm.
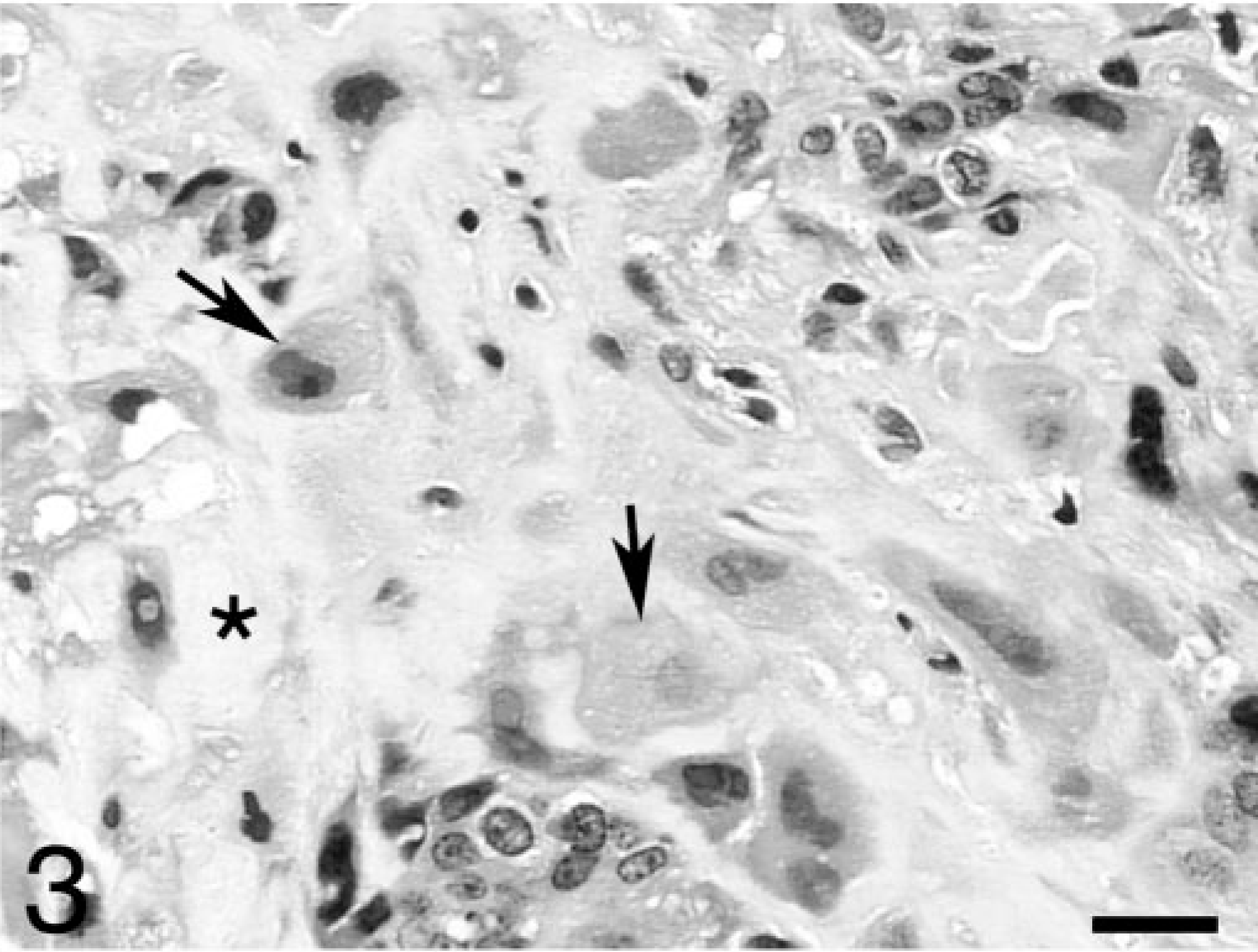
Ovary; rhesus monkey. Region of the ovarian tumor showing extravillous trophoblast (arrows) surrounded by fibrinoid (stars). HE. Bar = 30 μm.
The masses in the mesentery and lungs represented metastases with neoplastic features similar to those in the ovary. The metastatic lesions had fewer of the extravillous trophoblast and more hemorrhage and more prominent association of neoplastic cells in contact with red blood cells. With immunohistochemistry of the ovarian mass, some of the syncytiotrophoblast were positive for hCG (Fig. 4), some of the extravillous trophoblast were positive for hPL (Fig. 5), and almost all the tumor cells were strongly positive for CK (not shown). The immunohistochemistry results were similar to those in humans with choriocarcinoma. 12
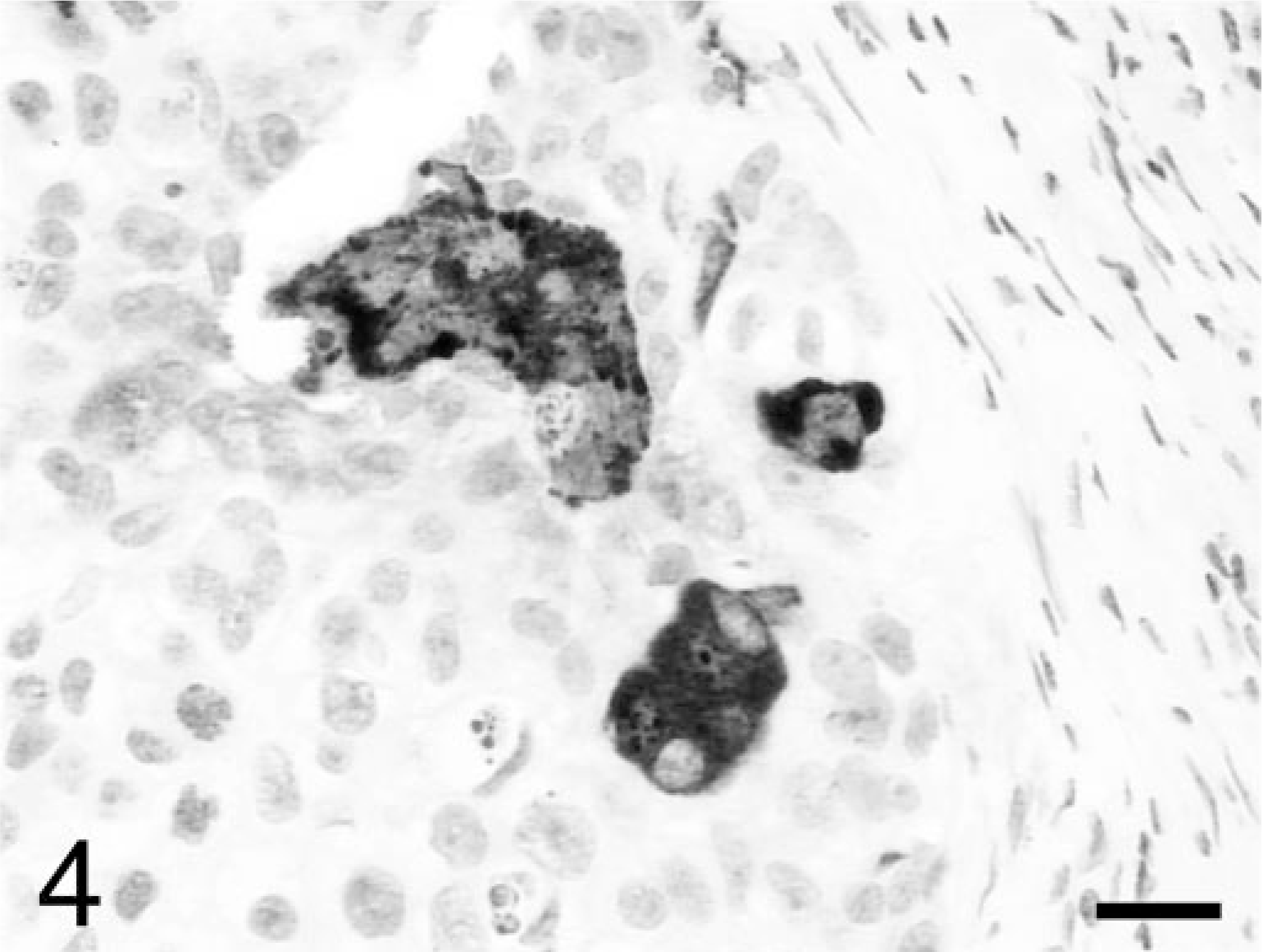
Ovary; rhesus monkey. Some of the syncitiotrophoblast are positive for human chorionic gonadotropin. Avidin–biotin–peroxidase complex method, hematoxylin counterstain. Bar = 30 μm.
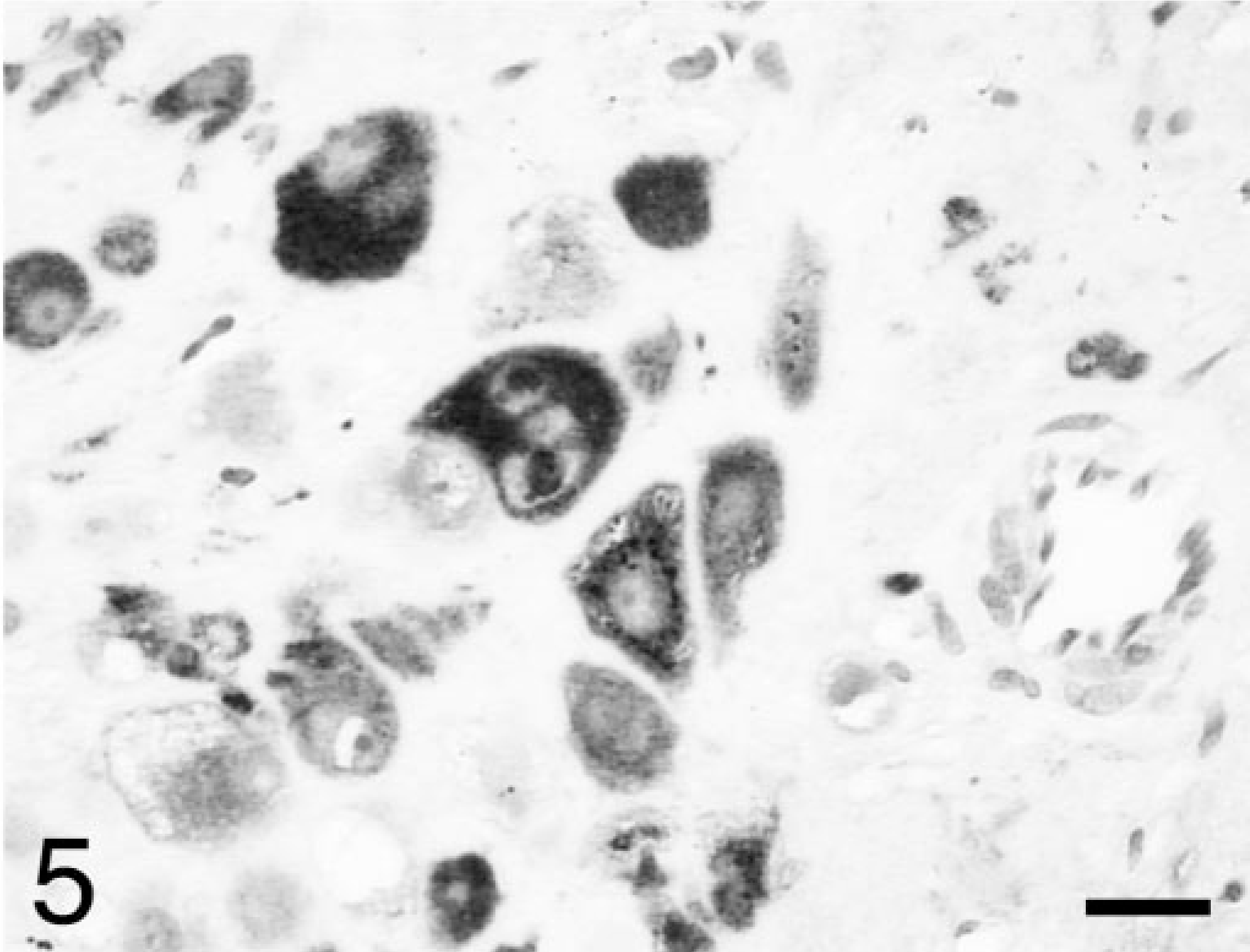
Ovary; rhesus monkey. Many of the extravillous trophoblast are positive for human placental lactogen. Avidin–biotin–peroxidase complex method, hematoxylin counterstain. Bar = 30 μm.
Blood remaining from samples drawn at the beginning of the study was tested for monkey chorionic gonadotropin (mCG) using an enzyme linked immunosorbent assay (ELISA), as previously described. 9 In women with choriocarcinoma, chorionic gonadotropin levels are often elevated. 12 The mCG level in this monkey was 1.90 ng/ml. The levels of mCG in the other females from this study, tested at the same time, ranged from undetectable to 0.109 ng/ml. Elevated mCG in the serum or urine of monkeys is usually diagnostic for pregnancy. However, because this monkey was not pregnant (the uterus was histologically normal, and she had not been exposed to a male), the elevated mCG levels were undoubtedly because of production by the tumor. The elevated mCG level before test article administration indicated that the neoplasm was likely preexisting at the start of the study. It did not prove the diagnosis of choriocarcinoma because elevated hCG levels have occasionally been associated with other tumor types in humans. 12 However, the histologic features and immunohistochemistry results were convincing of the diagnosis of choriocarcinoma in this monkey.
Choriocarcinomas of the ovary may occur from four different sources: primary neoplastic growth of ovarian germ cells; secondary to an ovarian pregnancy; metastases from a regressed or occult uterine primary; and in infants because of metastases from the placenta. 5 In humans, the source of the choriocarcinoma cannot be differentiated using immunohistochemistry or electron microscopy. However, DNA analysis may be performed to determine whether the choriocarcinoma contains only maternal DNA (primary germ cell origin) or both maternal and paternal DNA (subsequent to pregnancy). 12 DNA analysis was not performed of the tumor from the monkey of this report. However, because she was not currently pregnant and previous pregnancy was highly unlikely based on her previous history and housing arrangement, the neoplasm was considered a primary ovarian choriocarcinoma arising from germ cells.
In the previously reported case of a primary ovarian choriocarcinoma in a cynomolgus macaque, a teratoma was present in the opposite ovary. 13 In the monkey studied in this report, the opposite ovary appeared normal at necropsy, but was frozen for an assay as part of the study protocol, and was unavailable for histologic examination. The only other reported cases of choriocarcinoma in nonhuman primates were two instances of uterine choriocarcinoma in rhesus macaques that had been pregnant previously. 6, 10 Surveys of neoplasia (general or specifically ovarian) in nonhuman primates have indicated that the most common germ cell tumor of the ovary in monkeys is the teratoma. 7, 8 The more recent of those surveys indicated that regarding all ovarian tumors in nonhuman primates, sex cord stromal tumors were the most frequent, followed by relatively equal numbers of tumors of germ cell and surface epithelial origin. 8 The data in these surveys indicate that the type most likely to be malignant is the surface epithelial neoplasm.
Primary ovarian choriocarcinomas are also apparently rare in nonprimate species. Surveys of ovarian tumors in dogs, cats, and rats have failed to reveal any cases. 3, 4, 11 Eight cases have been reported in B6C3F1 mice, seeming to indicate an increased susceptibility in that strain. 1, 2 Beyond that, no case reports of ovarian choriocarcinoma in nonprimate species can be found.
Most choriocarcinomas in humans are of uterine origin and occur subsequent to a pregnancy. Ovarian choriocarcinomas usually do not follow a pregnancy and, as opposed to the uterine tumors of this type, are more typically seen in girls and young women (as young as 7 years of age and frequently in women younger than 20 years of age). 5 Primary ovarian choriocarcinomas in humans are almost always unilateral. However, more commonly, they occur in combination with other tumor types in the same or opposite ovary. 12 Ovarian choriocarcinomas historically have been associated with early metastasis and a poor prognosis, but newer therapies have resulted in an improvement of the outcome of girls and women with this neoplasm. 5
In addition to being the first report of a primary ovarian choriocarcinoma in a rhesus macaque, this article also documents the use of immunohistochemistry for demonstration of hPL in the tumor cells and ELISA for the measurement of serum chorionic gonadotropin to assist in the diagnosis of this unusual ovarian neoplasm.
Footnotes
Acknowledgements
We are grateful to Mr. Steven Claunch, University of California, San Diego, for his technical assistance in the immunohistochemistry slide preparation.
