Abstract
A cutaneous mass at the base of the retroauricular region of a 4-year-old, female Golden Retriever was examined pathologically. Histologically, the mass formed multiple nodules consisting of a proliferation of large clear cells with abundant cytoplasm. Mitotic figures among the neoplastic cells were very sparse. The large clear cells were intensely positive for cytokeratins (AE1/AE4, cytokeratin 8 and 18) and moderately positive for lysozyme and contained periodic acid-Schiff-positive granules in the cytoplasm. In addition, small flat cells lined the islands of neoplastic large clear cells, and these were strongly positive for alpha-smooth muscle actin and vimentin, and some were positive for cytokeratin (AE1/AE4), suggesting they were myoepithelial cells. No local recurrence or metastasis has been recognized during the 18 months since surgical excision. On the basis of these findings, the present tumor was diagnosed as apocrine sweat gland adenoma, clear cell variant. There have been few previous reports of canine apocrine adenomas showing a clear cell morphology.
Apocrine glands are the major type of sweat gland in dogs, and the distribution of eccrine sweat glands is limited to the footpad. Apocrine sweat gland tumors are rather common in dogs and tend to occur on the head, neck, and limb. The Golden Retriever is a breed that shows a predisposition to such tumors. 6 Approximately 70% of canine apocrine tumors are benign in nature, but malignant ones tend to recur locally and metastasize to regional lymph nodes and the lung. 5 The recent World Health Organization classification 4 categorizes apocrine sweat gland tumors in domestic animals into adenoma/carcinoma, complex or mixed adenoma/carcinoma (or both), and ductal adenoma/carcinoma. However, there is little information regarding clear cell morphology of these tumors.
In human sweat gland tumors, clear cell change of the neoplastic cells has been well demonstrated, 3, 8 and such morphologic features are found more frequently in eccrine gland tumors, especially nodular hidradenomas (hidradenocarcinomas) and poromas (porocarcinomas). 8 Moreover, similar clear cell change is also recognized in human salivary tumors. 3, 8 There has been a previous report of a clear cell variant of canine acinic cell carcinoma originating from a minor salivary gland in the oral cavity. 1 In this previous case, neoplastic foci were well circumscribed and were composed predominantly of sheets of large clear cells with vacuolar cytoplasm. The cytoplasmic vacuoles did not stain with periodic acid–Schiff (PAS), alcian blue, mucicarmine, or oil-red. Immunohistochemically, the neoplastic cells were also negative for several epithelial and mesenchymal markers including keratin and alpha–smooth muscle actin (SMA).
Recently, Mikaelian et al. 7 proposed a new type of canine tumor known as “follicular stem cell carcinoma” characterized by clear cells forming lobules and nests surrounded by a basement membrane. Among 30 dogs examined, 22 had trichoepithelioma-like lesions and 21 showed apocrine differentiations. On the basis of histologic, immunohistochemical, and ultrastructural findings, they concluded that the neoplasms might have originated from follicular stem cells. 7 The present article describes the morphologic features of a unique cutaneous canine tumor showing clear cell change and discusses the possible differential diagnoses.
A pedunculated mass was found at the base of retroauricular dermis in a 4-year-old, female Golden Retriever. The owner had been aware of the mass for at least 1 year before the dog was brought to the animal hospital, but during this period, there had been no clinical symptom. The surgically excised mass was hard and approximately 2.5 × 5 cm in diameter (Fig. 1). The cut surface was multilobulated in appearance and was white and partly red in color. The surgical margin at the deep subcutis was separated by a thin capsule, but the border between the tumor and dermis was unclear (Fig. 2). During the 18 months since the surgical excision, no local recurrence or metastasis has been recognized.
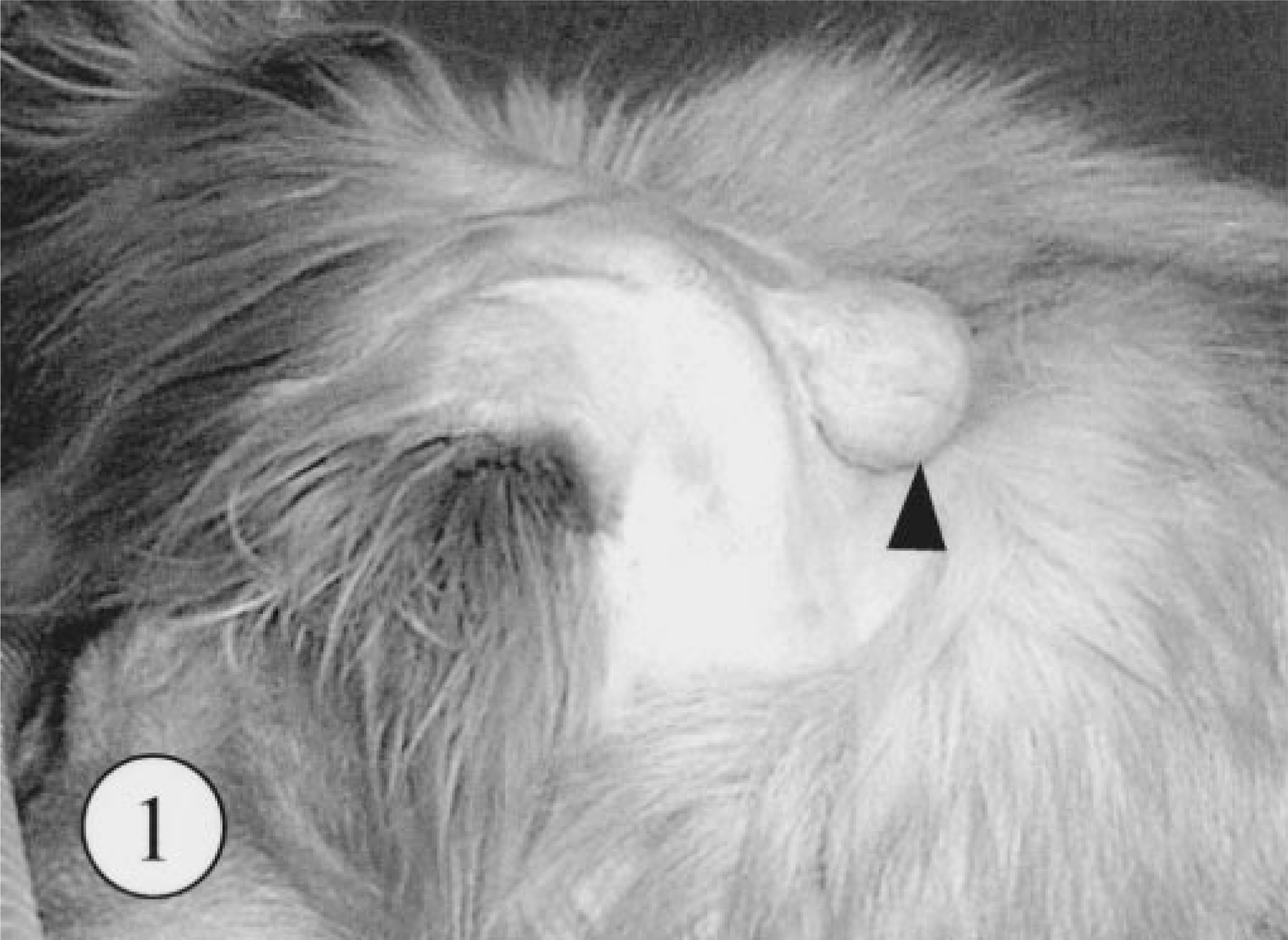
Skin tumor; dog. A pedunculated cutaneous mass on the base of retroauricular region (arrowhead).
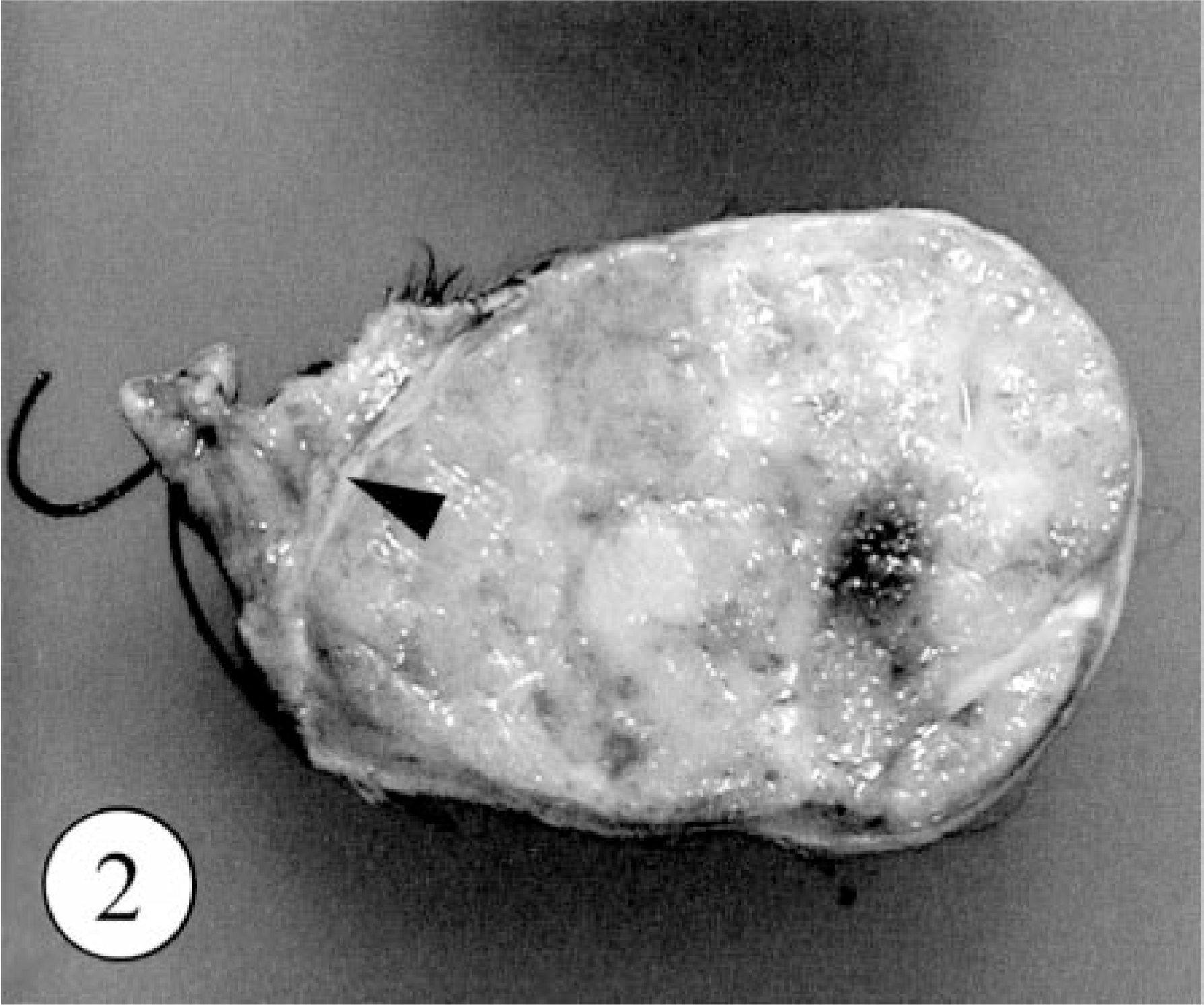
Skin tumor; dog. The cut surface of excised mass is multilobulated. The thin fibrous capsule between tumor and intact subcutis (arrowhead).
The mass was fixed with 10% formalin and embedded in paraffin for routine histologic examination. Paraffin sections of 2–4 μm thickness were stained with hematoxylin and eosin, PAS, and alcian blue (pH 2.5). Immunostaining was performed using Envision polymer reagent (Dako-Japan, Kyoto, Japan). Monoclonal antibodies against vimentin (prediluted, Dako-Japan), SMA, (prediluted, Dako-Japan), cytokeratin (AE1/AE3, prediluted, Dako-Japan), cytokeratin 8 (CK8, prediluted, Progen Biotechinik GmBH, Heidelberg, Germany), cytokeratin 18 (CK18, prediluted, Progen Biotechinik GmBH), lysozyme (1 : 300, Dako-Japan), and proliferating cell nuclear antigen (PCNA, prediluted, Dako-Japan) were used as primary antibodies. Pretreatment in a hydrated autoclave (121 C, 15 minutes) was performed for vimentin, cytokeratins, and PCNA immunostaining. Intact apocrine glands, eccrine glands in the footpad, parotid glands, and submandibular glands taken from several dogs at necropsy were used as the control tissues.
Microscopically, the neoplastic foci had formed in the dermis, and the border between tumor and the deep subcutis was separated by a thin fibrous capsule. The neoplastic foci were divided into multiple lobules by thin connective tissues (Fig. 3). Around the neoplastic foci, a small number of intact apocrine sweat glands were present. There were no ectopic tissues, including salivary glands. The dominant, proliferating neoplastic cells were large clear cells with a characteristic polygonal shape and abundant clear to eosiophilic cytoplasm occasionally containing PAS-positive and alcian blue–negative granules. The nucleus was varied in size from approximately 10 to 20 μm and sometimes contained distinct nucleoli. Mitotic figures among these cells were very rare. In addition, small flat cells lined the neoplastic foci of large clear cells. Single or a few cluster of the neoplastic cells were packed in the surrounding connective tissues (Fig. 4). There were no evidences for vascular invasion of neoplastic cells. The results of PAS and alcian blue staining in control tissues and the tumor are summarized in Table 1.
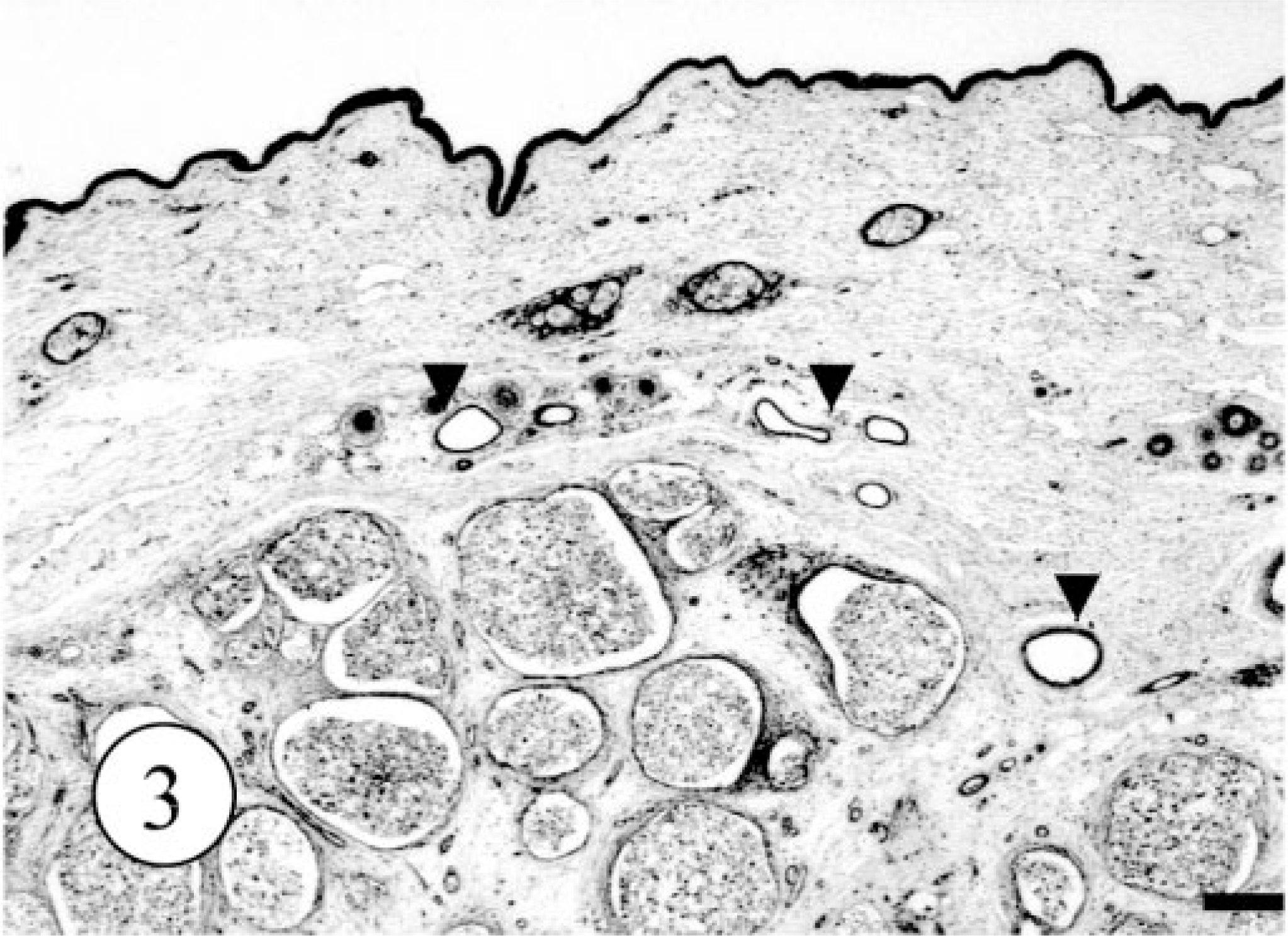
Apocrine gland adenoma; dog. Multiple nodules of neoplastic cells in the dermis and subcutis. Normal apocrine sweat glands are also present (arrow heads). HE. Bar = 500 μm.
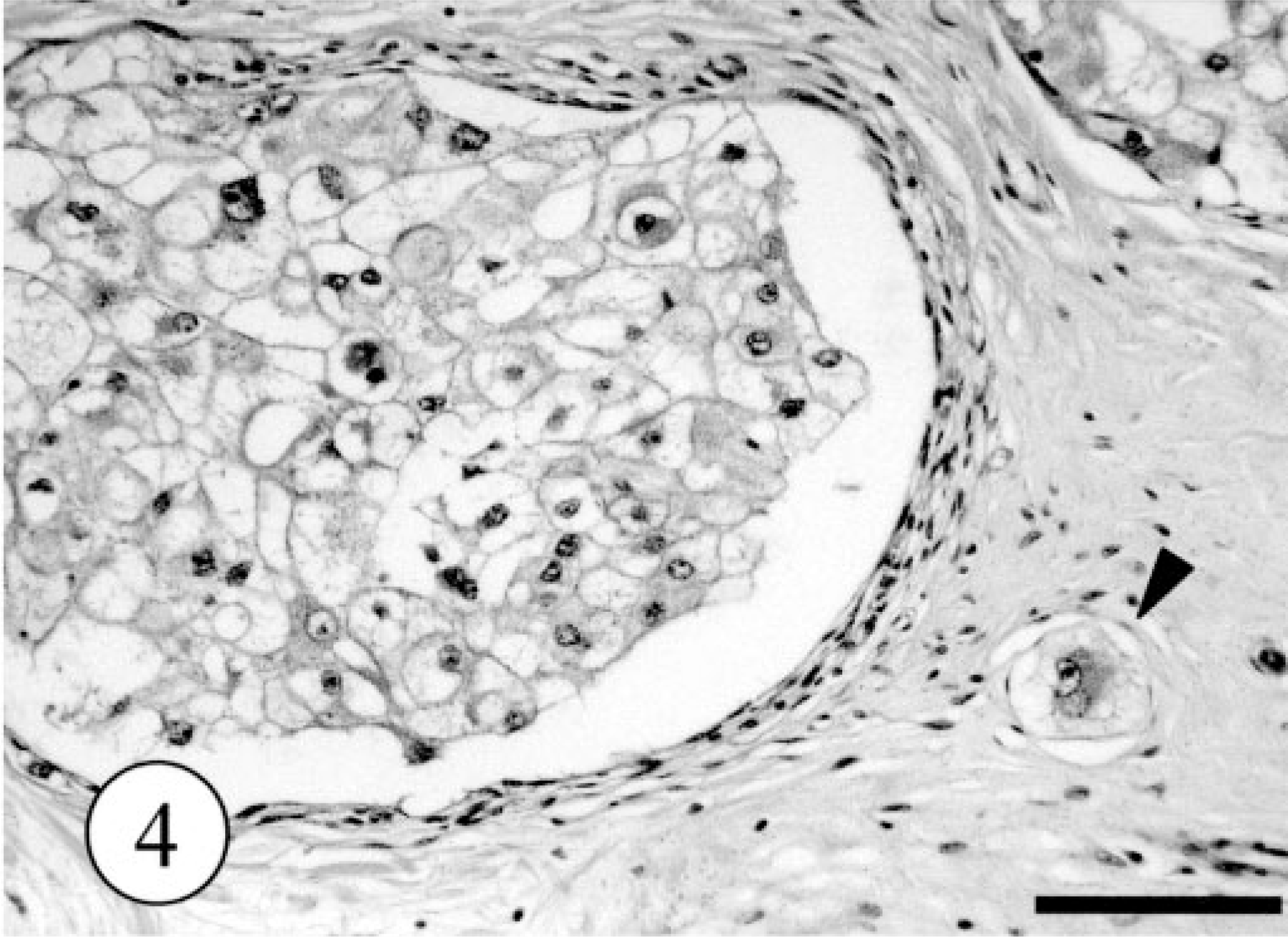
Apocrine gland adenoma; dog. The tumor cells with irregularly large, clear cytoplasm. Single neoplastic cell was packed in the surrounding connective tissues (arrow head). HE. Bar = 200 μm.
Results of special and immunostains of tumor and four control tissues.∗
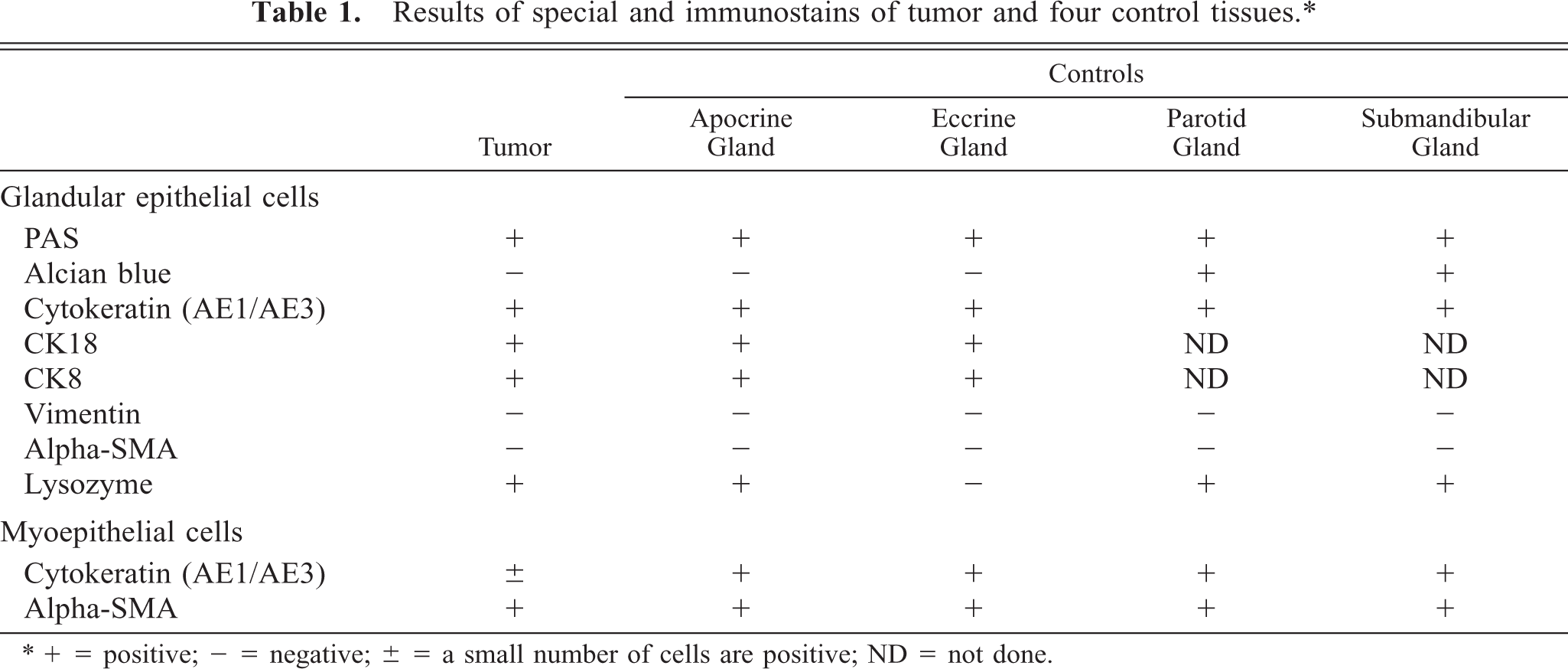
∗+ = positive; − = negative; ± = a small number of cells are positive; ND = not done.
In control tissues, most glandular epithelial cells were immunopositive for cytokeratin (AE1/AE4) and myoepithelial cells were positive for cytokeratin (AE1/AE4), SMA, and vimentin. Immunostaining for CK8 and CK18 labeled the single epithelium of apocrine and eccrine glands and did not react with the epidermis, hair follicles (Fig. 5), or sebaceous glands. Immunohistochemically, most of the neoplastic clear cells were strongly positive for cytokeratin (AE1/AE4, Fig. 6) and CK8 (Fig. 7) and some were also intensely positive for CK18. The small, flat cells around large clear cell were strongly positive for SMA and vimentin, and some were positive for cytokeratin AE1/AE4, suggesting that they were myoepithelial cells (Fig. 8). The neoplastic clear cells and glandular epithelial cells of both intact apocrine and salivary glands were moderately to weakly positive for lysozyme, 2 whereas those of eccrine gland were negative (Table 1). The immunohistochemical results showed that tumor had originated from secretory tissue, especially apocrine glands. There was no significant difference in the PCNA positivity index between the tumor cells and the cells in intact apocrine and salivary glands.
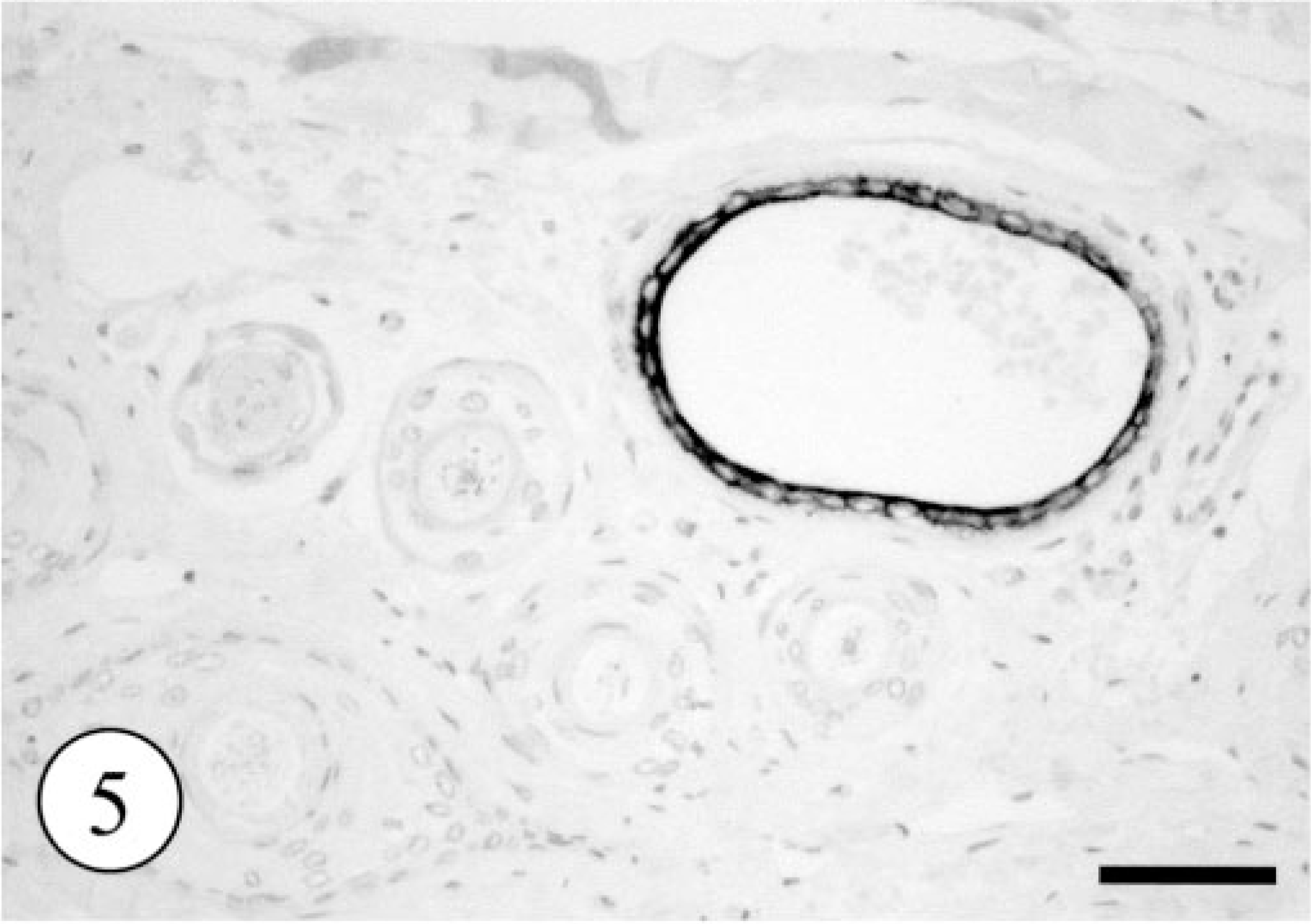
Normal skin; dog. CK8 immunostaining selectively labeled the single epithelium of canine apocrine gland. The follicular epithelium was completely negative for CK8. Envision polymer method. Bar = 200 μm.
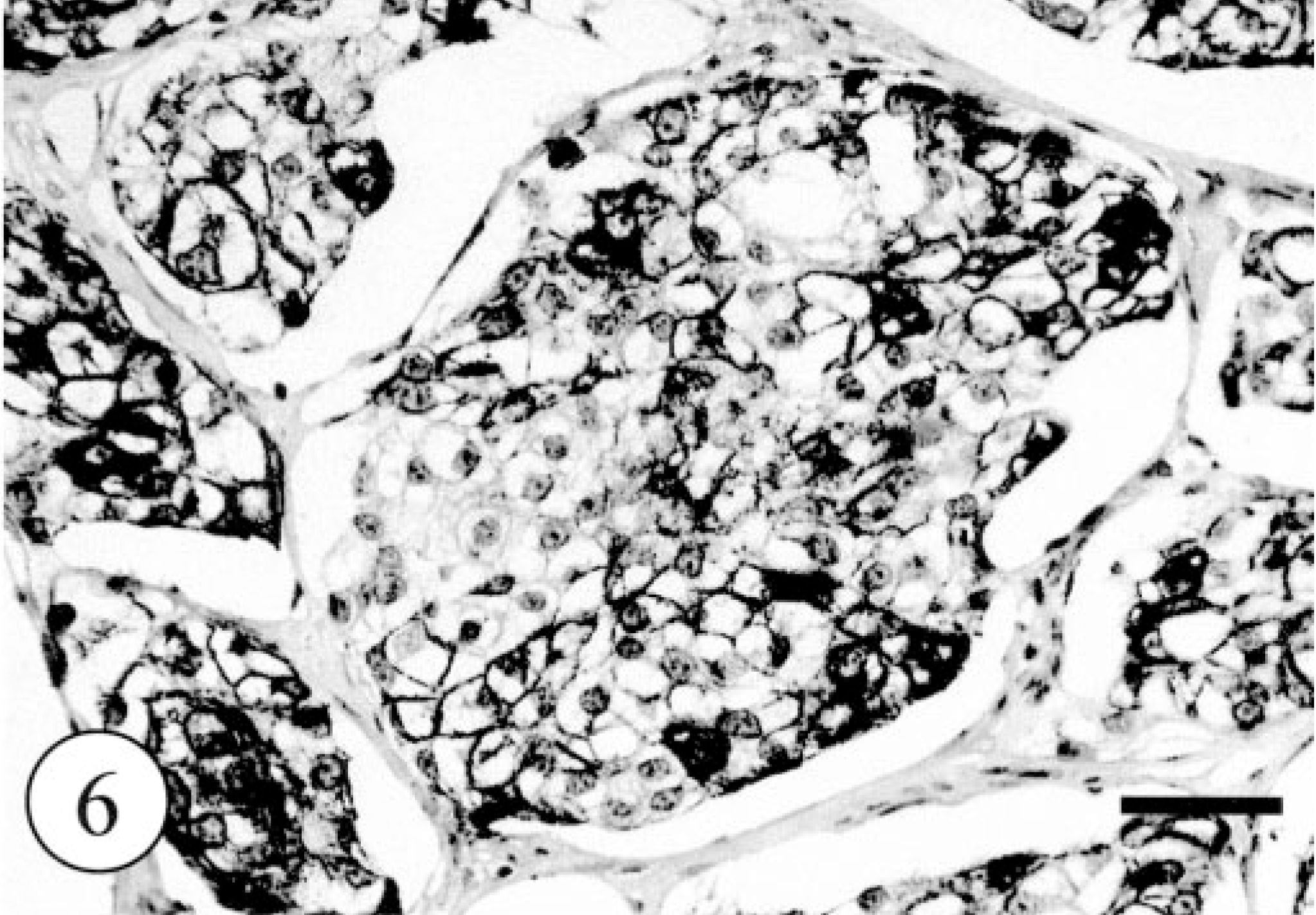
Apocrine gland adenoma; dog. Intense immunoreactivity of neoplastic large cells against cytokeratin (AE3/AE4). Envision polymer method. Bar = 200 μm.
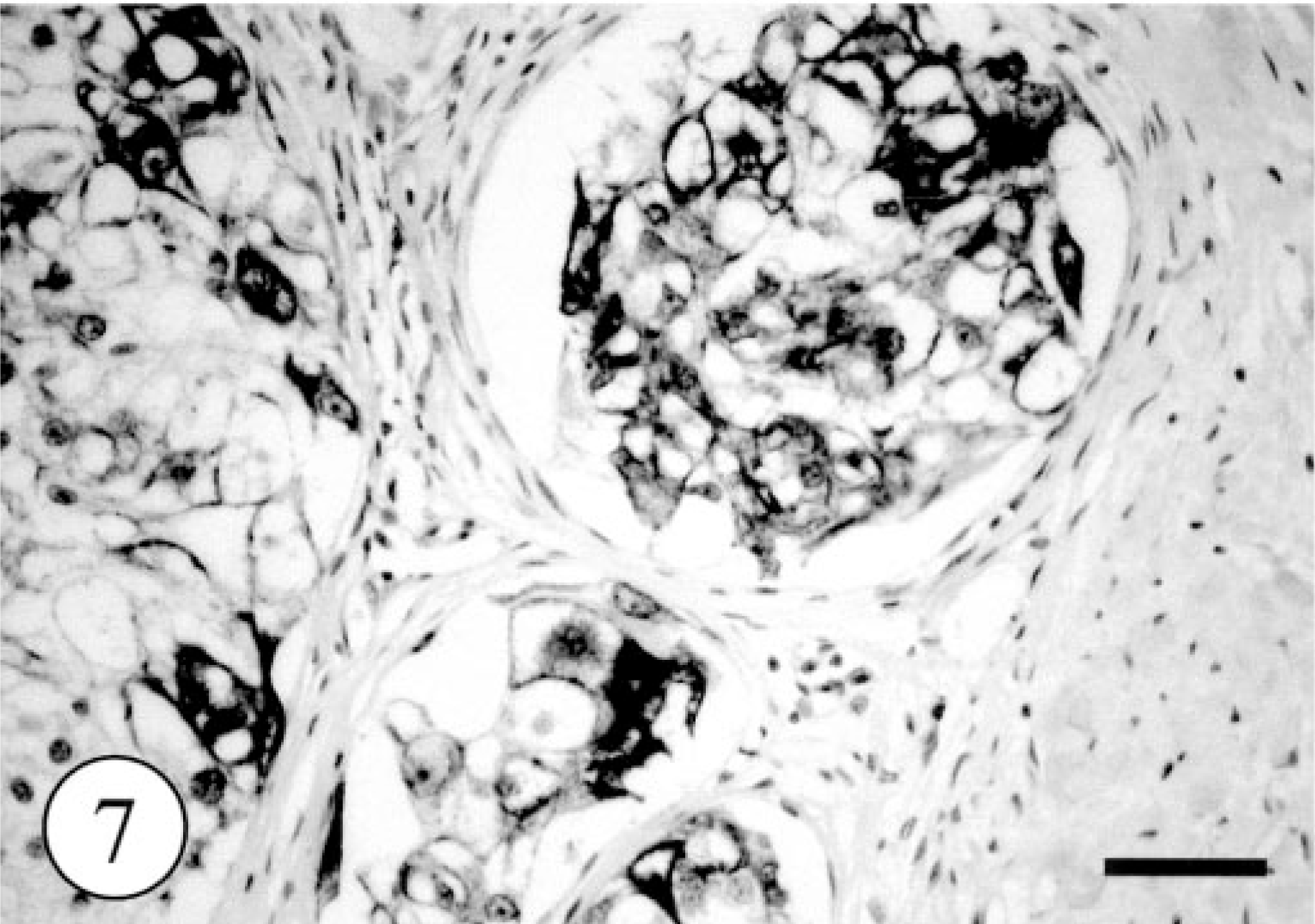
Apocrine gland adenoma; dog. Intense immunoreactivity of neoplastic large cells against CK8. Envision polymer method. Bar = 200 μm.
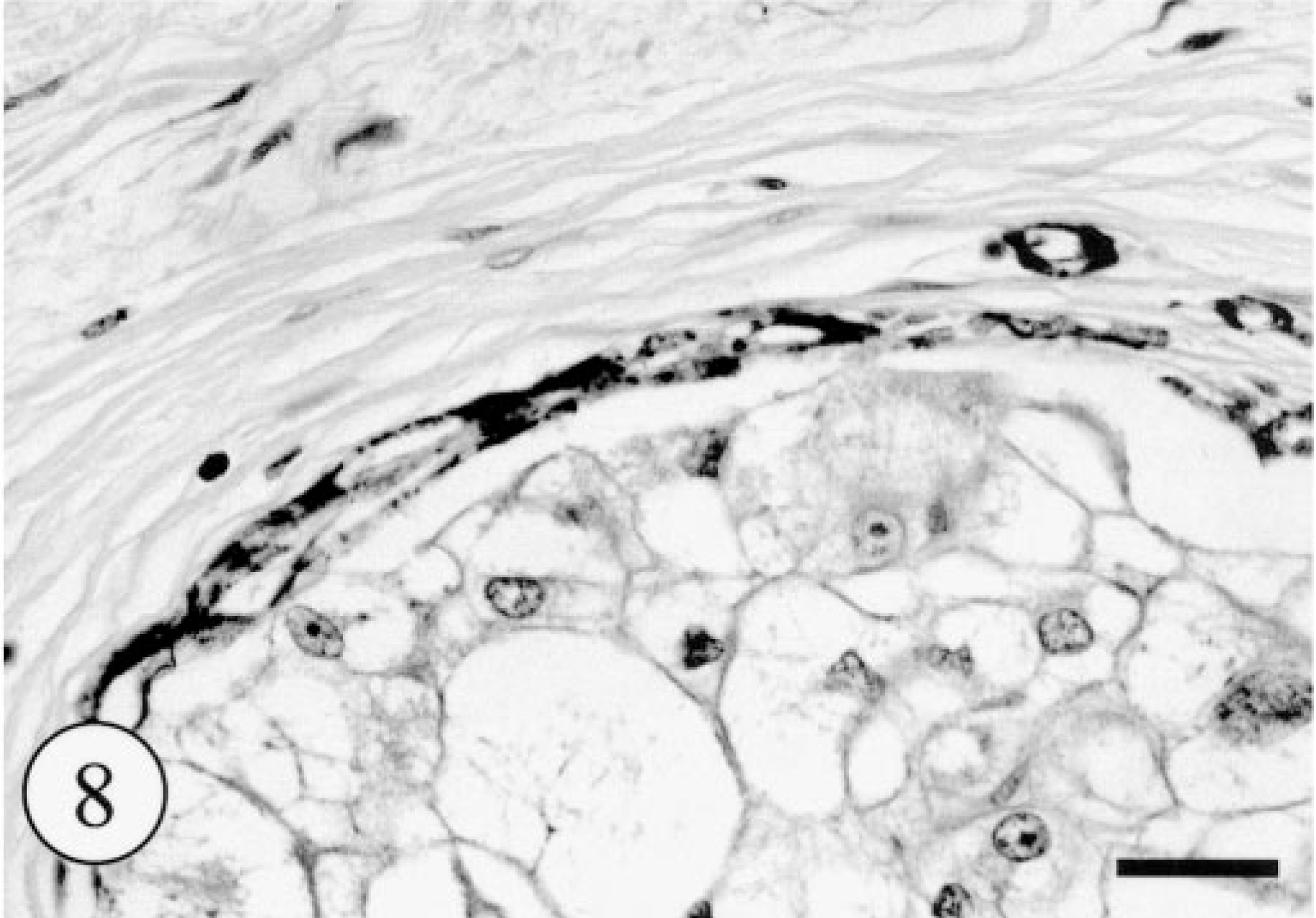
Apocrine gland adenoma; dog. Intense immunoreactivity of peripheral flat cells against α-SMA. Envision polymer method. Bar = 100 μm.
A clear cell morphology is unusual in tumors arising from skin appendage in domestic animals, 7 even though most skin appendages, including sebaceous, apocrine, eccrine glands, and hair follicle, have the potential to transform into clear cells. 8 The alteration to clear cell morphology occurs by accumulation of glycogen, mucosubstance, and lipid, according to the nature of original appendages. Generally, clear cell change of the neoplastic cells has been recognized most frequently in human sweat gland tumors. 8 These tumors are categorized as clear cell eccrine poromas arising from the intraepidermal portion of an eccrine or apocrine duct or acrosyringium or nodular hidradenomas (clear cell hidradenomas), including several alternative terms such as clear cell myoepithelioma, eccrine acrospriroma, and sweat gland adenoma. 8 However, the criteria for eccrine sweat gland tumors have not been well categorized in domestic animals 4 because of the limited distribution of eccrine glands and the low incidence of their tumors. In addition, the location of the present tumor was not consistent with the distribution of canine eccrine sweat glands.
Immunohistochemical examinations revealed that this neoplasm retained a double layer of acinar and myoepithelial cells. In addition, the neoplastic clear cells showed intense immunoreactivity for CK8 and CK18, which were expressed selectively in the single epithelium of apocrine and eccrine glands. These facts supported the likelihood that the neoplasm had arisen from glandular tissue, most likely an apocrine gland. Although the morphologic features of neoplastic cells were quite different from those of typical canine apocrine adenomas and the age of the affected dog was not typical, the present tumor was considered to be a benign apocrine sweat gland tumor with a clear cell morphology, for the subsequent reasons. First, the neoplastic clear cell proliferation was found in the dermis with normal apocrine glands. Second, the proliferation activity evaluated in terms of mitotic rate and PCNA immunostaining was very low and the clinical outcome was excellent, suggesting the benign natures of the tumor.
On the other hand, some neoplasms with clear cell morphology have been categorized in human salivary glands. Human glycogen-rich adenocarcinoma, derived from the salivary gland, has biologic features similar to those of the present tumor, such as PAS-positive and alcian blue–negative clear cytoplasm, and good prognosis despite microinvasion. 3 Even in dogs, similar clear cell changes have also been recognized in an oral tumor derived from minor salivary glands. 1 Thus, the salivary gland, especially parotid gland, could also be considered as the origin, although no ectopic salivary glands were observed within the tumor tissues examined.
The designation of “follicular stem cell carcinoma” recently proposed by Mikaelian et al. 7 might be also considered as a possible differential diagnosis. The histologic and immunohistochemical features of some previously reported tumor 7 are almost consistent with those in this study, although the dog in this study was considered to have a benign tumor because of its low-proliferative activity and excellent clinical prognosis. In addition, the tumor in this study had characteristic histologic and immunohistochemical features that indicated a sweat gland origin without follicular differentiation. Thus, “apocrine adenoma, clear cell variant” seems to be the most likely designation.
