Abstract
The prognostic significance of histologic and clinical features was evaluated in a retrospective study of 70 dogs treated with nephrectomy for renal cell carcinoma. Dogs presenting with hematuria and cachexia had significantly decreased overall and tumor-specific survival. Mitotic index (MI), nuclear size, nuclear pleomorphism, tumor differentiation, invasiveness, Fuhrman nuclear grade, and clear cell morphology were significantly associated with survival times (overall and tumor specific) in univariate analyses. A multivariate Cox proportional hazards model was constructed using stepwise selection to evaluate potential histologic predictor variables. This multivariate analysis revealed MI, defined as the number of mitotic figures in ten 400× fields, as the sole independent prognostic variable. Median survival for dogs with an MI >30 was 187 days compared with 1184 days for dogs with an MI of <10. Dogs with an intermediate MI of 10 to 30 had a median survival of 452 days. Canine renal carcinomas were categorized into the following subtypes based on histologic features and histochemical and immunohistochemical staining: (1) clear cell, (2) chromophobe, (3) papillary, and (4) multilocular cystic renal cell carcinomas. Clear cell carcinoma was diagnosed in 6 of 70 (9%) canine tumors and was associated with a significantly decreased median survival time. Papillary carcinomas were identified in 15 of 70 tumors (21%), chromophobe in 6 of 70 (9%), and the multilocular cystic variant of canine renal cell carcinoma in 3 of 70 tumors (4%). These findings facilitate uniform categorization of canine renal cell carcinoma and provide veterinary pathologists with criteria to determine prognostic information.
Keywords
Renal cell carcinoma (RCC) is the diagnosis given to a group of malignancies arising from epithelial cells of the nephron and is the most common primary tumor of the canine kidney. 26 RCC is a disease of variable histologic appearance and clinical behavior in dogs 1,5,14,17,24,26,31 and must be differentiated from other primary epithelial neoplasms, including renal adenomas, renal oncocytomas, nephroblastomas, and transitional cell carcinomas. 26,27 Primary renal neoplasia is estimated to be 0.3% to 1.5% of all canine neoplasms, 26 which is similar to the incidence of RCC in adult humans. 15 Males are overrepresented, with reported male to female ratios of 1.8:1 to 1.6:1, 14,17 which is similar to the 1.7:1 male to female ratio reported in people. 16 RCC is more common in middle-aged dogs, with an average age of 8 to 9 years at diagnosis. 26
A review of the literature identifies dogs with RCC that present with widespread metastases and have short survival times as well as cases with no evidence of metastasis and extended survival of up to 4 years following nephrectomy. 1,5,24 RCC is often diagnosed late in the course of disease, and presumed pulmonary metastases have been identified by thoracic radiographs in 18% to 48% of dogs at presentation. 5,17 The primary tumor often reaches considerable size before clinical signs are apparent, and 20% to 43% of dogs with primary renal tumors present with palpable abdominal masses. 5,17 Dogs commonly present with a history of hematuria, lethargy, anorexia, polyuria/polydipsia, abdominal pain, or cachexia. 5,17,24
Canine RCCs have previously been classified according to histologic (papillary, tubular, and solid type) or cytologic (chromophobic, eosinophilic, and clear cell type) subtypes, but differences in biologic behavior based on these classifications have not been previously observed. 26,27 The most common RCC subtypes in humans include clear cell, papillary, and chromophobe with less common subtypes including collecting duct and multilocular cystic RCCs. 9,20,23 Clear cell carcinomas are the most frequently diagnosed subtype in humans, comprising approximately 75% of human renal carcinomas; 16 clear cell carcinomas are rarely observed in dogs. 26,33 Case reports have also described canine subtypes with many similarities to human chromophobe 19 and collecting duct RCCs. 18 In humans, histologic grading has been repeatedly verified as the most important prognostic predictor in RCC following stage of disease, regardless of subtype. 7,12,16,28 Given the epidemiological and morphologic similarities of human and canine RCC, we hypothesized that histologic grading would correlate with survival in canine RCC. This retrospective study demonstrates the prognostic significance of histologic, epidemiological, and clinical features of canine RCC and establishes the incidence of clear cell, chromophobe, papillary, and multilocular cystic renal cell carcinomas in dogs.
Materials and Methods
Source of Cases and Criteria of Inclusion
For this retrospective study, cases were identified through a database search of the surgical pathology service at the Colorado State University Veterinary Diagnostic Laboratories. The diagnosis of renal cell carcinoma was made for 214 dogs between February 1996 and January 2012 on submissions received from 25 continental US states and 2 Canadian provinces. Of the 214 dogs, there was a lack of archived slides and tissue blocks for 4 cases. Diagnostic laboratory records for each dog included age, breed, and sex. Clinical information was retrieved via postoperative surveys, faxed medical records, and telephone interviews when necessary and was available for a subset of 82 dogs. Clinical information included presenting signs, date of initial diagnosis, diagnostic imaging results, lymph node status, postoperative treatment with chemotherapy, time from diagnosis to death or last follow-up, and cause of death. Cases excluded from survival analysis were those that died or were euthanized due to surgical complications (n = 6), those lost to follow-up within 20 days of surgery (n = 2), or those with diagnoses made via incisional biopsies (n = 4) as opposed to nephrectomies. Glass slides routinely prepared and stained with hematoxylin and eosin were available from the primary tumor for the cases included in the survival analysis. Categories for outcome included euthanasia or death related to RCC, euthanasia or death unrelated to RCC, lost to follow-up, or still alive at the conclusion of data collection. Survival time was defined as the time from nephrectomy until death or euthanasia. Animals were censored at the point of the last clinical visit if lost to follow-up or still alive. Categorical outcome and metastasis information was based on the interpretations of the primary care veterinarians and/or the surgical specialists involved in patient care and diagnosis.
Histologic Analysis
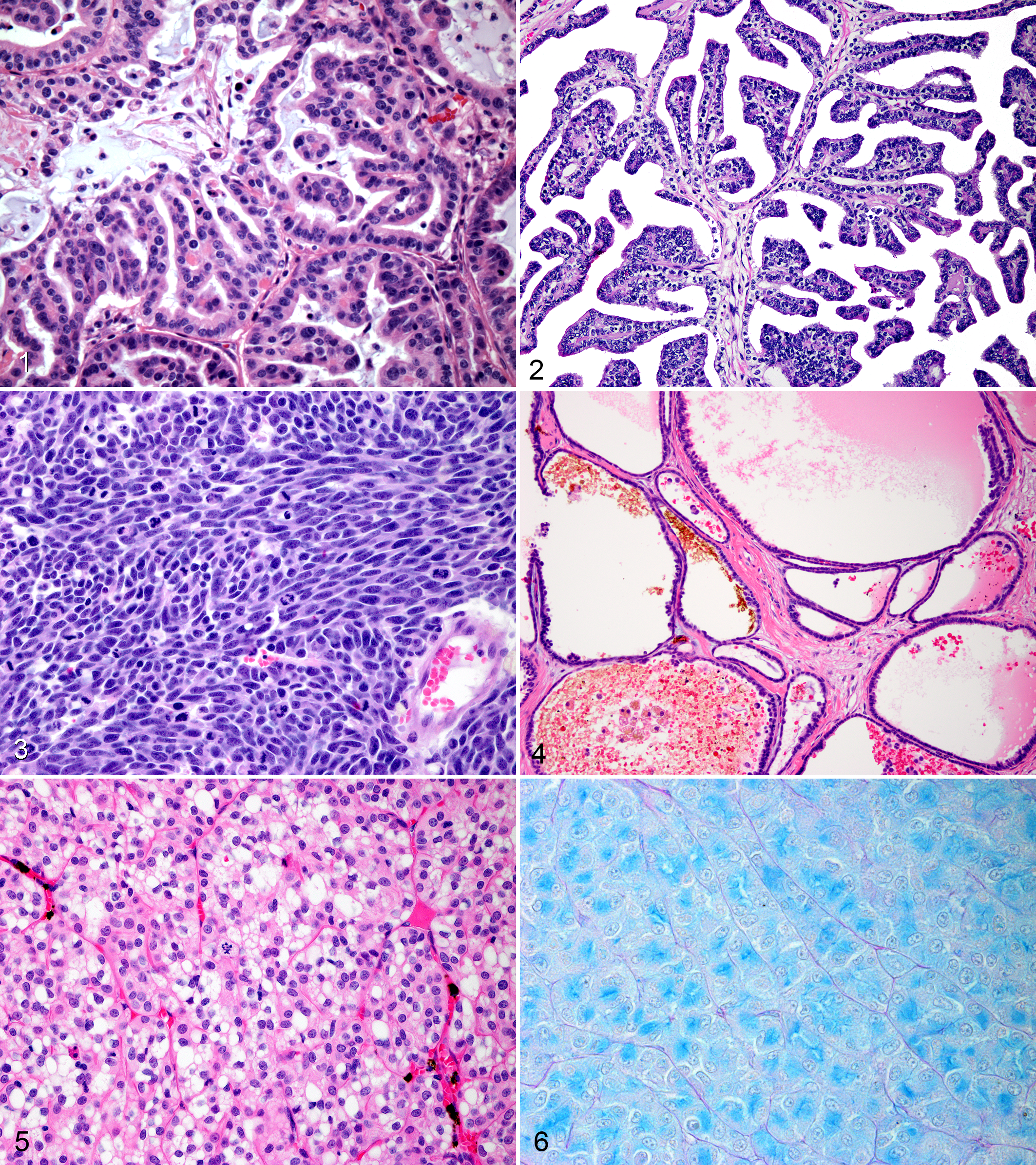
Histologic variables analyzed included mitotic index, histologic subtype (solid, tubular, papillary, cystic, multilocular cystic), cell type (clear cell and chromophobe), differentiation, vascular invasion, nuclear and nucleolar morphology, Fuhrman nuclear grade, 12 sarcomatoid differentiation, the presence of a pseudocapsule, and the graded presence of necrosis, hemorrhage, and invasiveness. Glass slides from cases examined for survival analysis were reviewed independently by 2 authors (E.F.E. and B.E.P.), and differences were discussed jointly. Mitotic index was defined as the number of mitotic figures counted in 10 consecutive 400× fields; areas of highest mitotic activity were selected. Nuclear pleomorphism was characterized as mild in tumors with uniform round nuclei, moderate in tumors with intermediate variation in nuclear shape and size, and marked in tumors containing karyomegalic nuclei, double the size of adjacent nuclei within the tumor, with marked variability in shape. Well-differentiated tumors contained cells organized in structures clearly identifiable as neoplastic renal tissue. Degree of invasion into the surrounding renal parenchyma varied from mildly infiltrative into adjacent parenchyma to marked invasion into adjacent normal tissues leading to foci of tumor cells distantly infiltrating normal kidney, often accompanied by desmoplasia. The 4-tiered Fuhrman nuclear grade, which is the most commonly used grading system by pathologists, was assigned as described by the original authors. 12 Solid tumors were characterized as having closely spaced carcinoma cells forming small nests, clusters, or sheets supported by minimal stromal elements. Tubular tumors were composed of neoplastic cells forming tubules separated by small amounts of fibrovascular stroma that frequently contained basophilic, mucinous material and apoptotic cellular debris (Fig. 1). Papillary tumors were composed of branching fibrovascular stroma lined by cuboidal epithelium (Fig. 2); papillary projections were commonly arranged in complex branching patterns. Cystic tumors were categorized based on the presence of a large, central space devoid of tumor cells containing hemorrhage, eosinophilic fluid, clear space, or cellular debris and contained a periphery composed of variably thick sheets of solid neoplastic cells or papillary projections. Sarcomatoid change was characterized by a gradual transition of neoplastic epithelial cells into atypical, elongated spindloid cells (Fig. 3). Multilocular cystic renal cell carcinomas were characterized by tumors composed of multiple, variably sized cystic spaces separated by fibrous septae lined by cuboidal epithelium (Fig. 4). Cystic spaces in multilocular cystic tumors contained eosinophilic homogeneous fluid, hemorrhage, or clear space, and neoplastic cells lining the septa contained uniform, round nuclei with hyperchromatic chromatin and indistinct nucleoli; cells were most commonly in a single layer, although regions containing cells in stacks of 2 to 3 were present as well as infrequent, blunted papillary projections.
Immunohistochemical and Histochemical Analysis
Immunohistochemistry for vimentin (Leica PA0033, monoclonal, 1:1000, Leica, Buffalo Grove, IL, ER2) and CD117 (Dako, Carpinteria, CA, polyclonal rabbit, 1:500, Leica ER2) and histochemical analysis using colloidal iron stain (acid mucopolysaccharides detection; American MasterTech, Lodi, CA, Colloidal Iron Stain Kit and procedures, item KTCIR) was performed on 5-μm sections from all primary tumors included in the survival analysis.
Criteria for Clear Cell RCC
Solid tumors composed of extensive areas of carcinoma cells with abundant, clear, vacuolated cytoplasm and an extensive network of small-caliber blood vessels were designated clear cell carcinomas (Fig. 5). In addition, these tumors each displayed strong, intracytoplasmic immunoreactivity for vimentin and were negative for expression of CD117.
Criteria for Chromophobe RCC
Solid tumors composed of polygonal cells with distinct cell borders and abundant, lightly eosinophilic cytoplasm that stained positively with colloidal iron stain for acid mucopolysaccharides (Fig. 6) were designated chromophobe RCC. Each of these tumors also was positive for intracytoplasmic CD117 protein.
Statistical Analysis
Clinical, epidemiological, and histomorphologic variables were analyzed. Clinical variables included presenting signs (hematuria, cachexia, anorexia, palpable abdominal mass) and treatment with chemotherapy (yes/no). Epidemiologic variables included sex (male or female), age (<6, 6–9, >9 years), and breed (small breed ≤20 kg, large breed >20 kg, mixed breed). Histomorphologic features evaluated included tumor pattern (solid, papillary, tubular, cystic, multilocular cystic), mitotic index, differentiation, vascular invasion, nuclear size, nuclear pleomorphism, nucleolar prominence, Fuhrman nuclear grade, cell type (clear cell, chromophobe), the presence of a pseudocapsule or sarcomatoid differentiation, and the graded presence of necrosis, hemorrhage, and invasiveness. Locally weighted scatterplot smoothing regression was used to evaluate breakpoints in mitotic indices in comparison to survival times. Univariate analyses with clinical, epidemiological, and histomorphologic features was done using both the Kaplan-Meier method and Cox proportional hazards models. Median survival times were derived from Kaplan-Meier analyses and 95% confidence intervals were derived when possible; in some cases, the 95% confidence interval was incalculable. All values were considered significant when P < .05. In addition to the univariate analyses, significant histomorphologic variables were evaluated in stepwise multivariate Cox proportional hazards analysis using overall and tumor-specific survival as dependent variables. SAS software (version 9.2; SAS Institute, Cary, NC) was used for Cox modeling and STATA software (version 11.2; StataCorp, College Station, TX) was used for Kaplan-Meier analysis.
Results
Outcome
The overall median survival time (MST) for dogs diagnosed with RCC was 743 days (95% confidence interval [CI], 410–1184). Death due to neoplastic disease occurred in 33 dogs (47%). Seven dogs (10%) died or were euthanized for reasons unrelated to RCC. Thirty dogs (43%) were still alive at the conclusion of data collection or lost to follow-up. When analyzing cases accompanied by surgery reports, 6 of 78 (8%) dogs undergoing nephrectomy for RCC experienced perisurgical mortality.
Signalment
For the 70 dogs with clinical follow-up, there was a male to female ratio of 1.3:1, including 40 males (9 intact) and 30 females (2 intact). There were 54 purebred dogs in the survival study, including 15 Labrador Retrievers, 3 Boxers, and 2 each of Golden Retrievers, Cocker Spaniels, Beagles, Poodles, Australian Shepherds, Rottweilers, Miniature Schnauzer, Bichon Frises, Chihuahuas, Border Collies, and Beagles, as summarized in Supplemental Table S1. When analyzing the signalment for the entire 214 cases retrieved from the database, there was a male predominance of 1.3:1, and the most common breeds were Labrador Retriever (n = 28), Golden Retriever (n = 13), Cocker Spaniel (n = 9), Boxer (n = 8), Border Collie (n = 6), Rottweiler (n = 6), Dalmatian (n = 5), and Bischon Frise (n = 5), with 4 each of Beagles, Dachshunds, Siberian Huskies, and Springer Spaniels. No significant survival differences were observed between small- and large-breed dogs (log rank, P = .278). The average age at diagnosis was 8.7 years (range, 3–15 years), and diagnosis at a young age was identified as a significant risk factor in univariate and Kaplan-Meier analyses. The 10 dogs diagnosed at younger than 6 years had a decreased MST of 187 days (P = .015; 95% CI, 68–452), as illustrated in Supplemental Table S2. In univariate analysis using tumor-specific survival, age <6 years at diagnosis was associated with a significantly elevated hazard ratio (HR) of 2.97 (P = .021; 95% CI, 1.18–7.50), but when using overall survival, age <6 years at diagnosis was not a significant risk factor (P = .187).
Clinical Data
The most commonly reported presenting signs were hematuria in 10 dogs (14%), a palpable abdominal mass on physical examination in 10 dogs (14%), cachexia in 6 dogs (9%), and anorexia in 5 dogs (7%). In 8 cases (11%), the renal tumor was an incidental finding during a health screening (n = 7) or, in 1 case, surgery to repair a gastric dilatation volvulus. Other presenting clinical signs included lethargy, vomiting, abdominal or back pain, diarrhea, urinary tract infection, polyuria and polydipsia, hemoabdomen, or collapse. Of the presenting signs, hematuria and cachexia were identified as significant risk factors. Hematuria was associated with decreased MST of 147 days (P < .001; 95% CI, 3–379) and an increased HR of 5.072 for experiencing death or euthanasia (P < 0.001; 95% CI, 2.183–11.786), as shown in Supplemental Table S3. Dogs presenting with cachexia had an MST of 76 days (P < .001) and an increased HR of 5.647 (P < .001; 95% CI, 2.248–14.187). Ultrasound reports often described tumors that invaded and replaced normal renal parenchyma and infiltrated the renal capsule and/or pelvis. In 1 case, sonographic evidence of invasion into the renal vein and caudal vena cava was present; surgical reports did not corroborate or refute this finding. For the 21 cases in which tumor size was provided, the average size was 7.1 cm in largest diameter with a range of 2 to 20.5 cm. No predilection for the left or right kidney was demonstrated, and bilateral renal involvement was observed in a single case.
Metastatic Disease and Local Recurrence
Thoracic radiograph results prior to nephrectomy were available for 44 dogs in this study. Presumed pulmonary metastasis was observed on chest radiographs in 5 of these 44 dogs (11%) prior to surgery. In addition to dogs with radiographic pulmonary nodules prior to surgery, 10 dogs developed radiographically visible pulmonary nodules attributed to RCC metastasis at an average of 10.2 months following surgery (range, 2–23 months). Two dogs had histologically confirmed nodal metastases at the time of diagnosis, and survival was 160 and 370 days for these dogs; both these tumors had mitotic indices of >30, and 1 case was classified as clear cell RCC. Other reported sites of presumed metastasis included liver (n = 3), lymph node (n = 3), mesenteric seeding or carcinomatosis (n = 3), and the fifth cervical vertebrae (n = 1). Presumed local recurrence was observed in 3 cases. Overall, metastases or local recurrence were observed clinically via imaging at an average of 8.5 months (range, 1–23 months) following nephrectomy.
Statistical Analysis
Kaplan-Meier survival method revealed a significant association between overall survival and tumor-associated survival with the following variables (P < .05): mitotic index (MI), nuclear size, nuclear pleomorphism, tumor differentiation, Fuhrman nuclear grade, clear cell morphology, vascular invasion, age <6 years at diagnosis, invasiveness, and the presenting clinical signs of cachexia and hematuria (Suppl. Table S2). Risk factors significantly affecting overall and tumor-specific survival using Cox proportional hazards revealed similar statistically significant variables (P < .05): MI, nuclear size, tumor differentiation, clear cell morphology, Fuhrman nuclear grade, and the presenting clinical signs of cachexia and hematuria (Suppl. Table S3). Using multivariate Cox stepwise regression analysis to elucidate potential combined predictor variables, MI alone was identified as the most significant predictor of risk (HR, 12.24; 95% CI, 5.149–29.123; P < .0001).
Prognostic Value of Mitotic Index
Analyzing the mitotic indices using locally weighted scatterplot smoothing (LOWESS) regression displaying the relationship between mitotic index and survival revealed differences in slope for cases with MIs less than 10 and greater than 30 (Suppl. Fig. S1). Cases were split into 3 mitotic groups as follows: <10, 10 to 30, and >30. The MI was <10 in 36 tumors and >30 in 20 tumors. Intermediate MIs between 10 and 30 occurred in 14 tumors. Statistically significant differences between mitotic groups and overall and tumor-specific survival were observed using Kaplan-Meier estimates analysis (P < .001). Dogs with an MI of >30 had a decreased MST of 187 days (95% CI, 57–352), as demonstrated in Fig. 7, and were associated with an increased HR of 12.245 for experiencing death or euthanasia (P < .001; 95% CI, 5.149–29.123). Dogs with an MI of <10 had a significantly increased MST of 1184 days (95% CI, 807–2567). Dogs with intermediate MIs between and including 10 and 30 had an MST of 452 days and an increased HR of 2.651 for experiencing death or euthanasia (P = .037; 95% CI, 1.060–6.631).
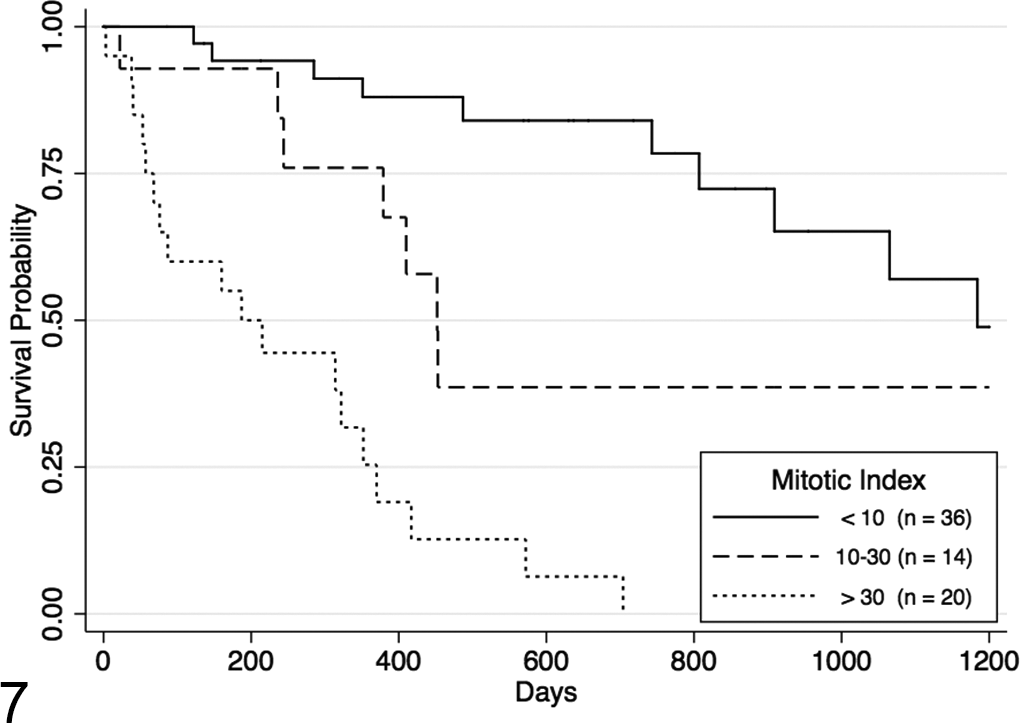
Kaplan-Meier survival curve for dogs in the 3 years following nephrectomy for renal cell carcinoma. The dogs are grouped according to mitotic index, which stratifies dogs based on overall survival (log rank, P < .0001). Censor: lost to follow-up or alive at the conclusion of study.
Clear Cell RCC
Tumors composed of extensive areas of carcinoma cells arranged in a solid growth pattern with abundant, vacuolated cytoplasm were observed in 6 dogs (9%). These tumors contained an extensive network of small-caliber blood vessels (Fig. 5), and each displayed strong, intracytoplasmic immunoreactivity for vimentin and were negative for CD117. Dogs with these tumor types had a significantly decreased MST of 87 days (P < .001; 95% CI, 3–410), as depicted in Fig. 8. Dogs with clear cell carcinoma had an increased HR of 6.463 for experiencing death or euthanasia (P < .001; 95% CI, 2.546–16.408). Of the 6 dogs with clear cell carcinomas, 3 were reported to have metastatic disease.
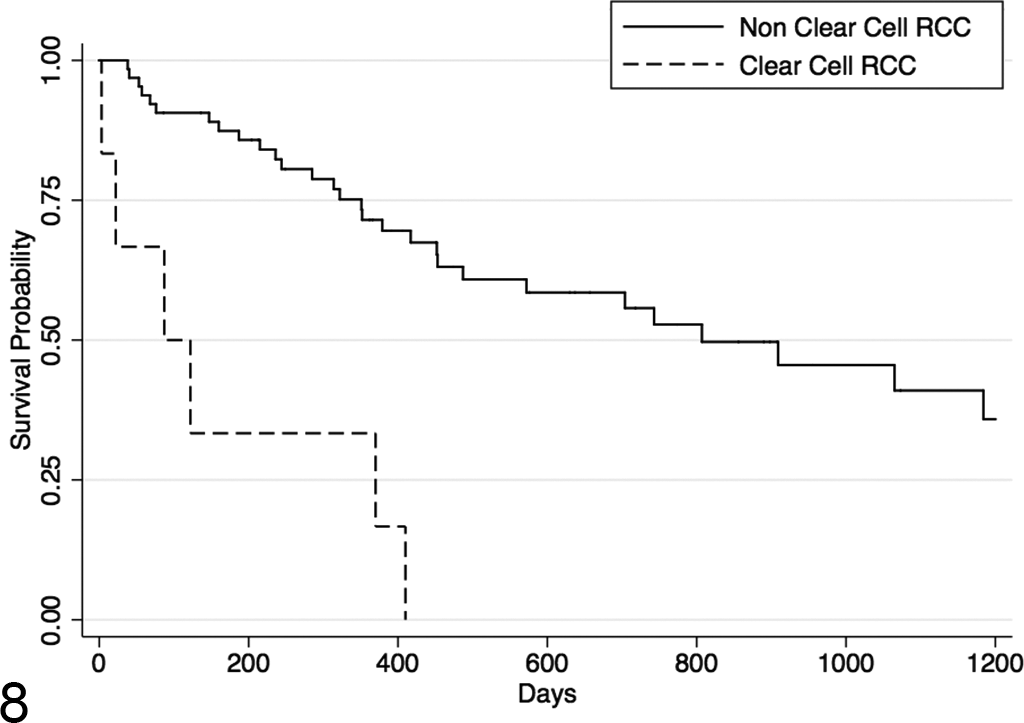
Kaplan-Meier survival curve for dogs in the 3 years following nephrectomy for renal cell carcinoma (RCC) based on overall survival. The clear cell carcinoma subtype is related to poorer survival (log rank, P < .0001). Censor: lost to follow-up or alive at conclusion of study.
Sarcomatoid Differentiation
Foci of sarcomatoid differentiation were observed in 5 of 70 tumors (7%). Sarcomatoid change was observed in solid (n = 3) and tubular (n = 2) tumors. Each of these tumors was positive for vimentin in areas of sarcomatoid differentiation and diffusely throughout the neoplasm. Significant differences in survival between sarcomatoid and nonsarcomatoid tumors were not observed (P = .148).
Multilocular Cystic RCC
Multilocular cystic tumors occurred in 3 dogs in this study (4%). These morphologically unique tumors contained multiple, variably sized cystic spaces separated by fibrous septa lined by cuboidal epithelium (Fig. 4). The 3 dogs with multilocular cystic RCC had prolonged survival times, but these findings were not statistically significant (P = .481); 1 case was lost to follow-up at 451 days, and the other 2 died of causes unrelated to RCC at 1065 and 1571 days following diagnosis.
Chromophobe RCC
Solid tumors composed of polygonal cells with distinct cell borders and abundant, lightly eosinophilic cytoplasm that stained positively for acid mucopolysaccharides with colloidal iron stain (Fig. 6) were diagnosed in 6 RCCs in this study (9%). Each of these tumors also were positive for intracytoplasmic CD117 protein expression. Survival analysis in these cases may have been obscured by the fact that each of the 6 dogs diagnosed with chromophobe RCC was lost to follow-up or still alive at the conclusion of data collection.
Additional Histologic Features
The Fuhrman 12 nuclear grade accounts for morphologic features of nuclei and nucleoli and is the most widely accepted human RCC histologic grading system. When applied to canine neoplasms, the Fuhrman nuclear grade was significantly correlated with outcome (P < .001). Five dogs had grade 4 tumors, which was associated with an MST of 87 days (95% CI, 3–160). Grade 3 tumors occurred in 31 dogs and was associated with an MST 379 days (95% CI, 244–572), and 28 dogs had grade 2 tumors associated with an MST of 1065 days (95% CI, 743–1571). Grade 1 tumors occurred in 6 dogs; the MST for this group of dogs could not be calculated due to censorship of patients still alive or lost to follow-up at 136, 362, 451, 718, and 955 days and due to non–tumor-associated death in 1 dog at 3703 days. Consistent with previous reports, tumor pattern (solid, papillary, tubular, cystic, multilocular cystic) was not significantly correlated with outcome (P = .085). Mixed histologic patterns were present in 29 cases (41%). Solid tumors occurred in 24 dogs (34%) with a male predominance of 1.7:1. Tubular tumors occurred in 17 cases (24%) and, unlike other histologic subtypes, had an increased number of female dogs (n = 9) in comparison to males (n = 8). Papillary tumors occurred in 15 cases (21%) with a male predominance of 1.5:1. Vascular invasion was observed in 4 tumors (6%) and was associated with a statistically significant decreased MST (P = .039).
Adjunct Therapy
In addition to surgery, treatment with chemotherapy was given to 8 dogs as follows: 3 dogs were treated with carboplatin, 1 with doxorubicin, 1 with cyclophosphamide, 1 with cisplatin, 1 with palladia, and 1 with mitoxantrone, intracavitary carboplatin, and palladia. The 3 dogs receiving carboplatin were also treated with toceranib phosphate (Palladia). In 45 of the cases, no chemotherapy or adjunct therapy was administered. In the remaining 17 dogs, there was no record of treatment with chemotherapy. No statistical difference (P = .132) in survival was observed between dogs treated and not treated with chemotherapy.
Discussion
The nephron is populated by a variety of epithelial cells that can give rise to distinct neoplastic entities, separable by careful histologic evaluation. There is a relative paucity of literature on canine RCC, particularly studies comparing histologic and clinical parameters with survival. The results presented here provide prognostic information based on overall and tumor-specific survival for histologic, epidemiological, and clinical parameters.
The mitotic index is an indirect proliferation measure, observable using routine diagnostic techniques, and was found to be the best predictor of survival for dogs with RCC in this study. Although several histologic parameters were significantly associated with overall and tumor-specific survival in univariate analysis, only mitotic activity was revealed to have independent prognostic value using multivariate analysis. Grouping dogs with RCC according to MI provides valuable information for clinicians since there are significant differences in median survival times for the 3 mitotic groups proposed. Also, using mitotic count alone minimizes variables that allow for intraobserver variation among diagnostic pathologists. In addition to revealing the prognostic significance of the MI in RCC, this study also provides the relative incidence and associated differences in survival for clear cell, chromophobe, papillary, and multilocular cystic variants of canine RCC; of these, recognition of clear cell morphology has the most significant clinical ramifications.
Due to the retrospective nature of this study, limitations to the data and statistical analysis were present. Metastatic disease detection was not performed consistently or in comparable intervals with one exception: radiographs preceding surgery were available for 44 dogs, 5 of which had evidence of metastatic disease. For this reason, conclusions about biologic behavior derived from metastatic information are limited. Size of the tumor, which is an important factor for stage of disease and outcome in humans, was available for only 21 cases in this study, making analysis of survival differences based on tumor size incomplete. Last, follow-up time was initiated at time of nephrectomy, not necessarily at the time the tumor was first appreciated or diagnosed. Inevitably, some amount of variation in survival analysis will exist between dogs with RCC due to differing intervals between tumor initiation, diagnosis, and nephrectomy; this likely had some effect on statistical analysis of survival.
Histologic grading is the most significant prognostic factor following stage of disease in human RCC. The Fuhrman nuclear grade, 12 first described in 1982, is the most widely used grading method to this day and is recommended for all subtypes of human RCC. 23 The Fuhrman grade is 4-tiered and based entirely on nuclear and nucleolar size and shape. The human grading scheme had similar success in predicting outcome for the dogs in our study, which corroborates the notion that human and canine renal cell carcinoma are similar.
RCC in humans is recognized to be a group of carcinomas with distinct morphologic features arising through assortments of genetic aberrations generally unique to each tumor type. 9,23 The most common human RCC morphologic-genetic subtypes are clear cell, papillary (types 1 and 2), chromophobe, collecting duct, Xp11 translocation carcinomas, and mucinous tubular and spindle cell carcinoma. 10 It is possible, given the morphologic and epidemiological similarities of human and canine RCCs, that orthologs of these human subtypes occur in dogs and that similar cytogenetic lesions are involved in the tumorigenesis for both species. In fact, this was shown to be the case in 2 similar inherited syndromes of humans and dogs, both of which are characterized by multiple renal tumors and benign skin tumors. The gene responsible for Birt-Hogg-Dubé syndrome in humans was shown to be responsible for the phenotypically similar renal cystadenocarcinoma and nodular dermatofibrosis syndrome in dogs. 2,22,30 Interestingly, the dog locus for this disorder was found prior to the human locus primarily due to the relatively narrow genetic variability in canine breeds (German Shepherds in this case), the large sizes of canine families, and the power of the breeding structure, all of which facilitate linkage mapping. 32
Clear cell RCCs arise from epithelial cells of the proximal convoluted tubules 10 and comprise a majority of human cases. In contrast, canine clear cell RCCs were observed in only 9% of cases (vs 77% of human cases 16 ). In most human clear cell carcinomas, there is somatic inactivation of VHL that may occur by allelic deletion, mutation, or epigenetic silencing. VHL protein normally inhibits hypoxia-inducible genes and functions as a tumor suppressor. These alterations in VHL are not commonly encountered in other RCC subtypes. 10 Exon mutation status in canine VHL was previously investigated in RCCs by polymerase chain reaction (PCR) and sequencing of 13 paraffin-embedded tumor samples, and no mutation was identified. 33 This may reflect the relative rareness of the clear cell variant in dogs in comparison to humans and suggests other pathways were likely involved in the canine RCCs analyzed by Pressler et al. 33 The dogs that were diagnosed with clear cell RCC in our study had significantly decreased survival times; clear cell carcinomas in humans also have decreased survival times in comparison to papillary and chromophobe subtypes, and the human clear cell variant more commonly produces metastatic disease. 10,16
Chromophobe RCCs arise from the intercalated cells of the collecting duct 23 and occur in approximately 5% of humans with renal cell carcinoma. 16,20 Human chromophobe RCC is associated with a better prognosis in comparison to clear cell and papillary subtypes. 16 Chromophobe carcinomas were observed in 9% of the canine RCCs in this study, but no significant differences in survival were observed compared with all other tumors. Prolonged survival in these cases may have been obscured by the fact that each of the 6 dogs diagnosed with chromophobe RCC was lost to follow-up or still alive at the conclusion of data collection. Chromosomal loss is the genetic hallmark of human chromophobe RCC with loss of heterozygosity and hypodiploidy of multiple chromosomes. 10 Diffuse cytoplasmic positivity for colloidal iron histochemical staining is a common diagnostic marker for human chromophobe carcinoma 25 and was successfully used for canine classification in this study and in a previous report of canine chromophobe RCC. 19 CD117 expression was present in all canine chromophobe RCCs, which is common in human chromophobe RCC (78% of cases) and less so in papillary, clear cell, and collecting duct carcinomas. 36
Papillary RCC has a human incidence of approximately 13% 16 and was observed in 21% of the dogs in this study. Trisomy or tetrasomy 7, trisomy 17, and loss of chromosome Y are the most common karyotypic changes in human papillary RCC. 10 Mutations in the proto-oncogene Met are known to cause the heritable form of human papillary RCC, 34 and chromosome 7, which contains MET, is duplicated in approximately 75% of spontaneous papillary RCC in humans. 8 The MET proto-oncogene encodes a receptor tyrosine kinase that binds hepatocyte growth factor and mediates cell migration, survival, and cycling. 4 A germline point mutation has been described in canine MET that alters the juxtamembrane domain of MET and increases migration of affected cells. 21 This canine germline mutation suggests that certain breeds harbor this activating MET mutation and that it may contribute to tumorigenesis in diseases such as RCC.
A rare form of RCC in humans, denoted multilocular cystic renal clear cell carcinoma (MCRCC), is recognized by the World Health Organization as a tumor composed entirely of cystic structures, the septa of which are lined by cells indistinguishable from grade I clear cell carcinoma cells; 9 no tumor with these features has been reported to recur or metastasize in humans. 35 In the 3 cases of multilocular cystic tumors observed in this study, the features were similar to the human counterpart. Canine multilocular cystic tumors are similar to MCRCC in that they appear to have a favorable clinical course, with no detection of metastatic disease and long survival times. Of the 3 dogs with multilocular cystic RCC in our study, 1 was lost to follow-up and the 2 others died of causes unrelated to RCC at more than 2 years following diagnosis.
Sarcomatoid change previously has been described in canine RCC 13,19 and was observed in 7% of the tumors in this study. In human RCC, sarcomatoid differentiation occurs in approximately 5% of cases and can occur in any histologic subtype. 6,25 There is evidence that sarcomatoid change in human disease is the result of epithelial to mesenchymal transformation 3 and is associated with an increased rate of metastasis and poor outcome in humans. 6,25 A statistically significant difference in survival for dogs was not observed in our study, but areas of sarcomatoid differentiation displayed positivity for vimentin, which is consistent with the epithelial to mesenchymal transition.
Pulmonary metastases have been reported in as many as 48% of dogs with RCC at the time of diagnosis in an early study of canine renal carcinoma. 17 Our finding of pulmonary metastases in 11% at diagnosis is comparable to the 18% reported in the most recent publication reporting the clinical aspects of canine RCC; 5 this lower percentage is likely more accurate and can be attributed to dogs living in an era of increased clinical detection of disease.
Renal cell carcinomas are remarkably resistant to chemotherapy, and development of drug resistance is common in humans. 11,29 Expression of the multidrug-resistant transporter in tumor cells may be the mechanism by which drug resistance occurs in RCCs. 8 The lack of response to chemotherapy in the RCC patient was corroborated in our canine study, where survival was not extended for dogs treated with chemotherapy compared with those not treated. In human medicine, therapy is continuously evolving, and immunotherapy, tyrosine kinase inhibitors, and monoclonal antibody therapies are currently used in the management of human RCC, including high-dose interleukin-2, vascular endothelial growth factor inhibitors, and mTOR inhibitors. 11
Conclusions
Our findings demonstrate marked differences in survival times for canine RCC that can be predicted using the mitotic index. In addition, recognition of the clear cell subtype of RCC has significant implications on survival, and a multilocular cystic variant of canine RCC is described that is associated with prolonged survival times. This account provides the incidences of canine variants of RCC, including papillary (21%), clear cell (9%), chromophobe (9%), and multilocular cystic (4%), as well as an incidence of sarcomatoid change (7%) in canine RCC.
Footnotes
Acknowledgments
The present work would not have been possible without the cooperation of the veterinarians who managed these cases. Those who contributed information on more than 3 cases are listed here: Amelia M. Simpson, DVM, DACVS; Britt Culver, DVM, DACVIM; Robyn Elmslie, DVM, DACVIM; Sarit Dhupa, DVSc, DACVS; Lorin A. Hillman, DVM, DACVIM; Linda Lew, DVM, DACVS; David Bostwick, DVM, DACVIM; Eileen Snakard, DVM, DACVS; Ken Moore, DVM, DACVS; and Wayne Davis, DVM.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Veterinary Diagnostic Laboratories at Colorado State University, particularly the histology laboratory (Todd Bass), which provided materials and methods for immunohistochemistry and histochemistry.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
