Abstract
A 15-year-old female Holstein cow was presented with a history of anorexia and weight loss. Abnormal physical examination findings included poor body condition, dehydration, icterus, and photodermatitis on nonpigmented areas of the skin of the dorsum. At necropsy, the common bile duct was greatly enlarged, with thickened walls, and tightly adhered to the hepatic capsular surface and serosa of adjacent loops of the small intestine. Two sessile, yellowish, friable, well-circumscribed, cauliflower-like, bulging masses (14 and 8 cm in diameter) were attached to the inner surface of the common bile duct blocking its lumen. Microscopically, the tumor was composed of well-differentiated, columnar epithelial cells with oval or round, vesicular, basal nuclei arranged in papillary projections. Periodic acid-Schiff-positive material accumulated in the apical cytoplasm of the neoplastic cells. Immunostaining of the tumor cells was positive for cytokeratin (CK) of wide-spectrum screening and for CK 7. The diagnosis of papillary adenocarcinoma of the extrahepatic bile duct was based on the morphologic features of the neoplasm and evidence of local invasion. To the best of our knowledge, extrahepatic bile duct carcinomas have not been described in cattle.
Keywords
Tumors arising from intrahepatic bile ducts and gallbladder have been described in several species of domestic animals. 4 In cattle, epithelial tumors of the intrahepatic biliary ducts and gallbladder have been reported as incidental findings in retrospective studies carried out in slaughterhouses. 1 , 14 Epithelial tumors of the extrahepatic biliary tree have been reported in animals on rare occasions, including bile duct carcinomas in bears, 7 sea otters, 13 cats, 2 , 12 and dogs. 3 This is in contrast to the much higher prevalence of carcinomas of the extrahepatic biliary tract reported in humans. 5 , 10 Tumors of the extrahepatic biliary tree have not been reported in cattle. This report describes the clinical, pathologic, and immunohistochemical aspects of a single case of papillary adenocarcinoma of the extrahepatic bile duct in a Holstein cow.
A 15-year-old female Holstein-Friesian cow was presented to a local university veterinary teaching hospital with a history of anorexia and weight loss. On physical examination, poor body condition, dehydration, icterus, and photo-dermatitis on the nonpigmented areas of the skin on the dorsum of the body were observed. Laboratory analyses were not done. The cow was humanely killed because of poor prognosis.
A complete necropsy was performed immediately after euthanasia. The carcass was thin. Grossly, the common bile duct was greatly enlarged and converted into a large out-pouching (30 cm in diameter) due to marked dilation of its lumen. The extrahepatic biliary tree was dissected and carefully scrutinized to find out the site of origin of the neoplasm. The common bile duct was tightly adhered to the ventral surface of the liver and serosa of the adjacent loops of the small intestine. The extrahepatic duct was markedly distended with an orange liquid (exudate and bile) mixed with necrotic debris. The common bile duct was greatly thickened, and its internal surface was shriveled and had dark gray discoloration. Two small, separate openings arising from the neoplasm within the common bile duct established new, patent communications with the adjacent proximal duodenum. One of the openings measured 5 cm in diameter, whereas the other had 2.5 cm in diameter. The outlet of the larger abnormal communication between the neoplastic extrahepatic duct and the small intestine had yellow, raised edges. No gross changes were observed in the normal aperture of the common bile duct into the proximal duodenum. Two yellowish, friable, well-circumscribed, cauliflower-like, bulging masses measuring approximately 14 and 8 cm in diameter were attached to the inner surface of the common bile duct blocking its lumen (Figs. 1, 2). The walls of the other extrahepatic ducts were markedly thickened, dilated, and filled with inspissated, discolored orange bile. The gallbladder was greatly enlarged, measuring 25 cm in diameter, had thickened walls, and was distended with thick and lumpy bile. The liver was firm and diffusely orange with arborizing dilated and distorted biliary ducts throughout the capsular surface. On cut section, the larger intrahepatic bile ducts were thickened because of fibrous tissue proliferation and ectatic and discolored orange because of bile accumulation. In addition, jaundice was observed in the mucous membranes, internal organs, intima of large vessels, and fasciae of muscles. No significant gross lesions were found in the pancreas or other organs.

Common bile duct; cow. Papillary adenocarcinoma. A friable well-circumscribed, cauliflower-like, bulging mass is attached to the inner surface of the common bile duct blocking its lumen. The wall of the common bile duct (A) is markedly thickened and shriveled. Bar = 2.3 cm.
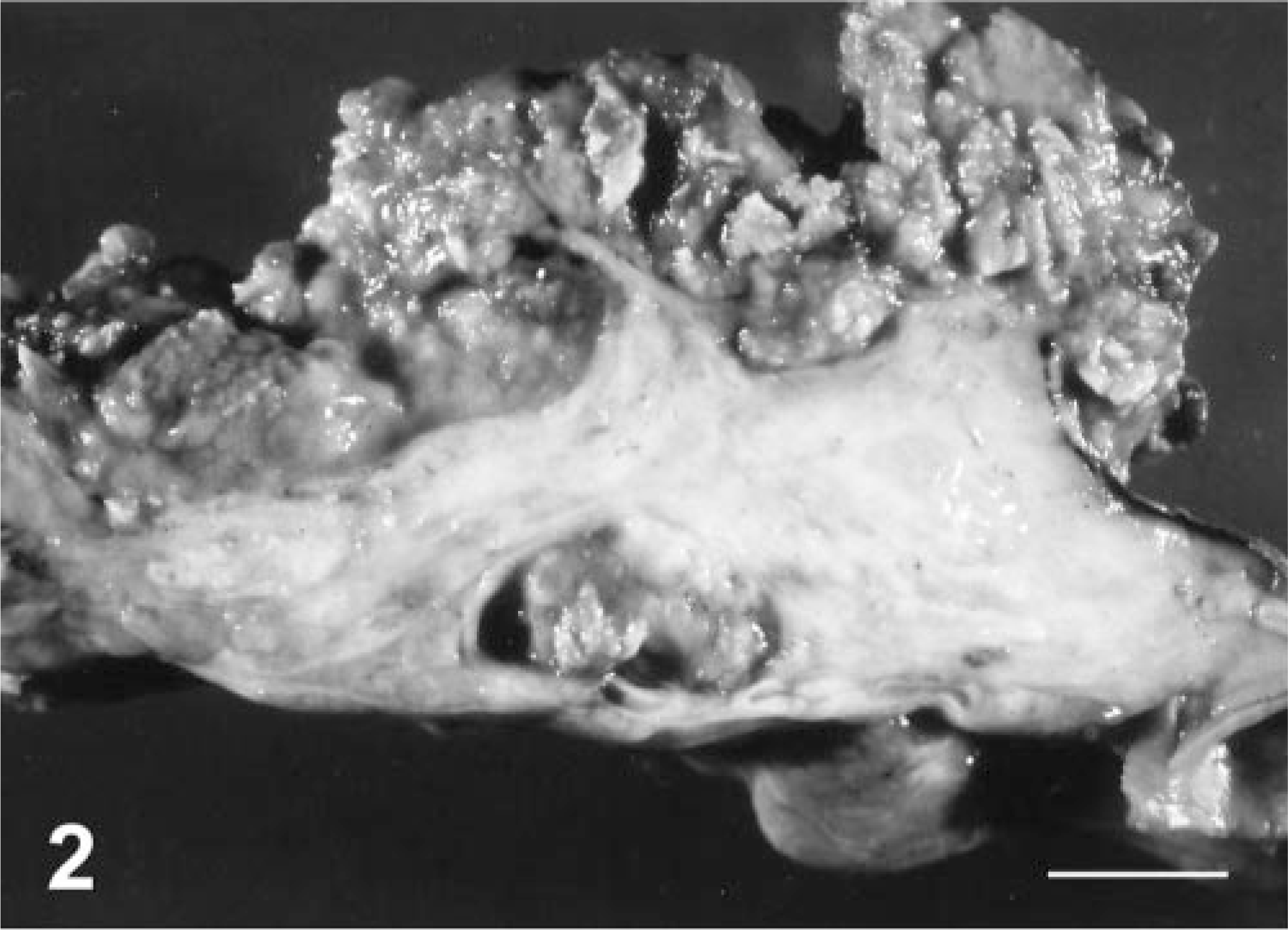
Common bile duct; cow. Papillary adenocarcinoma. Cut surface of the mass showed on Fig. 1. The tumor has a wide base with a scirrhous core that is surrounded by friable neoplastic tissue. Bar = 1.8 cm.
Samples of the extrahepatic bile duct neoplasm, the central nervous system (CNS), and different tissues and organs, including the liver, hepatic lymph nodes, and pancreas, were collected during necropsy, fixed in 10% buffered formalin, and submitted to routine processing methods. Histologic sections of the neoplasm were also stained with periodic acid–Schiff (PAS). In addition, routine immunohistochemistry was performed, with positive and negative controls. Monoclonal antibodies used against cytokeratins (CKs) were AE1/AE3 (CKs 1–8, 10, 14–16, 19, dilution 1 : 100) and OVTL (clone OVTL12/30, Dako, CK 7, dilution 1 : 50).
Microscopically, the extrahepatic bile duct tumor was composed of well-differentiated epithelial cells, with columnar shape, weakly eosinophilic, slightly granular cytoplasm, and oval to round, vesicular, basal nuclei, with multiple nucleoli. Mitotic rate ranged from 0 to 2 per high-power field. Tumor cells were arranged in numerous irregular, papillary projections formed by a single or double layer of epithelial cells assuming a pseudostratified pattern. The neoplastic outgrowth had a wide base with a scirrhous core. Larger papillary projections branched gradually into smaller ones (Fig. 3). Exophytic growths were interspersed with scarce fibrovascular stroma. Neoplastic cells were also arranged in a glandular or pseudoacinar pattern. Pseudoacinar structures were filled with necrotic and cellular debris admixed with an eosinophilic material. PAS-positive material accumulated in the apical cytoplasm of the tumor cells and inside the lumen of the pseudoacini (Fig. 4). Mixed inflammatory infiltrates were found scattered throughout the stroma. Multiple areas of coagulative necrosis in the neoplasm were also observed. The external surface of the neoplastic mass was ulcerated and covered by necrotic debris, bacteria, and eosinophilic PAS-positive material.
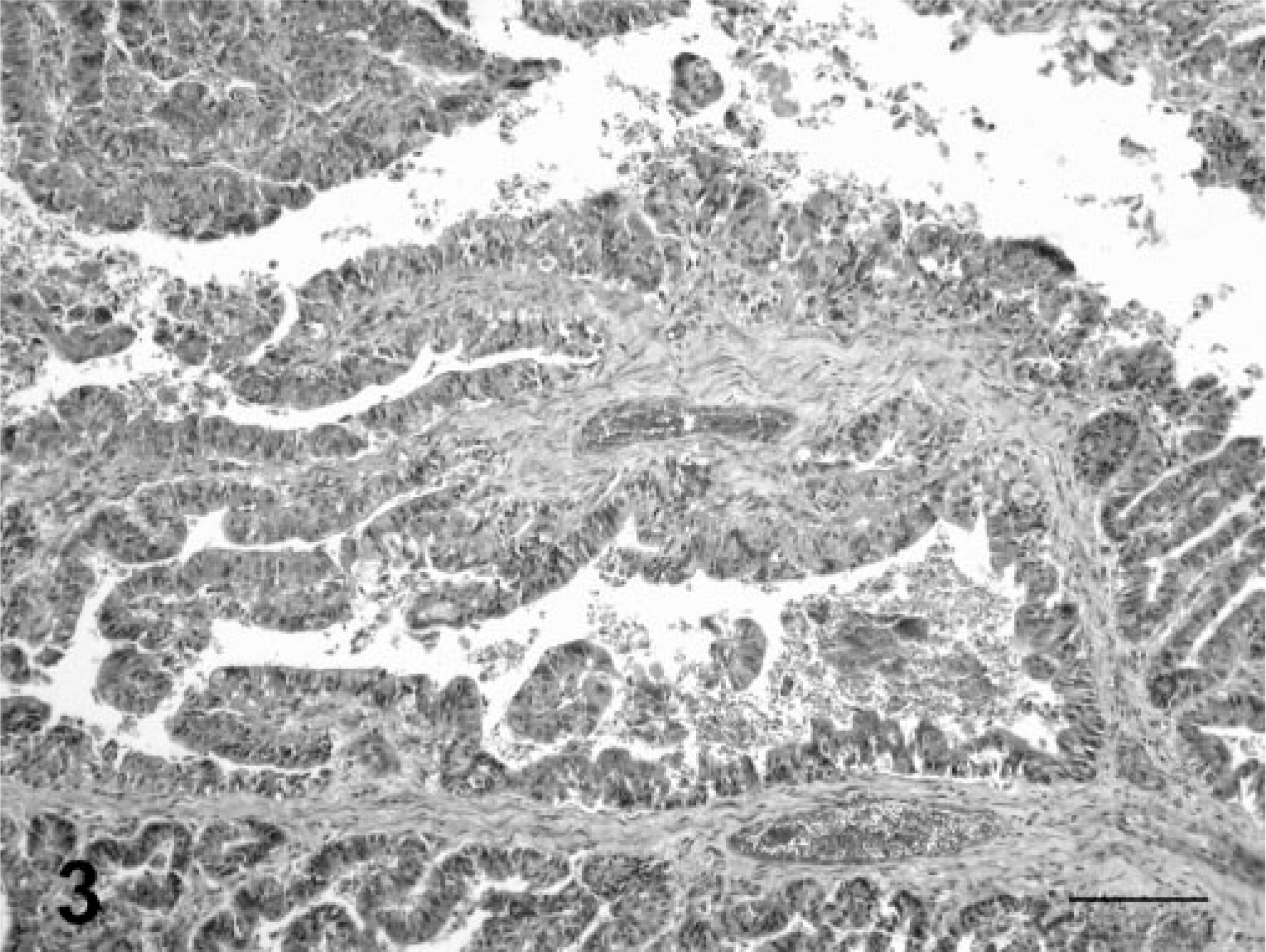
Common bile duct; cow. Papillary adenocarcinoma. The tumor cells are arranged in numerous irregular, papillary projections. Larger papillary projections branch gradually into smaller ones. Exophytic growths are interspersed with sparse fibrovascular stroma. HE. Bar = 330 µm.
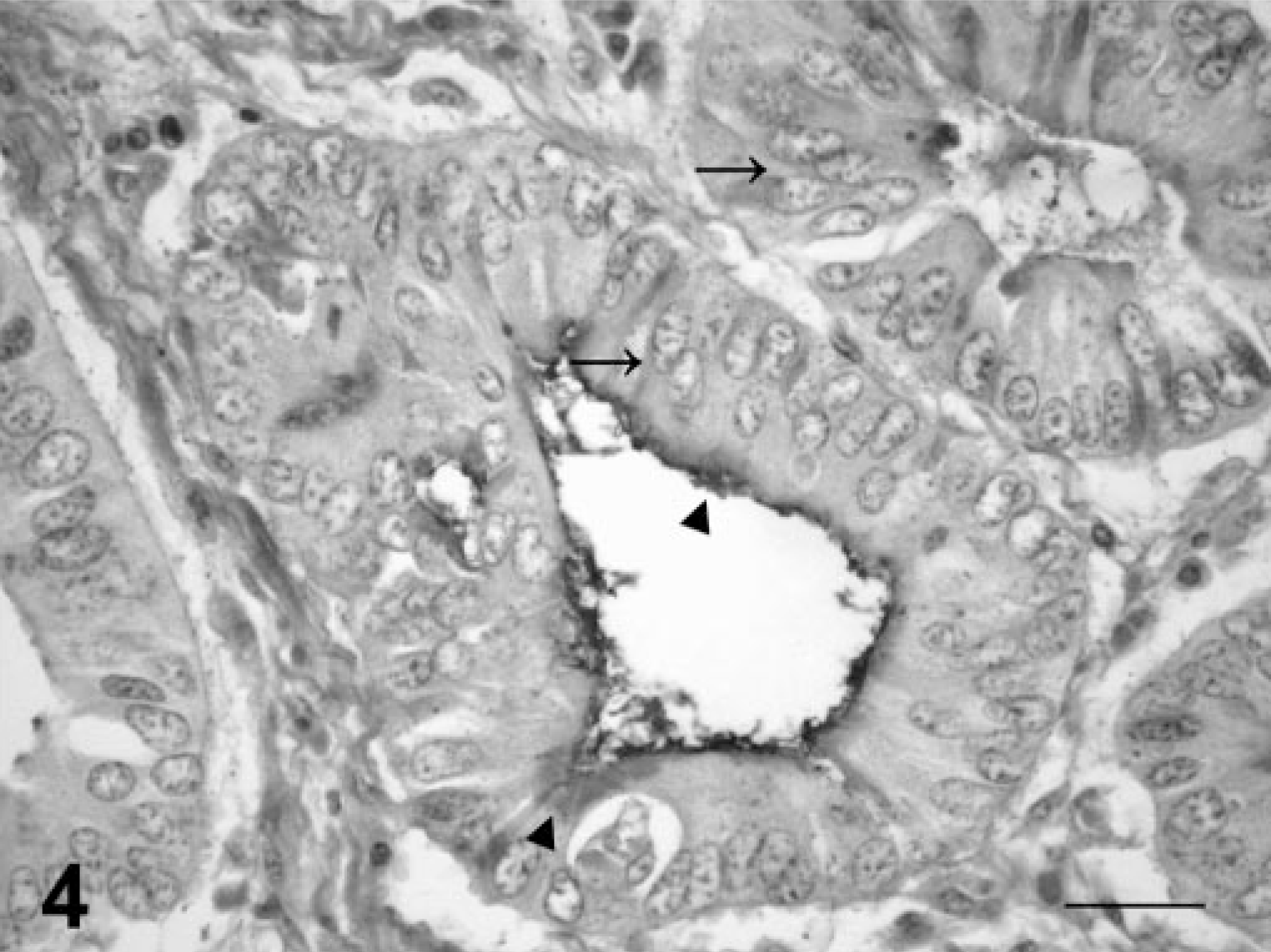
Common bile duct; cow. Papillary adenocarcinoma. The tumor is composed of well-differentiated epithelial cells, with columnar shape, slightly granular cytoplasm, and oval to round, vesicular nuclei, with multiple nucleoli. Epithelial tumor cells are arranged either in a single layer with basal nuclei or in a pseudostratified pattern (arrows). PAS-positive material accumulates in the apical cytoplasm of the tumor cells (arrowheads). PAS. Bar = 55 µm.
Histologic examination of the raised edges from the outlet of the new, patent communication established between the neoplastic common bile duct and the proximal duodenum revealed similar neoplastic epithelial cells infiltrating the muscular and submucosa tunicae of the small intestine and extending into the mucosal layer establishing an alternative outlet. Metastases were not observed in the hepatic lymph nodes or other organs.
Additional histologic findings included severe bile ductular proliferation and fibrosis associated with mononuclear cell infiltration accumulated in the portal triads. Intrahepatic cholestasis and hepatocellular fatty degeneration were also observed. No histologic changes were seen in the pancreas, CNS, and other organs.
Immunostaining of tumor cells was positive for CK of wide-spectrum screening and for CK 7.
In this report, the diagnosis of papillary adenocarcinoma of the extrahepatic bile duct was based on the necropsy findings and histopathology. Close examination and identification of the anatomic structures involved by the neoplastic mass in which the common bile duct was entangled played a decisive role in determining the exact origin of the tumor. In humans, the identification of the tissue of origin for tumors arising in and around the biliary tract is particularly problematic because of the intricate anatomy of the region. 5 In humans, the common bile duct and the pancreatic duct lie in close proximity to each other, whereas in cattle the outlet of the common bile duct into the lumen of the proximal duodenum is 30 cm apart from the sphincter of the pancreatic duct. 8 The histologic similarity between pancreatic carcinomas and carcinomas arising from the extrahepatic biliary tract in humans may raise additional histologic diagnostic problems. 5 Similarly, immunohistochemistry is not of much help in these cases 5 because many markers expressed by bile duct carcinomas, such as CK 7, 5 CK 19, 11 and CK 20, 5 glucose transporter protein glut-1, 16 and carcinoembryonic antigen 11 are also expressed by pancreatic epithelial tumors. 5 , 6 , 9 , 15 In this study, the reaction of neoplastic cells to a panel of polyclonal and monoclonal human CKs (CK 7) did not rule out the possibility of a pancreatic tumor. However, localization of the neoplasm affecting the walls of the common bile duct and lack of involvement of pancreatic tissue confirm the diagnosis of extrahepatic bile duct neoplasm.
Several risk factors have been suggested for the occurrence of extrahepatic biliary carcinoma neoplasms in humans, including cystic dilatation of bile ducts, chronic exposure to the radiologic contrast agent thorotrast, smoking, primary sclerosing cholangitis, liver flukes, chronic intraductal gallstones, bile duct adenomas, and biliary papillomatosis. 10 In bears, genetic predisposition and diet changes have been incriminated in the development of extrahepatic bile duct carcinomas. 7 However, there is no evidence that any of these risk factors are involved in the pathogenesis of the neoplasm described here. Carcinomas of the extrahepatic bile ducts in humans usually develop in patients more than 65 years old. 10 Advanced age may have been a predisposing factor in this case, although carcinomas of the gallbladder and intrahepatic cholangiocarcinomas have been described as an incidental finding in abattoirs in younger cattle. 1
In the present case, gross and microscopic changes in the liver and associated signs of icterus and photodermatitis can be attributed to partial to complete obstruction of normal outflow of the bile through the biliary system. Obstructive icterus has also been described in bears, 7 humans, 10 and cats 2 , 12 affected by carcinomas of extrahepatic biliary ducts. Longstanding, extrahepatic cholestasis can cause progressive dilation of bile ducts with accompanying biliary hyperplasia in the portal areas and hepatocellular injury due to retention of bile constituents. Bile may accumulate inside distended biliary canaliculi or inside ectatic bile ducts. The leakage of bile into portal areas induces an inflammatory reaction and fibrosis that can progress to extensive scarring of the portal triads (biliary fibrosis). 4
The multifocal nature of the neoplasm could represent local spread or multicentric neoplasia. No distant metastases were identified, but cytologic features and local invasion are indicative of malignancy.
Footnotes
Acknowledgements
We thank Mr. João Francisco Nunes (Faculty of Veterinary Medicine, Universidade Federal de Pelotas, Pelotas, RS, Brazil) for preparation of the histologic sections and Dr. David Driemeier (Faculty of Veterinary Medicine, Universidade Federal do Rio Grande do Sul, Porto Alegre, RS, Brazil) for technical assistance in the photomicrographs.
