Abstract
A retrospective study was performed on primary cardiac tumors found in 44 cattle during meat inspection in Japan, using histology, immunohistochemistry, and electron microscopy. The age of affected cattle ranged from 10 to 129 months; 82% were less than 3 years old. In 38 hearts, the tumor was solitary; the remaining 6 hearts had multiple masses. All tumors were subendocardial and involved the papillary muscles and/or cardiac valves. Two histologic patterns were recognized; both included giant cells. Pattern 1 was characterized by interlacing fascicles of spindle-shaped cells; pattern 2 resembled cavernous hemangioma. The 2 patterns coexisted and were linked by transitional zones. Given the immunohistochemical reactivity and ultrastructure, the spindle-shaped cells were identified as smooth muscle cells and the giant cells as endothelial cells. Single cell and multicellular angiogenesis by giant cells was observed. The tumors were classified as cardiac angioleiomyoma. Subendocardial multipotential mesenchymal cells, persistent tissue of the endocardial cushion, or valvular interstitial cells were suspected as the origin.
A wider range of primary cardiac tumors has been reported in cattle than in any other animal. Examples include rhabdomyoma, 20,21,29 rhabdomyosarcoma, 19,24,29,34 peripheral nerve sheath tumors (neurofibroma, schwannoma, neurinoma, neurofibroma), 11,18,20,22 –24,26,29,31 hemangioma, 22,29,37 hemangiosarcoma, 6,29 fibroma, 18 fibrosarcoma, 18,29,33 angiolipoma, 8 leiomyoma, 29,33 leiomyosarcoma, 29 adenomatoid tumor, 35 hamartoma, 29 mesothelioma, 29,32 and myxoma. 3,20
In Japan, 53 primary cardiac tumors have been identified during routine meat inspection of slaughtered cattle since 1982. Of these 53 tumors, 1 group, in 44 animals, stood out because of its common features: the tumor was mainly in young cattle and occurred in subendocardial tissue of cardiac valves and papillary muscles but not within the myocardium. Metastasis was not encountered. In the past, these tumors have been diagnosed as schwannoma (neurofibroma), hemangioma, hemangiosarcoma, or rhabdomyosarcoma.
To clarify the true character of these tumors, we retrospectively evaluated the 44 cases histologically, immunohistochemically, and ultrastructurally. Based on common features, in particular the cytologic appearance of the neoplastic cells and the pattern of neoplastic tissue, we reclassified all 44 tumors as cardiac angioleiomyoma.
Methods
Tissue Samples
Formalin-fixed tumors from 51 cattle (1982–2009) were evaluated along with accompanying photographs and inspection reports by meat hygiene centers throughout Japan. Forty-four tumors were chosen for further study because of common consistent gross and histologic findings: subendocardial location in valves and over papillary muscles (but not in the myocardium) and composition of spindle-shaped cells and giant cells. The prevalence of this tumor averaged 0.00113% (21 of 1,855,544 slaughtered cattle) in 4 centers that tallied the number of cattle slaughtered. The frequency of occurrence and the distribution of the cardiac tumors were confirmed from records and photographs provided by each meat inspection center.
Case profiles of the 44 cattle are in Table 1. Age, recorded for 33 animals, ranged from 10 to 129 months, average, 37.1 months; 27 of the 33 (82%) were younger than 3 years. Sex distribution was 25 castrated males, 16 females, and 3 unknown. The breed distribution was 23 Holstein-Friesian, 6 Japanese Black, and 12 crossbreds (Holstein-Friesian/Japanese Black). Breed was not designated for 3 animals. Almost all animals were clinically healthy. The cardiac tumors had been diagnosed as rhabdomyosarcoma, hemangiosarcoma, hemangioma, or schwannoma (neurofibroma). The 7 tumors that were excluded from this study included 1 epicardial leiomyoma, 1 multicentric myocardial angioleiomyoma, 3 leiomyosarcomas, 1 atrial rhabdomyoma, and 1 diffuse subendocardial neurofibroma.
Signalment, Gross Features, and Original Diagnosis in 44 Cases of Bovine Cardiac Angioleiomyoma
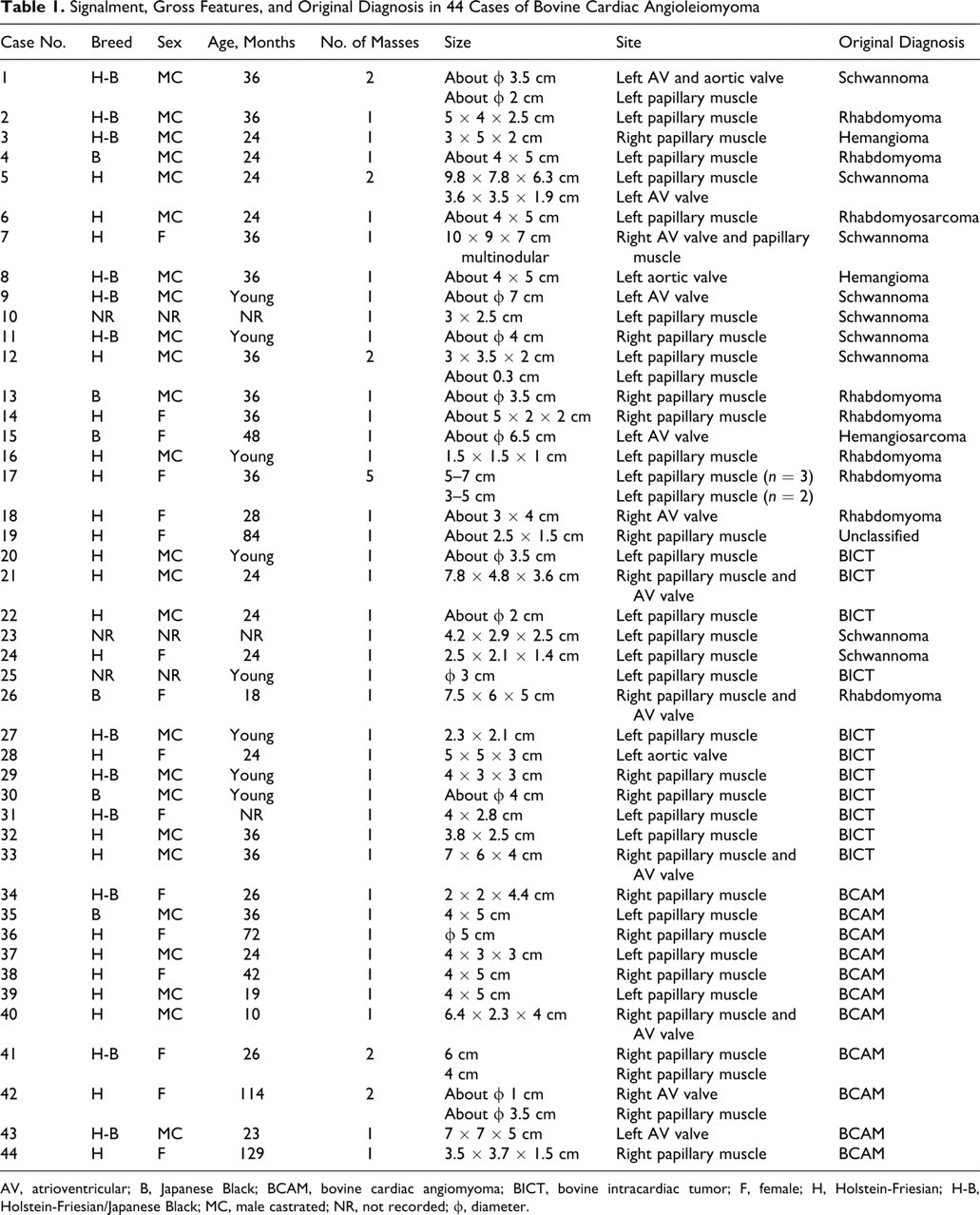
AV, atrioventricular; B, Japanese Black; BCAM, bovine cardiac angiomyoma; BICT, bovine intracardiac tumor; F, female; H, Holstein-Friesian; H-B, Holstein-Friesian/Japanese Black; MC, male castrated; NR, not recorded; ϕ, diameter.
Histologic Examination
All formalin-fixed tissue specimens were processed routinely, embedded in paraffin, and stained with hematoxylin and eosin (HE). Selected sections were also stained with periodic acid–Schiff (PAS), Alcian blue (pH, 2.5), Watanabe’s silver impregnation (for reticulin fibers), phosphotungstic acid hematoxylin (PTAH), and Azan-Mallory stain.
Immunohistochemistry
Immunohistochemistry was performed by conventional methods with diaminobenzidine as chromogen and hematoxylin as counterstain. Antibodies against cytokeratin, vimentin, desmin, S-100 protein, pan-actin, smooth muscle actin, type IV collagen, proliferating cell nuclear antigen (PCNA), Ki67, and factor VIII–related antigen were used. The profiles of primary antibodies and pretreatments for antigen retrieval are listed in Table 2 . The immunoreactants were visualized by Histofine Stravigen BSA-PO kit (Nichirei, Japan).
Details of Immunohistochemical Reagents
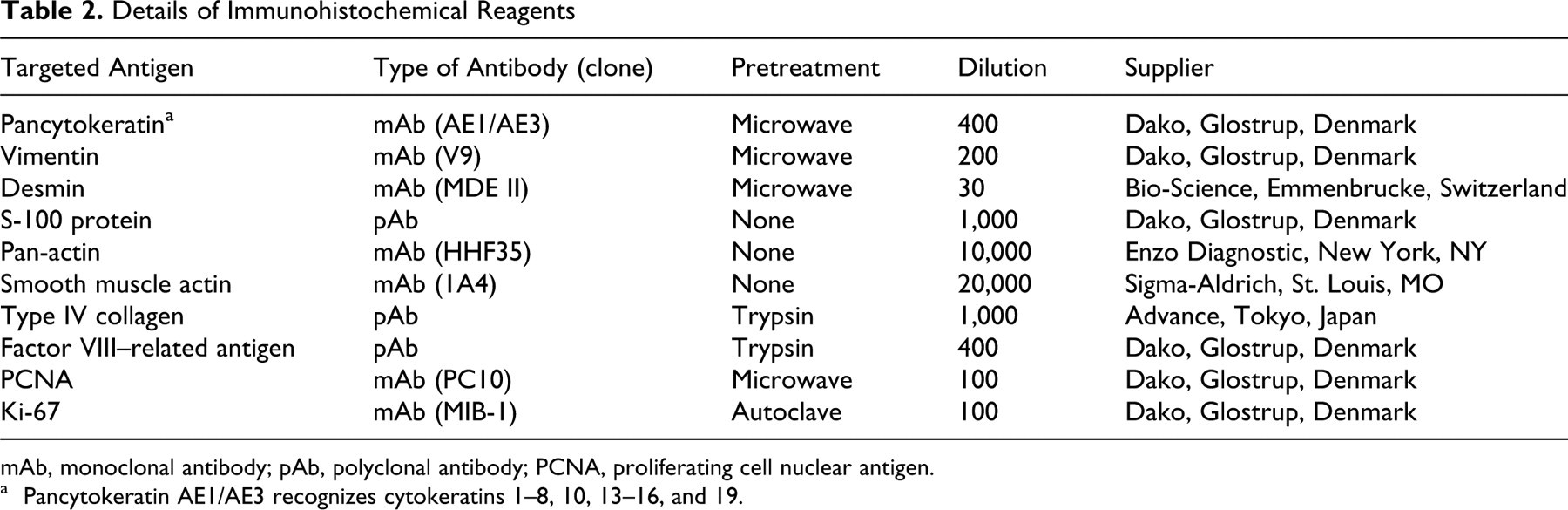
mAb, monoclonal antibody; pAb, polyclonal antibody; PCNA, proliferating cell nuclear antigen.
a Pancytokeratin AE1/AE3 recognizes cytokeratins 1–8, 10, 13–16, and 19.
Tissue from the hearts of bovine fetuses and from soft tissue tumors of cattle (leiomyoma, leiomyosarcoma, rhabdomyosarcoma, hemangiosarcoma, and peripheral nerve sheath tumor) were prepared as positive controls. For negative controls, the primary antibody was omitted.
Ultrastructural Examination
For transmission electron microscopy, 11 formalin-fixed tumors were postfixed in 1% osmium tetroxide in 0.1 M cacodylate buffer (pH 7.2). The tissues were dehydrated in graded ethanols, cleared with propylene oxide, and embedded in Epon 812. One-micrometer sections were stained with toluidine blue. Ultrathin sections from selected areas were contrasted with uranyl acetate and lead citrate and examined with a Hitachi transmission electron microscope (H300, Tokyo, Japan).
Results
Morphologic Features
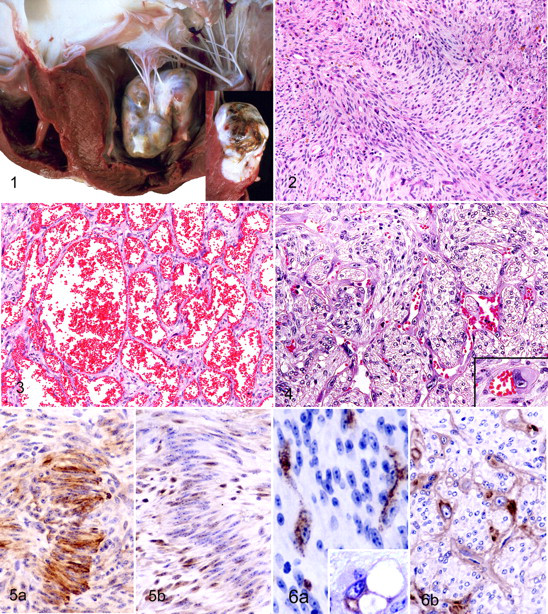
Of the 44 cardiac tumors, 38 were solitary and 6 were composed of multiple masses. Tumor distribution is in Table 1. Nineteen tumors were in the right ventricle and 25 in the left ventricle (Fig. 1); none involved both ventricles. Metastasis was not documented in any case. All tumors were in subendocardial tissue and projected into the ventricular lumen. The overlying endocardium was smooth, without thrombosis. The position of the tumors was highly characteristic: all involved the valve complex (valves, chordae tendineae, or papillary muscles). Thirty-eight masses involved the papillary muscles (in particular the tip of the papillary muscles), 7 were on the cardiac valves (5 on the atrioventricular valves and 2 on the semilunar valves), and another 8 large masses involved both the valves and papillary muscles. Their diameter ranged from 2 mm to about 10 cm. The cut surface of tumors was white or red, or both. The border with myocardium was distinct (Fig. 1).
Histologic Features
There were 2 main histologic patterns, both of which included giant cells (Figs. 2 and 3). The relative proportion of the 2 patterns varied among cases. Pattern 1, predominant in many cases, was characterized by prominent proliferation of spindle-shaped cells arranged in interlacing fascicles that intersected at 90° angles in a herringbone pattern (Fig. 2). The cytoplasm of the spindle-shaped cells was weakly to strongly eosinophilic, fibrillar, and red with Azan-Mallory stain; some cells contained PTAH-positive fibrillar material. Focally, pattern 1 included palisading arrangements and myxomatous areas. Pleomorphic giant cells with eosinophilic cytoplasm surrounded by a PAS-positive basement membrane-like material were scattered or clustered in pattern 1 areas. The cytoplasmic processes of giant cells formed tubular structures with a slit-like or broad lumen. The tubes bifurcated and anastomosed to form networks or vascular channels (Fig. 4). Many giant cells had single or multiple cytoplasmic vacuoles that contained erythrocytes.
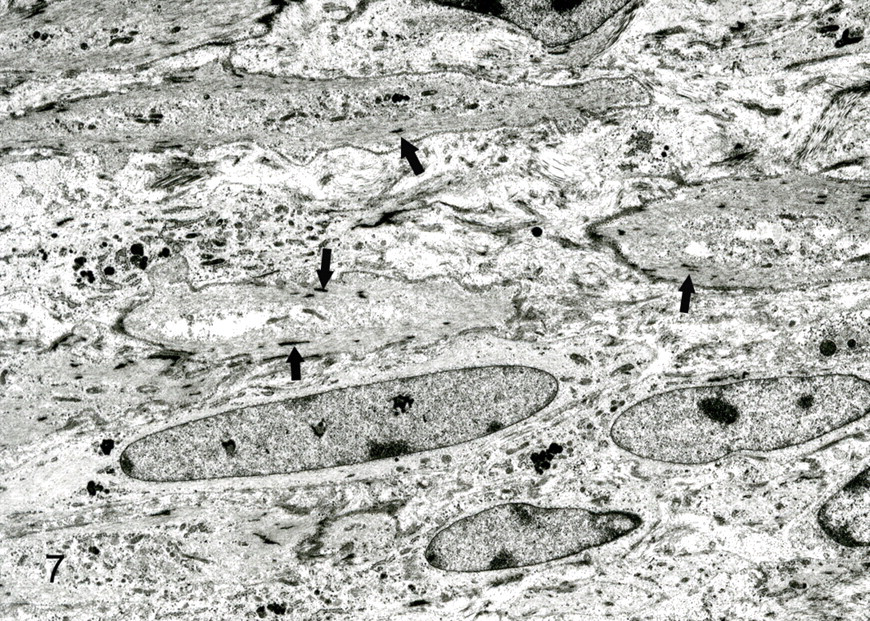
Bovine heart; No. 30. Spindle-shaped cells have an oblong nucleus and intermediate filaments with small focal densities (arrows). Transmission electron micrograph.
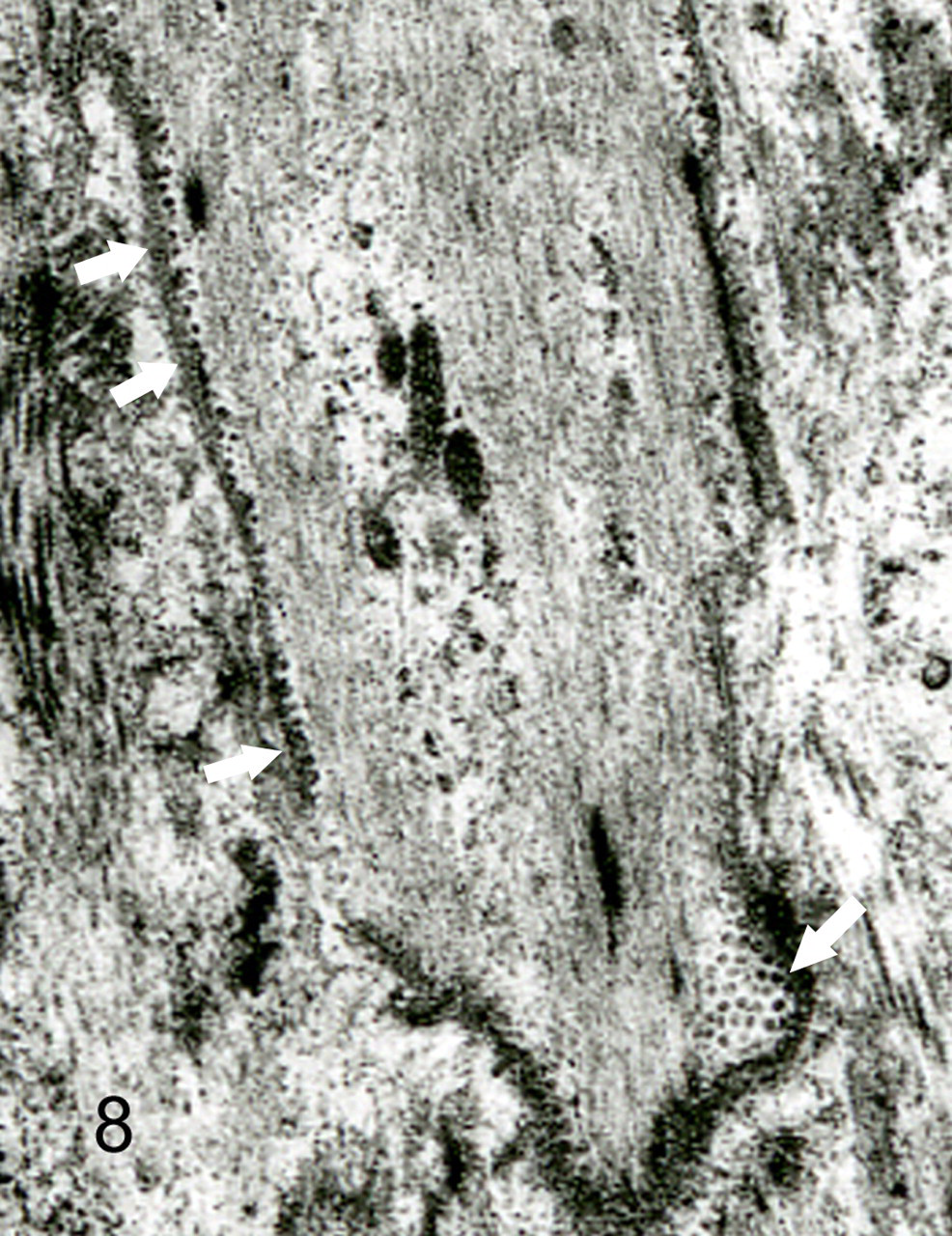
Bovine heart; No. 30. Spindle-shaped cell has intermediate filaments with small focal densities and many pinocytotic vesicles (arrows). Transmission electron micrograph.
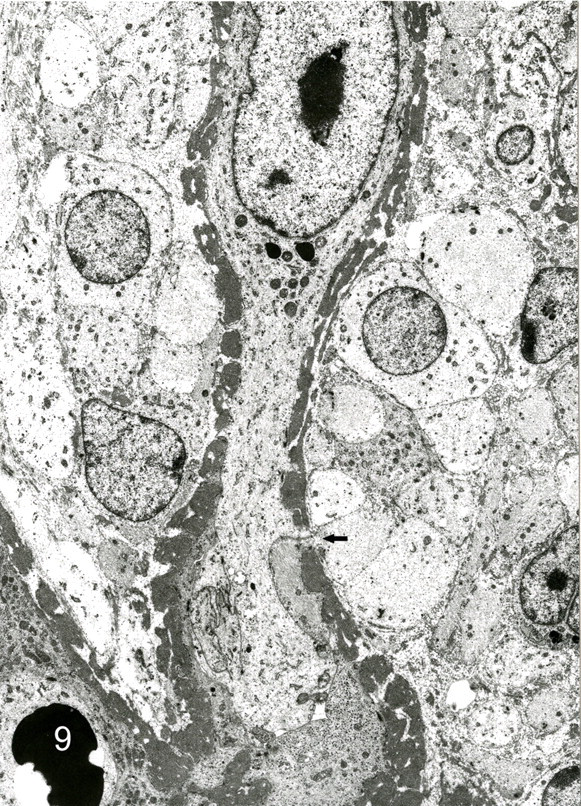
Bovine heart; No.30. Vascular channels formed by giant cells. Basal lamina is thick and discontinuous (arrow). Spindle-shaped cells surround the channels. Transmission electron micrograph.
Pattern 2 had numerous uniformly sized vascular spaces like those of a cavernous hemangioma (Fig. 3). Many vascular spaces were lined by flattened endothelial cells or hypertrophied endothelial cells with large, bizarre, and hyperchromatic nuclei. Patterns 1 and 2 were combined in many tumors with no border between patterns. Spindle-shaped cells proliferated between vascular spaces. Smooth muscle bundles were noted in the stroma. Mitotic figures were rare and observed in only a few cases. There was no vascular or capsular invasion by tumor cells. Necrosis was negligible or absent. No tumors were seen anywhere but in the heart.
Immunohistochemical Examination
Immunohistochemically, most of the spindle-shaped cells, the major component of pattern 1, expressed pan-actin and smooth muscle actin (Fig. 5a) but not factor VIII-related antigen. The spindle-shaped cells between vascular spaces in pattern 2 had the same immunohistochemical reactivity. Immunoreactivity for desmin was detected in smooth muscle bundles but not in other elements. The spindle-shaped cells in both pattern 1 and pattern 2 and the giant cells had diffuse and strong immunoreactivity for vimentin. Scattered reactivity for S-100 protein was observed in the nucleus and cytoplasm of spindle-shaped cells (Fig. 5b) in both patterns, but the intensity was weaker than that of the schwannoma control. The cells in myxomatous areas had positive immunoreactivity for S-100 protein, similar to that in cells of the cardiac trigonum fibrosum.
Giant cells in both patterns expressed factor VIII–related antigen in their cytoplasm (Fig. 6a) without positive reaction for any myogenic markers and were enveloped by type IV collagen-positive, basement membrane-like material (Fig. 6b). Immunoreactivity for cytokeratins was absent in all tumors. The immunoreactivity of neoplastic cells for PCNA and Ki67 was the same as that of mesenchymal cells in peritumoral cardiac tissue.
Mesenchymal cells in the cardiac trigonum fibrosum and subendocardial tissue of the normal bovine adult and fetal heart (positive control) expressed S-100 protein and smooth muscle actin. All primary antibodies recognized appropriate antigens in normal control tissues and all tumor tissues evaluated.
Ultrastructural Features
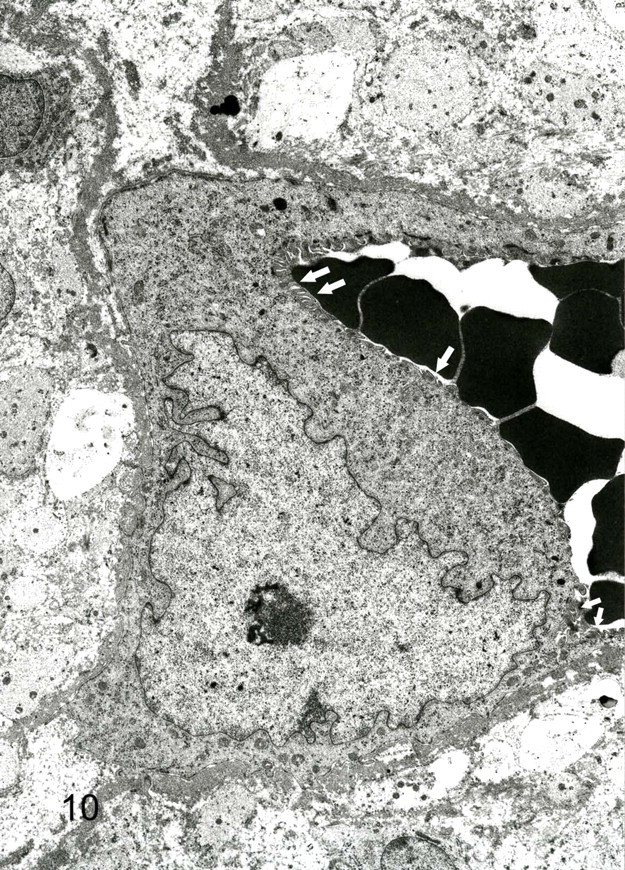
The predominant spindle-shaped cells of pattern 1 had an oval to oblong nucleus, various quantity of filaments with small focal densities, and a few organelles and pinocytotic vesicles and were surrounded by a delicate and discontinuous basal lamina (Fig. 8). Intercellular collagen fibers were not observed. Giant cells around vascular channels had cytoplasmic interdigitations, pinocytotic vesicles, and dense attachments to the basal lamina. The basement membrane of vascular channels varied in thickness from 0.3 to 1.9 μm (Fig. 9). Abrupt thinning and disruption of the basement membrane were associated with insertion of cytoplasmic processes by the giant cells. Polyptychial arrays of degraded basement membrane were scattered along the length of more mature vascular channels (Fig. 10). The spindle cells were always outside the basement membrane of the vascular channels. Giant cells had intracytoplasmic lumens with few microvilli, intermediate filaments, and basal lamina. In cavernous hemangioma-like areas (pattern 2), the flattened cells that lined vascular spaces had a few microvilli, marginal folds, and cytoplasmic interdigitations. Hypertrophied lining cells had numerous microvilli and organelles.
Bovine heart; No. 30. Vascular channel with erythrocytes. Microvilli (arrows) project from the luminal surface of the giant cell. Transmission electron micrograph.
Discussion
Although these 44 primary bovine cardiac tumors had been variably classified, we concluded that all were the same type of tumor because of consistent and characteristic pathologic features. The location, and the histologic, immunohistochemical, and ultrastructural features of the spindle-shaped neoplastic cells were consistent with those of smooth muscle cells. 9,10,39 The giant cells expressed factor VIII–related antigen, a marker of endothelial cells, and had ultrastructural features of endothelial cells. 10,11 Giant cells with vacuoles are also seen in human epithelioid hemangioendothelioma, 40 in some hemangiosarcomas, 30 and in the early stage of angiogenesis in chicken embryos, where they are said to represent single-cell angiogenesis. 7 In the present study, many of the giant cell vacuoles contained erythrocytes. Where pleomorphic giant cells were clustered, networks of cellular processes formed slit-like lumens or broader spaces that contained erythrocytes. A similar feature is seen in neovascularization of granulation tissue 36 and in angiogenesis in human budding-type hemangiosarcoma. 28 Considering these findings, we identified the giant cells as endothelial cells.
Continuity was evident between the vascular spaces of pattern 1 and the more cavernous spaces in pattern 2. In both patterns, the immunohistochemical and ultrastructural features of the giant cells were almost identical. Therefore, we concluded that the single-cell and multiple-cell angiogenesis of pattern 1 progressed to the cavernous hemangiomatous state of pattern 2. We propose that these tumors are derived from proliferation of smooth muscle cells and endothelial cells of blood vessels.
These bovine cardiac angioleiomyomas are distinct from the cardiac schwannomas reported in cattle 11,18,20,22 –24,26,29,31 and rats. 25 The spindle cells of bovine cardiac angioleiomyoma had limited expression of S-100 protein with concurrent expression of actin and smooth muscle actin, reactions that are not expected in schwannoma. In addition, the ultrastructural characteristics of the neoplastic cells are consistent with those of smooth muscle and distinct from those of Schwann cells. The more difficult problem is to distinguish whether the tumors are angiogenic or leiomyogenic.
The World Health Organization Histological Classification of Tumors of Domestic Animals includes the Kaposi-like vascular tumor, 12 which has been reported in a dog 30 and a cow. 27 Human Kaposi’s sarcoma is believed to be derived from capillary endothelial cells. 41 In contrast, cytologic features of the tumors in the present study are consistent with origin from 2 types (smooth muscle and endothelial) of cell and, thus, are distinct from Kaposi’s sarcoma.
Human soft tissue tumors with proliferation of smooth muscle cells and angiogenesis include angiomyomas (also called vascular leiomyomas or angioleiomyomas). These tumors are said to be derived from vascular smooth muscle, are characterized by the proliferation of smooth muscle cells, and are rich in blood vessels. 38 In animals, angiomyomas have been reported in dogs and cats 1,4,13,17 and, as in the present study, have proliferation of both smooth muscle cells and blood vessels. However, the present tumors contain smooth muscle cells in various stages of differentiation, from desmin-negative immature cells to mature cells with all muscle markers including desmin and forming muscle bundles. In addition, angiogenesis ranges from single to multiple cells. This variable differentiation of the myogenic and angiogenic components of the bovine cardiac tumors has not been described in reported cases of angioleiomyoma.
Bovine cardiac angioleiomyoma shares some features with hamartoma, but angioleiomyomas in this study were classified as neoplastic, based on the immaturity of most components, whereas in hamartomas, aberrant differentiation results in a mass of disorganized but mature specialized cells. 14
The angioleiomyomas of the present study are distinct from those reported in other species and may be unique to the bovine heart. These bovine cardiac angioleiomyomas were considered benign because of the well-demarcated growth without invasion of vessels or the tumor capsule, absent or negligible necrosis, absence of metastasis, and low mitotic (or proliferation) index.
The spindle-shaped cells in the cardiac tumors expressed smooth muscle actin and S-100 protein, as did those in valvular stroma and subendocardium of the fetal and adult bovine hearts used as immunohistochemical control tissues. The neoplastic cells resembled the interstitial cells that constitute 30% of the valvular membrane and are thought to contribute to valvular contractions. 5 Considering the age of affected cattle and the location of the tumors, they may be derived, like human cardiac myxoma, 2,15,16 from subendocardial pluripotential mesenchymal cells, with the capability to differentiate into smooth muscle cells and form vascular structures or into valvular interstitial cells. In this series, cardiac angioleiomyoma affected mainly young cattle; breed predilection was not apparent.
Although bovine cardiac tumors have been variably classified, some reported cases of fibrosarcoma 33 or rhabdomyosarcoma 24 resemble the angioleiomyomas of this report. Therefore, angioleiomyoma should be included in the differential diagnosis for bovine cardiac tumors. The subendocardial location of these spindle cell tumors, the immunohistochemical expression of smooth muscle actin and S-100 protein, and the formation of vascular structures by giant cells are consistent findings that facilitate diagnosis.
Footnotes
Acknowledgements
This study was carried out with help from the National Meat Inspection Office of Japan. We thank also the members of the Meat Inspection Laboratory, Gunma Prefecture, and Mr. Kitada Hanjiro of Osaka City Meat Inspection Office, who provided useful material and information.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
