Abstract
Bovine ocular squamous cell carcinoma (OSCC) is the second most common cause of rejection due to neoplasia in slaughterhouses on Sao Miguel Island, Azores, and accounts for significant economic losses. To obtain a better insight into the genesis and neoplastic transformation process of bovine OSCC, abnormal protein expression and proliferation index were assessed by the immunohistochemical evaluation of p53 and Ki67, respectively. OSCC samples were collected from 15 bovines and were classified histologically according to the degree of differentiation into three categories: poorly, moderately, and well differentiated. Immunohis-tochemistry using polyclonal anti-human p53 antibody and polyclonal anti-human Ki67 antibody was performed. Ten of 15 tumors tested were immunoreactive for p53. Twelve tumors demonstrated Ki67 expression. As in human squamous cell carcinoma, p53 overexpression is frequent in bovine OSCC, providing support for a possible role of the protein in the pathogenesis of this neoplasia. No correlation between the percentage of p53 stained nuclei and the degree of differentiation was observed, although different patterns of staining were seen according to the degree of keratinization of the tumor cells. With the exception of the moderately differentiated OSCC group, Ki67 index showed significant correlation with the histologic pattern, increased proliferation being found in poorly differentiated OSCC (P = 0.013).
Ocular squamous cell carcinoma (OSCC) is a general designation for a group of primary neoplasms of keratinocytes emerging from ocular tissues, especially the eyelids and particularly the third eyelid. In cattle, OSCC has been diagnosed all over the world with high incidence. It is the most common bovine tumor and the one that causes the most significant economic losses. 6 , 13 , 15 , 33
In Portugal, the frequency of this tumor is particularly high in the Azores. On Sao Miguel Island, large numbers of cattle affected with OSCC are rejected for consumption at slaughter, and OSCC is the second most frequent neoplasia after urinary bladder tumors, representing 21% of all cases of rejection due to neoplasia. 25 , 26
Histologically, OSCC may range from well-differentiated to undifferentiated anaplastic carcinoma. The degree of invasiveness of these tumors is variable, and occasionally metastases occur, especially to the parotid lymph node. 8
Although a common tumor, the genesis of bovine OSCC has not been fully investigated, and several theories have been advanced to explain its development. 14 Nevertheless, there is ample evidence that ultraviolet (UV) radiation is the major epidemiologic risk factor for the development of squamous cell carcinoma of the skin in humans 10 and animals. 9 , 11 In cattle, OSCC prevalence is directly related to exposure to UV light and is most commonly found in unpigmented areas of the ocular region 2 , 17 because melanin plays a photo-protective role in epidermal and mucosal surfaces. 16 , 18 , 24 , 27 , 32 Prolonged exposure to sunlight is in fact believed to be the most important factor in the genesis of bovine OSCC in Sao Miguel, an island with very rich pastures where cattle are bred for milk production and animals stay in pasture year-round with the benefit of few shelters.
As with other carcinogens, UV radiation causes mutation in oncogenes and tumor-suppressor genes, and specific p53 mutations have been detected in UV light–induced cutaneous squamous cell carcinomas. 5 , 23 , 31 This gene functions in the control of the cell cycle, DNA repair, and apoptotic pathway, 28 giving rise to a nuclear phosphoprotein that serves as a negative regulator of cell proliferation. 5 , 7 , 23 Mutation of the p53 gene, resulting in loss of normal function or acquisition of abnormal functions of its protein product, may thus contribute to unrestricted cellular proliferation by several mechanisms. 7 , 23 In most cases of p53 gene mutation, the translated protein has an altered conformation and accumulates in the nuclei of tumor cells in amounts detectable by immunohistochemistry. 31 Despite its importance, there are in fact very few reports on the detection of p53 specifically on bovine OSCC. 30 , 31
In normal skin, differentiation and proliferation are tightly coupled to maintain normal architecture in a continually renewing tissue. 4 Yet, temporal and spatial relationships between these two processes are disrupted in neoplastic skin, with gradual loss of tumor cell competence to differentiate, accompanied by increased cell proliferation. 4 , 19 Some authors refer to a reversal of the quantitative ratio of proliferation to differentiation in human skin neoplasms, in comparison with normal skin, that correlates with the degree of histologic differentiation, 4 with significantly higher Ki67 index values in poorly than in well-differentiated squamous cell carcinomas. 19 Ki67 antigen is a nuclear protein expressed during all active stages of the cell cycle (G1, S, G2, and M phases) but is absent in resting, noncycling cells (G0 phase) and is an excellent marker for determining the growth fraction or proliferation index of a given cell population. 3 , 28 , 29
Given the influence of p53 mutations in the balance between cell growth and death (apoptosis), controlling not only cell numbers but also cell differentiation in neoplastic conditions, 19 it was decided to investigate whether the degree of differentiation of bovine OSCC could be correlated with p53 overexpression and with proliferative activity of tumor cells. Hence, for this purpose, immunohistochemical techniques were used to determine the expression of p53 and Ki67 in samples of bovine OSCC showing various degrees of histologic differentiation.
Materials and Methods
Sampling and routine histopathologic diagnosis
Fifteen eye tumors were collected from an abattoir on Sao Miguel Island, Azores, from Holstein Friesian cattle aged 4–10 years (mean age 7.4 years). Tumors presented the following anatomic origins—eyelids, seven; third eyelid, five; the whole of the ocular region, three. Most animals showed lack of melanin pigment in the skin of the face and eyelashes (Fig. 1).
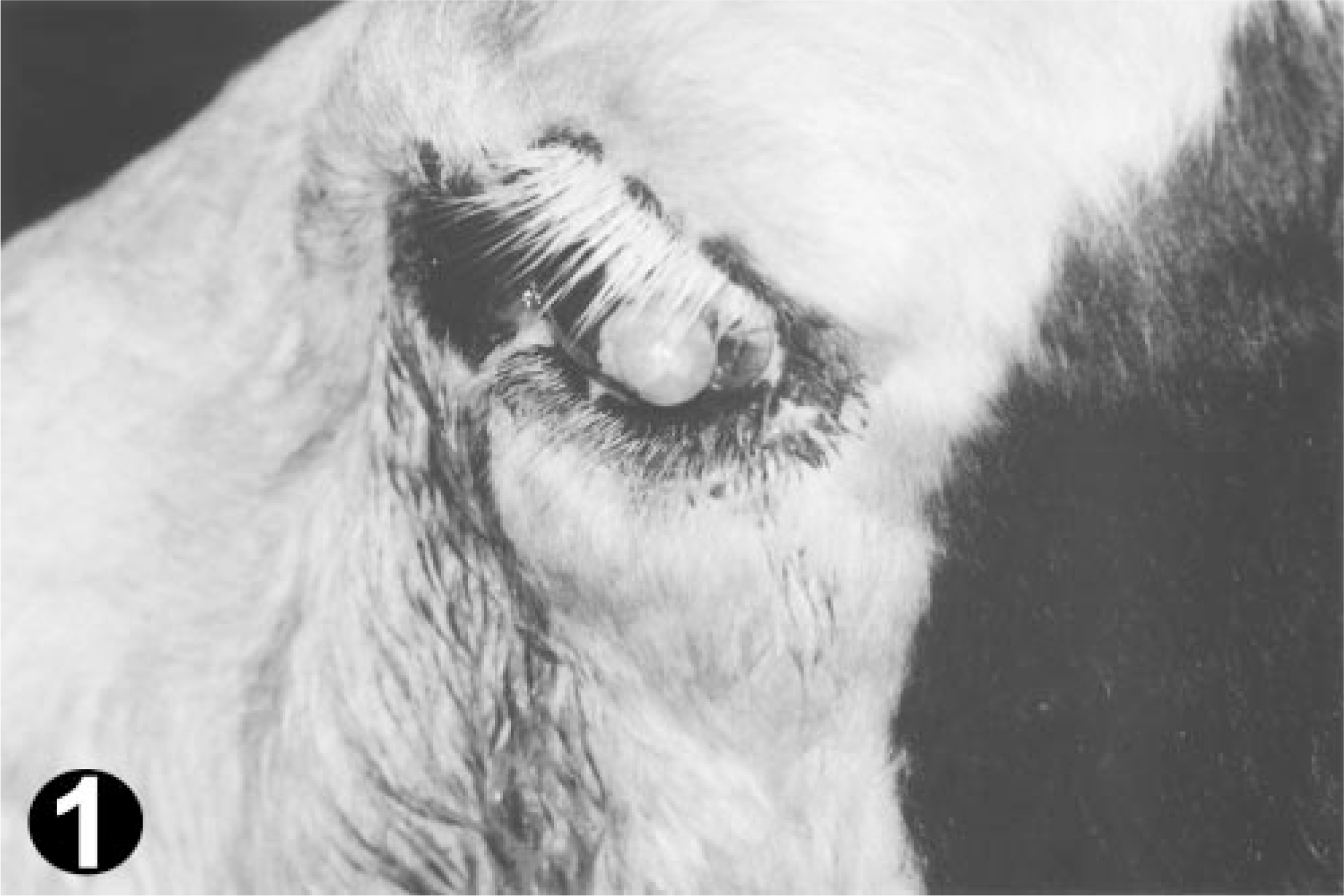
Ocular region; bovine. OSCC in a Holstein-Friesian cow with nonpigmented face and eyelashes.
Samples were fixed in 10% formalin for a maximum of 48 hours, embedded in paraffin, and 5-µm sections were stained for routine histopathologic diagnosis with hematoxylin and eosin.
All OSCC were classified with regard to macroscopic and microscopic features (Table 1). Differentiation was evaluated according to the presence and intensity of keratinization, squamous differentiation of neoplastic cells, and island formation. The maximal score (+++) was attributed to well-differentiated neoplasms containing numerous large keratin pearls and large islands formation, with obvious squamous differentiation. The intermediate score (++) referred to poorly differentiated neoplasms with a moderate degree of keratinization and differentiation, exhibiting small- to medium-sized keratin pearls, smaller islands, still with spinous differentiation but with increased number of poorly differentiated cells. The minimal score (+) referred to neoplasms with only individual cell keratinization, few small-sized islands, and poor cellular differentiation. Regarding invasiveness, the highest score (+++) corresponded to deep invasion far from the main tumor, the intermediate score (++) to neoplasms with small independent islands of neoplastic tissue surrounding the main tumor, and the lowest score (+) was assigned to neoplasms with minimal signs of invasion. Mitotic index was evaluated as the number of mitotic figures per high-power field, ranging from 0 to 2 (+), 3 to 6 (++), and > 6 (+++).
Features for histopathological classification of bovine OSCC.

Immunohistochemistry
Immunohistochemical staining of Ki67 and p53 was performed using the streptavidin–biotin–peroxidase method and two commercially available polyclonal antibodies directed against human antigens (NCL-Ki67p and NCL-p53-CM1, Novocastra Laboratories, Newcastle upon Tyne, UK). In brief, deparaffinized and rehydrated 5-µm-thick tissue sections mounted on Vectabond (Vector Laboratories, Burlin-game, CA)-coated glass slides were microwaved in 10 mM sodium citrate buffer (pH 6.0) for 15 minutes at 750 W. After cooling down in a cold water bath for 20 or more minutes, endogenous peroxidase activity was abolished with 0.5% hydrogen peroxide in methanol for 30 minutes, and nonspecific staining was blocked by preincubation with normal swine serum (1 : 5 dilution, 60 minutes). For the staining of Ki67 and p53, sections were incubated with primary antibodies, both diluted 1 : 1,000, overnight at room temperature. Sections were extensively washed in Tris-buffered saline (TBS, pH 7.4) and then incubated with a biotinylated swine anti-rabbit immunoglobulin, diluted 1:,300, for 60 minutes at room temperature. Sections were again washed before incubation with streptavidin–biotinylated horseradish peroxidase complex (Dako Corporation, Copenhagen, Denmark) for 60 minutes, and the reaction was developed with 0.075% 3,3-diaminobenzidine tetrahydrochloride (Sigma Chemicals, St. Louis, MO) and 0.02% hydrogen peroxide for 10 minutes. Finally, sections were counterstained with Mayer's hemalumen (Merck), dehydrated, and mounted in Entelan (Merck, Darmstadt, Germany).
Corresponding negative control slides, for both p53 and Ki67, were prepared by replacing primary antibody with TBS.
To test the specificity of the immunostaining, the polyclonal anti-human p53 antibody was simultaneously applied to a human transitional cell carcinoma of the bladder, known to show strong p53 immunoreactivity (kindly provided by the Portuguese Institute of Oncology Francisco Gentil, Lisbon), and to a selected sample of bovine OSCC (tumor No. 3). Strong p53 immunostaining was obtained in both tissues and the bovine OSCC sample subsequently used as a positive control.
Normal bovine lymph node was used as a positive control for Ki67. Reactive lymphoid follicles located in the perineo-plastic tissue provided an additional positive internal control.
Evaluation of immunostaining
For both p53 and Ki67, immunopositivity was indicated by the presence of distinct brown nuclear staining.
Positive cells were counted by two independent observers using an Olympus BX 51 microscope and Olympus DP-Soft software. In each tumor, positive and negative nuclei accounting for a minimum of 1,000 neoplastic cells were counted in 8–12 representative high-power fields to determine the proportion of labeled nuclei. For scoring, preference was given to areas presenting more intense and homogenous staining.
For p53, immunoreactivity scores ranged from 0 to 3 according to the percentage of stained nuclei (0% = 0; < 10% = 1; 10–50% = 2; > 50% = 3). Because staining of more than 10% of tumor nuclei has been associated with p53 mutation, 31 tumors were considered positive if they received a score of 2 or 3.
For Ki67, the proliferation index, or proportion of positive neoplastic cells, was calculated in every tumor. Statistical comparison of the proliferation indexes between the three different groups of differentiation, well, moderately, and poorly differentiated, was performed using the Student's t-test. A probability of error (P) of < 0.05 was considered significant.
Results
Histopathologic diagnosis
All tumors sampled were diagnosed as OSCC. Their distribution in the abovedescribed categories was as follows: seven well differentiated, four moderately differentiated, and four poorly differentiated (Table 2, Figs. 2–4).
Histopathological characterization and p53 and Ki67 immunostaining in Bovine OSCC.

∗ + + + = maximum; ++ = intermediate; + = minimum (for more details see Table 1).
† p53 score: 0% = 0; <10% = 1; 10-50% = 2; >50% = 3.
‡ ND= not determined.
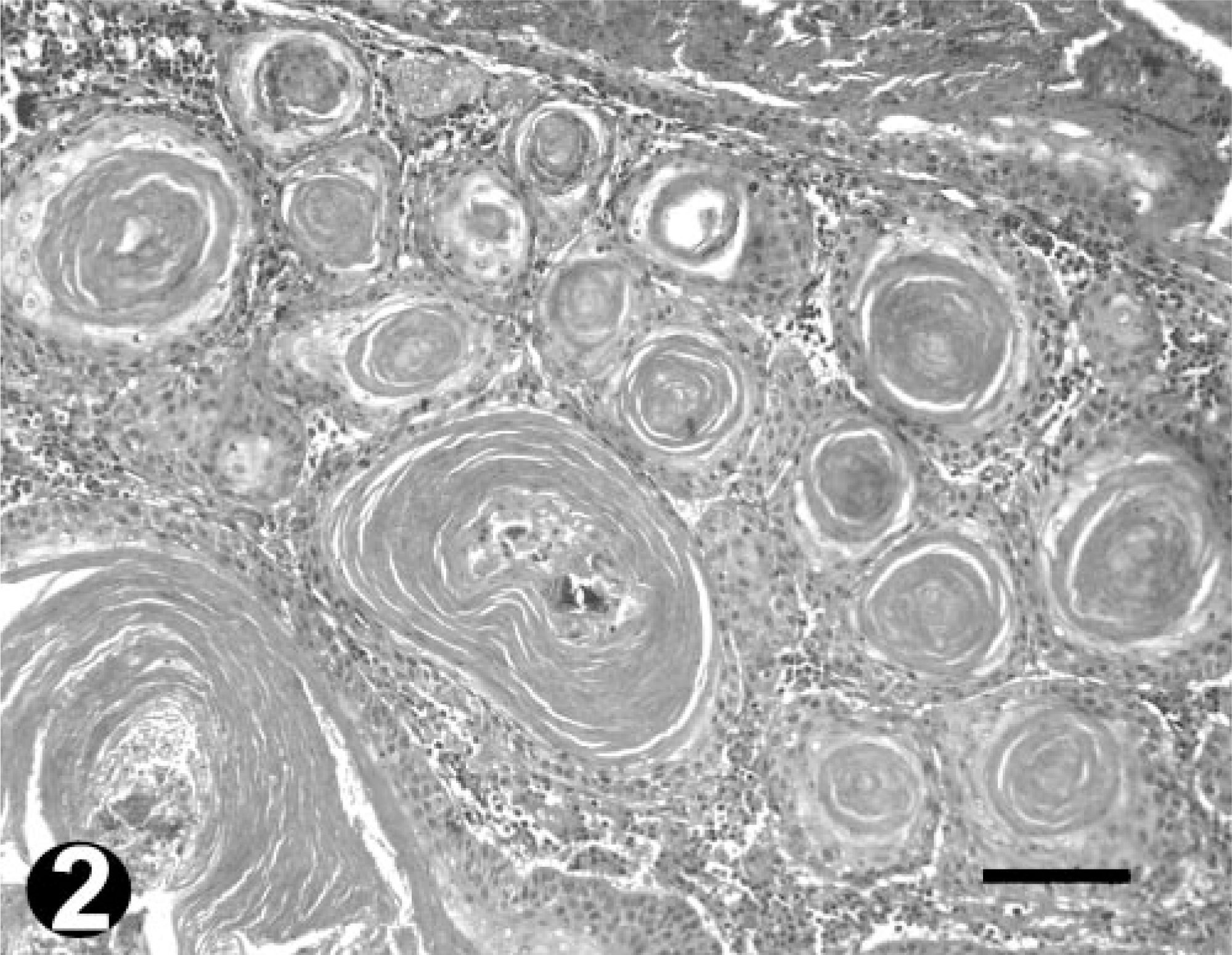
Well-differentiated OSCC; bovine. HE. Note the extensive keratin pearl formation. Bar = 100 µm.
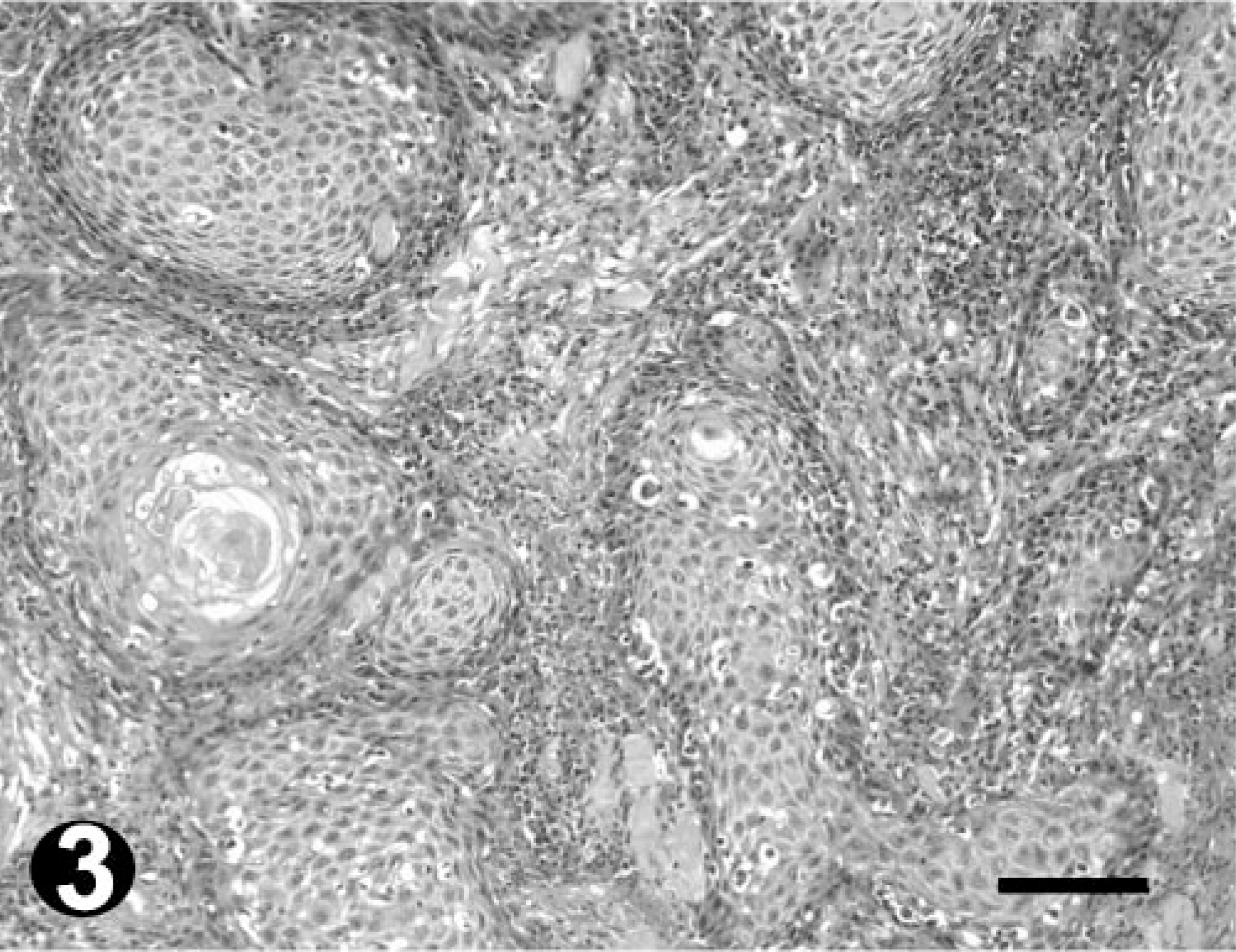
Moderately differentiated OSCC; bovine. HE. Note the island formation but with few keratin pearls. Bar = 100 µm.
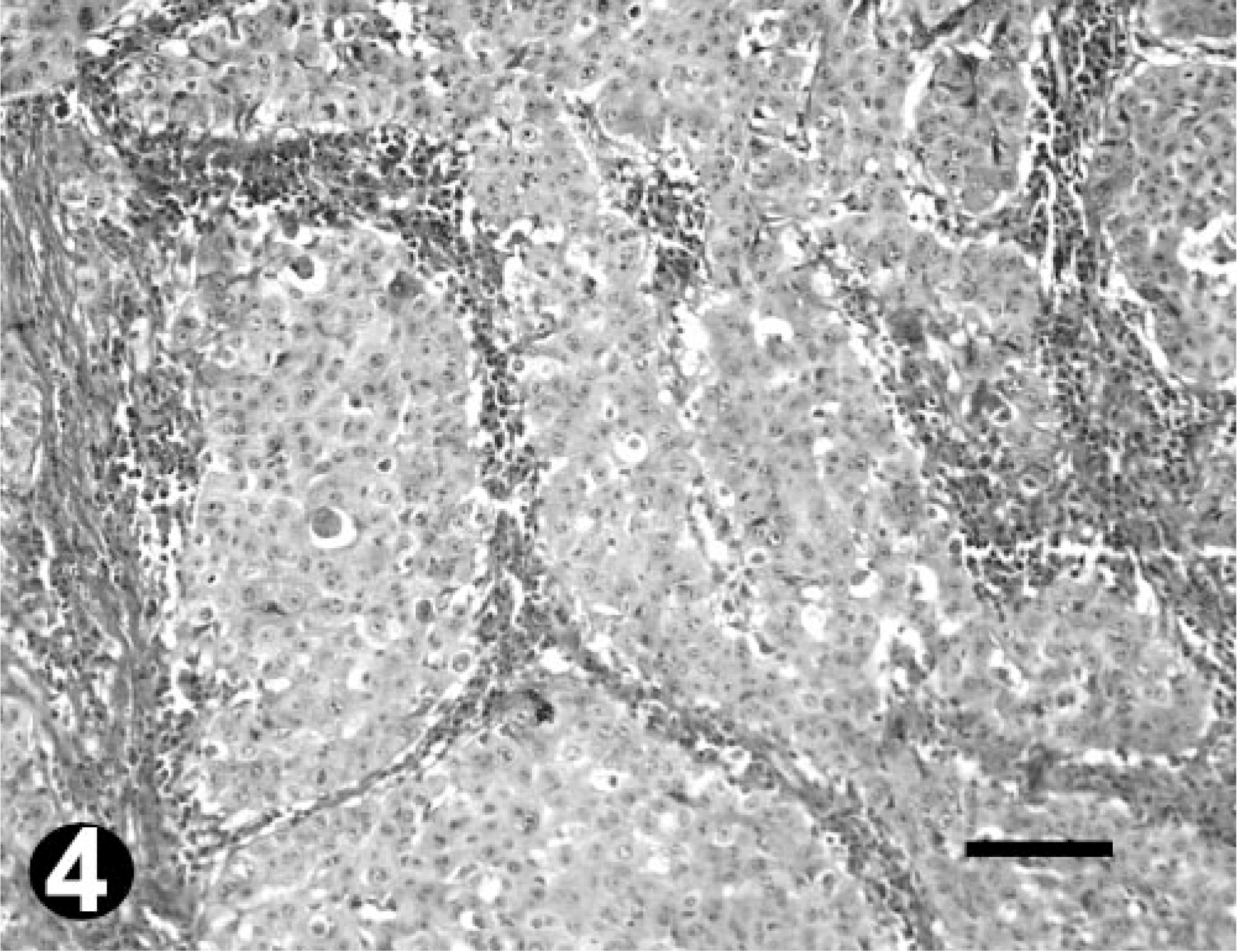
Poorly differentiated OSCC; bovine. HE. Note keratinization of individual cells and the severe des-moplastic reaction. Bar = 100 µm.
Immunohistochemistry
Sections stained for specificity control in the absence of the primary antibodies failed to show immunoreactivity for both p53 and Ki67. Positive controls showed strong nuclear staining.
Ten of 15 collected tumors (67%) were immunoreactive for p53. Eight scored 3 (> 50% positive cells) and two scored 2 (10–50% positive cells). Of the remaining five tumors, two scored 1 (< 10% positive cells), with very few positive cells, and the other three scored 0 (Table 2). Tumors scoring 3 included all degrees of histologic differentiation. The only two tumors scoring 2 were well-differentiated OSCC. The p53 stain was typically intense and homogeneous. Although no correlation between the percentage of stained nuclei and the degree of tumor differentiation was detected, different patterns of p53 staining were observed within the same tumor. Positive cells were found predominantly in the periphery of the neoplastic islands, with gradual loss of p53 staining intensity as the cells differentiated toward the keratinized centers (Fig. 5). Poorly differentiated areas showed a more diffuse and homogenous pattern of staining (Fig. 6).
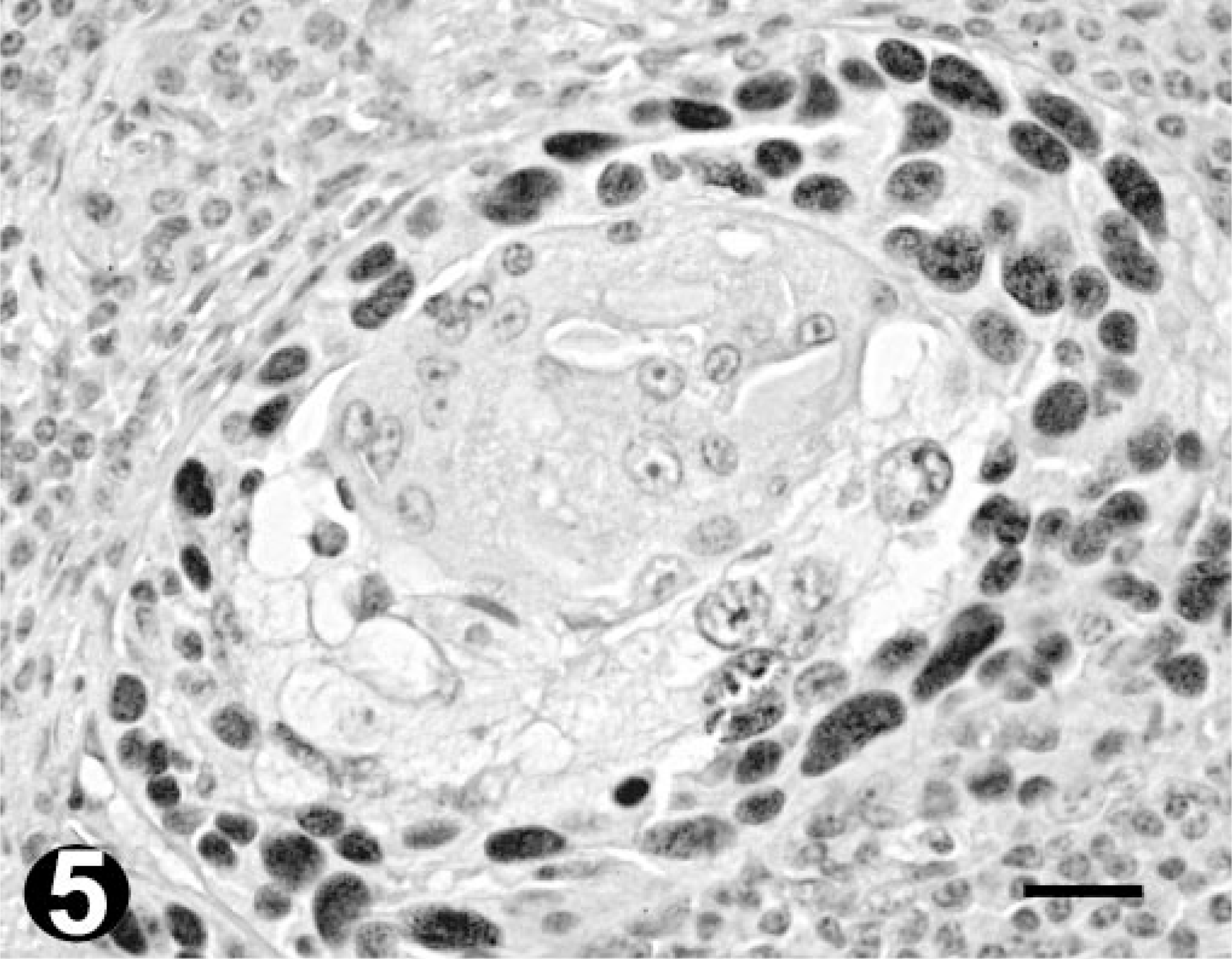
Well-differentiated OSCC; bovine. Immunohistochemistry. Nuclear p53 immunoreactivity is mainly seen in the peripheral neoplastic cells of tumor islands, sparing the central keratinized areas. Bar = 20 µm.
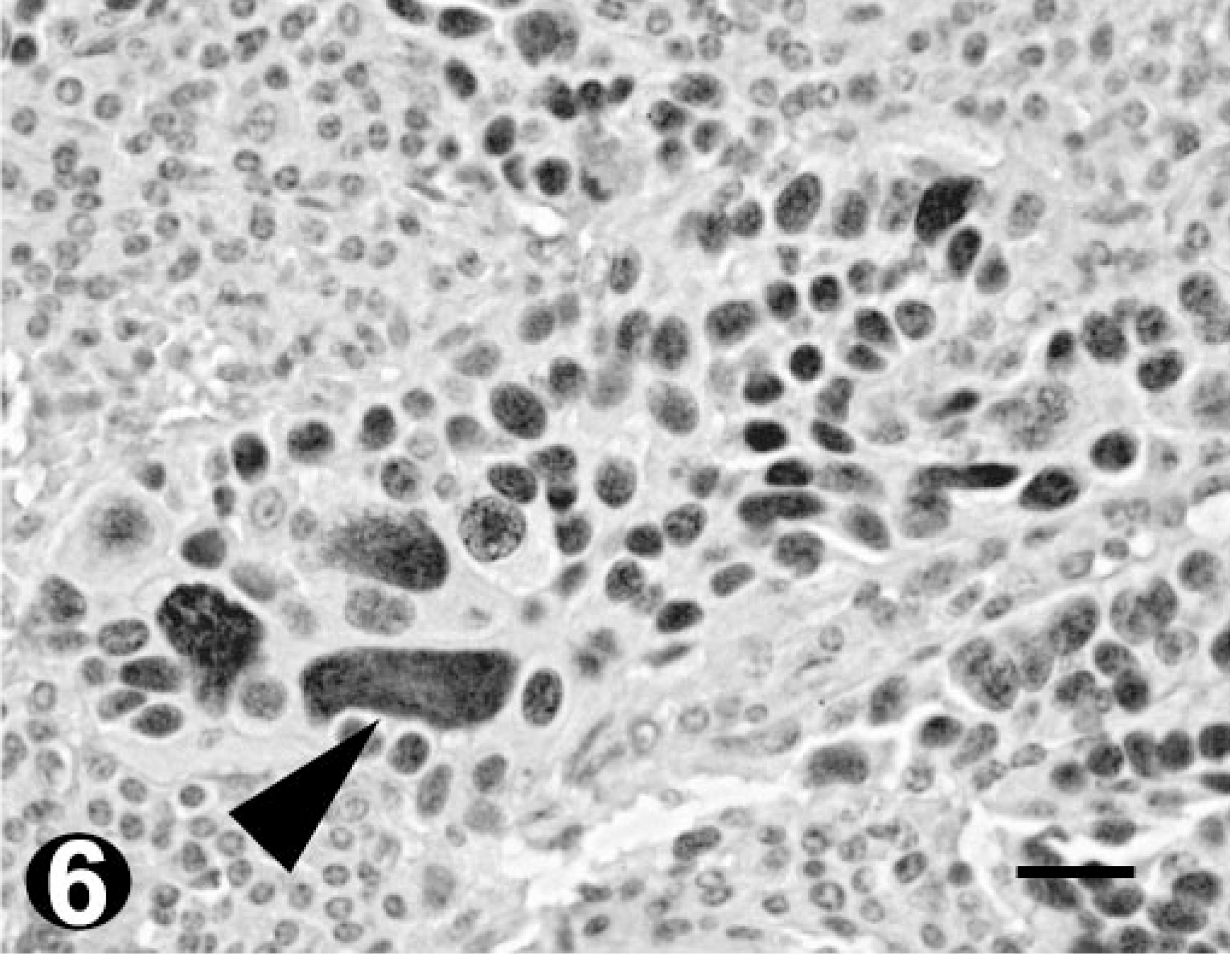
Poorly differentiated OSCC; bovine. Immunohistochemistry. Diffuse pattern of p53 immunostaining within the tumor island. Note the macronuclei (arrowhead). Bar = 20 µm.
Immunostaining for Ki67 was heterogeneous in both distribution and intensity. Mitotic figures were always strongly labeled. All the tumors tested demonstrated Ki67 expression. The Ki67 index ranged from 14.2 to 21.6% (mean value = 19.3%) in well-differentiated OSCC (Fig. 7) and from 19.3 to 34.6% (mean value = 27.7%) in poorly differentiated OSCC (Fig. 8). Comparing the indexes for the well- and poorly differentiated tumors, a significant difference was found (P = 0.013, Fig. 9). Conversely, no significant differences were observed between the Ki67 indexes obtained for moderately differentiated tumors (range 4.5–26%; mean value = 13.3%) and the ones for well-and poorly differentiated tumors.
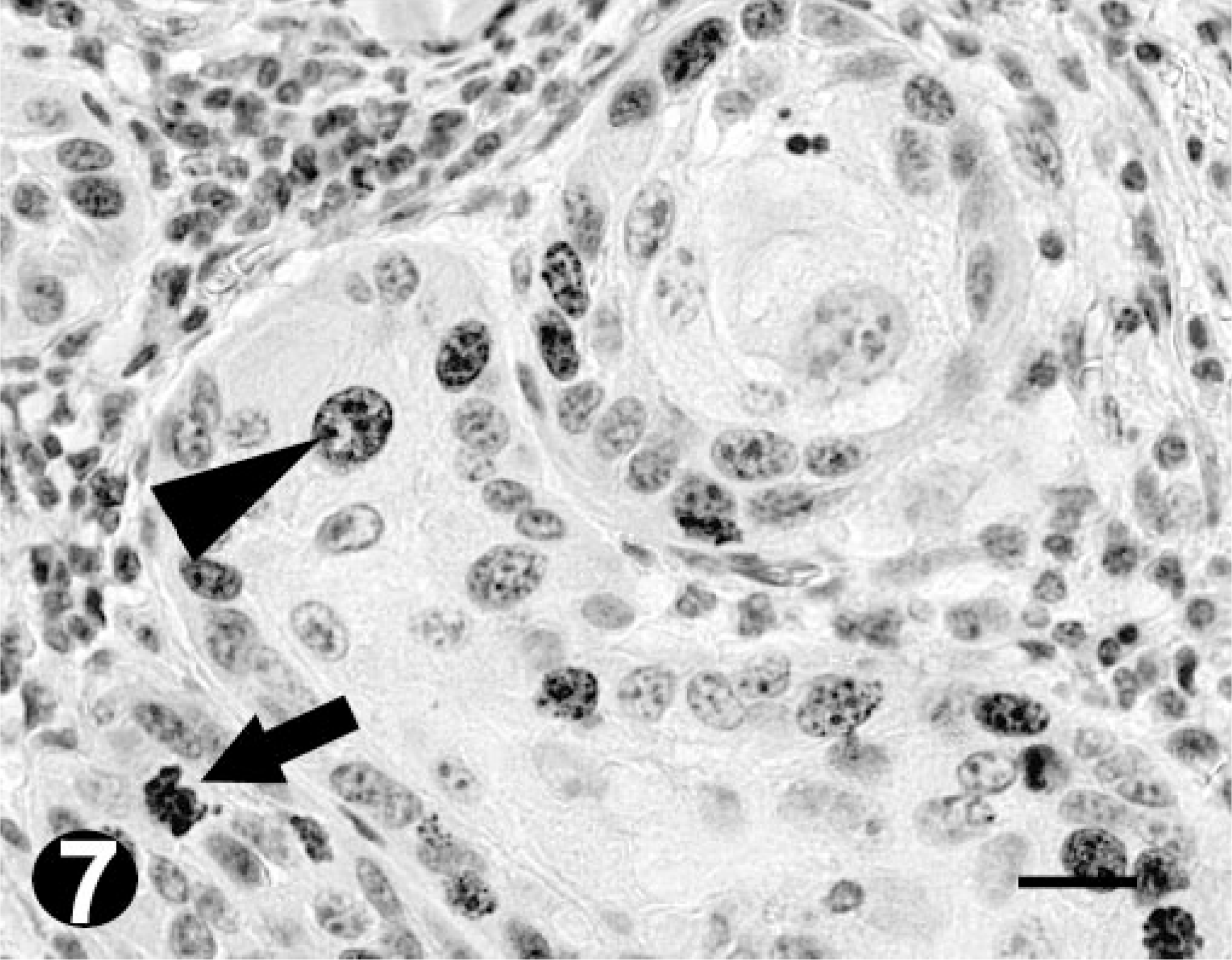
Well-differentiated OSCC; bovine. Immunohistochemistry. Positive reaction for Ki67 is indicated by the presence of nuclear staining (arrowhead). Note the mitotic figure (arrow). Bar = 20 µm.
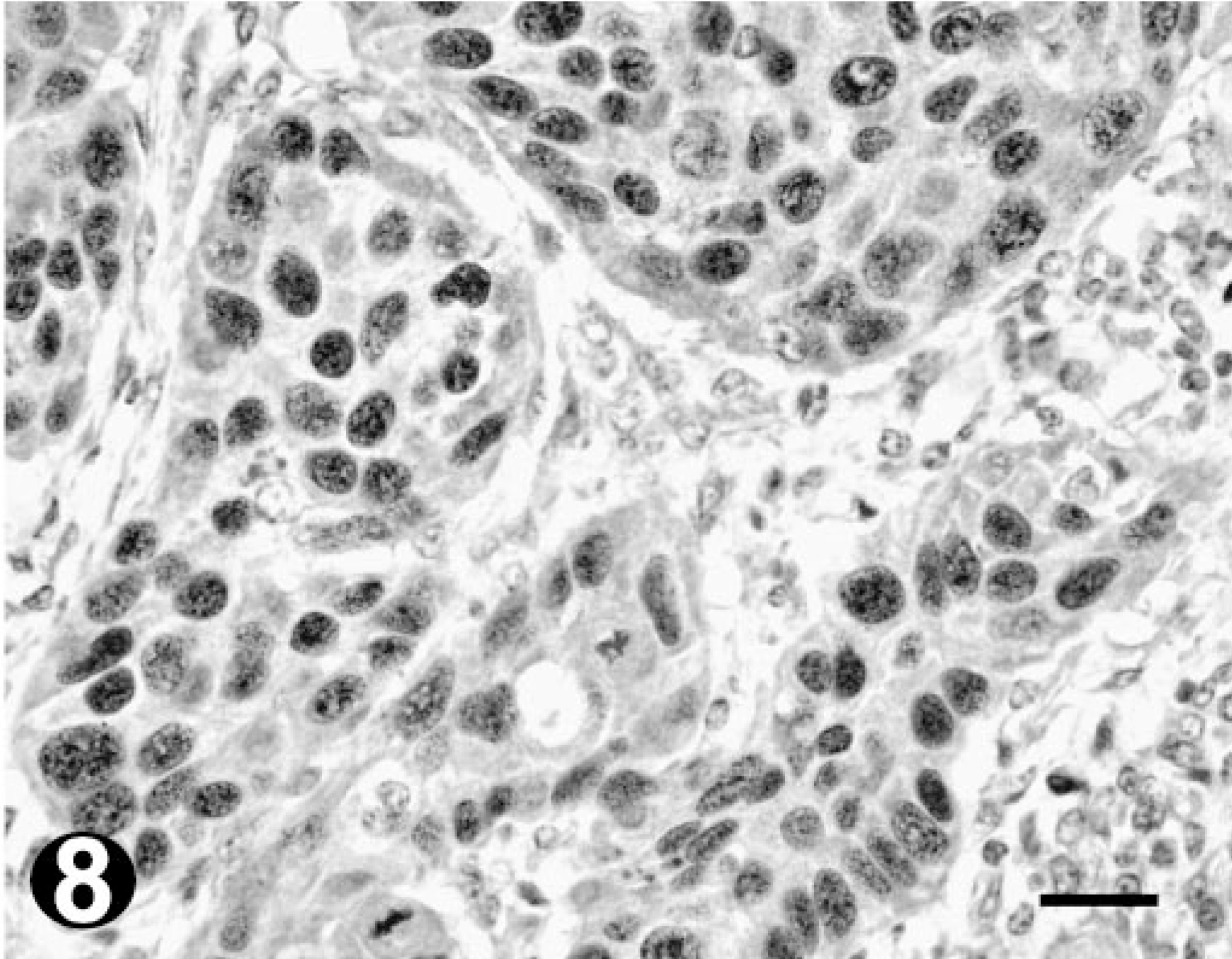
Poorly differentiated OSCC; bovine. Immunohistochemistry. Note the increased number of stained neoplastic cells in comparison to Fig. 7. Bar = 20 µm.
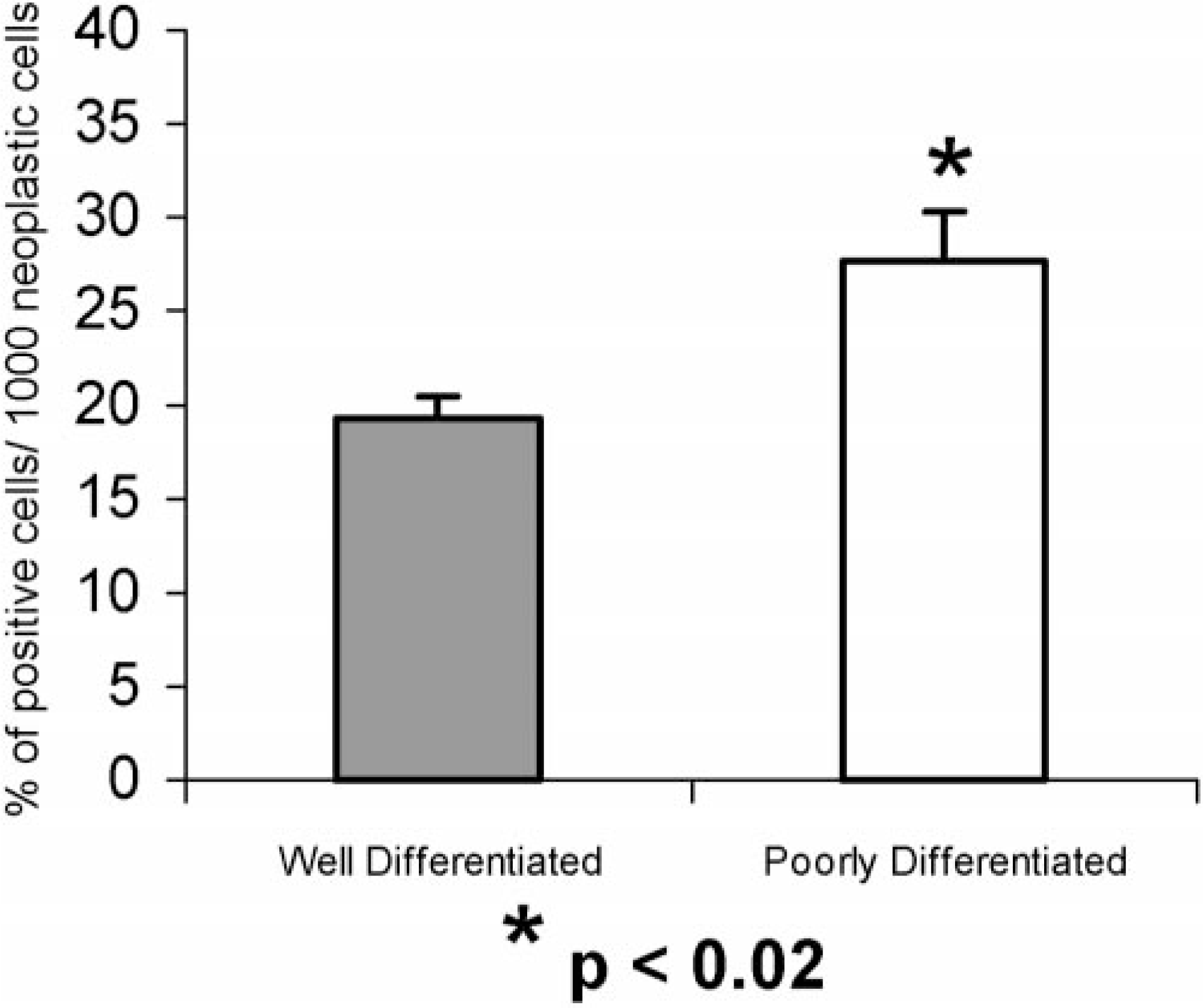
Comparison of Ki67 indexes (mean × SEM) between well (n = 7) and poorly (n = 4) differentiated bovine OSCC.
No association was noted between p53 immunoreactivity and the proliferation index as measured by Ki67 immunohistochemistry.
Discussion
In this study, high levels of labeling were obtained with anti-human p53 CM-1 polyclonal antibody in 67% of the bovine OSCCs. Albaric et al. 1 had already tested CM-1 antibody in one bovine OSCC, also with good staining results. In all positive cases, p53 immunoreactivity was restricted to the nuclei of tumor cells. These data indicate that p53 accumulation in the nucleus of cells is common in bovine OSCC. The percentage of neoplasms expressing p53 was similar to that found by Teifke and Löhr 31 in bovine OSCC (63%), with a distinct antibody, and also by Gamblin et al. 7 in canine squamous cell carcinoma (68%) with the same CM-1 polyclonal antibody, which recognizes both wild and mutated p53 protein.
In humans, cutaneous squamous cell carcinoma induced by radiation has been found to contain specific mutations in the p53 gene. 23 In fact, p53 tumor-suppressor gene plays an important role in the carcinogenesis of various tissues, 21 being the most commonly altered gene in human cancer. 7 Bovine OSCC prevalence has already been directly related to exposure to UV radiation 2 , 17 and, as with humans, it seems highly probable that a mutation in the gene that encodes for this protein is, at least, one of the factors involved in the genesis of this tumor. 22 , 23
Correlation between p53 nuclear immunostaining and gene mutation is highly significant in numerous solid human cancers. 1 However, overexpression or accumulation of wild-type p53 protein can occur in a variety of situations, such as induction by normal cellular stimuli (DNA damage, apoptosis) or protein binding that leads to alteration of p53 degradation pathways or its stabilization. 1 Although molecular analysis of positive cases in this study remains to be done, to confirm whether the high values of p53 immunostaining obtained correspond effectively to the accumulation of the mutated protein, this study provides support for a role of p53 in the pathogenesis of bovine OSCC.
Statistical analysis of the results obtained revealed no significant correlation between the percentage of p53 stained nuclei and the degree of tumor differentiation, which is in accordance with other studies in OSCC, in humans 23 and also in several domestic animals. 31 Nevertheless, we found variations in patterns of p53 immunoreactivity between individual tumor cells. Staining was more prominent in less well-keratinized cells, commonly present in poorly differentiated OSCC and in the advancing proliferating zones of the well-differentiated ones, as well as in the periphery of the tumor islands, with a gradual loss of positivity as the cells integrated keratin whorls. Despite the referred absence of correlation between percentage of p53 immunoreactive cells and degree of tumor differentiation, our results indicate that p53 overexpression is associated with an immature cell phenotype, a finding also reported by other authors. 12 , 20 , 23 , 31 A possible explanation for this fact will be possible only after molecular genetic studies of p53 mutations, performed simultaneously with p53 protein analysis in the same tumors in a large series, as suggested by Oram et al. 23
Normal tissue adjacent to the tumor lesions was consistently negative for p53, with the exception of one sample where intense nuclear staining was seen in apparently nondysplastic squamous mucosa, adjacent to the neoplasm. Similar observations were made by Coulter et al. 5 in human squamous cell carcinoma, supporting the possibility that p53 mutation is an early molecular event in the development of squamous cell carcinoma.
Successful immunostaining of Ki67 was also obtained in this study with a polyclonal anti-human Ki67 antibody. Ki67 index was significantly correlated with the histologic differentiation, that is, high proliferation generally corresponded to a low degree of differentiation (P = 0.013). These findings match the ones described by several other authors that report significantly higher Ki67 index values in poorly than in well-differentiated squamous cell carcinomas 4 , 19 and a reversal in the normal ratio of proliferation to differentiation that is seen with increasing anaplasia. 4 The proliferation indexes obtained by Makino et al. 19 in poorly and in well-differentiated human squamous cell carcinomas, 31.9 and 20.0%, respectively, are in fact very similar to the ones obtained in this study. Nevertheless, it should be noted that, in this study, this correlation is only valid on a group basis, and not discriminatory on an individual animal basis, because some overlap in the values was seen. Thus, increased Ki67 index in a bovine OSCC may not represent by itself a reliable prognostic parameter of malignant behavior.
Likewise, the inconsistency of the values obtained in moderately differentiated OSCC revealed that it is not possible to predict the proliferating index of these tumors on the basis of exclusively the histologic pattern. This may be attributed to the fact that OSCC in this category corresponds to neoplasms that touch both ends of the spectrum, either being more similar to the well-differentiated or to the poorly differentiated tumors, and consequently, significant tumor group heterogeneity is present, including a broad range in Ki67 proliferation index.
Finally, no relationship seems to exist between p53 immunoreactivity and Ki67 index, as also reported in human epidermal squamous neoplasia. 19
In summary, our results show that p53 gene product is highly expressed in bovine OSCC, providing further support for its role in bovine OSCC tumorigenesis. Nevertheless, further studies must be performed to identify the nature of that protein and the presence of gene mutation. In addition, this study demonstrates that there is an inverse relationship between the degree of tumor differentiation and the rate of cellular proliferation in bovine OSCC, as measured by Ki67 proliferation index.
Footnotes
Acknowledgements
We wish to thank all the technical support provided by the laboratory technicians from both Lisbon and Barcelona Veterinary Faculties and also thank the technicians from the Portuguese Institute of Oncology Francisco Gentil, Lisbon, for the assistance in immunohistochemistry. We gratefully acknowledge Dr. Yvonne Schulman for reviewing the manuscript. This work was supported by CIISA (Interdisciplinary Investigation Centre for Animal Health) from the Faculty of Veterinary Medicine, Lisbon.
