Abstract
A newborn Longhorn heifer calf presented with generalized tremors, muscle fasciculations, ataxia, and nystagmus. At necropsy, no gross central nervous system lesions were observed. Histologically, the brain and spinal cord had mild to moderate diffuse microgliosis and astrocytosis, minimal nonsuppurative encephalitis, and decreased myelin staining. Ultrastructural examination revealed thinning and absence of myelin sheaths. Various cell types were immunohistochemically positive for bovine viral diarrhea virus (BVDV). Noncytopathogenic BVDV was isolated from the brain and identified as BVDV type 2 by phylogenetic analysis. BVDV-induced hypomyelination is rare and analogous to lesions in neonates infected with border disease and classical swine fever viruses. This is the first documented case of hypomyelination in a calf specifically attributed to BVDV type 2 and the first description of the ultrastructural appearance of BVDV-induced hypomyelination.
Bovine viral diarrhea virus (BVDV) is a single-stranded, enveloped, positive-sense RNA virus grouped in the family Flaviviridae, genus
History and Clinical Findings
A newborn female Longhorn calf was presented to the Texas A&M Veterinary Teaching Hospital with a history of congenital neurologic signs. The calf was unable to rise, and it exhibited generalized tremors and muscle fasciculations, grade 4 generalized ataxia, 9 and horizontal and vertical nystagmus. Spinal and cranial nerve reflexes, the pupillary light reflex, muscle strength, and mentation were normal. Because of a poor prognosis, the owner elected euthanasia, and the calf was euthanized at 8 days of age.
Necropsy Findings
The brain and spinal cord were grossly normal at necropsy. Gross lesions in other tissues were limited to mild cranioventral consolidation of the lungs and a small amount of purulent exudate within the thickened umbilical cord. Tissue samples were fixed in 10% neutral buffered formalin, processed routinely for light microscopy, and stained with hematoxylin and eosin (HE). Selected tissue sections were stained with the Weil and Luxol fast blue methods; others were stained with the streptavidin–biotin–immunoperoxidase method and the following primary antibodies: microglia (Iba1; 1:000 dilution; Biocare Medical, Concord, CA), glial fibrillary acidic protein (1:1000 dilution; DakoCytomation, Carpinteria, CA), and BVDV (undiluted; New York State Animal Health Diagnostic Center, Ithaca, NY). Fresh samples of brain and spinal cord were immersed overnight in fixative (2.5% glutaraldehyde, 2.5% paraformaldehyde in 0.1M sodium cacodylate buffer), then washed and processed for electron microscopy. Briefly, after washing in buffer, the tissue was en bloc stained with 1% osmium tetroxide and 0.5% potassium ferrocyanide for 1.5 hours, then dehydrated in an ascending alcohol series followed by propylene oxide. The tissue was then embedded in epoxy resin. Control brain and spinal cord tissue for immunohistochemistry and electron microscopy were obtained from a 2-day-old female Charolais calf that presented for bilateral rear limb fractures and did not have clinical signs or lesions consistent with neurologic disease.
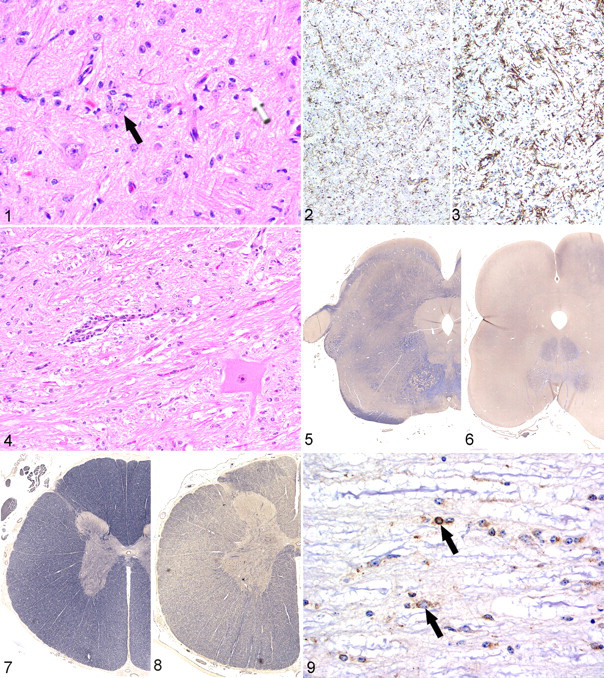
Histologically, a mild to moderate gliosis was evident throughout the brain and spinal cord, most notably in the cerebral white matter, cerebellar white matter, and brain stem (Fig. 1). The increased glial cell population included astrocytes and microglia. The astrocytes had large pale nuclei, often formed clusters, but generally did not have an increased amount of visible cytoplasm on HE staining. However, immunohistochemical expression of glial fibrillary acidic protein by astrocytes was increased in comparison with the control. Microglial cells were larger than normal and often had irregularly shaped nuclei. Compared with that of the control calf, microglial immunohistochemical reactivity was increased in intensity in the midbrain and spinal cord (Figs. 2, 3). A few swollen axons were evident in the medulla oblongata. Rare parenchymal blood vessels were surrounded by a small number of lymphocytes, generally not more than one cell layer in thickness (Fig. 4), and endothelial cells were often swollen. Weil and Luxol fast blue stains revealed decreased myelin staining in the cerebral white matter, midbrain, and spinal cord white matter, when compared with that of the control calf (Figs. 5–8). Other histologic findings included necrosuppurative omphalophlebitis and arteritis with intralesional bacteria and suppurative bronchopneumonia with intralesional plant material. Minimal multifocal neutrophilic inflammation was in the heart, liver, esophagus, omasum, and colon. Mild lymphocytic inflammation was evident in the dermis. The following tissues were unremarkable: kidney, spleen, adrenal gland, thyroid gland, eye, trachea, radial nerve, common peroneal nerve, tibial nerve, spinal nerve roots, dorsal root ganglia, skeletal muscle, urinary bladder, abomasum, rumen, small intestine, thymus, and bone marrow.

This dendrogram shows the phylogenetic location of the bovine viral diarrhea virus isolate in this case (strain No. N06-419) based on the comparison of sequences from the 5′ untranslated region.
Immunohistochemistry for BVDV antigen in sections of spinal cord, medulla oblongata, cerebellum, and cerebrum revealed positive reaction in a variety of cell types, including astrocytes, oligodendrocytes, microglia, pericytes, ependymal cells, and endothelial cells (Fig. 9 ) but only rarely in neurons. Immunoreactivity was strongest in the spinal cord and medulla oblongata. In the cerebrum, BVDV immunoreactivity was relatively weak and confined to pericytes and occasional endothelial cells and glial cells within the white matter. In the skin, weak multifocal BVDV immunoreactivity was evident in the epidermis and within follicular epithelial cells. Strong BVDV immunoreactivity was evident in thyroid follicular epithelial cells. Immunohistochemistry of the control calf brain for BVDV was negative.
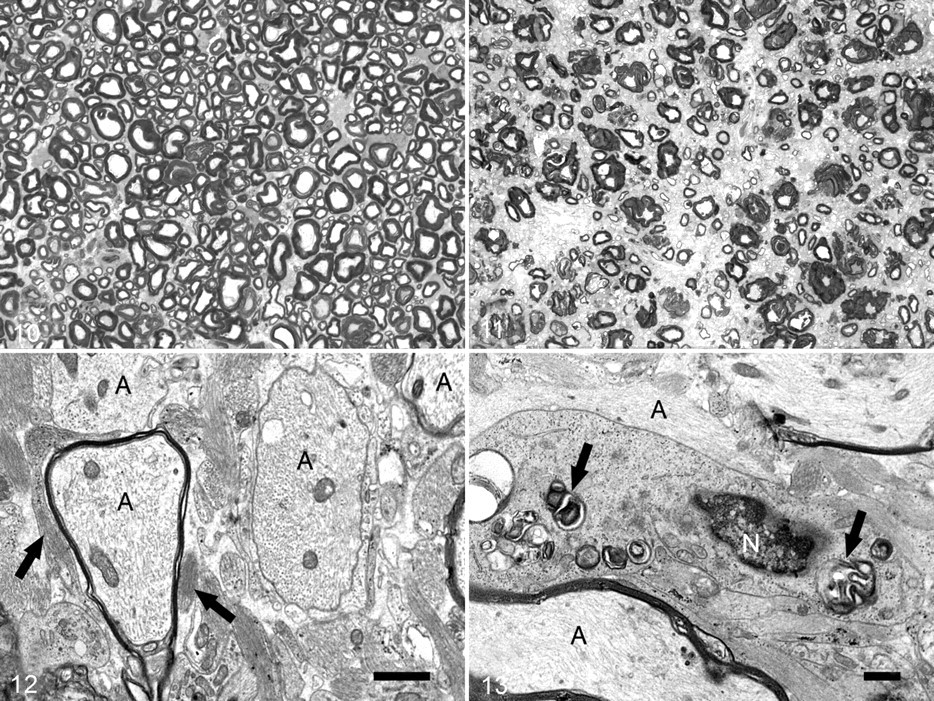
In semithin sections of the spinal and cerebral white matter prepared for ultrastructural examination, myelin sheaths were absent or markedly thinner in comparison with those in tissue from the control calf (Figs. 10, 11). Ultrastructurally, the thinning of myelin sheaths was accompanied by an increase in astrocytic processes, which were identified by the presence of cytoplasmic fibril bundles (Fig. 12). Astrocytic processes were often closely apposed to myelin sheaths or bare axons. An increase in microglial processes was noted, as identified by the presence of long endoplasmic reticulum cisternae; these processes were also seen in close apposition to myelin sheaths and axons. Amorphous accumulations of electron-dense material, interpreted as myelin debris (Fig. 13), were infrequently observed in the cytoplasm of microglia. Small, round, clear vacuoles were rarely seen within microglial cytoplasm.
BVDV was isolated from the brain with standard methods. The virus was of the noncytopathic biotype based on serial passage in cultured epithelial cells. 13 Phylogenetic analysis (Fig. 14 ), based on sequences generated from the 5′ untranslated region, was performed as described. 22 Segregation of BVDV strains based on comparison of sequences from the 5′ untranslated region is an accepted method for determining BVDV genotypes. 25
Discussion
Hypomyelination is an uncommon outcome of transplacental BVDV infection. Most fetal infections with BVDV result in persistent subclinical infection, fetal death, or defects such as cerebellar hypoplasia, hydranencephaly, internal hydrocephalus, microencephaly, and porencephaly. 5,10,14 Neuropathologic descriptions of natural in utero BVDV infection resulting in hypomyelination are limited. In a dairy farm in the Netherlands, an outbreak of BVD resulted in multiple calves being born with neurologic signs similar to those described here, and histologic findings included widespread loss of myelin staining, an increased number of microglia and astrocytes with enlarged vesicular nuclei, and mild mononuclear cell cuffing of blood vessels. 5 Another report describes a single case of BVDV in a newborn Holstein calf that resulted in ataxia, hypermetria, and intention tremors of the head. 23 Hypomyelination was reported in that case but not accompanied by gliosis or inflammation. A recent comprehensive British study suggests that hypomyelination is a relatively common outcome of BVDV infection in some herds; 20 it documented 23 herds over a 17-year period in which hypomyelination was the primary lesion in calves with fetal BVDV infection, resulting in neurologic signs. The pathologic findings were similar to those reported here. The reason for the apparent higher incidence of hypomyelination in some herds in unclear. Hypomyelination can occur in conjunction with other CNS defects in BVDV-infected calves. Cerebellar hypoplasia was the most common lesion in a study of natural transplacental BVDV infection in Germany; in fact, 5 of 25 animals showed evidence of concurrent hypomyelination. 14
Experimental in utero BVDV infection has only rarely produced hypomyelination. In a study by Done et al, 10 pregnant cows injected with BVDV either aborted or gave birth to calves with morphologic defects in the CNS. The defects ranged from structural brain malformations to hypomyelination, and some calves had both. More recent studies involving in utero infection with BVDV produced varying lesions but no histologic evidence of reduced myelin. 4,19 In contrast to congenital BVDV infections, hypomyelination is relatively more common in congenital border disease and classical swine fever infections. Histologic lesions in both diseases typically consist of severe hypomyelination, gliosis, and a lack of inflammatory cell infiltration. 3,7,18
The pathogenesis of hypomyelination resulting from in utero pestivirus infection is poorly understood. A deleterious effect on oligodendrocyte development or function is possible, although studies of experimental border disease indicate that BDV does not have a selective tropism for oligodendrocytes. Cell types infected by BDV include neurons, astrocytes, oligodendrocytes, endothelial cells, ependymal cells, and meningeal fibroblasts,
11,15
and studies with BVDV have produced comparable results.
4,19
Similarly, a variety of cell types were infected with BVDV in the case reported here. BDV-induced interference with thyroid function may play a role in pathogenesis. In one study, lambs with border disease had significantly lowered levels of thyroid hormones and the myelin-associated thyroid hormone–dependent enzyme CNP (2′,3′-cyclic nucleotide-3′-phosphodiesterase) in comparison with normal lambs.
1
Like this calf, the lambs in that study had viral antigen within thyroid follicular cells without histologic lesions. The role of CNP in myelinogenesis is questionable given that
The variation in the nervous system defects in fetuses infected transplacentally by BVDV is intriguing. The outcome of BVDV in utero infection varies with the timing of the infection. Infections before immunocompetency at 150 days can result in abortion, congenital defects, or persistent infection, whereas those occurring after 150 days are typically cleared. 17 Few experimental transplacental BVDV infections have resulted in hypomyelination; thus, determining why it occurs in some cases is difficult. In the previously noted study, the viral inoculum was a mixture of BVDV strains, and all cows were inoculated at 100 days of gestation. 10 The variety of resulting lesions in that study suggests that the virus strain plays more of a role in determining the type of defect than the timing of infection. As a single-stranded RNA virus, BVDV lacks proofreading ability and is prone to a high mutation rate; the resulting viral strains have varying levels of virulence. 21 The tropism and effects of different viral strains on the fetal nervous system have only begun to be examined. Experiments suggest that type 2 BVDV strains infect the fetal brain more readily than do type 1 strains, although both types have similar tissue tropism. 4 In a previously reported case of BVDV2-induced neurologic disease in a 15-month-old heifer, the lesions were limited to meningoencephalitis, neuronal necrosis, and gliosis. 6 In the cited British study, isolated viruses classified by molecular techniques were either BVDV1a or BVDV1b. 20
The ultrastructural appearance of the CNS changes in BDV- and CSFV-infected neonates has been described. As with the calf reported here, an increased number of astrocytic processes in close proximity to axons has been noted in BDV-infected lambs.
3
Cancilla and Barlow
8
suggested that astrocytic processes may retard myelin formation by inhibiting contact between axons and oligodendrocytes; but whether astrocytes truly play a role in the pathogenesis or if their proliferation is simply a response to injury remains to be determined. Microglial processes adjacent to thinned myelin sheaths containing myelin-like debris and clear vacuoles have been observed in BDV and classical swine fever infection.
3,7,8
Although rare, this finding suggests that destruction of preformed myelin (demyelination) or abnormally formed myelin (dysmyelination) may play a role in the pathogenesis and that the most appropriate term for the lesion may not be
Most previously described cases of BVDV-induced hypomyelination were not linked to a specific viral genotype. Phylogenetic analysis verified that the virus in this case was a strain of type 2 BVDV. Further study is required to determine the specific viral and host factors required for the development of hypomyelination in calves infected with BVDV in utero.
Footnotes
Acknowledgements
We thank Sarah Jones for assistance with cytochemical stains, Joy Cramer and Dr Andy Ambrus for immunohistochemistry, and Dr Tom Lester for virology support.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
