Abstract
A neonatal foal with signs of rectal bleeding was diagnosed with an intraluminal rectal mass and intussusception on surgical exploration of the abdomen. Histologically, the mass consisted of cystic spaces lined by simple columnar epithelium with numerous goblet cells and was surrounded by thin bands of smooth muscle in a myxomatous stroma. Although the mass shared similarities with retrorectal cystic hamartoma (tailgut cyst) and juvenile polyps, described in human medicine, location and histologic findings were not entirely consistent with either condition.
An 18-hour-old Thoroughbred colt presented to the University of Pennsylvania George D. Widner Hospital with the complaint of bleeding from the rectum thought to originate from a rectal tear after administration of two enemas.
Upon presentation, a large amount of blood was noted on the foal's tail and perineum, and mild signs of neonatal encephalopathy were evident. During the initial evaluation, the foal exhibited continuous straining to defecate resulting in prolapse of hemorrhagic rectal mucosa. On the basis of digital palpation of the rectum, a full thickness rectal tear was suspected, other differential diagnoses at the time included intussusception and necrotizing enterocolitis. Upon surgical exploration of the abdomen, an intraluminal tubular soft tissue mass was identified in the most orad aspect of the rectum. The mass was attached to the rectal wall by a stalk 24 cm orad from the anus. The adjacent small colon had formed an intussusception with the intussusceptum bypassing the mass. The affected small colon and rectal segment, including the mass was resected, placed in 10% neutral buffered formalin, and submitted for histopathologic examination.
Grossly, the mass was cylindrical with a tapering apex and measured 6.5 × 5.0 × 3.0 cm (Fig. 1). It had a cylindrical stalk, 1.5 cm long, 1.0 cm in diameter, attached to the rectal mucosa. The surface was smooth, gray-green, and covered with meconium. On cut surface, it consisted of numerous small cysts (up to 1.5 cm in diameter) filled with clear fluid. Thin septa of firm, pink-white stroma separated the cysts.
Hemisectioned polypoid rectal mass; foal. The mass consists of numerous small cysts (up to 1.5 cm in diameter) filled with clear fluid. Thin septa of firm, pink-white stroma separate the cysts.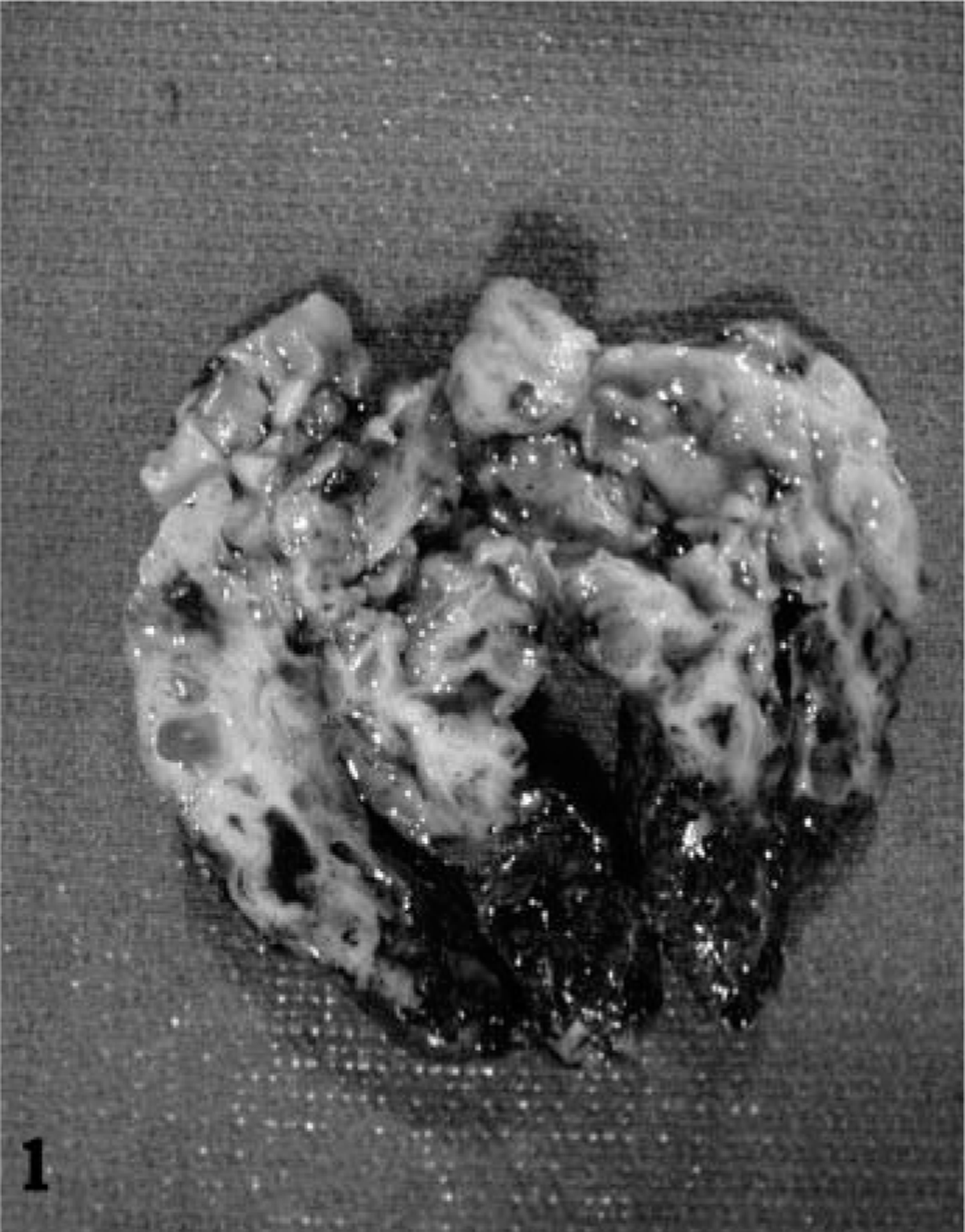
On histopathologic examination, the mass was covered with a single layer of normal rectal mucosa (Figs. 2, 3). It contained numerous large cystic spaces lined by a well-differentiated simple columnar epithelium with frequent admixed goblet cells. The cysts were separated by abundant, myxomatous stroma. Thin bands of well-differentiated but unorganized smooth muscle were scattered throughout the stroma and often surrounded the cystic spaces. A diagnosis of cystic polypoid rectal hamartoma was made.
Cystic polypoid rectal hamartoma; foal. Large cysts are separated by abundant, myxomatous stroma. Thin bands of well-differentiated but unorganized smooth muscle are scattered throughout the stroma and often surrounded the cystic spaces. HE. Bar = 500 µm.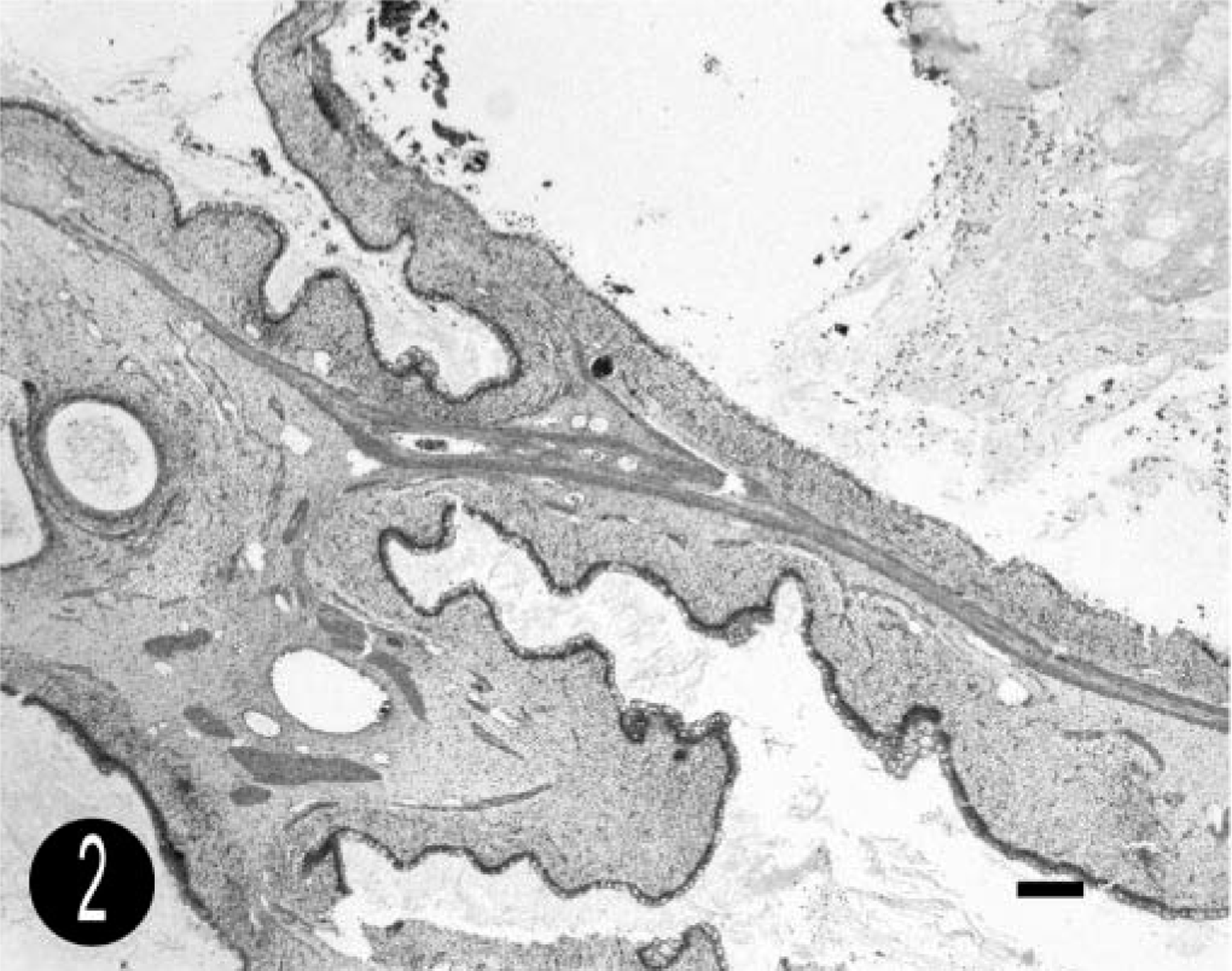
Cystic polypoid rectal hamartoma; foal. The cysts are lined by simple columnar epithelium with numerous goblet cells (arrowhead). There is adjacent myxomatous stroma (solid arrow), with thin bands of smooth muscle (open arrow). HE. Bar = 25 µm.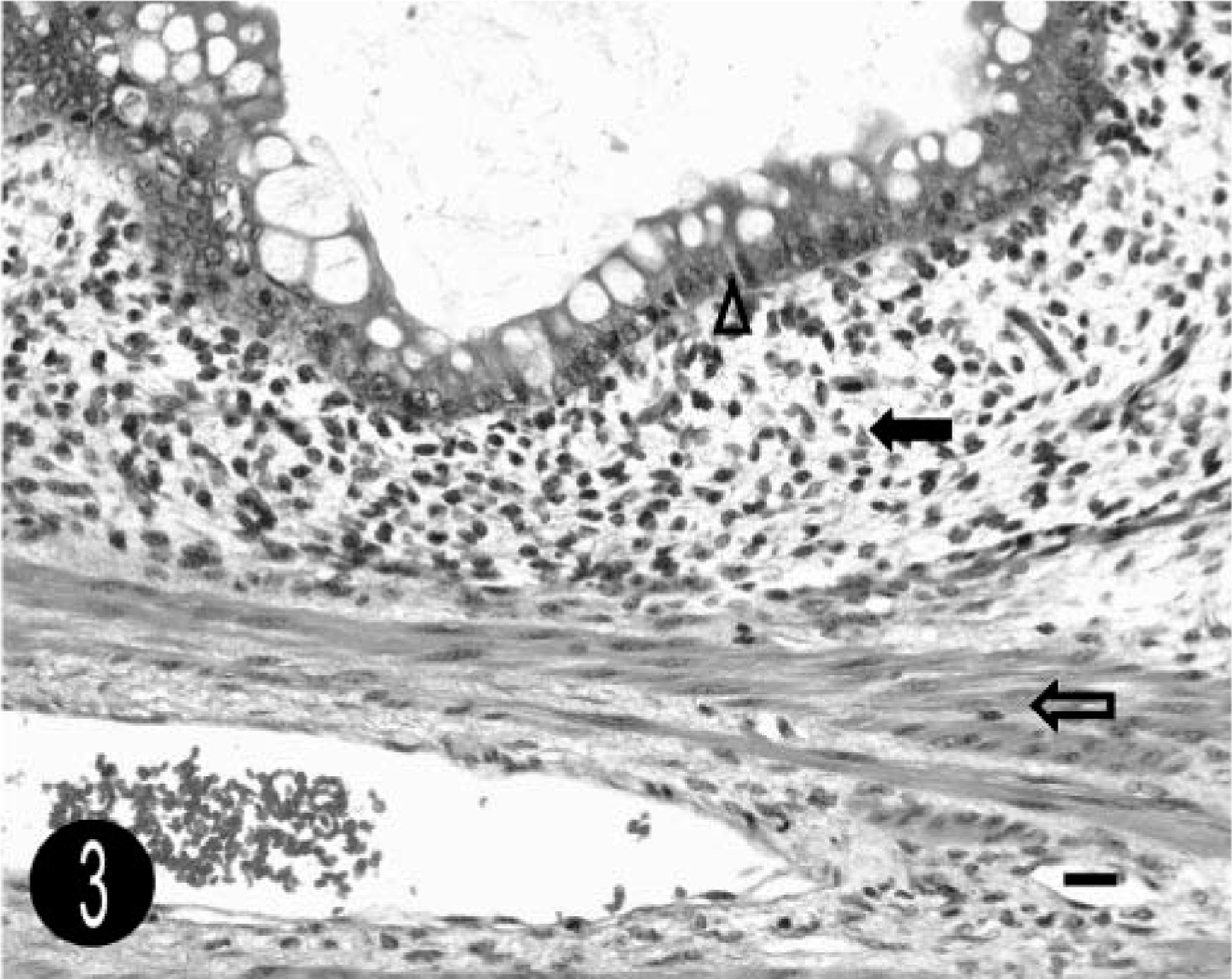
The foal was euthanatized because of fibrinous peritonitis and incisional dehiscence 4 days after surgery. On postmortem examination, there was perforation of the small colon approximately 1.5 cm orad to the anastomosis site, severe fibrinous peritonitis with adhesion formation, and herniation of omentum into the subcutis through a gap in the abdominal incision site. The anastomosis site and the site of small colon perforation were examined histologically, and both had severe subacute fibrinous peritonitis. The overall histologic structure of both sites was normal, with no remnants of the mass retained.
The foal's rectal mass shares characteristics with two types of congenital tumors described in humans: retrorectal cystic hamartomas (tailgut cysts) and juvenile polyps. Its histologic characteristics are most similar to tailgut cysts, although, in humans, these are typically located within the retrorectal space and do not communicate with the rectum. Grossly, they are characterized as well-circumscribed but unencapsulated, multicystic or unilocular masses with adhered surrounding fibroadipose tissue. Histologically, the cysts are lined by various types of epithelium; most commonly stratified squamous epithelium. Other types include transitional, pseudostratified columnar, ciliated columnar, goblet cell–rich simple columnar, stratified columnar, and simple cuboidal epithelium. 9 Scattered smooth muscle bundles are almost always present in the cyst wall, and in about 50% of cases, these concentrically surround the periphery of the cysts. 3,6 Acute or chronic inflammation may be present. 3 Tailgut cysts are rare and are thought to derive from the persistence of embryonic remnants of the postanal gut (tail-gut), which appears in the human embryo at the 3.5-mm stage and usually completely regresses by the 35-mm stage. Remnants of the tailgut may result in congenital cysts in this region. 4,5 Differential diagnoses for a mass in this location in humans include teratomas, dermoid cysts, duplication cysts, and anal gland cysts. 4 Teratomas contain tissue from all three germ cell layers; skin appendages, neural components, immature elements or other mesenchymal tissues are usually present. 9 Epidermoid and dermoid cysts result from closure defects of the ectodermal tube with inclusion of skin. The cyst lining consists of squamous epithelium and in the case of dermoid cysts also contains dermal appendages such as sweat glands, hair follicles, or sebaceous glands. 4,7,9 Duplication cysts are characterized by the presence of all intestinal wall layers, including a well-formed smooth muscle layer and neural plexus. Bundles of smooth muscle are usually present in tailgut cysts, and in most cases, an apparent relationship exists between the orientation of the muscle fibers and the epithelium. However, tailgut cysts lack neural plexuses, and the smooth muscle does not form a complete, organized muscular layer. 3,4,9 Duplications of the colon have been reported infrequently in dogs and a horse and can resemble retrorectal cysts in appearance. 1
Juvenile polyps are another type of congenital tumor that share some characteristics with the mass found in this foal. Juvenile polyps are found mainly in the rectum and can occur as solitary masses, which are not precancerous, or as part of a precancerous polyposis syndrome with multiple tumors present. Grossly, they are smooth, hemispherical, pedunculated, and usually under 3 cm in diameter. Histologically, they consist of large, cystic and tortuous glands lined by goblet cell–rich columnar epithelium on a fibrous connective tissue stalk. The stroma generally contains acute and chronic inflammatory cells and granulation tissue. Smooth muscle bundles generally are not seen in these polyps. 8
Several syndromes in which numerous intestinal, colonic, or rectal hamartomatous polyps are present in young patients have been described in humans. These include juvenile polyposis syndrome (as mentioned above), Peutz-Jeghers syndrome, and Cowden disease. These are associated with specific genetic mutations and have multiple other neoplasms in a wide variety of tissues. 11 Colonic hamartomas have been reported to occur in Cdx2 knockout mice, and one report describes hamartomatous polyps in the small intestine of two dogs. 2 The polyps described in the mice were pedunculated and consisted of a stalk of arborizing bands of smooth muscle covered with polypoid proliferations of columnar epithelium whereas in the dogs, they were characterized by extension of the smooth muscle from the muscularis mucosae into the lamina propria. 2,10 With the exception of these knockout mice, this is the first report of a congenital large intestinal polypoid hamartoma in domestic animals.
Footnotes
Acknowledgements
We acknowledge Drs. Dukti, Southwood, Palmer, and Habecker for their assistance in case management and for intellectual guidance.
