Abstract
This report describes the fetoplacental pathology of Chlamydia psittaci-associated abortion, premature birth, and neonatal loss in 46 of 442 equine abortion investigations between 2015 and 2019. Seven abortions, 26 premature births, and 13 neonatal deaths with positive C. psittaci polymerase chain reaction (PCR) were evaluated. In 83% of cases (38/46), C. psittaci infection was considered as the primary cause of loss based on quantitative PCR (qPCR) confirmation, pathological findings, and exclusion of other causes, and was supported by Chlamydia spp immunolabeling in fetoplacental lesions. Lymphohistiocytic placentitis with vasculitis (36/38) affected the amnion, umbilical cord, and chorioallantois at the umbilical vessel insertion and/or cervical pole. Lymphohistiocytic chorionitis in the subvillous stroma extended to the allantois mostly without villous destruction. Lymphohistiocytic amnionitis and funisitis occurred at the amniotic cord attachment. Lymphohistiocytic hepatitis was observed in 19/38 cases and pneumonia was identified in 26 cases. Chlamydia spp immunolabeled in placenta, lung, liver, or splenic tissue in the cases that were tested (14/38). C. psittaci infection was not the cause of loss in 2 cases with other diseases and of uncertain significance in 6 cases with no conclusive cause of loss. immunohistochemistry (IHC) was negative for 6 of these cases (6/8). The highest Chlamydia load was detected in pooled placental tissues by qPCR. qPCR and IHC had 83% congruence at a qPCR cut-off of 1 gene copy. IHC limits of detection corresponded to infections with 2 × 102 gene copies identified by qPCR. This study confirms the etiological role of C. psittaci as a cause of naturally occurring equine reproductive loss.
Chlamydia psittaci, a member of the Chlamydiaceae family, is a gram-negative obligate intracellular pathogen with a broad host range. 21 Birds are the major reservoir species globally. 20 Several Chlamydia species are recognized to cause reproductive disease and abortion in animals. C. abortus and C. pecorum cause abortion in sheep, goats, cattle, and pigs with C. pecorum additionally causing serious reproductive disease and infertility in koalas.6,14,23,27,30,42,44 C. suis similarly causes a range of reproductive disease in pigs. 32 The role of C. psittaci in reproductive disease is less clear. Since the reclassification of certain strains of C. psittaci as separate species, C. abortus and C. pecorum, well-defined reproductive disease descriptions of abortion and fetal loss due to C. psittaci in any animal species are lacking. 13 In 2 reports of equine abortion with chlamydial infection, the Chlamydia were not speciated and C. abortus was not excluded.1,5 In 3 other reports, the causative role of C. psittaci infection in the equine reproductive loss was unclear either due the presence of other infectious or noninfectious causes of abortion or the infection was not associated with pathological lesions.4,16,37 A Hungarian study identified C. psittaci as the most likely cause of equine reproductive loss in 14% of horses (11/77cases) using immunohistochemistry (IHC) and polymerase chain reaction (PCR). 37 However, 89% (53/64) of Chlamydia-positive cases had other causes of reproductive loss or the causative role of C. psittaci infection was unclear. 37 The pathology of C. psittaci-induced equine reproductive disease is not well described.
In 2014, a parrot C. psittaci strain from aborted equine fetal membranes was associated with a cluster of human psittacosis in Australia. 8 Subsequent screening of equine abortion cases in 2016 identified an epizootic of C. psittaci equine reproductive loss in New South Wales thought to be associated with suspected spill over from native Australian parrots. 19 During this epizootic, C. psittaci infection also caused acute respiratory disease in neonatal foals. 15 A pigeon-type C. psittaci was identified in a single equine abortion. 18 Since 2016, routine screening of equine fetal tissues and placenta from the Hunter Valley, New South Wales for C. psittaci by PCR identified more infections and histopathology suggested a distinctive pattern of placentitis. More recently a retrospective survey identified historical evidence of C. psittaci infection by PCR in equine abortion cases in Australia, but the significance of infection was unknown without pathological evidence confirming a causative role in the pregnancy loss. 2 In this retrospective study of cases from the Hunter Valley infected with C. psittaci from 2015 to 2019, we confirm the etiological role of C. psittaci in equine pregnancy loss; describe the fetoplacental pathology of C. psittaci-induced equine abortion, premature birth, and neonatal loss; and develop criteria for the diagnosis of the disease.
Materials and Methods
Case Selection
The Scone Equine Hospital Laboratory provides a necropsy diagnostic service for abortion and foal loss to the thoroughbred industry in the Hunter Valley, Australia. As part of a surveillance program for C. psittaci infection, a total of 442 cases of equine abortion and premature or neonatal foal loss investigated at Scone Equine Hospital Laboratory between 2015 and 2019 were tested for C. psittaci using PCR. During 2016, a distinctive pattern of placentitis was recognized in association with C. psittaci infection. A total of 49 cases were positive for C. psittaci by PCR. Forty-six cases that fulfilled the following conditions were selected for review: (1) necropsy with histopathology of fetus and placenta, if available, was performed and (2) fetal and/or placental tissues were positive for C. psittaci using PCR. An additional 8 cases negative for C. psittaci by PCR were included as control samples; 3 were controls for IHC and 5 cases were negative controls for individual tissue quantitation of C. psittaci load. The 46 cases included 36 cases previously reported.15,19 The clinical history of the fetal or foal loss was recorded. The diagnosis made for each case was reviewed including review of the gross pathology and the Hematoxylin and eosin (HE) sections for microscopic pathology. IHC and quantitative PCR (qPCR) tests were performed on selected cases and tissues. Diagnostic criteria for C. psittaci-induced equine abortion, premature, or neonatal foal loss were developed based on typical placental microscopic pathology, positive PCR, and exclusion of other diseases and supported by IHC.
Gross and Microscopic Pathology
A standardized necropsy was performed at the laboratory necropsy facility by specialist equine medicine veterinarians experienced in equine fetal pathology and normal equine fetal anatomy within 24 h of abortion or death, as previously described. 38 Placental features, including the weight of the entire placental unit, umbilical cord length, the vascular pattern insertion on the chorioallantois, and the presence of any abnormalities, such as the presence of twists or abnormal vascular patterns, were routinely examined and recorded.9,45,46,48 The fetus and placenta were examined simultaneously except for retained placentas which were submitted separately up to 48 h later. After the collection of sterile samples for PCR and bacteriology, samples of lung, liver, spleen, thymus, amnion, umbilical cord, chorioallantois at the cervical pole near the cervical star, and umbilical vessel insertion were collected and fixed in 10% phosphate buffered formalin for a minimum of 12 h. The placental collection sites sampled were modified from Todhunter, 39 and are outlined in Fig. 1. Kidney was collected in 2 cases and chorionic sites in the body or horns with gross pathology were sampled in 3 cases.
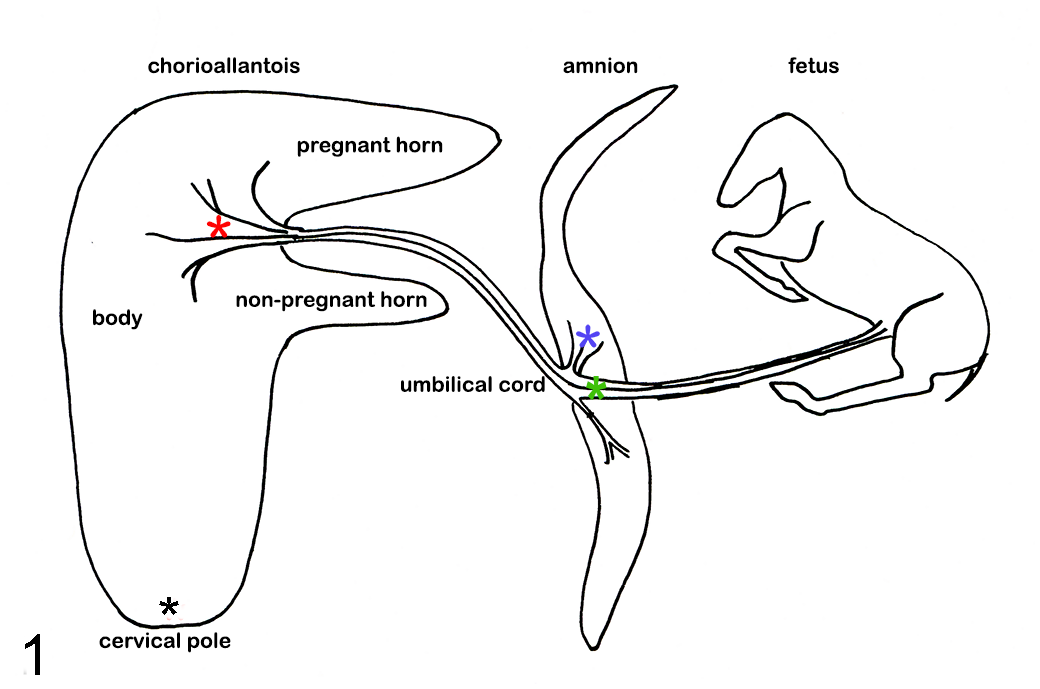
Chlamydia psittaci abortion, horse. Diagram of the 4 sites in the placenta routinely sampled for microscopic examination; the body of the chorioallantois near the insertion of the umbilical vessels (red
All tissues were processed (Laverty Vetnostics, North Ryde, New South Wales 2113) using standard histologic techniques to produce 6 µm sections stained with Wright’s modification of HE. Other stains such as Gram, periodic acid-Schiff (PAS) for fungi, and Steiner stain for spirochaetes were performed on selected tissues.
Bacteriology
Tissue aspirates of lung (42/46 cases), stomach contents (40/46 cases), and liver (2 neonates/46 cases) were routinely cultured for 5 days at 37°C for aerobic, facultative, and capnophilic bacteria as previously described, without broth enrichment. 39 Bacteriology was not performed on 4 treated term foals. Agars used were horse blood (HBA), HBA + CNA, and MacConkey no salt. Capnophilic conditions were achieved using CO2GenTM2.5L (all from Oxoid Thermo Fisher Scientific, South Australia, Australia). Smears of the aspirates were routinely assessed for bacteria using Gram stain and cells using Quick Dip stain (POCD Scientific, New South Wales, Australia). Bacteria were considered significant if grown in cultures of 1 or 2 isolates and smear examination revealed consistent bacteria and inflammatory cells. Mixed cultures of more than 2 isolates of environmental or enteric bacteria were not considered significant. Significant bacteria were identified to genus level using standard identification systems of commercially available biochemical test and serological typing kits. Once identified, bacterial isolates were correlated with microscopic pathology to determine final significance.
Immunohistochemistry
Formalin-fixed, paraffin-embedded tissues were immunolabeled for Chlamydia lipopolysaccharide (LPS) using a primary mouse monoclonal antibody against Chlamydia spp group-specific LPS from C. trachomatis that cross-reacts with C. psittaci and C. pneumoniae (Clone 4B10F; Thermo Fisher Scientific). Endogenous peroxidases were blocked with hydrogen peroxide and trypsin digestion was used for antigen retrieval. Sections were incubated with primary antibody diluted to 1/50 for 1 h, washed, and sequentially incubated with antimouse serum (Dako EnVision™), and 3-Amino-9-ethylcarbazole (AEC) substrate–chromogen (Agilent). Slides were counterstained with Mayer’s hematoxylin and mounted with Dako Faramount aqueous mounting medium.
Cases for IHC were selected based on the integrity of the tissues as determined by histologic assessment; tissues that displayed advanced autolysis were excluded. Twenty cases were examined and included 1 case where PCR produced a discrepant result due to incongruence in results when tissues versus swabs were tested. Tissues from an avian case of psittacosis (lung, liver, spleen, and kidney) were used as positive IHC controls. Nine tissues from 3 equine abortion cases with negative C. psittaci qPCR were used as negative IHC controls. Two had bacterial placentitis; 1 with focal suppurative chorionitis and the other with equine amnionitis and fetal loss (EAFL). Tissues from the third case were grossly and histologically normal and the cause of the abortion was uncertain. Tissues tested by IHC included lung, liver, spleen, thymus, amnion, and chorioallantois.
Polymerase Chain Reaction
Samples for PCR were collected aseptically via swabs and small tissue pieces before any other samples were collected. The sample collection protocol varied during the period reviewed due to changes in testing procedures and concerns over environmental chlamydial contamination of placental samples. The samples were variably pooled as follows: (1) pooled swab from fetal organs and placenta, (2) pooled swab from fetal organs and pooled swab from placental sites, and (3) pooled fetal tissue samples and pooled placenta tissue samples. During 2018, all fetal and placental tissues were collected individually using sterile procedures and separate sterile instruments for each organ and then were stored individually to prevent the cross-contamination of organs. The sites sampled were the same as for histology. In addition, both the chorionic and allantoic surfaces of the chorioallantois and the fetal and allantoic surfaces of the amnion were sampled at the respective collection sites. All swabs were suspended in phosphate-buffered saline containing 0.1% gelatin (PBGS) for transport and storage at -80°C. Tissue samples were stored at -80°C. Swabs were used for initial PCR testing. The stored tissue samples were used for quantification of chlamydial load in cases from 2016, 2017, and 2018, and confirmation or exclusion of a negative swab PCR where microscopic pathology was suggestive of chlamydial infection. The 5 cases with negative C. psittaci qPCR used as negative controls for the individual quantification of C. psittaci load included 3 cases of perinatal death due to hypoxia and 2 cases with long-term placentitis of unknown origin.
DNA was extracted from all samples using InstageneTM matrix (Bio-Rad). Twenty-five mg of tissue or a 100-µL aliquot of PBGS (for tissue and swab samples, respectively) was added to 200 µL of premixed matrix and incubated at 56°C for 15–30 min. The sample tubes were vortexed vigorously and then placed in a heating block at 100°C for 8 min. Samples were then centrifuged at 13,000 r/min and the DNA-containing supernatant collected for PCR analysis. Samples from most cases (39 cases) were tested in a qPCR assay, while some historical cases had been tested by conventional PCR (cPCR) and were no longer available for retest using qPCR (7 cases from 2015).
The qPCR for C. psittaci was performed as previously described using PCR primers and probe targeting the C. psittaci OmpA gene.19,28 Dilutions of pET21a+ plasmid containing the OmpA gene were used as standards for quantifying the number of gene copies in each sample as described in a previous study. 19 Gene copies were calculated using the DNA concentration derived from comparison with the standards as previously described. 19
Gene copy numbers were compared across tissues to determine where the organism was most concentrated. A Wilcoxon test was conducted on gene copy data from paired fetal and placental samples yielding a 2-tailed P value.
The cPCR for C. psittaci was adapted from Madico. 25 Briefly, the PCR was performed in a 20-µl volume containing 0.25 µM CPS100 and CPS101 primers, 0.2 mM dNTPs 2.5 mM MgCl2, 0.7 U of Taq polymerase, and 1 µL of DNA template. Cycling conditions consisted of a 3-min denaturation step at 94°C followed by 35 cycles of denaturation (94°C, 30 s), annealing (53°C, 30 s), and extension (72°C, 40 s), and a final extension step of 72°C for 5 min. PCR amplicons were visualized on a 2% 0.5× Tris-borate ethylenediaminetetraacetic acid (TBE) gel.
Quantitative PCR for Leptospira spp was also performed to investigate this organism as an alternative cause of abortion in most cases, subject to the availability of samples (36/46). Instagene™ DNA extracts were tested for pathogenic leptospires using a qPCR targeting the 16S rRNA gene as previously described. 35
EHV1 PCR was performed on all cases as previously described. 12
Results
The 46 C. psittaci PCR-positive cases represented 10% of the equine abortions, premature births, and neonatal foal losses investigated during 2015–2019. The reported diagnostic categories for the remaining 396 cases were placentitis EAFL (12%), ascending placentitis (6%), long-term active placentitis (including nocardioform, 4%), unknown placentitis (8%), equine herpesvirus (5%), perinatal death (11%), cord occlusion (20%), poor perfusion (5%), other (congenital abnormality, twin, fetal infection, 2%), and unknown diagnosis (17%). C. psittaci PCR-positive cases were divided into 3 groups based on the necropsy findings, histologic findings, IHC results, and the presence or absence of other infectious or noninfectious disease. Group 1 was designated as fetal or foal loss due to C. psittaci infection (38/46 cases). This was based on the identification of Chlamydia by PCR, a predominant lymphohistiocytic placentitis, if placenta was available, positive IHC results, if performed, and the absence of any other infectious or noninfectious cause for the fetal or foal loss. Group 2 was designated as fetal or foal loss due to other infectious or noninfectious disease (2/46 cases). This was based on the presence of another disease as indicated by necropsy findings, histologic findings, tests for other infectious agents, absence of a lymphohistiocytic placentitis, and negative IHC results . Group 3 was designated as fetal or foal loss of uncertain cause (6/46 cases). This was based on necropsy findings, absence of a lymphohistiocytic placentitis, negative IHC results, if performed, and the absence of a conclusive cause for the foal or fetal loss. The individual data for each case is in Supplemental Table S1.
Clinical History
The 46 cases of equine reproductive loss were all from thoroughbred mares, resident in the Hunter Valley region of NSW across 15 studs. Eight studs had 1 sporadic case. Seven studs had multiple cases and of these, 5 had between 4 and 8 cases in 2016 and 2018. The numbers of cases observed each year, and the stage of pregnancy loss are listed in Table 1. Seven cases (7/46, 15%) were abortions and the remainder of the cases were premature foals (26/46, 57%) or term foals (13/46, 28%, Table 1). All abortions occurred in group 1 (Table 1). Of the 7 abortions, only 2 mares showed clinical signs of impending abortion or premature lactation. Of the 39 premature or term births, 27 mares were reported to have acute delivery of a dead premature or term foal; 1 of which delivered a term foal without initiating lactation. Pregnancy compromise was detected by placental ultrasound in 2 mares; 1 delivered a dead premature foal shortly after and the other was treated for 14 days with antibiotics before delivering a dead premature foal (cases 37 and 45, respectively, Supplemental Table S1). Eight mares delivered a severely compromised premature (1) or full-term foal (7), which received intensive care but died rapidly (Supplemental Table S1). History for the remaining 2 mares was not recorded. The weight of the 21 complete fetal membranes varied from 2.8 to 16 kg.
Numbers and stage of pregnancy loss of equine fetuses and foals with Chlamydia psittaci infection from 2015 to 2019.

Infection determined by positive C. psittaci PCR.
Abbreviations: IHC, immunohistochemistry; PCR, polymerase chain reaction.
Stage of pregnancy is gestational age in days calculated as the days since last service date. Abortion is pregnancy loss at < 290 days, premature birth is birth at 290–320 days of gestation, neonatal loss is term foals at >320 days. Neonatal loss includes 8 compromised foals born alive and treated in intensive care.
No cases occurred in 2019.
Group 1 fetal or foal loss due to C. psittaci infection (38/46 cases), based on the presence of a positive C. psittaci PCR, a predominant lymphohistiocytic placentitis if placenta was available or positive Chlamydia spp IHC results if done and the absence of any other infectious or non-infectious cause for the fetal or foal loss.
Gross Pathology
The complete fetus or foal was examined in all cases. Placental tissue was examined in most cases (43/46) but was unavailable for 3 full-term foals presented for intensive care. Parts of the placenta were missing in 22 cases (22/43). These included parts of the chorioallantois, amnion, or umbilical cord. In 1 case, only the amnion and umbilical cord was available. Consequently, a necropsy of a complete fetoplacental unit was performed on half the cases (23/46, 50%).
A summary of the gross lesions in all groups with the numbers examined is in Table 2. Most cases had minimal autolysis. Across all groups, some fetuses or foals were small or thin (50%, Table 2) with liquid intestinal contents and meconium staining of the fetus, amnion, umbilical cord and sometimes the allantois (51%, Table 2). One premature foal had a deformity of the right front leg involving the radius and ulna with fetlock contracture (case 43, Supplemental Table S1). Lung lesions were more common in groups 1 and 2 (76% and 100%, respectively, Table 2) and were similar in appearance across all groups. Affected lungs were variably uninflated or inflated and heavy with congestion, interstitial edema, and serosal petechiation of varying severity. No other abnormalities of fetal organs were detected.
Summary of gross pathology in aborted fetuses and premature births or neonatal foal deaths with Chlamydia psittaci infection.
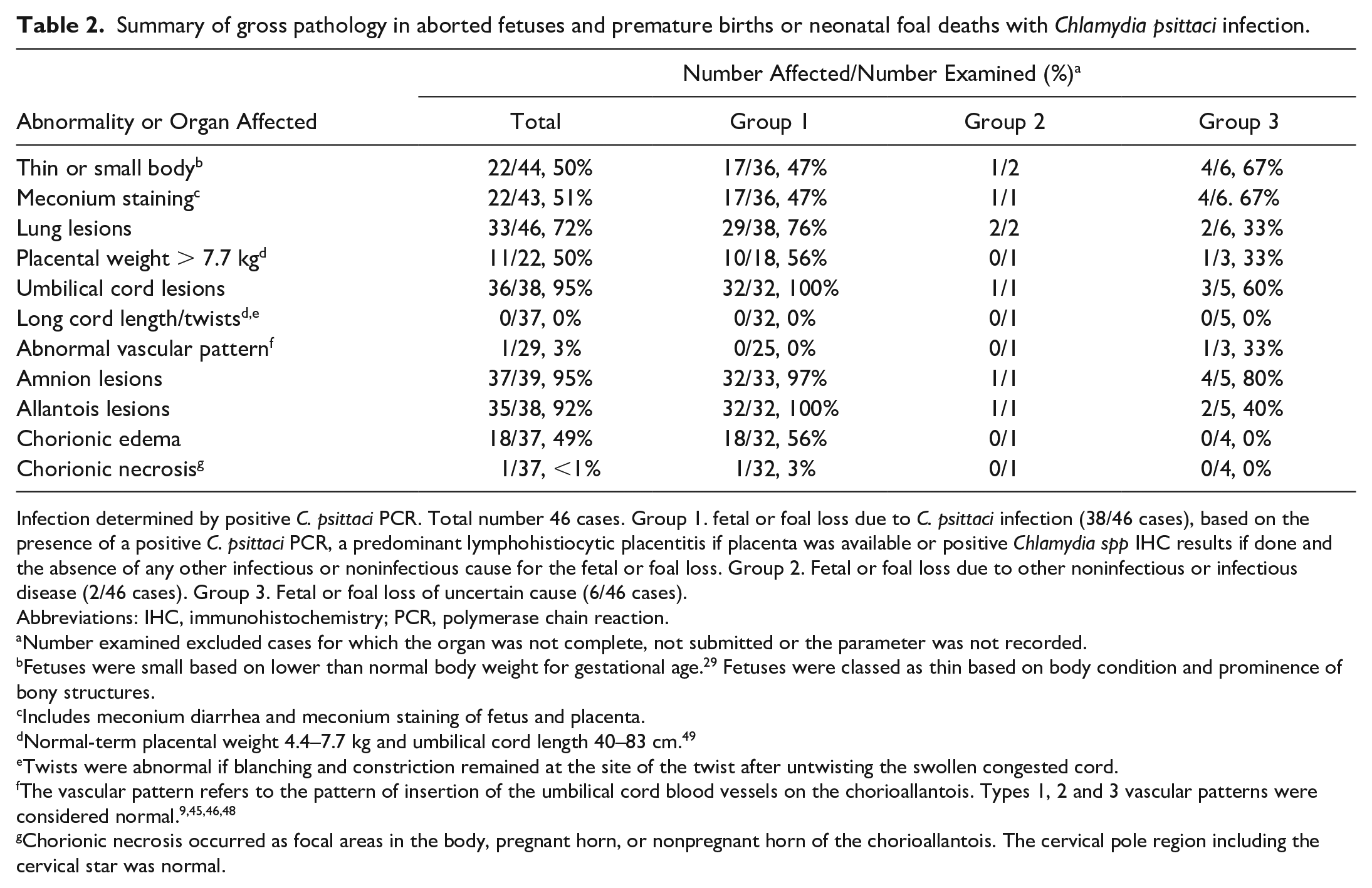
Infection determined by positive C. psittaci PCR. Total number 46 cases. Group 1. fetal or foal loss due to C. psittaci infection (38/46 cases), based on the presence of a positive C. psittaci PCR, a predominant lymphohistiocytic placentitis if placenta was available or positive Chlamydia spp IHC results if done and the absence of any other infectious or noninfectious cause for the fetal or foal loss. Group 2. Fetal or foal loss due to other noninfectious or infectious disease (2/46 cases). Group 3. Fetal or foal loss of uncertain cause (6/46 cases).
Abbreviations: IHC, immunohistochemistry; PCR, polymerase chain reaction.
Number examined excluded cases for which the organ was not complete, not submitted or the parameter was not recorded.
Fetuses were small based on lower than normal body weight for gestational age. 29 Fetuses were classed as thin based on body condition and prominence of bony structures.
Includes meconium diarrhea and meconium staining of fetus and placenta.
Normal-term placental weight 4.4–7.7 kg and umbilical cord length 40–83 cm. 49
Twists were abnormal if blanching and constriction remained at the site of the twist after untwisting the swollen congested cord.
The vascular pattern refers to the pattern of insertion of the umbilical cord blood vessels on the chorioallantois. Types 1, 2 and 3 vascular patterns were considered normal.9,45,46,48
Chorionic necrosis occurred as focal areas in the body, pregnant horn, or nonpregnant horn of the chorioallantois. The cervical pole region including the cervical star was normal.
Placental lesions were present in most cases across all groups often involving the amnion, umbilical cord, and allantoic surface of the chorioallantois and were more common in these regions in group 1 (97%–100%, Table 2). Placentas were greater than 7.7 kg in 10/18 premature or term foals in group 1 (Table 2, normal-term placental weight 4.4–7.7 kg 49 ). The amnion, umbilical cord, and the allantois of the chorioallantois in group 1 placentas were edematous with opaque, red surfaces, congested vasculature, and petechial or ecchymotic hemorrhages (Figs. 2, 3). In addition, 9/35 placentas were meconium stained. Group 1 cases had more severe allantoic lesions with concurrent chorionic edema and congestion (18/32, 56% Table 2). Chorionic edema and congestion were not seen in groups 2 and 3. The chorioallantoic lesions in group 1 were often extensive over the body near the insertion of the umbilical vessels and extended into the pregnant horn and cervical pole. Chorionic necrosis, characterized by extensive brown necrotic exudate covering thickened areas of the body and pregnant horn tip, was only present in 1 case (group 1, case 38, Supplemental Table S1). The amniotic, umbilical, and allantoic lesions of the 1 case with placenta in group 2 (case 39 with EAFL, Supplemental Table S1) were similar to those in group 1 except that the amnion had a sharply delineated area of pallor due to infarction. Placental tissues in group 3 cases were thickened, opaque and yellow due to meconium staining, without reddening, congested vasculature, hemorrhage, or edema. No other abnormalities of the umbilical cord including excessive length (>83 cm49) or twists causing vascular obstruction were detected (Table 2). One case in group 3 had an abnormal type 3 vascular pattern where one set of umbilical vessels supplied almost the entire chorioallantois with hypoplasia of the other vessels (Case 43, Supplemental Table S1). No other abnormalities of the placental unit were detected in any case.
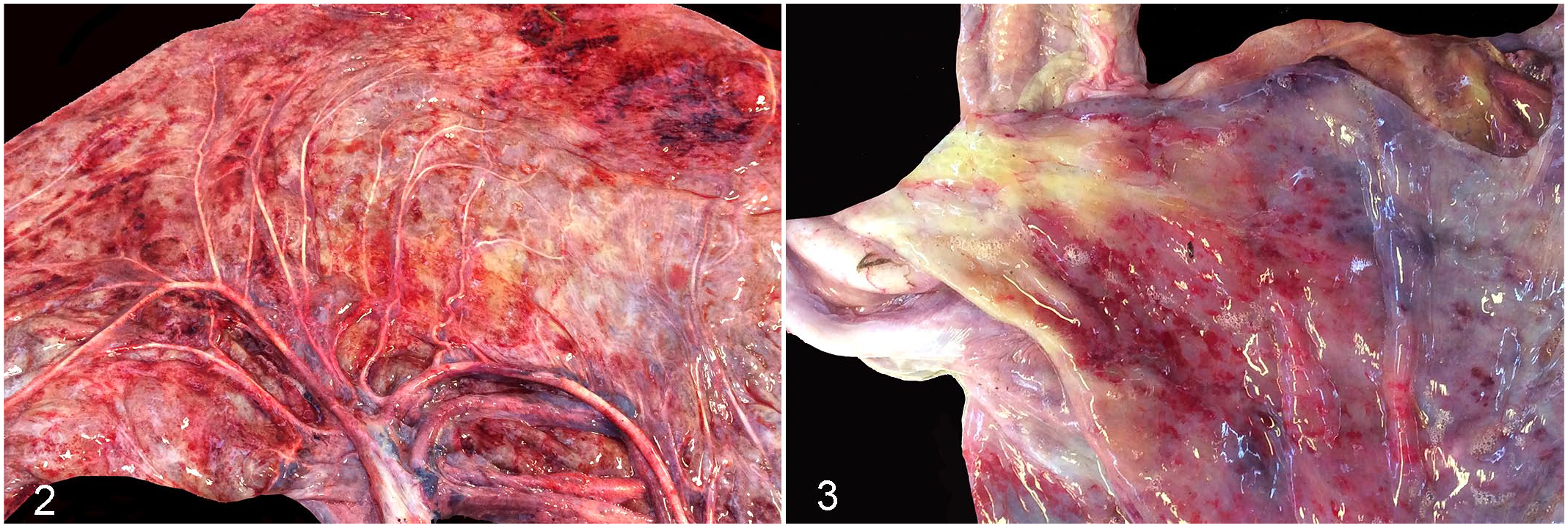
Chlamydia psittaci abortion, horse, placenta from term foal 32.
Microscopic Pathology
A summary of the main microscopic pathology findings in the 46 cases is in Table 3. Definitions for terms used in the microscopic pathology of lesions are in Supplemental Table S2.
Summary of microscopic pathology and IHC in the lung, liver, amnion, umbilical cord, and chorioallantois of aborted fetuses, premature births and neonatal foal deaths with Chlamydia psittaci infection.
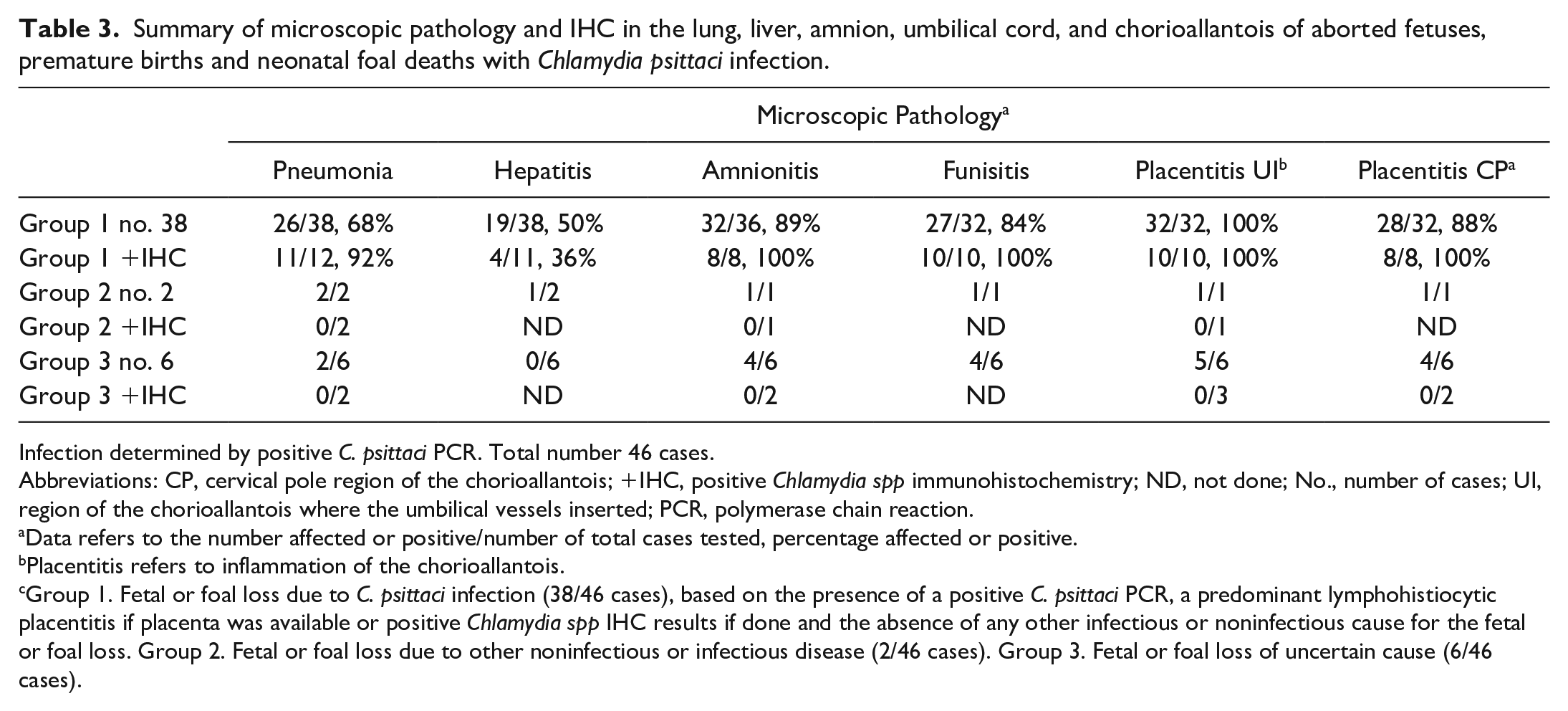
Infection determined by positive C. psittaci PCR. Total number 46 cases.
Abbreviations: CP, cervical pole region of the chorioallantois; +IHC, positive Chlamydia spp immunohistochemistry; ND, not done; No., number of cases; UI, region of the chorioallantois where the umbilical vessels inserted; PCR, polymerase chain reaction.
Data refers to the number affected or positive/number of total cases tested, percentage affected or positive.
Placentitis refers to inflammation of the chorioallantois.
Group 1. Fetal or foal loss due to C. psittaci infection (38/46 cases), based on the presence of a positive C. psittaci PCR, a predominant lymphohistiocytic placentitis if placenta was available or positive Chlamydia spp IHC results if done and the absence of any other infectious or noninfectious cause for the fetal or foal loss. Group 2. Fetal or foal loss due to other noninfectious or infectious disease (2/46 cases). Group 3. Fetal or foal loss of uncertain cause (6/46 cases).
Placenta
In group 1, all cases with placenta available had a similar pattern of predominant lymphohistiocytic placentitis, involving the amnion, umbilical cord and/or chorioallantois (36/38). Lymphohistiocytic placentitis was most frequently seen in the chorioallantois at the umbilical insertion region (100%), followed by the cervical pole region (88%) and as amnionitis (89%) and funisitis (84%, Table 3). In the chorioallantois, lymphohistiocytic chorionitis of the subvillous chorionic stroma extended to allantoitis with variable involvement of the coelom and the allantoic vessels, sometimes with extensive hemorrhage (Fig. 4). The severity varied between the 2 regions of the chorioallantois examined (Supplemental. Table S1) with the more severe reactions corresponding to areas of the chorioallantois with more prominent gross lesions. The inflammatory infiltrate consisted of mononuclear infiltrates of varying size (interpreted as lymphocytes and macrophages [lymphohistiocytic]) that surrounded blood vessels and extended to a diffuse infiltration pattern in more severely affected placentas. The pattern was a perivascular to diffuse predominantly lymphohistiocytic infiltration admixed with neutrophils in some placentas. There was variable perivascular to diffuse edema and vasculitis affected the veins radiating from villi to coelom and the allantoic arteries (Figs. 4, 6, 7). The vasculitis occurred as endothelial swelling, mural, and adherent luminal leukocytes with rare thrombosis, and rare fibrinoid necrosis of the media of arteries. The chorionic villi were generally intact with only small focal superficial lymphohistiocytic infiltrates in scattered villous stalks in less than half the placentas (Fig. 8) and rare aggregates of sloughed pyknotic or karyorrhectic trophoblasts and degenerate neutrophils. In some cases, examination of the villi was hampered by autolysis (5/38, cases 5, 6, 8, 9, 30, Supplemental Table S1). The coelom often contained scant fibrin, low numbers of macrophages, and lessor neutrophils with hemorrhage in some cases. The lymphohistiocytic placentitis was also found in the chorioallantoic horns in the 3 cases where additional sites were examined. The amnionitis and funisitis had predominantly perivascular lymphohistiocytic infiltration with perivascular edema, focal hemorrhage, and congestion (Figs. 5, 6, Supplemental Fig. S1) in all affected cases (89 and 84% respectively, Table 3). Vasculitis was present in many 20/38 cases (Supplemental Fig. S2). The inflammation was often more pronounced around the insertion of the amnion on the cord. The coelom less commonly contained scant fibrin, low numbers of macrophages, and scattered neutrophils (Fig.5, Supplemental Fig. S3). In 4 cases, there was scant superficial interstitial neutrophilic infiltration on the fetal surface (cases 3, 13, 16, 20, Supplemental Table S1). Variable congestion, hemorrhage, and mild edema were present in the amnion and umbilical cord in cases without inflammation and only 1 case had no abnormalities (case 17, Supplemental Table S1). Two cases had concurrent amniotic and/or chorioallantoic fibrosis indicating a long-term process (cases 37, 38 Supplemental Table S1). A prominent feature was marked irregular hyperplasia of the trophoblasts with layering/piling of trophoblasts (16/38, 42%), particularly where there was less edema and hemorrhage. This tended to affect the deep half to one-third of the villi, extending to intervillous areas and was usually diffuse, but sometimes multifocal (Fig. 8). Only 1 case (case 38, Supplemental Table S1) had locally extensive villous loss from necrosis with a focal long-term active placentitis as described (Supplemental Table S2). 17 Centrally, chorionic villi were replaced by a layer of squamous epithelium or stunted distorted villi covered in hyperplasic squamous epithelium. Thick layers of superficial, necrotic, neutrophilic exudate covered the area of villous loss and dense locally extensive lymphocytes and plasma cells with fibrosis expanded the underlying chorionic stroma. More acute placentitis was present at the edge with villous necrosis, dense neutrophilic inflammation and thick layers of necrotic exudate on the surface.
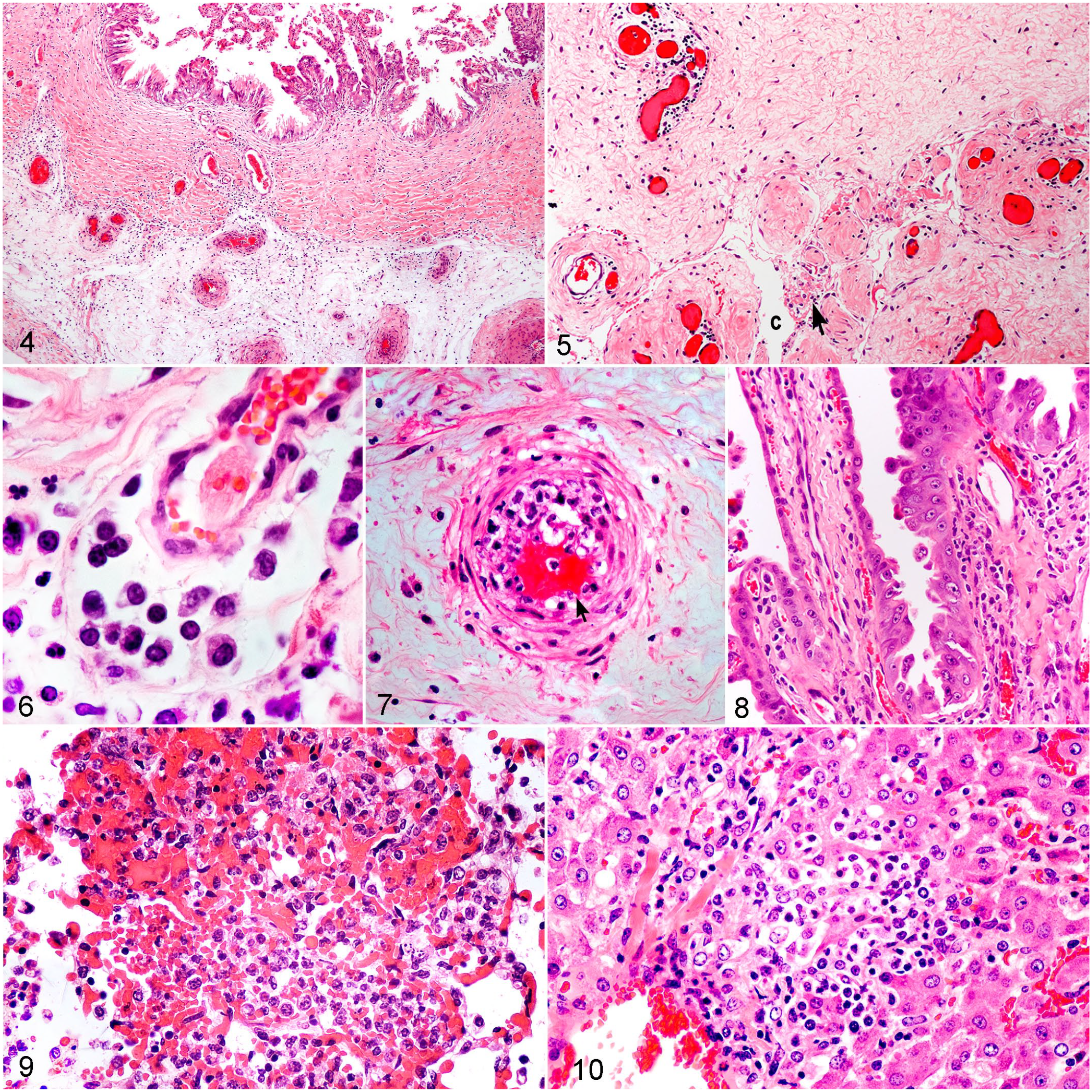
Chlamydia psittaci abortion, horse. Fetoplacental tissues from aborted fetuses, premature and term foals. Hematoxylin and eosin (HE).
In group 2, microscopic lesions were detected in the placenta of the only case with placenta available (case 39, Supplemental Table S1). This case had neutrophilic placentitis with fibrinopurulent amnionitis and funisitis with intralesional bacterial rods, leucocytoclastic vasculitis, and amniotic infarction. In the chorioallantois at both the umbilical insertion and cervical pole, fibrinopurulent inflammation encompassed the coelom and allantois only. This pattern of inflammation is typical of EAFL.
In group 3, all cases (6/36) had other patterns of placentitis which were not specific for any causes. The microscopic lesions were often mild (4/6) and frequently long-term (5/6) as indicated by fibrosis and allantoic epithelial hyperplasia (Supplemental Table S2). Mild superficial neutrophilic amnionitis and funisitis and fibrosis was observed in 2 cases (cases 42, 43). Case 43 also had patchy mineralization of the amniotic surface and mild lymphohistiocytic allantoitis associated with allantoic fibrosis and mineralization. Case 42 only had microscopic lesions in the chorioallantois as a diffuse interstitial lymphocytic infiltrate in low numbers in all layers and allantoic stromal fibrosis of the chorioallantois with mild papillary allantoic epithelial hyperplasia. Cases 45 and 46 were thin or small fetuses with severe meconium staining and autolysis hampering examination. Both had autolysed amnions, long-term superficial foreign body funisitis due to meconium with variable allantoic fibrosis. Case 46 additionally had multifocal adenomatous allantoic epithelial hyperplasia of the chorioallantois, chorionic vascular congestion with mineralization of trophoblasts and trophoblastic hyperplasia (Supplemental Table S1).
Fetus
Fetal and foal pneumonia occurred across all groups, but hepatitis was only seen in group 1 and in 1 group 2 case with EHV1 infection (Table 3, Supplemental Table S1). Across all groups, microscopic lesions in the lungs varied considerably often depending on the presence of meconium aspiration from fetal diarrhea or whether foals were born alive and treated intensively. Most cases had squamous epithelial cells within airways as evidence of amniotic fluid aspiration whilst intrauterine, although in 7 cases without pneumonia, this was considered within normal limits for equine fetal lungs (Supplemental Table S1). Meconium aspiration was also seen across all groups (17/46, Supplemental Table S1). In group 1, pneumonia occurred in 68% of cases (26/38, Table 3). In aborted fetuses and untreated premature or term foals (19/38), the pattern varied from multifocal bronchointerstitial or interstitial pneumonia with alveolar or bronchiolar necrosis, hemorrhage, and fibrin accumulation progressing to diffuse bronchointerstitial or interstitial pneumonia with more dense diffuse cellular infiltrates Mixed cellular infiltrates variably filled alveoli or bronchioles with predominant macrophages and lessor neutrophils (Fig. 9). Five cases had multifocal bronchopneumonia with neutrophilic infiltration; 3 of these cases had a large amount of aspirated squamous epithelial cells (Supplemental Table S1). All of the treated premature or term foals (7/7, Supplemental Table S1) had multifocal to diffuse bronchointerstitial or interstitial pneumonia, microscopically similar to the untreated cases, although this was more severe in 6 foals resulting in diffuse lung consolidation. Meconium aspiration was evident in 11/38 cases, but the amount of aspirated meconium did not always correlate with the severity of the pneumonia (Supplemental Table S1). Two cases (29, 37 Supplemental Table S1) had a severe diffuse foreign body pneumonia with luminal multinucleated giant cells containing meconium and squamous epithelial cells, and lymphocytes and plasma cells expanding the interstitium (Supplemental Fig. S4). One case had a large amount of recently aspirated material in airways with only mild inflammation (case 26, Supplemental Table S1.). The hepatitis in group 1 was characterized by scant lymphohistiocytic infiltration and perivascular edema in portal areas (19/38, Table 3). Occasional small inflammatory foci of less than 10 to 50 cells of mixed lymphocytes, macrophages, and fewer neutrophils were present in portal, perivascular, and midzonal areas (Fig. 9). Focal vasculitis of a larger portal vein was present in 1 case (case 11, Supplemental Table S1). In group 1, thymic and splenic lymphoid tissue was well-developed. Lymphoid atrophy was present in only 3 cases (cases 27, 28, 37 Supplemental Table S1). No lesions were detected in the kidney, when examined (cases 1, 2, Supplemental Table S1).
In group 2, case 40 with EHV1 infection had a multifocal necrotizing hepatitis, thymic necrosis, necrotizing bronchiolitis, and interstitial pneumonia. Intranuclear viral inclusion bodies were present in thymic reticuloendothelial cells, hepatocytes, and bronchiolar and alveolar epithelium. Case 39 with EAFL had multifocal to diffuse neutrophilic bronchopneumonia with aspirated meconium and foreign body multinucleated giant cells. Bacterial rods were evident within the neutrophilic exudate.
In group 3, pneumonia was in only seen in 2/6 cases (Table 3) with a large amount of aspirated meconium. One had acute bronchointerstitial pneumonia (case 46) and the other had diffuse long-term bronchointerstitial pneumonia with chronicity indicated by interstitial fibrosis, (case 45, Supplemental Table 1S1). The only treated case in this group, a premature foal, had a moderate amount of aspirated squamous epithelial cells and meconium without pneumonia (case 43, Supplemental Table S1). No liver lesions were detected in group 3 cases. Marked lymphoid atrophy of the spleen and thymus was present in 3/6 cases (cases 41, 43, 46, Supplemental Table S1).
Immunohistochemistry
IHC was performed on 78 selected tissues of 20 cases: specifically, 14/38 cases in group 1, 2/2 cases in group 2, and 4/6 cases in group 3. Thirteen tissues from 3 PCR negative cases were included as negative controls. Autolysis prevented IHC being performed on some cases and some tissues (Supplemental Table S1). In 4 cases in group 1 (33, 34, 35, 36, Supplemental Table S1), most tissues were tested to correlate the IHC data with the qPCR results on these individual tissue samples. IHC results for each group and tissues are summarized in Table 3, not including thymus and spleen. No positive immunolabeling was seen in any of 13 tissues examined from groups 2 and 3 or in the 9 tissues from the 3 Chlamydia negative control cases. Positive immunolabeling was seen in all 14 of the cases tested in group 1 (52/65 tissues). These cases had positive immunolabeling in all placental tissues examined and in 2 or more sites of the amnion, umbilical cord or both sites of the chorioallantois in each case (Table 3). In the chorioallantois, positive immunolabeling was predominantly in macrophages and some stromal spindle cells (Figs. 11, 12). Immunolabeling of villous trophoblasts was not as common (6/10 cases) as individual trophoblasts with positive cytoplasmic inclusions (Fig. 13). Interpretation was sometimes complicated by pigmented trophoblastic inclusions that are common in equine placentas, although these were not positive in negative control cases. The only case with extensive villous destruction (case 38, Supplemental Table 1) had strong positive immunolabeling of the sloughed necrotic trophoblasts and surface inflammatory exudate. In all cases, immunopositive interstitial macrophages in the amnion and umbilical cord were present near the amniotic fetal surface, around and free within the coelom, and occasionally concentrated in areas of inflammation, particularly at the amniotic attachment to the cord (Fig. 14). In 1 case, immunopositive labeling lined the fetal amniotic surface (case 11, Fig. 15).
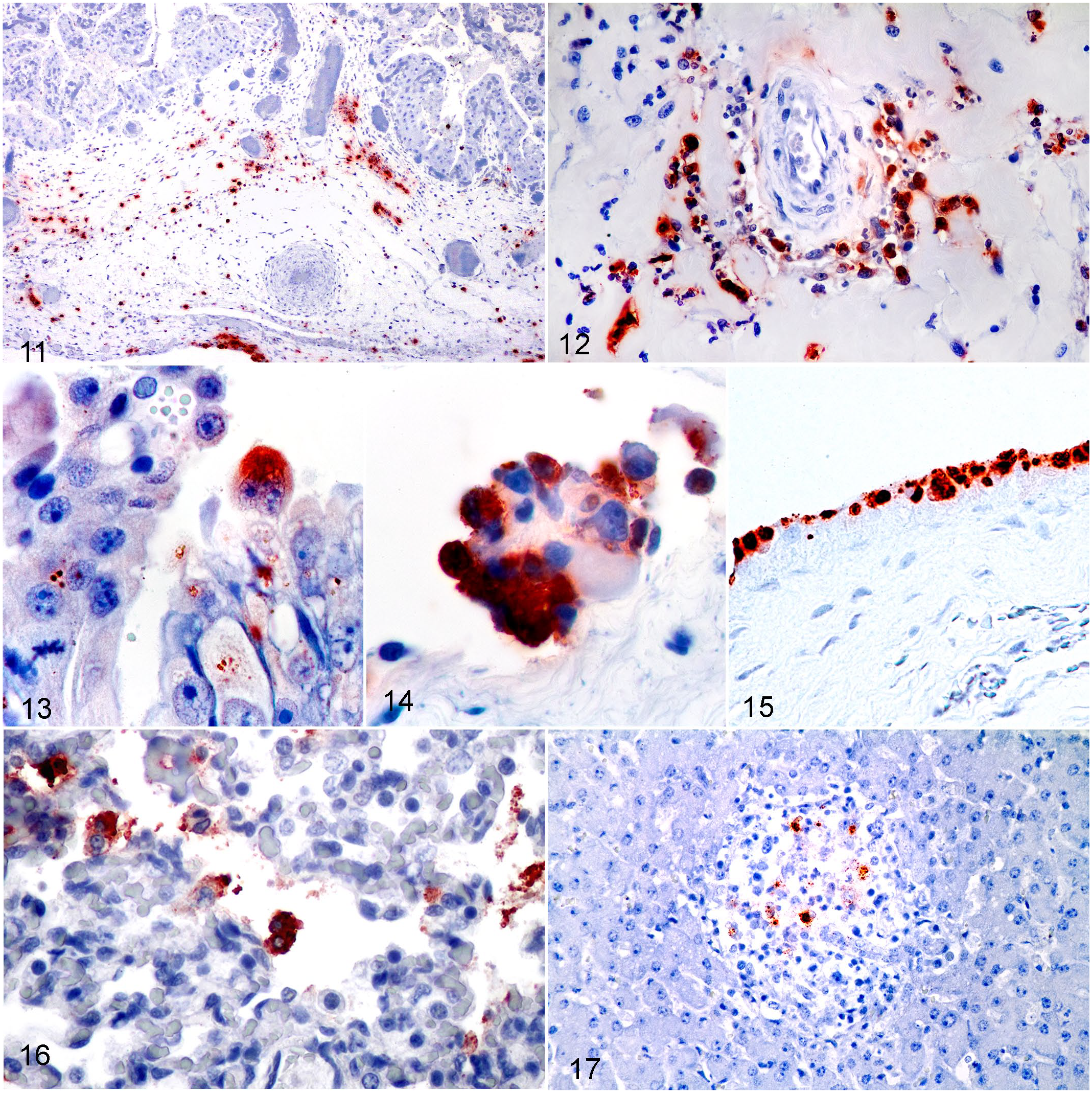
Chlamydia psittaci abortion, horse. IHC for Chlamydia spp in fetoplacental tissues form aborted fetuses, premature, and term foals.
Lung tissue was positive in 85% of group 1 cases tested (11/12), the negative case being case 37 with severe diffuse long-term bronchointerstitial pneumonia and marked meconium aspiration. Alveolar macrophages and epithelial cells were variably immunopositive within inflammatory foci or with immunopositive aspirated debris in alveolar spaces and bronchioles (Fig. 16). Case 11 with the immunopositive layer over the amniotic fetal surface had a large amount of immunopositive material in alveoli indicating immunopositive aspirated amniotic fluid. Liver tissue was positive in 40% of group 1 cases tested (4/10), with positivity reflecting the presence of hepatitis in all but 1 case with moderate hepatitis. This case had long-term active placentitis (case 38, Supplemental Table S1). The immunopositive mononuclear cells were present within hepatic lymphohistiocytic foci (Fig. 17) and occasionally in mononuclear cells in sinusoids. Rare immunopositive mononuclear round cells were seen in the splenic red pulp in the 3 group 1 cases tested (cases 33, 34, 35), but no positive immunolabeling was seen in the thymus of these cases.
C. psittaci PCR and Quantification of Chlamydial Load in Tissues
PCR was used to screen samples from equine pregnancy loss cases for the presence of C. psittaci DNA. qPCR was performed on 40 cases from 2015, 2016, 2017, and 2018. Six cases from 2015 were positive using cPCR, but stored tissues were not available for retesting by qPCR (Supplemental Table S1). qPCR was also used to determine the number of gene copies in pooled placental versus pooled fetal tissues and to examine whether any specific fetal or placental tissues were more likely to contain high loads of the pathogen. Comparison of paired pooled fetal and pooled placental tissues from all cases showed that the mean load of C. psittaci was statistically higher in placental rather than fetal tissues (Fig. 18). However, when individual tissues were examined in 4 cases in group 1 (Fig. 19), the load of C. psittaci was generally highest in the lung (>106 gene copies), followed by placental tissues, and the lowest in the thymus and spleen (<104 gene copies). When the load in pooled fetal and placental tissues of individual cases was examined, 89% of group 1 cases (30/34) had loads of C. psittaci greater than 1 × 103 in fetal tissues and/or placental tissues, with placental tissues typically being much higher. In comparison the loads of C. psittaci in group 2 and 3 cases were always less than 1 × 103 and mostly less than 1 × 102.
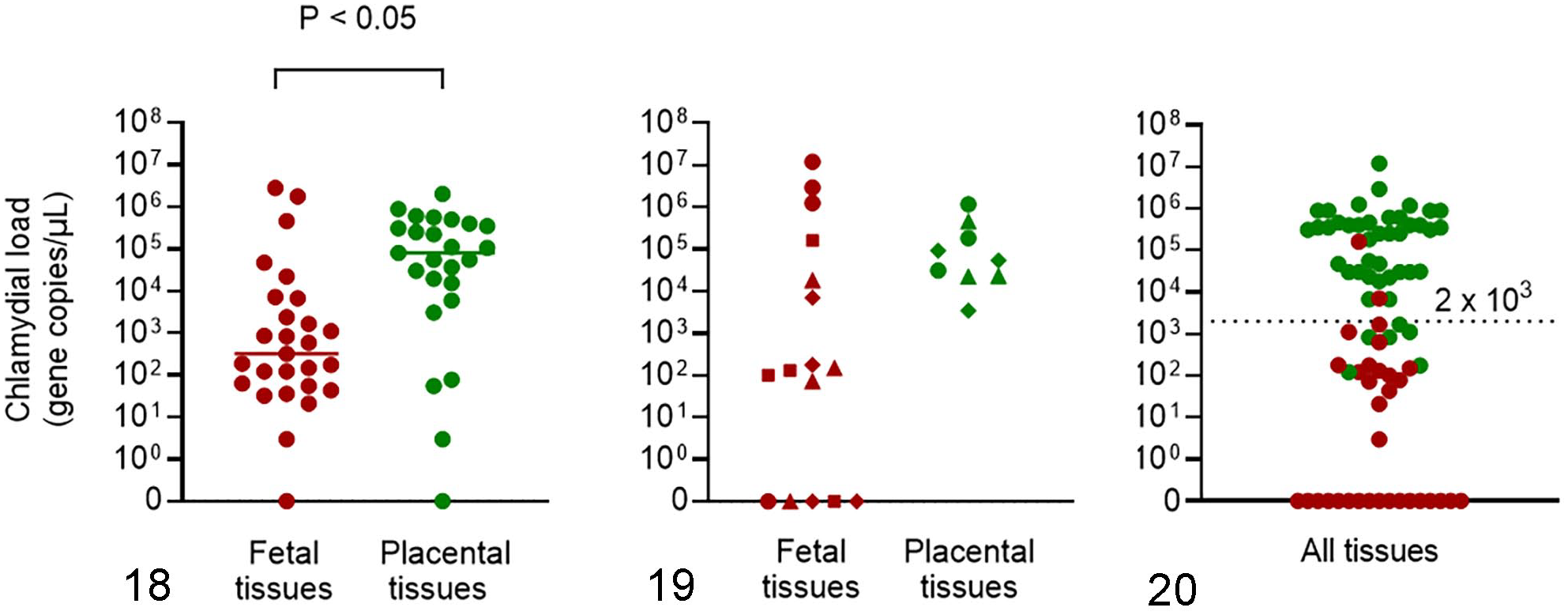
Quantification of C. psittaci qPCR load (gene copies/µl) in fetal (red •) and placental tissues (green •) from equine aborted fetuses, premature, or term foals (n = 27). The mean load was statistically higher (P < 0.05) in pooled placental tissues (n = 25) compared with pooled fetal tissues (n = 27, Fig. 18). In individual tissues (n = 4, Fig. 19), the load was generally highest in the lung (red •) followed by amnion (green •), umbilical cord (green ▲), chorioallantois (green ♦), liver (red ■), spleen (red ▲) and thymus (red ♦), but the variation was not statistically significant (mean not shown, P = 0.07).
Comparison of qPCR and IHC for Detection of C. psittaci
Of all the tissue samples examined, 75 had results for both qPCR and IHC. Comparison of each method for positive C. psittaci detection showed 83% congruence (Table 4) when a qPCR cut-off of 1 gene copy was used. IHC detected C. psittaci in 46/62 qPCR-positive tissues and was negative in C. psittaci qPCR-negative control cases (3 cases, 10 tissues). When test congruence was examined in relation to chlamydial load as determined by qPCR, samples with lower loads of C. psittaci were less likely to be detected as positive via IHC. Of the samples that were qPCR positive and IHC negative (8 samples), all except one sample had a chlamydial load below 2 × 102 gene copies (Fig. 20). Therefore, 2 × 102 gene copies appeared to approximate the limit of detection of the IHC.
Congruence between PCR and IHC on tissues from aborted fetuses, premature, or term foals with Chlamydia psittaci infection.
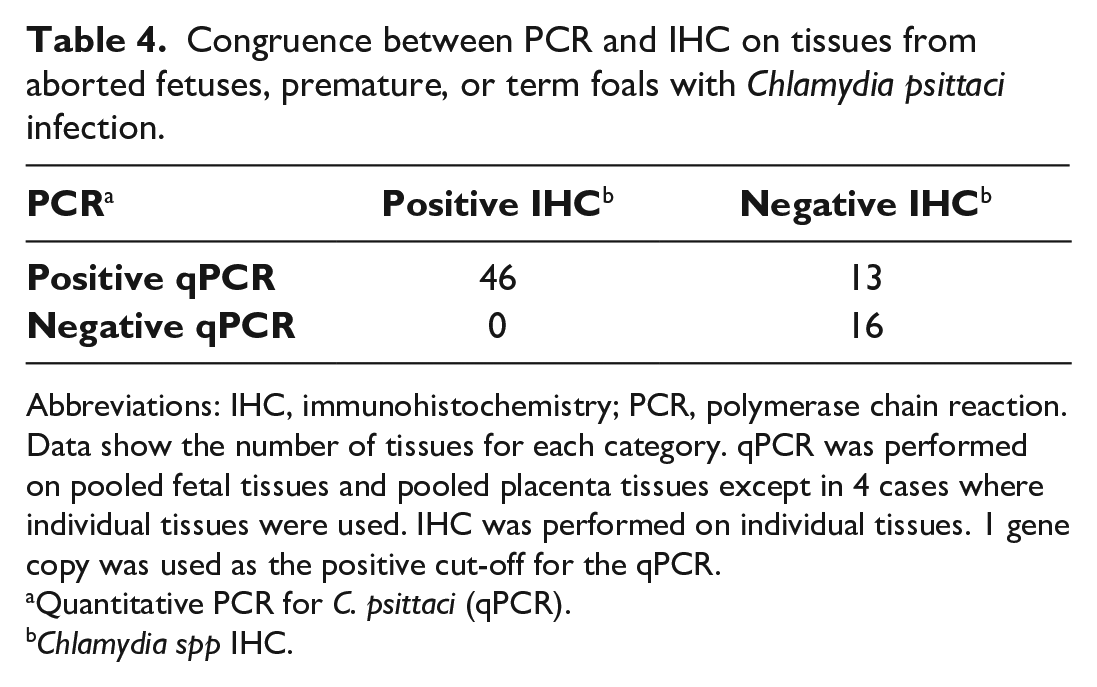
Abbreviations: IHC, immunohistochemistry; PCR, polymerase chain reaction.
Data show the number of tissues for each category. qPCR was performed on pooled fetal tissues and pooled placenta tissues except in 4 cases where individual tissues were used. IHC was performed on individual tissues. 1 gene copy was used as the positive cut-off for the qPCR.
Quantitative PCR for C. psittaci (qPCR).
Chlamydia spp IHC.
In the PCR-positive cases in group 2 and group 3, IHC failed to confirm chlamydial infection in all group 2 cases (2/2) and all of group 3 cases that were tested (4/6, Table 3, Supplemental Table S1). Of the group 3 cases, 2/6 had discrepant PCR results with disagreement between a negative historical cPCR and a positive qPCR when retested (cases 41, 46, Supplemental Table S1). This may reflect differences in assay sensitivity as the gene copy numbers in these samples were very low (between 3 and 78 gene copies) and were close to the limit of detection of the qPCR and thus not detected by the cPCR. The case in group 2 with EHV1 (case 40) was positive in both cPCR and qPCRs, but also had a very low gene copy number of 21, indicating that the presence of C. psittaci was not related to the pathology observed. qPCR was not available for the final 3 IHC negative cases (cases 39, 42, 44) in groups 2 and 3 as only the results from historical testing (cPCR) were available; 1 of these group 2 cases (case 39) was diagnosed as EAFL, consequently the Chlamydia PCR detection was unlikely to be the cause of foal loss.
Other Infectious or Non-infectious Diseases
Other infectious disease was identified in 3 cases: 2 in group 2 and 1 in group 3. EHV1 PCR tests were negative in 45/46 cases. One tested positive in all tissues (group 2, case 40). This positive test and accompanying microscopic pathology confirmed EHV1 as the cause of foal loss in this case. Leptospira PCR tests were negative for the 36 cases tested (36/46), across all groups. Samples were not available for the remainder.
Significant bacterial infections were identified in 2 of the cases tested (2/42 cases). The remainder (40/42) either had no bacteria or no significant bacteria isolated. Pantoea agglomerans was isolated from the lung and stomach contents of case 39 in group 2. This together with the microscopic pathology, including intralesional bacterial rods confirmed EAFL as the cause of foal loss. Enterococcus spp and Streptococcus agalactiae were isolated from the stomach contents, but not the lung of case 41 in group 3. The microscopic pathology of this case indicated a mild neutrophilic amnionitis without intralesional bacteria. This together with the negative IHC and low Chlamydia PCR load meant that the cause of the foal loss was uncertain.
Other noninfectious disease was only found in 1 case (group 3, case 43). This thin small premature foal had a congenital deformity of 1 front leg, an abnormal type 3 vascular pattern in the chorioallantois without microscopic evidence of placental ischemia or lymphohistiocytic placentitis. The placental lesions were long-term and nonspecific and marked autolysis meant that IHC was not done. The cause of loss was uncertain and significance of the C. psittaci infection was unclear.
Discussion
The main aim of this study was to describe the pathology of equine abortion, premature birth, and neonatal loss due to C. psittaci. A group of C. psittaci PCR-positive cases of equine reproductive loss was identified in which C. psittaci was confirmed as the cause of loss by a pattern of consistent pathological findings, immunolabeling of fetoplacental lesions for Chlamydia spp, quantification of chlamydial load, and exclusion of other causes. The percentage of cases (83%) for which the etiological role of C. psittaci was confirmed was much higher than in previous reports of C. psittaci infection in equine reproductive loss (14%). 37 In previous studies, a high prevalence of either concurrent other infectious or noninfectious causes, or absence of a conclusive cause of reproductive loss (89%) 37 meant that the etiological significance of the C. psittaci infection was questioned.4,37 In the series reported here, other concurrent infectious or noninfectious causes of loss were very low (4%) as were the cases without a conclusive cause of loss (13%). In addition, these factors were excluded in the cases where the conclusion was made that the C. psittaci infection was the cause of the reproductive loss.
A consistent pattern of lymphohistiocytic placentitis involving the chorioallantois, umbilical cord and amnion without chorionic trophoblastic and villous destruction was identified in association with C. psittaci infection in most cases in this series. The selection of cases based on a positive C. psittaci PCR introduced a bias as other cases of placentitis with a negative C. psittaci PCR were not reviewed for this pattern in this study. However, this pattern of lymphohistiocytic placentitis was not recognized in the Hunter Valley before C. psittaci infection was identified nor was it reported in the histopathology of any of the C. psittaci negative cases investigated during 2015–2019. Furthermore, the lymphohistiocytic pattern has been described in the chorioallantois previously in the only other report of substantial cases with confirmed C. psittaci infection by PCR and IHC. 37 We also identified lymphohistiocytic amnionitis and funisitis.
The pattern of lymphohistiocytic placentitis was different to most recognized infectious causes of equine placentitis. Most bacterial infections cause a necrosuppurative response with villous necrosis initially, irrespective of the mode of entry to the pregnant equine uterus as occurs in ascending placentitis and focal long-term active placentitis with variable fetal infection.7,17,43 A lymphoplasmacytic and histiocytic component may develop as lesions and infections become long-term. In leptospiral abortion in horses, the placentitis may be lymphohistiocytic, but is often mixed with predominant neutrophilic infiltrates and concurrent lesions including chorionic necrosis, chorionitis, nodular cystic allantoic adenomatous hyperplasia, fibrinopurulent funisitis, nephritis, giant cell hepatitis, pneumonia, and myocarditis.31,34,36 Leptospira infection was excluded in these cases by PCR. Caterpillar-associated placentitis recognized in North America as Mare Reproductive Loss Syndrome (MRLS) and in Australia as Equine Amnionitis and Fetal Loss (EAFL), results in bacterial infection causing funisitis in MRLS and amnionitis, funisitis and allantoitis in EAFL mostly without chorionic destruction.33,38–40,50 These are predominantly a suppurative response to bacteria that gain entry to the allantois, amnion, and umbilical cord likely spreading via the coelom with bacteria evident microscopically in the coelom and on surfaces of the amnion and cord as well as in the lungs.
The placental pathology found in confirmed cases in this study is different to ovine enzootic abortion due to C. abortus in sheep and to a lesser extent sporadic chlamydial abortion in goats and sheep due to C. pecorum.6,10,14,26,44 Both of these Chlamydia spp in natural and experimental abortions in small ruminants cause extensive necrosis of chorionic trophoblasts and villi with marked fibrinopurulent inflammation and prominent vasculitis, although some aborted ovine fetuses with C. pecorum primarily have a mild lymphohistiocytic placentitis.10,22,26,44 In this series, extensive chorionic villous necrosis was only found in 1 case with long-term active chorionitis. The presence of a dense lymphoplasmacytic cell population in the deeper chorion of this case suggested development of a fetal immune response, as was shown in experimental studies in sheep with C. abortus infection.6,22
Pneumonia and hepatitis were a feature of C. psittaci infection in this series occurring in 68% and 50% of cases, respectively. Fetal pathology as lymphohistiocytic hepatitis and lung necrosis has only been briefly described in 1 other equine abortion due to C. psittaci, although they were reported in some cases where the Chlamydia was not speciated.5,37 The lymphohistiocytic hepatitis was mild. The pneumonia in most cases was similar to that described in the neonatal foals previously as an acute interstitial or bronchointerstitial pattern with multifocal necrosis and mixed inflammatory cell infiltrates. 15 Differentiation from the necrotizing bronchointerstitial pneumonia caused by EHV1 infection is needed, although the chlamydial cases did not have necrotizing bronchiolitis, a feature of fetal EHV 1 infection. Fetal defecation, diarrhea, and meconium aspiration was a common finding, and this likely complicated the pattern when it was present for sufficient time to create an immunologic response. Fetal defecation with meconium staining of the fetus, fetal fluids, and placenta is a long recognized sign of fetal distress and hypoxia in people and many domestic animals.11,24,41 It commonly occurs in compromised equine pregnancies with placentitis, other placental abnormalities and birth hypoxia, so it was not considered a specific change of C. psittaci infection.41,47 Aspiration of meconium-stained amniotic fluid with keratin, squamous epithelial cells, and meconium initially causes a mild alveolitis with a few neutrophils and macrophages. 24 A diffuse pneumonia with large numbers of multinucleated foreign body-type giant cells often develops with time in equine fetuses, concurrent with reduced fetal growth, and this was present in a number of cases in this series both with significant and insignificant C. psittaci infection.
IHC and qPCR confirmed infection with C. psittaci as the cause of the placentitis and fetal loss in cases with the lymphohistiocytic placentitis. Quantitative PCR proved to be a useful screening test due to its high sensitivity and was able to detect down to 3 gene copies in the samples examined in this study. The chlamydial loads greater than 1 × 103 were comparable to that seen in aborted lambs with C. pecorum infection.10,44 Although different qPCR methods were used, loads of C. psittaci detected in cases of reproductive loss in this study (0.30 × 101–2.84 × 106; Fig. 18) were comparable with those detected in a recent retrospective survey of equine abortion cases across 3 Australian states (1.28 × 101–1.19 × 107). 2 In contrast, IHC was less reliable in detecting the presence of C. psittaci, and was mostly positive when higher loads of the organism were present (>2 × 102 gene copies). IHC was nonetheless useful for confirming that histopathological observations were a result of C. psittaci infection. The other cases in this series, including those with other diseases, were positive by C. psittaci PCR, but were not confirmed by IHC when it was performed. Most of these cases had a low C. psittaci load determined by qPCR. In some cases, the presence of significant other disease meant the chlamydial infection had not caused the pregnancy loss, and in others where no diagnosis was made, the chlamydial infection was of uncertain significance. Some of these cases may have represented a long-term manifestation of C. psittaci-induced loss as one of these cases had been treated with antibiotics and came from a stud with 7 cases. Environmental C. psittaci DNA has been demonstrated in avian feces on stud farms and this could contaminate equine placentas, which almost always have environmental contamination. 3 This could result in a positive PCR test that has no significance as a cause of loss and may affect the significance of C. psittaci detection in tissues where samples from the fetus and placenta are comixed or where poor aseptic collection technique allows cross-contamination. A positive qPCR on separately pooled fetal or placental tissue and positive IHC are likely needed to confirm significant infection where the placental pathology is not typical. From a zoonotic infection risk, a test with high sensitivity is desirable and so a lower specificity and some false positives may be acceptable.
How C. psittaci causes equine pregnancy loss is not fully understood. However, in this study, IHC demonstrated that infection of the chorioallantois resulted in invasion of chorionic trophoblasts similar to the route of fetoplacental infection with C. abortus in sheep.6,22,23,26 IHC further demonstrated subsequent spread to deeper tissues, the amnion cord and ultimately the fetus in contrast to the previous report of C. psittaci equine abortion where infection was restricted to the chorionic trophoblasts. 37 The lack of widespread or strong immunopositivity in trophoblasts and predominant immunopositivity in stromal macrophages in the subvillous stroma, coelom, amnion, and cord, and in pulmonary alveolar macrophages, suggested pregnancy loss occurred sometime after initial uterine infection and correlated with the subacute inflammation seen in many cases. This may also explain the lack of trophoblastic necrosis and the presence of trophoblastic hyperplasia.
IHC also suggested spread to the amnion and cord may be by infection of the coelom. The coelom is a space that is a remnant of the extraembryonic coelom and is extensive over large areas of the chorioallantois, the allantoic section of the umbilical cord and extends into the amnion for a short distance at the amniotic attachment to the cord. 49 Fetal infection can occur by aspiration of infected amniotic fluid or by hematogenous spread via the umbilical vein, which returns oxygenated blood from the placenta to the fetus. The presence of strong immunopositivity and the higher bacterial load by qPCR in lung tissue of some foals rather than liver tissue suggested fetal infection mainly occurred by aspiration of amniotic fluid, although the liver pathology and immunopositivity in some cases indicated hematogenous spread was also possible.
Conclusions
This report confirms the abortogenic effect of C. psittaci in naturally infected horses and describes a characteristic pattern of lymphohistiocytic placentitis and fetal infection. The findings confirm the need for a detailed fetoplacental pathological examination to confirm the etiological significance of C. psittaci infection as detected by PCR. Definitive diagnosis requires a positive C. psittaci-specific PCR, preferably qPCR on separate fetal and placental tissues combined with a thorough necropsy, fetoplacental histopathology, and exclusion of other infectious and non-infectious causes including leptospirosis. IHC may be helpful in confirming significant infection but is not as sensitive as PCR. Chorioallantois, amnion, umbilical cord, and lung are the preferred tissue samples.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221120008 – Supplemental material for Fetoplacental pathology of equine abortion, premature birth, and neonatal loss due to Chlamydia psittaci
Supplemental material, sj-pdf-1-vet-10.1177_03009858221120008 for Fetoplacental pathology of equine abortion, premature birth, and neonatal loss due to Chlamydia psittaci by Angela P. Begg, Joan Carrick, Catherine Chicken, Anna Blishen, Kristen Todhunter, Kieran Eamens and Cheryl Jenkins in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Laverty Vetnostics, Dr Bruce Duff and the histology staff for access to the historical HE slides and Scone Equine Hospital for access to historical records. The help of the staff of Scone Equine Hospital Laboratory is gratefully acknowledged.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by AgriFutures Australia. Project No. PRJ-011628
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
