Abstract
Tailgut cysts, also known as retrorectal cystic hamartomas, are rare congenital lesions with the potential for malignant transformation into adenocarcinoma, which presents significant diagnostic and therapeutic challenges. We present the case of a 41-year-old woman with a large presacral mucinous adenocarcinoma that arose from a tailgut cyst. Due to the tumor’s size and location, a complete surgical removal was not possible, resulting in an R2 resection. The patient subsequently received adjuvant chemotherapy with the CapeOX regimen, which led to a significant decrease in the size of the primary tumor from 11.5 cm to approximately 2 cm. This was followed by adjuvant radiotherapy. The patient currently has no evidence of radiological progression and remains on close follow-up. This case highlights that in situations where complete surgical excision of a malignant tailgut cyst is not feasible, adjuvant systemic chemotherapy and radiotherapy can be a highly effective treatment option for disease control.
Keywords
Introduction
Presacral tumors, which arise in the complex embryological junction of the retrorectal space, represent a rare and clinically challenging group of neoplasms. With an estimated incidence of 1 in 40,000, these lesions are pathologically diverse, encompassing both benign and malignant subtypes. The clinical presentation is often characterized by nonspecific symptoms such as pelvic pain, constipation, or urinary complaints, which can mimic more common anorectal or gynecological disorders, leading to misdiagnosis rates exceeding 30% and significant delays in treatment. 1 Though malignant transformation of a tailgut cyst in HIV-infected individuals is a rare occurrence, it is fundamental to understand the underlying mechanism for better management. The presence of HIV could potentially ameliorate the immune system and affect the behavior of these cysts, although specific knowledge on this mechanism is limited.
Tailgut cysts, also known as retrorectal cystic hamartomas, are among the most frequently reported types of primary retrorectal tumors. They are rare congenital lesions believed to be caused by persistent remnants of the embryonic postanal gut. These lesions typically develop in the retrorectal area and are most commonly identified in middle-aged women, with a female-to-male ratio of approximately 3:1. 2
The clinical presentation is highly variable, ranging from asymptomatic incidental findings to the aforementioned nonspecific symptoms resulting from mass effect.
While historically considered benign, there is a growing body of evidence that the risk of malignant transformation in tailgut cysts is substantial and may have been previously underestimated. 3 Early reports suggested a malignancy rate of approximately 8%–13%. 4 However, more recent systematic reviews of the literature indicate a neoplastic transformation rate as high as 26.6%, fundamentally shifting the clinical perspective toward a higher index of suspicion. 5
Adenocarcinoma, neuroendocrine tumors, and squamous cell carcinoma are among the reported malignancies. Mucinous adenocarcinoma is a particularly rare variant that presents significant diagnostic and therapeutic issues due to its histological overlap with other presacral or gastrointestinal malignancies.
The cornerstone of management for tailgut cysts, whether benign or malignant, is complete surgical excision to provide a definitive diagnosis, alleviate symptoms, and prevent recurrence or malignant progression. However, in cases of large, locally advanced, or unresectable malignant tumors, the optimal therapeutic strategy is not standardized. 6
This report describes a unique instance of mucinous adenocarcinoma that developed from a tailgut cyst in a 41-year-old woman, where an incomplete (R2) resection was followed by a remarkable response to adjuvant chemotherapy and radiotherapy. This case contributes valuable evidence to the limited literature on managing residual disease in this rare malignancy and highlights the efficacy of a multidisciplinary, multimodality approach.
Case presentation
This case report has been prepared in accordance with the CARE (CAse REport) guidelines. 7
Patient information and clinical findings
A 41-year-old female patient presented with a chief complaint of lower urinary tract symptoms along with constipation persisting for the last 5–6 months, for which she sought evaluation. She had no significant past medical or family history. On physical examination, abdominal fullness was noted, associated with a hard, palpable lump in the pelvic region.
Timeline of care
The key events in the patient’s diagnosis, treatment, and follow-up are summarized in Table 1.
Timeline of patient care.
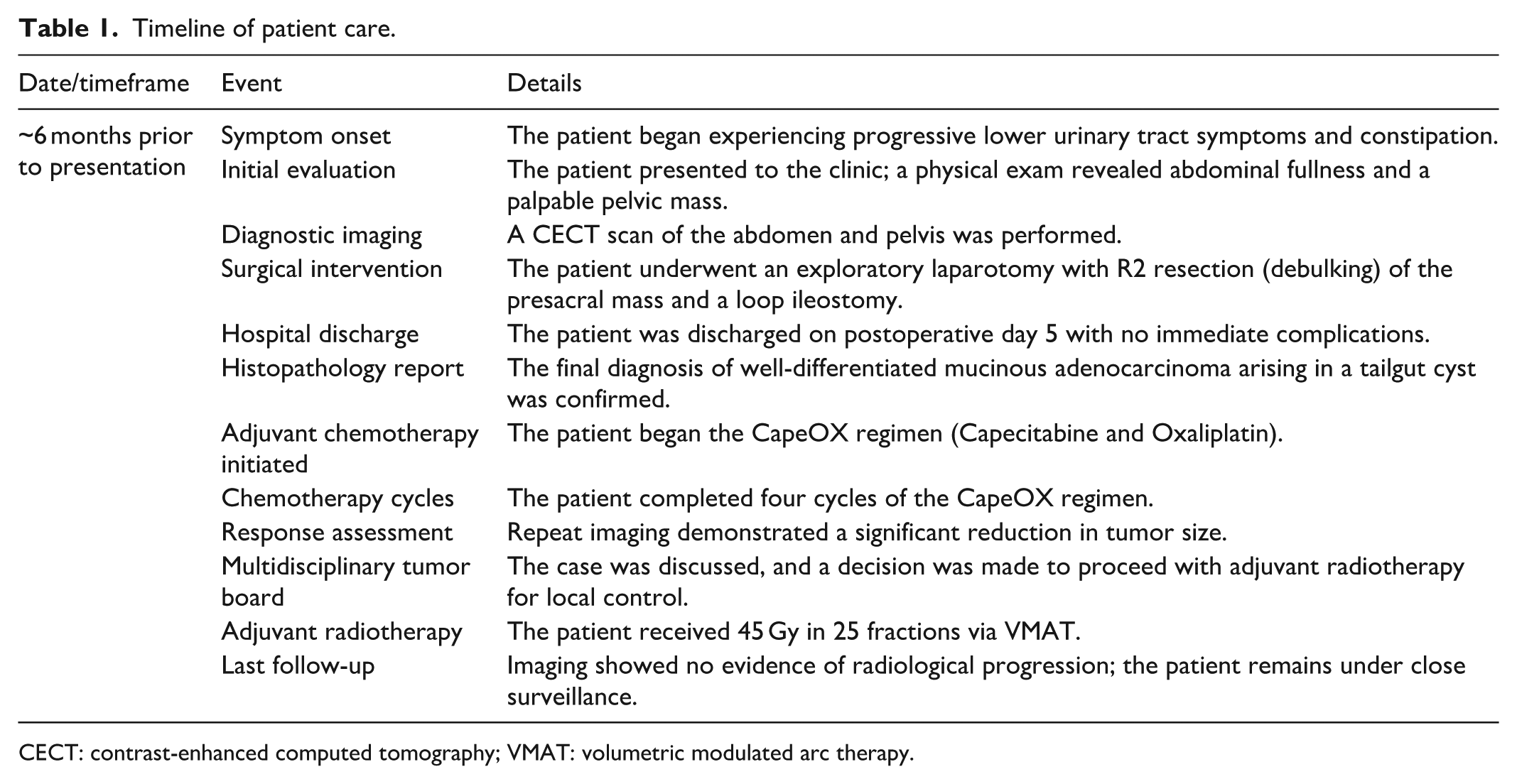
CECT: contrast-enhanced computed tomography; VMAT: volumetric modulated arc therapy.
Diagnostic assessment
A contrast-enhanced computed tomography (CECT) scan of the abdomen and pelvis revealed a multi-loculated, well-defined, heterogeneous cystic lesion (HU 28) with peripheral wall calcification in the presacral region, extending from the S1 to coccygeal level and measuring 11.5 × 10.3 × 9.4 cm (AP × T × CC). The lesion anteriorly displaced the uterus and displaced the rectum/recto-sigmoid colon toward the left. Posteriorly, the lesion was seen to closely abut the sacrum. Two additional, similar cystic lesions without wall calcification were observed anterior to the above-described lesion; the anterior-most lesion closely abutted the posterior wall of the uterus, and the other lesion was in close proximity to the sigmoid colon. The lesion also extended into the right ischioanal fossa. No obvious extension into the sacral foramen was noted (Figure 1).

Contrast-enhanced CT showing large presacral mass (11.5 × 10.3 × 9.4 cm) with local extension.
The patient subsequently underwent surgical intervention, and the resected tissue was sent for histopathological examination.
The gross biopsy showed a multiloculated cyst with cystic spaces filled with abundant mucin, separated by fibrocollagenous septae. The mucin was also seen infiltrating the cyst wall and intervening septae. The septae were, in places, lined by dysplastic columnar epithelium, displaying foci of papillaroid configuration. The lining cells in these areas displayed moderate nuclear pleomorphism, coarse chromatin, conspicuous nucleoli, and a moderate amount of eosinophilic cytoplasm, with foci of nuclear pseudostratification. Several papillaroid fragments were also seen embedded within the mucin. Scattered cells having a signet ring cell morphology were also seen embedded within the mucin pools. The fibrocollagenous cyst wall was surrounded by fibro-adipose tissue. Mucin was seen infiltrating the surrounding adipose tissue, inciting an extensive inflammatory reaction, comprising sheets of foamy macrophages, lymphocytes, and several multinucleated giant cells, along with lymphoid aggregates. In places, cholesterol cleft formation was noted. Multiple foci of calcification were also identified. Focally, the mucin was seen reaching the inked outer surface as well.
On immunohistochemistry (IHC), the tumor cells were immunopositive for cytokeratin 7 (CK7), cytokeratin 20 (CK20), and caudal-type homeobox 2 (CDX2). No nuclear staining was noted with PAX8. This IHC profile, particularly the positivity for CK20 and CDX2, is characteristic of a gastrointestinal phenotype, confirming the diagnosis of mucinous adenocarcinoma of hindgut origin (Figure 2).

(a) Pools of extracellular mucin are seen infiltrating the cyst wall, which shows hyalinization and fibrosis. Within these pools of mucin-embedded epithelial cells (arrow heads) are seen, arranged as fused glands, in a cribriform pattern and clusters. The cyst wall is lined in places by dysplastic columnar epithelium, with foci of papillaroid growth (black arrow; Hematoxylin and Eosin [H&E], 20×). (b) The lining cells in these areas display nuclear pseudostratification, moderate nuclear pleomorphism, coarse chromatin, conspicuous nucleoli and a moderate amount of eosinophilic cytoplasm (H&E, 200×). (c) Few foci show irregular, fragmented glands, infiltrating the cyst wall (H&E, 100×). On immunohistochemistry the tumor cells are positive for CK7 (D, 100×), CK20 (e, 100×) and CDX2 (f, 100×).
In view of CD20/CDX2 positivity, she underwent a full gastrointestinal evaluation, including colonoscopy, which showed no evidence of colorectal primary. Baseline serum CEA Serum Carcinoembryonic Antigen was also within normal limits (2 ng/ml).
Therapeutic intervention
Following the diagnostic workup, the patient underwent an exploratory laparotomy with an R2 resection (indicating macroscopic residual disease was left behind) with the creation of a loop ileostomy, with no post-procedural complications. She was discharged on postoperative day 5.
Post surgery, she was started on adjuvant chemotherapy with the CapeOx regimen (consisting of intravenous Oxaliplatin 130 mg/m2 on day 1 and oral Capecitabine 1250 mg/m2 twice daily on days 1–14, administered in 3-week cycles). She received a total of four cycles of chemotherapy. Adverse events were assessed per CTCAE v5.0: the patient experienced grade 1 nausea and transient grade 2 neuropathy, with no hematologic toxicity > grade 1.
After four cycles of the CapeOX regimen, a follow-up assessment showed that the size of the primary residual tumor had decreased to 18 × 21 mm, representing a significant clinical and radiological response.
After completion of chemotherapy, a multidisciplinary tumor board discussion was held to determine the next steps in management. Based on the initial incomplete resection (R2), the significant residual tumor burden post surgery, and the excellent response to chemotherapy, the final decision was made to proceed with adjuvant radiotherapy to consolidate local control. Radiotherapy (45 Gy/25#) was delivered to the residual presacral mass and postoperative bed with margins, using volumetric modulated arc therapy (VMAT). Clinical Target Volume (CTV) encompassed the gross residual and operative bed, with a 0.5 cm Planning Target Voluma (PTV) margin. Organ At Risk (OAR) constraints: rectum V45 <50%, bladder V45 <35%, small bowel V45 <15%. The patient tolerated treatment well, with only grade 1 diarrhea and no late toxicity at 6-month follow-up.
Follow-up and outcomes
Currently, the patient has no evidence of radiological progression at her last follow-up (post 6 months of treatment completion) and remains on close surveillance with pelvic magnetic resonance imaging (MRI) q6monthly.
Discussion
This case report details the successful management of a rare and complex clinical entity: a large mucinous adenocarcinoma arising in a presacral tailgut cyst. The key features of this case include the initial inability to achieve a complete (R0) surgical resection and the subsequent remarkable response to a multimodality adjuvant treatment strategy involving both chemotherapy and radiotherapy.
The diagnostic process in this patient highlights several important clinical considerations. The presentation with nonspecific pelvic symptoms is typical for tailgut cysts and often contributes to a delayed diagnosis. 8
A critical finding on the initial CECT scan was the presence of “peripheral wall calcification.” While not a pathognomonic feature, calcification is uncommon in benign tailgut cysts, and its presence should significantly raise the clinical suspicion for an underlying malignancy or a teratoma. This radiological “red flag,” combined with the large size and heterogeneous nature of the mass, strongly suggested a complicated or malignant process preoperatively. While CECT is valuable, MRI is often considered the preferred modality for evaluating such lesions due to its superior soft-tissue contrast, which aids in delineating the tumor’s relationship with adjacent structures such as the rectum, sacral nerves, and pelvic floor muscles, thereby facilitating surgical planning. 9
The cornerstone of treatment for malignant tailgut cysts is complete surgical excision with negative margins (R0 resection). 4 This approach offers the best chance for cure and prevents local recurrence. In this case, however, the large tumor volume and likely adherence to vital pelvic structures precluded a complete resection, resulting in an R2 (macroscopic residual disease) debulking surgery. This oncologically incomplete resection created an absolute indication for adjuvant therapy to manage the significant residual tumor burden.
The choice of systemic chemotherapy was guided by the tumor’s histopathology. Because tailgut cyst cancers are so uncommon, there are no established, evidence-based guidelines for the use of systemic chemotherapy. However, the histological and immunohistochemical findings of a mucinous adenocarcinoma with a gastrointestinal phenotype (positive for CK20 and CDX2) support a logical extrapolation from colorectal cancer therapy protocols. The CapeOX regimen (capecitabine plus oxaliplatin) is a standard, widely used first-line treatment for advanced and high-risk colorectal cancer, with proven efficacy. 10
In our case, four cycles of CapeOX resulted in a significant reduction in residual tumor mass from 11.5 cm at its largest diameter to approximately 2 cm, indicating a high level of chemosensitivity. This finding is consistent with a few isolated case reports in the literature in which fluoropyrimidine-based regimens (with or without oxaliplatin) have shown encouraging responses in adenocarcinomas developing from tailgut cysts. 11
Such positive results support the standardization of adjuvant chemotherapy for malignant tailgut cysts, particularly in patients with residual disease, high-grade histology, or positive surgical margins.
Following the excellent response to chemotherapy, the multidisciplinary team recommended adjuvant radiotherapy. This decision was made to provide consolidative local control for the remaining, albeit much smaller, tumor mass. The use of radiotherapy in this setting is a standard oncologic principle aimed at reducing the risk of local recurrence, which is a significant concern after an R2 resection. 12 Although radiotherapy in the pelvis presents challenges due to the proximity of sensitive structures, such as the rectum, bladder, and sacral nerves, modern techniques such as VMAT allow for highly conformal dose delivery, minimizing toxicity while effectively targeting the tumor bed.
This case demonstrates several important clinical lessons:
Tailgut cysts should be considered in the differential diagnosis of any presacral mass, particularly in adult women.
Radiological features such as large size, heterogeneity, and especially calcification should raise suspicion for malignant transformation, prompting referral to a specialized center.
In circumstances when complete surgical excision is not possible, adjuvant systemic chemotherapy with a colorectal cancer regimen such as CapeOX can be a highly effective treatment, capable of inducing a significant tumor response.
A multidisciplinary management approach involving surgeons, medical oncologists, radiation oncologists, pathologists, and radiologists is essential for optimizing outcomes in these rare and complex malignancies.
Conclusion
Mucinous adenocarcinoma developing from a tailgut cyst is an extremely rare clinical condition with insufficient data to guide a standard of care. This case highlights the necessity of contemplating malignant transformation in patients who present with large, complex presacral cystic lesions, particularly middle-aged women. While complete surgical excision remains the ideal therapeutic option, this case provides compelling evidence that adjuvant chemotherapy can offer significant benefit in cases with residual disease. Our patient had an excellent clinical and radiological response to the CapeOX regimen following debulking surgery, which was consolidated with radiotherapy, implying that a multimodality approach can play an important role in disease control for this patient population. This case emphasizes the importance of increased awareness, early suspicion, multidisciplinary therapy, and ongoing documentation of such unusual cases to establish a stronger evidence base for future therapeutic decisions.
Footnotes
Ethical considerations
The authors affirm that the case report titled “Presacral Mucinous Adenocarcinoma Originating from a Tailgut Cyst: Diagnostic and Therapeutic Challenges in a Rare Entity” has been prepared in accordance with ethical standards. Written informed consent was obtained from the patient for the publication of the clinical details and accompanying images. The patient’s identity has been anonymized to ensure confidentiality.
This report does not contain any experimental interventions and is based solely on standard diagnostic and therapeutic procedures. Institutional ethics committee approval was not required, as per the local regulations for retrospective single-case reports without intervention.
The authors declare no conflict of interest and confirm that the preparation and publication of this case report complies with the principles outlined in the Declaration of Helsinki.
Consent for publication
Written informed consent was obtained from the patient for the publication of this case report, including all relevant clinical information and images. The patient was informed about the nature and purpose of the publication and was assured that all personal identifiers would be removed to maintain anonymity. The patient understands that while efforts will be made to ensure complete confidentiality, absolute anonymity cannot be guaranteed. The patient has given full permission for this case to be used for academic and publication purposes.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this case report are available from the corresponding author upon reasonable request. Due to patient confidentiality and privacy concerns, supporting clinical documents and imaging cannot be publicly shared. However, anonymized data may be provided for academic or research purposes upon request, in accordance with institutional and ethical guidelines.
