Abstract
Schistosome infections in mammals cause chronic proliferative vascular lesions associated with the presence of adult parasites in the lumen of mesenteric and portal veins. In birds, however, this has never been reported. In this study, we found obliterative endophlebitis associated with the presence of adult schistosomes (Trichobilharzia sp., probably Trichobilharzia filiformis) as the main pathologic finding in five of eight mute swans (Cygnus olor). On histologic examination, the intestinal and portal veins of these swans showed moderate to severe, diffuse, hyperplastic endophlebitis, characterized by myointimal hyperplasia, often with obliteration of the vascular lumen. In addition, moderate to severe lymphocytic and granulocytic enteritis occurred in all eight swans associated with the presence of schistosome eggs in the intestinal mucosa. Other findings included hepatic and splenic hemosiderosis and high hepatic copper levels. The vascular lesions associated with Trichobilharzia sp. infection may have contributed to the emaciation and death of those mute swans by obstruction of venous return in the intestinal and portal veins.
Schistosomes of the genus Trichobilharzia commonly occur in various avian species, including waterfowl. In humans, they are known to cause cercarial dermatitis or swimmer's itch, an allergic response after repeated skin penetration of Trichobilharzia spp. cercariae. 9,25,28 The mute swan (Cygnus olor) is known to be the final host of Trichobilharzia filiformis, 2,15,22,26,28
Although this parasite has been found regularly in the viscera of mute swans, 22 associated lesions have not been reported.
In mammals, schistosome infections are often associated with serious disease caused by the presence of adult worms in the blood vessels and by their eggs, which pass through the intestinal mucosa or lodge in other tissues as foreign bodies. 9,20,24 Despite the known pathogenicity of some schistosomes for humans and domestic mammals, there is relatively little information of the effects of schistosomes on their avian hosts. 31 The largest genus of avian Schistosomatidae, covering more than 40 species, is Trichobilharzia. 8 Based on their predilection site, Trichobilharzia spp. can be divided in visceral and nasal species. Visceral species migrate through the viscera and can be found in mesenteric, renal, cloacal, and portal blood vessels, whereas nasal species also may display a neurotropic mode of migration. 9 The known lesions of infection with visceral Trichobilharzia spp.—to which T. filiformis belongs—vary according to the parasite and host species involved. For example, Trichobilharzia physellae infection in three species of ducks was associated mainly with portal fibroplasia in the liver, with viable adult schistosomes in the portal veins and dead schistosomes in the bile ducts. 18 In contrast, the main lesions of Trichobilharzia brantae infection in Atlantic brant geese (Branta bernicla hrota) were thrombosis of the caudal mesenteric vein and its branches associated with adult schistosomes in serosal and mesenteric blood vessels and fibrinohemorrhagic colitis associated with eggs in the intestinal wall. 32
The objective of this study was to determine the lesions associated with Trichobilharzia sp. infection in mute swans and to evaluate its role as a mortality factor.
Materials and Methods
Unusually high morbidity and mortality of mute swans were observed in the peninsula Walcheren, in the province Zeeland, The Netherlands (51.33°N, 3.35°E), between September 2002 and January 2003. In that period, 56 mute swans were submitted to the Bird and Hedgehog Sanctuary “De Mikke,” in Middelburg, from an area within 20 km of Middelburg, compared with 6 mute swans in the same period in 2000/2001 and 10 in 2001/2002. Of these 56 animals, 14 were submitted dead and 42 severely or moderately emaciated and dehydrated.
Necropsies were performed according to a standard protocol on eight swans, of which four were found dead (swan Nos. 1, 2, 7, and 8), three died in the Sanctuary (swan Nos. 3, 5, and 6), and one, with central nervous signs, was euthanatized (swan No. 4). Birds were categorized as male or female on the basis of the presence of testes or an ovary. Birds were categorized as immature if they still had brown feathers in their plumage, a pink-gray beak, and gray feet, whereas birds with a fully white plumage, an orange beak, and black feet were categorized as mature. 4
Samples of brain, liver, kidney, spleen, heart, trachea, lung, adrenal, thyroid, pancreas, crop, proventriculus, ventriculus, jejunum, ileumceca, colon, and cloaca were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4 µm, and stained with hematoxylin and eosin (HE) for light microscopic examination (swan Nos. 1–8). Sections of liver were also stained with rhodanine for copper and Perls' stain for iron (swan Nos. 6 and 7).
Virus culture attempts were performed using 10% homogenates of brain, liver, and kidney tissue inoculated in the chorioallantoic fluid of 10-day-old embryonated duck eggs and onto chick fibroblast cultures (swan Nos. 1 and 4). Eggs were monitored daily for embryo survival for a total of 7 days. Ten days after inoculation, the chorioallantoic fluid was harvested and a hemagglutination assay was performed as described previously. 29 Chick fibroblast cultures were monitored daily for 5 days for appearance of cytopathic effects. Cloacal swabs were examined for the presence of Newcastle disease virus and influenza virus by polymerase chain reaction (PCR) after the extraction of RNA using the High Pure RNA isolation kit (Roche Molecular Biochemicals, F. Hoffmann-La Roche Ltd., Basel, Switzerland) and using PCR conditions as described previously 6 (swan Nos. 1–7).
Bacteriologic examination was performed on liver and colon samples (swan Nos. 1–5) and lung and air sac samples (swan No. 1), using standard aerobic culture methods. Any parasites found were stored in 70% alcohol or 10% neutral buffered formalin and identified according to standard determination keys. 5,12,27
Frozen liver samples were tested for botulinum toxin by the mouse toxicity test (swan Nos. 2–7), and dry matter concentrations for copper and zinc were determined by inductively coupled plasma–optical emission spectrometry, in accordance with the method deduced from ISO/DIS 6869 (1997) (swan Nos. 1–7) and for lead by graphite furnace–atomic absorption spectroscopy, based on AOAC official method 972.25 (swan Nos. 1–7).
Results
Of all birds submitted to the Center, 55% (31/56) were juvenile, 28% (16/56) were adult, and the age category of the rest (16%, 9/56) was not determined. Of the eight mute swans examined, three were male (swan Nos. 1, 2, and 4) and four were female (swan Nos. 3, 6–8; the sex of swan No. 5 was not determined; Table 1). Six swans were immature (swan Nos. 1–4 and 6–7) and one was mature (swan No. 8; the age category of swan No. 5 was not determined; Table 1). Grossly, all eight necropsied swans were severely emaciated, as indicated by pectoral muscle atrophy, absence of fat depots, serous atrophy of pericardial fat, and hepatic atrophy. All swans had a multifocal reddening of the intestinal mucosa, most prominent in ileum, ceca, and colon. The main histologic lesions were found in the intestinal tract, liver, and lung (Table 1).
Summary of main lesions in eight mute swans.

In the intestine, lesions were found in the veins and in the mucosa. The intestinal veins showed moderate to severe, diffuse, hyperplastic endophlebitis characterized by myointimal hyperplasia, often with obliteration of the vascular lumen (swan Nos. 2, 5–8; Fig. 1). These changes were found throughout the length of the intestine and in all layers but appeared to be most severe in ileum, ceca, and colon, and in the serosa. Multiple adult schistosomes, including a pair with the female held within the gynecophoric canal of the male, were present in the lumen of the serosal veins of the ileum and ceca of swan No. 7 (Fig. 2), and multiple adult schistosomes were found in the serosal veins of duodenum, ileum, and ceca of swan No. 8. Adult schistosomes were identified as Trichobilharzia sp. on the basis of their specific morphologic features 5,15 and on host, site in the host, and earlier records of this genus in mute swans. 2,15,22,26,28 Characteristic morphologic features in parasite fragments from the mesenteric veins were a filamentous body, an oral and ventral sucker, and a spatulated body end. The width of adult parasites was about 0.1 mm, the length was more than 10 mm, and they had tandem testes. Based on the similarity to Szidat's 26 and McMullen's 15 species description, we tentatively identified these schistosomes as T. filiformis.
Cecum; swan No. 7. The serosal vein (arrow) has marked myointimal hyperplasia, with virtual occlusion of the vascular lumen. The wall of the accompanying serosal artery (arrowhead) is normal in thickness. HE. Bar = 37 µm.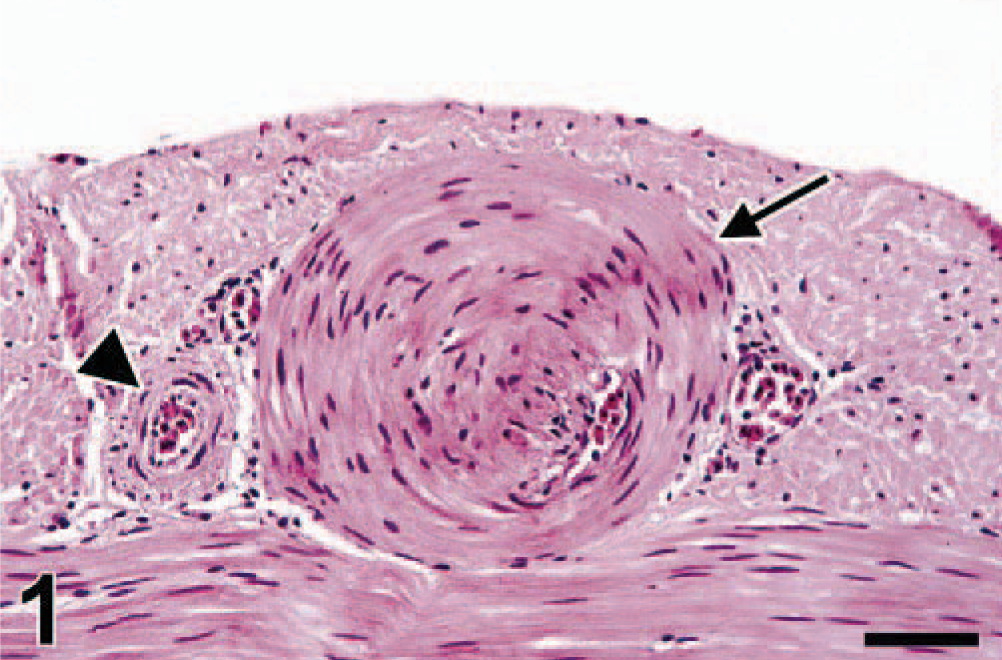
Cecum; swan No. 7. The distended serosal vein contains multiple cross sections of adult schistosomes (Trichobilharzia sp.). Some of these occur in pairs, with the male (arrow) carrying the female (arrowhead) in a ventral groove, the gynecophoric canal. HE. Bar = 72 µm.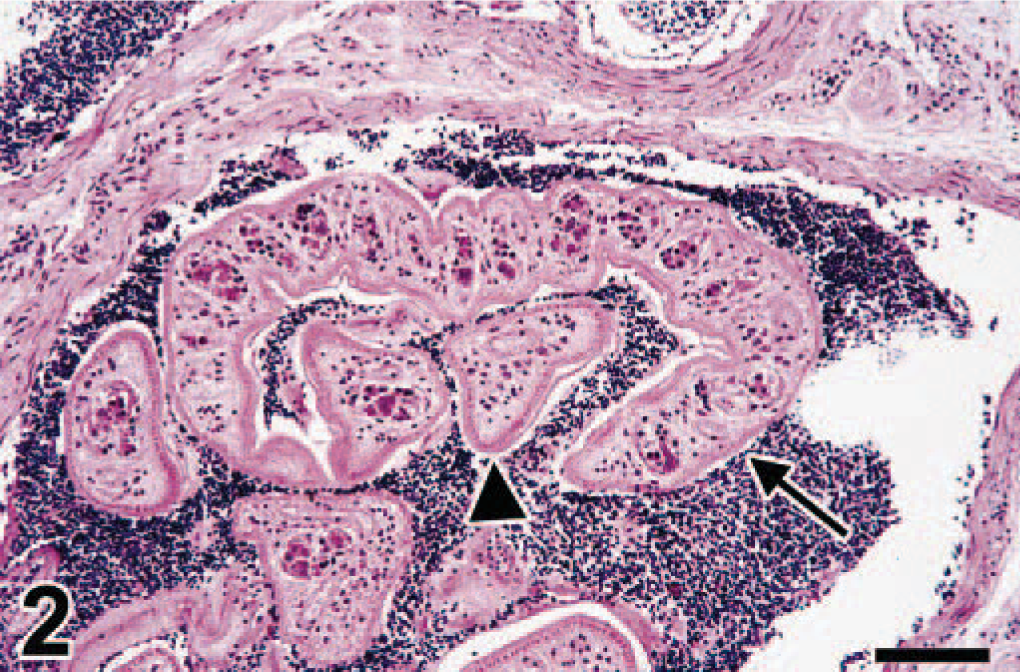
Intestinal mucosal lesions consisted of a moderate to severe superficial enteritis (swan Nos. 1–8), characterized by diffuse infiltration of the lamina propria of jejunum and ileum with moderate to large numbers of lymphocytes and plasma cells and fewer heterophils and eosinophils and superficial congestion (swan Nos. 1–8; Fig. 3). All swans also had mild to severe villous atrophy, characterized by villous blunting, fusion, and edema (swan Nos. 1–8; Fig. 3). Oval, thin-walled embryonated schistosome eggs, about 40 by 60 µm in size (swan Nos. 5–7; Fig. 3), were found multifocally in the lamina propria of small or large intestine, or both. Some of these contained a miracidium or had a small spike on one pole (Fig. 4). The inflammatory response around these eggs varied from none to severe granulomatous reaction, with macrophages, multinucleated giant cells, and granulocytes.
Ileum; swan No. 6. The lamina propria is diffusely infiltrated by lymphocytes and plasma cells, and the intestinal villi are markedly blunted. Superficially in the lamina propria are several schistosome eggs (arrows), surrounded by macrophages and multinucleate giant cells. HE. Bar = 147 µm.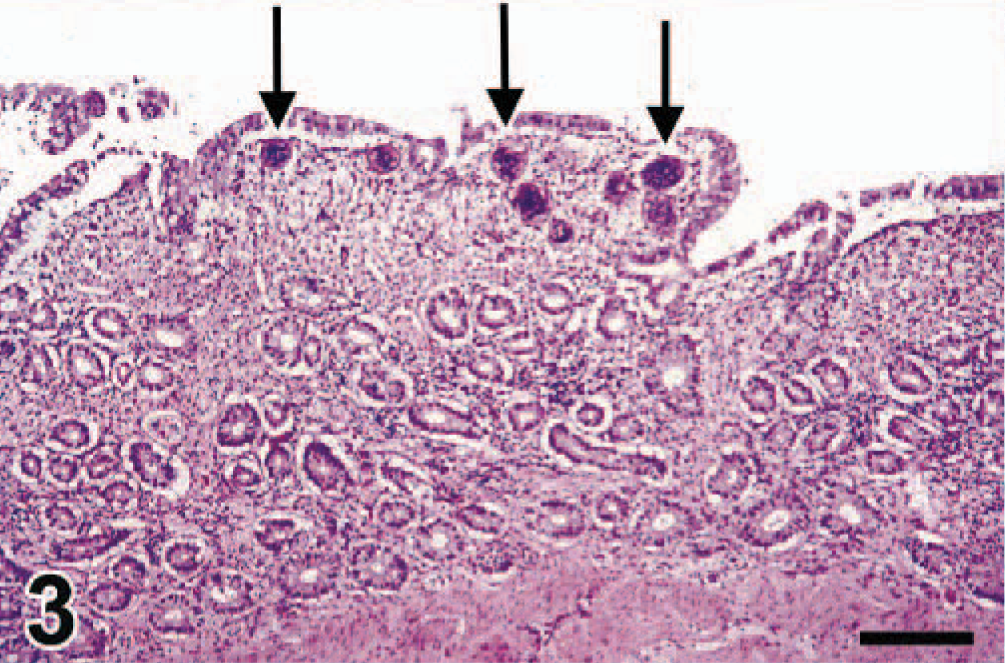
Duodenum; swan No. 7. Granuloma in lamina propria, centered around schistosome egg with characteristic terminal spine (arrow). HE. Bar = 14.7 µm.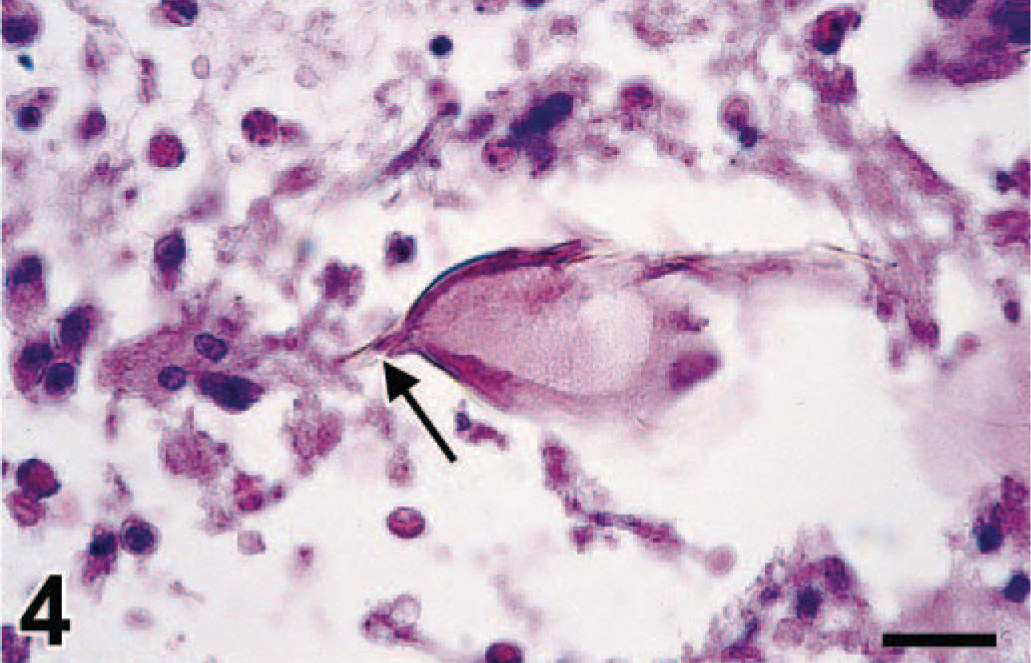
In the liver, the most prominent lesions were found in the portal venules in the portal triads. These had severe, diffuse, hyperplastic endophlebitis, similar to that in the intestinal veins (swan Nos. 5, 7, and 8; Fig. 5). A less prominent hepatic lesion was pigmentation of hepatic Kupffer cells by intracytoplasmic brown granules (swan Nos. 1–7), which were iron positive by Perls' stain for iron (moderate to severe hepatic hemosiderosis). A similar pigment was present in splenic macrophages (swan Nos. 2, 4–7; mild splenic hemosiderosis). Another pigment was visible diffusely in hepatocytes (swan Nos. 1–7) and consisted of intra-cytoplasmic light golden-brown granules, which were copper positive by rhodanine stain. Finally, there was mild portal fibrosis with periportal lymphocytic hepatitis (swan Nos. 1–3) and mild bile duct hyperplasia (swan Nos. 1 and 2).
Liver; swan No. 7. Within the portal tract, the branch of the portal vein (arrow) has marked myointimal hyperplasia, with virtual occlusion of the vascular lumen. The wall of the branch of the hepatic artery (arrowhead) is normal in thickness. There is mild bile duct hyperplasia (curved arrows) and lymphocytic infiltration of the portal tract. HE. Bar = 37 µm.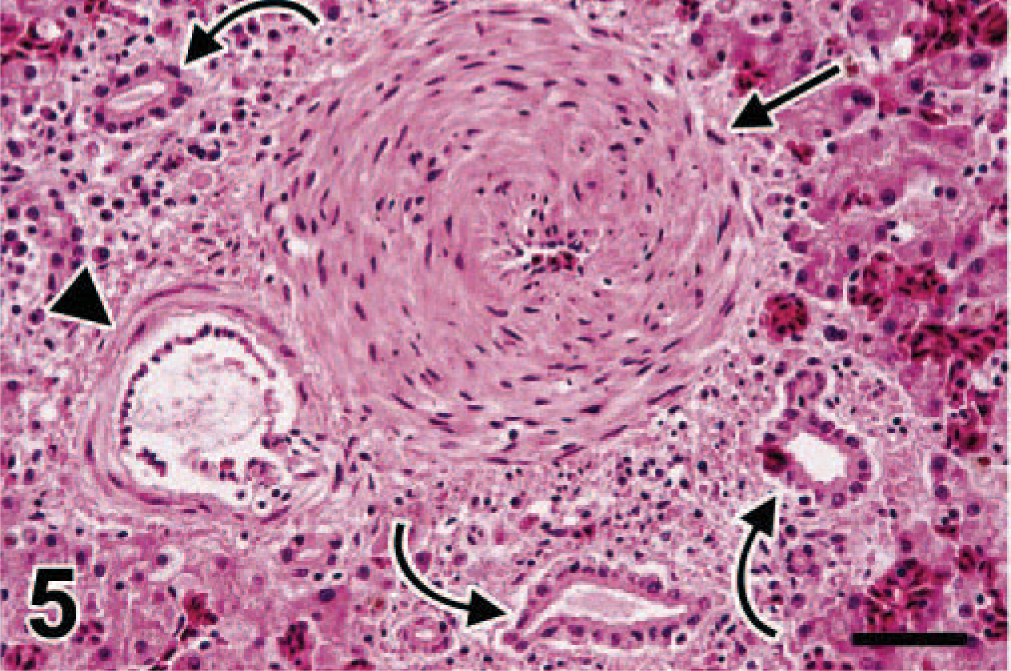
In the lung, swan No. 6 had multifocal granulomas associated with the presence of degenerate schistosome eggs, similar to those in the intestine (Fig. 6). Lung lesions not associated with schistosome infection were severe, chronic, multifocal to coalescing airsac-culitis combined with a moderate to severe, chronic, multifocal, hemorrhagic pneumonia associated with either characteristic Aspergillus-like hyphae or plant remains admixed with numerous aggregates of bacteria (food aspiration) (swan Nos. 1, 3, 7). Swan No. 1 had lymphocytic perivasculitis in the cerebrum without any obvious etiology (Table 1).
Lung; swan No. 6. There is a degenerate schistosome egg (arrow) in the lung parenchyma surrounded by foreign body–type multinucleate giant cells and epithelioid macrophages. HE. Bar = 37 µm.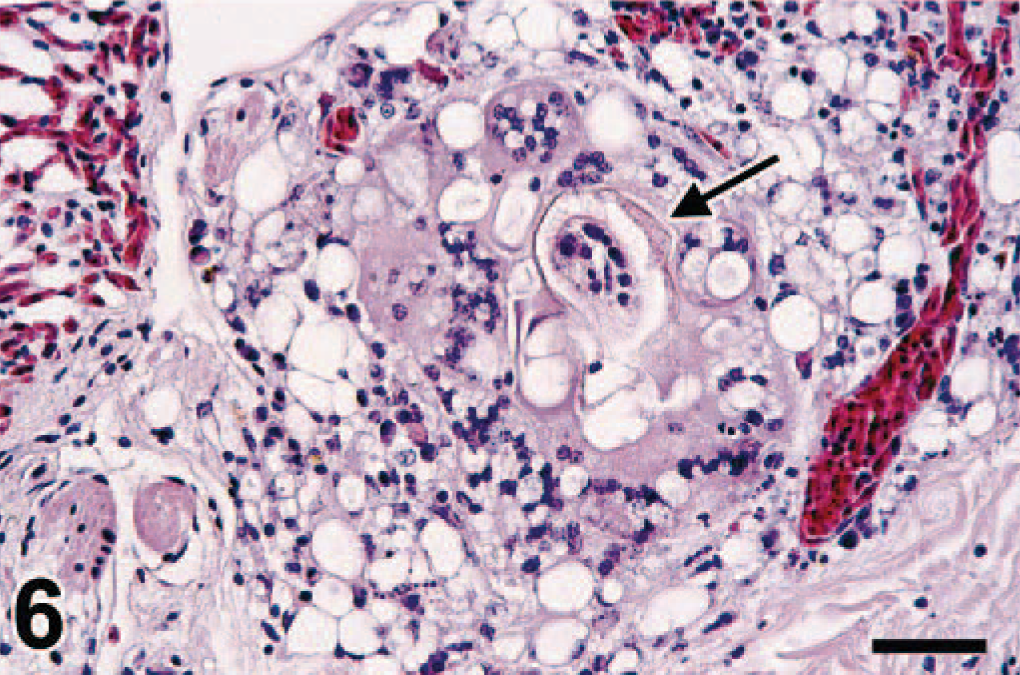
The hepatic copper levels were high compared with those reported previously in mute swans (Table 2) 3,7,13,16,24 and showed a greater range than those of zinc and lead (Table 2). 3,7,13,19,24 Neither virus infections nor significant bacterial infections were detected by virologic and bacteriologic examination, and liver samples were negative for botulinum toxin by the mouse toxicity test.
Dry matter concentrations (ppm) of copper, zinc, and lead in liver of mute swans from different locations and different studies (mean ± SD [range]).
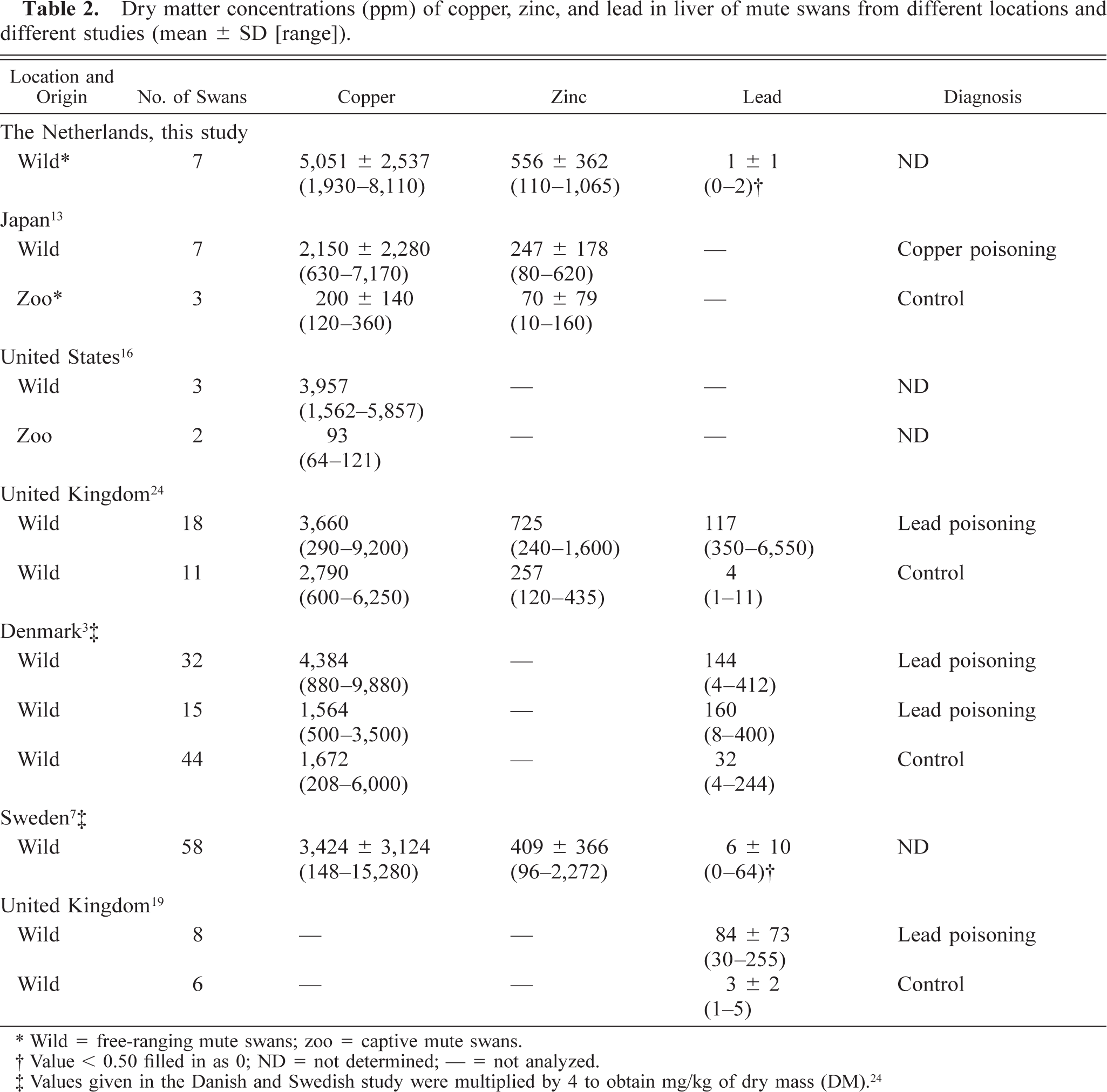
∗ Wild = free-ranging mute swans; zoo = captive mute swans.
† Value < 0.50 filled in as 0; ND = not determined; — = not analyzed.
‡ Values given in the Danish and Swedish study were multiplied by 4 to obtain mg/kg of dry mass (DM). 24
Discussion
In this article, we provide the first description of lesions associated with Trichobilharzia sp. infection in mute swans and schistosome-associated obliterative endophlebitis in an avian host. The detection of adult schistosomes at the site of the vascular lesions in two of five swans, together with the similarity in character and localization of the lesions to those reported in mammalian schistosomiasis, 20 indicate that infection with mature Trichobilharzia sp. likely caused the endophlebitis in these swans.
In mammals, adult schistosomes in mesenteric and portal veins elicit eosinophilic endophlebitis, often with irregular intimal proliferation. 20 In different experimental studies in mammals and birds, it is suggested that viable adult schistosomes cause proliferation of the vascular intima either by mechanical injury of the intima or by the secretion of antigens. 14,17,23,30 In mammals, severe cases of schistosomiasis may result in occlusion of the lumen of the veins, excluding the adults from the smaller venules. Furthermore, lesions may extend to the intrahepatic branches of the portal vein, leading to prominent portal fibrosis. 20 In this study, the extent and distribution of the lesions in intestinal and hepatic veins were consistent with the above described findings of schistosomiasis—or, more correctly, schistosomosis—in mammals.
The lesions associated with Trichobilharzia sp. infection in these mute swans show both similarities and differences to previously published lesions of visceral Trichobilharzia infection in other species of waterfowl. The lesions associated with Trichobilharzia sp. (probably T. brantae) in serosal and mesenteric veins of Atlantic brant geese consisted of thrombosis, extensive perivascular hemorrhage, and mononuclear cell infiltration. They were believed to be the result of the reaction to both schistosome eggs and adults. 32 These lesions are similar in location but more acute in character than those we observed in the intestinal veins of mute swans. A possible explanation is that the brant geese had been translocated and, therefore, may have been infected by a species of Trichobilharzia that they would not normally encounter. In contrast, Trichobilharzia sp. infection may be less pathogenic for mute swans because they are probably the normal final host. Three species of duck infected with T. physellae showed medial hyperplasia and periphlebitis of the portal veins. 18 This could be a more acute stage of the obliterative hyperplastic endophlebitis that we observed in the portal veins of the mute swans. The ducks with T. physellae infection also had mild to extensive fibroplasia of the portal triads, attributed in part to the aberrant localization of female schistosomes in the bile ducts. Although we saw mild portal fibrosis in three mute swans, we found no evidence of schistosomes in bile ducts.
Minor lesions caused by schistosomes are often associated with the presence of their eggs lodged in tissues. 32 On one hand, the migration of schistosome eggs through the intestinal wall causes chronic enteritis, which is a common histologic finding in waterfowl. 31,32 On the other hand, as occurs with human schistosomiasis, some eggs of Trichobilharzia sp. fail to reach the intestinal lumen and are instead disseminated through the circulatory system to the liver, lungs, and other organs where they cause reactions ranging from inapparent to marked granulomatous inflammation. 9 Antigens released by the schistosome eggs induce a delayed hypersensitivity response and formation of small granulomas, initially characterized by leukocytes, eosinophils, and mononuclear leukocytes, giant cells and reactive fibrosis. 20 The presence of Trichobilharzia sp. eggs in intestine and lung in these mute swans resulted in multifocal granulomatous inflammation and moderate to severe enteritis, similar to that caused by schistosome eggs in humans, 21 domestic mammals, 20 and other waterfowl species. 31,32
It is noteworthy that 55% (31/56) of all submitted swans and 75% (6/8) of the necropsied swans in this study were juvenile. This is in agreement with results from the field, where the prevalence of Trichobilharzia sp. infections in juvenile birds is higher than in adults because of increased immunity after repeated exposure to Trichobilharzia parasites. 8
Although it is likely that the Trichobilharzia sp. infection in these mute swans had a negative effect on their health, we cannot determine whether it was the primary cause of the increased morbidity and mortality of mute swans observed in Walcheren in the fall and winter of 2002/2003. The clinical signs of emaciation and weakness in these swans may have resulted from severe enteropathy associated with Trichobilharzia sp. infection. This enteropathy was caused on the one hand by obliterative vascular lesions attributable to the presence of adult schistosomes and on the other hand by enteritis with villous atrophy attributable to the migration of schistosome eggs. However, emaciated birds often carry heavy burdens of endoparasites, and it is simplistic to assume that the parasites caused the emaciation. Identification of the cause of emaciation in such cases requires a knowledge of body weight and parasitic burden in apparently healthy birds, and efforts must be made to rule out the presence of other primary diseases, particularly chronic lead poisoning. 31 Although we were able to rule out other primary diseases, we did not have data on the bodyweight and parasite burden in apparently healthy mute swans from this region for comparison.
The average copper concentration (Table 2) in the liver of these mute swans from Walcheren is remarkably high and up to 3.2 times higher than the average concentrations found in mute swans in other studies. 3,7,16,24 The effect of these high copper levels on the health of these mute swans is not clear. On the one hand, Kobayashi et al. 13 diagnosed copper poisoning in free-ranging mute swans in Japan based on focal necrosis of the liver in these swans and high hepatic copper levels (Table 2). On the other hand, several studies suggest that swans can accumulate high liver levels of copper without significant hepatic damage being caused. 3,7,16 The latter studies correspond with our study, in which the swans had hepatic copper levels twice as high as free-ranging mute swans in Japan, but no evidence of hepatic necrosis. The high copper levels found in this study correspond to the severe contamination (up to 5,300 mg/kg, 1,495% in excess of the maximum permissible risk) of the sediment in the canals of Walcheren, probably caused by contamination from an old factory for fittings. The average lead concentration (Table 2) in the liver of these swans from Walcheren was 100 times lower than the average levels diagnostic for chronic lead poisoning in mute swans. 24 The average hepatic zinc concentration (Table 2) in the liver of these swans from Walcheren was comparable with those in other studies in free-ranging mute swans 7,24 but two times greater than levels described by Kobayashi 13 as nonharmful. The zinc levels considered toxic in swans are not clear.
In conclusion, in this study we have described severe obliterative endophlebitis of veins in the intestinal wall and hepatic veins as the primary lesion of Trichobilharzia sp. (probably T. filiformis) infection in mute swans. Further studies on this schistosome infection in mute swans and their intermediate snail host are required to confirm the Trichobilharzia species involved and to better characterize its prevalence and pathology in mute swans. In this way, we will better understand the contribution of this schistosome infection to the health of mute swan populations.
Footnotes
Acknowledgements
We thank J. Beekman, F. H. M. Borgsteede, H. Cremers, B. E. E. Martina, J. van der Hiele, B. Nolet, A. van der Laan, F.H. van Kalteren, V. Munster, M. van Zuijlen, J. van Steenbergen, G. van Zuijlen, P. E. F. Zoun, and everyone else from Bird and Hedgehog Sanctuary “de Mikke” for their help with this study.
