Abstract
Falconry is a long-standing tradition in the United Arab Emirates (UAE), and in 2016, falconry was included by UNESCO in the list of intangible cultural heritage of humanity. The health and wellness of falconry raptors is a priority for the local culture; plasma chemistry analysis plays an important role in monitoring, assessing, and managing diseases in avian patients. Imbalances of Cu, Mg, and Zn have been linked to several diseases in avian species; therefore, determining the reference intervals (RIs) of these minerals has important implications in the clinical management of falcons. We determined the RIs in plasma for Cu, Mg, and Zn in captive (falconry-managed) falcons according to the Quality Assurance and Laboratory Standards Committee of the American Society for Veterinary Clinical Pathology, and the Clinical Laboratory Standards Institute guidelines. Blood chemistry analyses were carried out on 252 clinically healthy falcons examined at the Dubai Falcon Hospital in the UAE: 124 gyrfalcons (Falco rusticolus) and 128 falcons of other species. We observed no significant variation in RIs of Cu (1.5–7.0 µmol/L), Mg (0.49–0.78 mmol/L), or Zn (11.8–34.1 µmol/L) based on different species or sex.
Falconry is a long-standing tradition in the United Arab Emirates (UAE) and a hallmark of the Emirati culture. Many Emiratis own falcons that are used for hunting, speed competitions, heritage cultural shows, and even falcon beauty pageants. Health and wellness of the raptors used in falconry is a high priority for the local culture. Falcons in the UAE undergo routine monitoring to assess their health and the presence of possible pathologic conditions. In addition to routine checks, the most common reasons for Emirati falconers to seek veterinary attention for their birds is when abnormal respiratory signs are noted. Respiratory diseases are the most common cause of morbidity and mortality in falcons in the Middle East. Falconry-managed falcons, however, may also suffer from a variety of conditions such as metabolic, parasitic, viral, fungal, and bacterial diseases.1,16,18,20,21 Since its establishment in 1983, >23,000 falcons have been treated in the Dubai Falcon Hospital, and ~700 individual falcons are examined yearly. The admitted captive falcons belong commonly to the following species: gyrfalcon (Falco rusticolus), gyrfalcon-hybrids (F. rusticolus × F. peregrinus, F. rusticolus × F. cherrug), peregrine falcon (F. peregrinus), saker falcon (F. cherrug), and Barbary falcon (F. peregrinoides).
Plasma chemistry analysis plays an important role in monitoring, assessing, and managing diseases in avian patients. Therefore, reference intervals (RIs) of clinically healthy falcons are necessary to interpret biochemistry changes that result from specific pathologic conditions. Plasma chemistry RIs for captive and hybrid falcons have been published.12,13,24 These studies covered other trace elements, minerals, and micronutrients (e.g., Ca, K, Na, Cl, Fe). However, RIs specifically for plasma copper (Cu), magnesium (Mg), and zinc (Zn) elements in captive falcons have not been reported, to our knowledge. Indeed, several diseases are associated with an imbalance of these minerals in birds.
Cu is an essential mineral involved in several physiologic functions; Cu deficiency can affect the immune system and normal growth in animals. Cu toxicity is rarely described in free-living or pet birds, but it has been recorded in swans and eider ducks. 6 The plasma threshold concentration needs to be determined for Cu toxicity in avian species. In American kestrels (F. sparverius) fed Cu bullets, the hepatic Cu concentrations increased, although no difference was observed in blood concentrations between the treated and control birds. 4 Furthermore, Cu supplementation of broiler chickens produced a positive effect on bone metabolism accompanied by improved cartilage development, if combined with phytase diet supplementation; however, blood Cu concentrations did not differ from those of control birds. 15
Mg is an important cofactor in several enzymatic reactions, for example in kinase-catalyzed reactions, glycolysis, and in normal nerve function. Hypomagnesemia has been observed in birds. 5 In a case of hypomagnesemia in an African grey parrot, the plasma Mg concentration was 0.78 mmol/L (1.9 mg/dL; RI: 0.86–1.40 mmol/L [2.1–3.4 mg/dL]), and the bird was unable to right itself and to perch, vocalized frantically, was depressed, and had declining ionized calcium concentrations despite treatment. 10 To our knowledge, there are no studies reporting hypomagnesemia in raptors, although some “neurologic syndromes” might be associated with such deficiencies. There is some indication that hypomagnesemia could play a role in intermittent seizures in raptors after intense training along with hypocalcemia, hypovitaminosis, and hypoglycemia. The lack of RIs for Mg has prevented confirmation of hypomagnesemia in captive falcons.
Zn is another element essential for the function of several enzymes. Zn toxicosis is well recorded in pet parrots 14 and waterfowl, 6 but rarely in captive falcons. Zn can cause pancreatitis in waterfowl, 22 pancreatic atrophy in ostriches, 3 and has a role in molting, immunity, and reproduction in poultry. 19 Plasma Zn concentrations have not been reported in captive falcons. We had a suspect case of Zn toxicosis in a gyrfalcon after the bird was fed a pigeon that had ingested a piece of metal wire, but the lack of species-specific plasma RIs has hindered the evidence-based diagnosis of Zn toxicity.
It is fundamental to establish RIs for Cu, Mg, and Zn in falcons to supplement existing data on plasma chemistry on Falconiformes. According to the Quality Assurance and Laboratory Standards Committee (QALS) of the American Society for Veterinary Clinical Pathology (ASVCP), and the Clinical Laboratory Standards Institute (CLSI) guidelines for the determination of RIs in veterinary species, a minimum of 120 reference individuals should be analyzed for determining nonparametric RIs and 90% confidence intervals (CIs) of reference limits. 9
Our objectives were to establish RIs in plasma for Cu, Mg, and Zn in falcons, and assess potential variation based on sex, age, and species. For this purpose, we carried out blood chemistry analyses on 252 clinically healthy falcons examined at the Dubai Falcon Hospital in UAE.
Our study was approved by the Dubai Falcon Hospital director and the ethical committee of the American University of Sharjah, Sharjah, UAE. We collected blood samples from 252 falcons (176 females [16 adult, 137 juvenile, 23 immature], 75 males [3 adult, 62 juvenile, 10 immature]) that had been admitted to the Dubai Falcon Hospital, Dubai, UAE, between August 2018 and September 2019 either for health checks or for pre-purchase examinations. We included 124 gyrfalcons, 72 gyr × peregrine hybrids, 32 peregrine falcons, 11 saker falcons, 8 gyr × saker hybrids, 2 gyr × Barbary hybrids, and 2 Barbary falcons. Health checks were performed on all falcons at the Dubai Falcon Hospital, and clinical status was assessed by routine diagnostic procedures. The birds had not received any medication for a minimum of 2 wk before blood sampling and were fasted for 12–24 h prior to blood collection. We included only healthy falcons based on clinical, laboratory, radiographic, and endoscopic (laparoscopy, tracheoscopy) examinations.
The falcon diet in the UAE consists mainly of thawed or fresh quail, pigeon, and, in smaller quantities, lamb and beef. The quails and pigeons are fed after removing the skin, feathers, and internal organs; wings from the elbow level and the legs from the metatarsal level are also discarded. Each falcon according to stage of training may receive a complete body of a quail or the full or half of a pectoral muscle of a pigeon. The mean weight of meat per falcon can be 50–150 g, depending on the species and the level of training.
Falcons were pre-medicated with medetomidine–ketamine and maintained under anesthesia with isoflurane and oxygen as carrier gas. Blood samples were collected at 8–10 am, from the medial metatarsal vein of 252 healthy falcons, using a 27-ga needle and 1-mL syringe; ~ 0.5 mL for each sample were transferred into pediatric lithium–heparin tubes (Mini Collect; Greiner). Lithium–heparin samples were immediately placed on a roller mixer (Coulter) for 10–15 min, then centrifuged at 2,100 × g for 5 min (Micromax; Thermo IEC), and the plasma was immediately frozen at −40°C. No hemolysis or other plasma alterations were observed. Plasma samples were stored at −40°C until analysis and were analyzed within 3 mo of collection. Cu, Mg, and Zn were analyzed by colorimetric methods (Miura 200 chemistry analyzer, ISE; Cu: 3,5-dibromo-PAESA, Mg: xylidyl blue, Zn: 5-Br-PAPS, Dialab; Suppl. material). Dialab controls and calibrators were used daily before analysis. Control checks were repeated after every 20 samples. Despite being used widely in birds, validation studies of those methods in birds have not been published, and validation is highly desirable in the near future.
We determined 95% RIs according to the ASVCP guidelines for veterinary RIs using either a parametric or nonparametric method. 9 Normality of the analytes was assessed using a Shapiro–Wilk test with normality for p > 0.2. Outliers were detected using the Horn method and evaluated on scatterplots. The influence of species, age, sex, and their interactions on mean element concentrations was assessed using a 3-way ANOVA. Assumptions of normality and homogeneity of variance of residuals were assessed on residual and quantile plots. An alpha of 0.05 was used for statistical significance. Partitioning by sex and species was also performed, and reference limits were compared between groups by examining the CIs of the reference limits. R foundation for statistical computing (https://www.r-project.org) was used for statistical analysis and the R-package ‘Reference Intervals’ (https://CRAN.R-project.org/package=referenceIntervals) for RI determination.
RIs were calculated for all 252 falcons: 124 gyrfalcons, and 128 falcons of other species grouped. One Cu outlier (11.4 mmol/L [72.4 µg/dL]) was detected and removed from the calculations. Age (data not shown) and sex (176 females, 75 males) had no effect on the mean concentrations of Cu, Mg, or Zn (p > 0.05; Table 1).
Reference intervals for Cu, Mg, Zn, determined in plasma of healthy falcons.
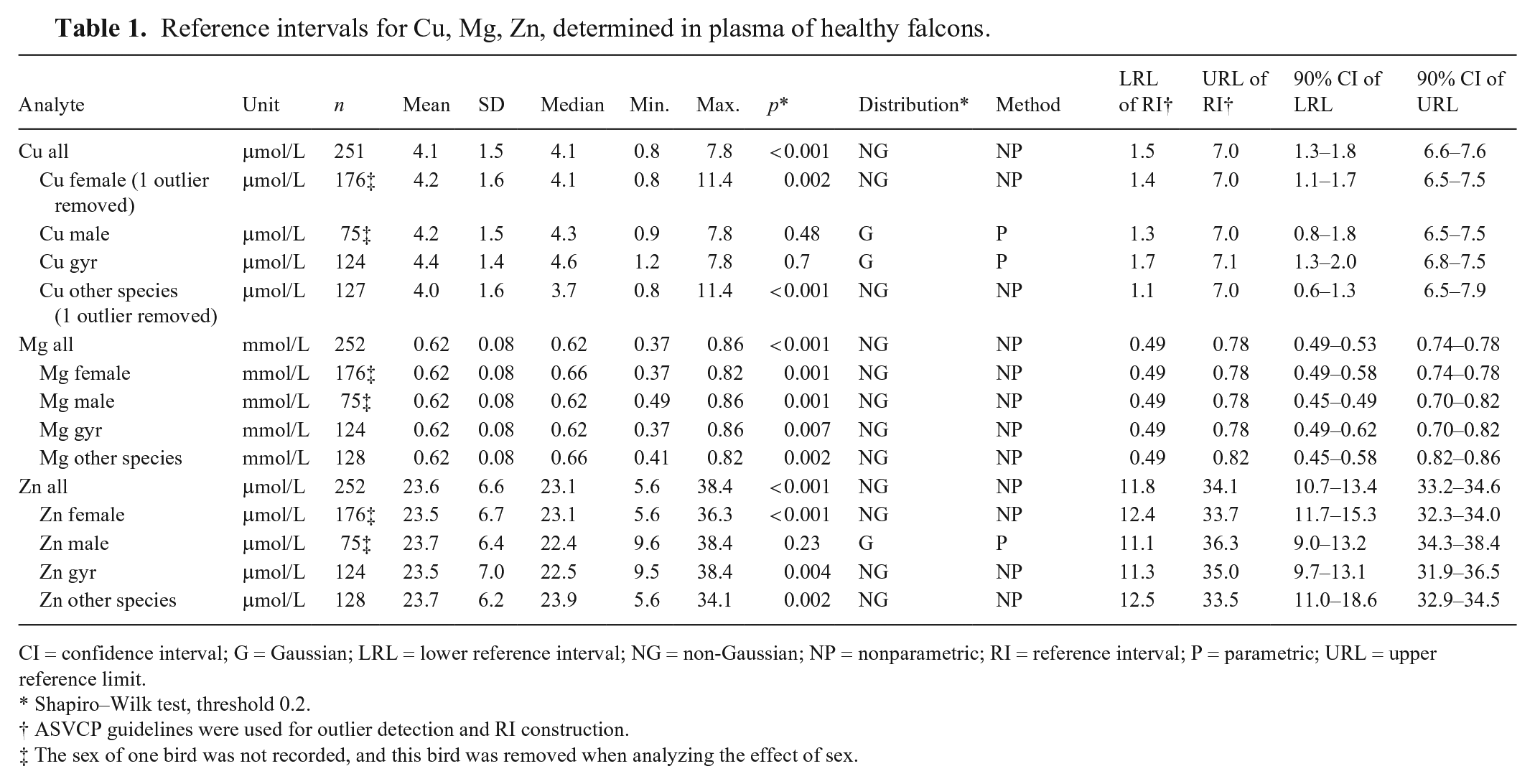
CI = confidence interval; G = Gaussian; LRL = lower reference interval; NG = non-Gaussian; NP = nonparametric; RI = reference interval; P = parametric; URL = upper reference limit.
Shapiro–Wilk test, threshold 0.2.
ASVCP guidelines were used for outlier detection and RI construction.
The sex of one bird was not recorded, and this bird was removed when analyzing the effect of sex.
Our RIs for Cu (1.46–7.03 mmol/L [9.3–44.7 mg/dL]) were consistent among different species and sex of captive falcons (Table 1). The mean value for Cu (4.1 mmol/L [26.4 mg/dL]) was close to the value determined in captive greater flamingos (Phoeniconaias phoenicopterus rubeus; 5.6 mmol/L [35.7 mg/dL]), 2 in free-living griffon vultures (Gyps fulvus; 3.2 mmol/L [20.4 mg/dL]), 8 and in free-living nestling northern goshawks (Accipiter gentilis; 3.8 mmol/L [24 mg/dL]). 7
Our RIs for Mg (0.49–0.78 mmol/L [1.2–1.9 mg/dL]) were consistent among different species and sex of captive falcons (Table 1). The mean Mg concentration (0.62 mmol/L [1.5 mg/dL]) was also consistent with mean values determined earlier in gyrfalcons and its hybrids, 13 although it was lower in falcons than in parrots (0.99 mmol/L [2.4 mg/dL]). 5
Our RIs for Zn (11.8–34.1 mmol/L [77–223 mg/dL]) had no significant differences among different species and sex in captive falcons (Table 1). To our knowledge, plasma Zn RIs have not been reported for captive falcons. Isoflurane anesthesia lowers Zn plasma concentrations in humans 23 ; it is important to note that we determined RIs in anesthetized falcons as per routine diagnostic procedures. The mean value that we determined for Zn (23.6 mmol/L [154 mg/dL]) was close to that determined in captive lesser flamingos (24.5 mmol/L) 2 and within the RI (19.1–35.0 mmol/L [125–229 mg/dL]) for Hispaniolan Amazon parrots (Amazona ventralis). 17 In raptor species, the Zn concentration in free-living griffon vultures (50.8 mmol/L [332 mg/dL]) and in Egyptian vultures (Neophron percnopterus; 55.3 mmol/L [362 mg/dL]) was 2× higher than in captive falcons. 8 Free-living nestling northern goshawks had a 3× higher Zn concentration (76.5–85.7 mmol/L [500–560 mg/dL]) than free-living adult northern goshawks (19.7–35.2 mmol/L [129–230 mg/mL]) 7 and the captive falcons in our study.
Zn toxicity has been recognized as a problem in birds, and serum or plasma concentration >30.6–45.9 mmol/L (200–300 mg/dL) have been suggested as diagnostic for Zn toxicosis in psittacines. 17 However, further studies are necessary in falcons to determine the Zn concentrations that cause acute or chronic Zn toxicity in these birds. A study suggested that quails fed Mg and Zn supplements had reduced heat-stress effects when kept at 35°C for 8 h/d. 11 The UAE climate is quite harsh, with temperatures reaching 50–55°C during summer months; falcons are flown outdoors during October to March, in the early morning or late evening, and are exposed to temperature up to 35°C. No studies have been reported on correlation between the heat-resistance performance of falcons with their intake or plasma concentration of Mg and Zn, which could be of importance for gyrfalcons, which are arctic species and are more prone to heat stress. Determining the RIs for these minerals would set the stage also for such studies in falcons.
Supplemental Material
sj-pdf-1-jvd-10.1177_10406387211015655 – Supplemental material for Reference intervals for Cu, Mg, and Zn in captive gyrfalcons and other falcon species in the United Arab Emirates
Supplemental material, sj-pdf-1-jvd-10.1177_10406387211015655 for Reference intervals for Cu, Mg, and Zn in captive gyrfalcons and other falcon species in the United Arab Emirates by Lucia Pappalardo, Christu-Das Silvanose, Hugues Beaufrère, Ambilli Binoy and Panagiotis Azmanis in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our work was supported by the Dubai Falcon Hospital and the American University of Sharjah.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
