Abstract
Four young broiler chickens affected by multiple melanotic tumors are described. Grossly, there were multiple tumors composed of melanocytes within the skin, skeletal muscle, and multiple visceral organs. Tumors ranged from flattened macules to masses that extensively replaced viscera. Microscopically, melanocytes were often well pigmented, and while there was moderate nuclear anisokaryosis, mitotic rates were low. Immunohistochemical staining of some melanomas with antibodies to S100 proteins, Melan-A, vimentin, or neuron-specific enolase after bleaching of tumor cells with potassium permanganate revealed lack of immunostaining of tumor cells with antibodies to S100, strong positive staining of tumor cells for neuron-specific enolase, moderate staining with antibodies to vimentin, and faint staining for Melan-A. Only neuron-specific enolase staining was evident in unbleached tumor cells. Attempts to identify exogenous avian leukosis viruses in these tumors were unsuccessful.
Melanomas are relatively common in dogs and in some breeds of pigs and horses but are less commonly identified in other mammals. 10 Melanomas in birds are less commonly reported, although these tumors have been identified in a variety of avian species, including several breeds of ducks,1,5,7,10,14 raptors,1,10 penguins, 7 gray parrots, 1 pigeons,7,13,15 a zebra finch, 6 a pheasant, 5 a seagull, 4 a comorant, 9 an umbrella cockatoo, 18 and a budgerigar. 7 In addition, melanomas have been briefly described in several chickens.2,3,5,15 We describe the gross, microscopic, immunohistochemical, and ultrastructural appearance of widely metastatic melanomas in 4 young broiler chickens.
Affected tissues were obtained from affected broiler chicken carcasses, fixed in 10% neutral buffered formalin, routinely processed for histology, and embedded in paraffin. Four-μm-thick sections were cut from paraffin blocks, placed on glass slides, deparaffinized with xylene, rehydrated, and stained with hematoxylin and eosin. Replicate sections from 3 cases (Nos. 1–3) were used for immunohistochemical stains utilizing antibodies to S-100 proteins (Biogenex, San Ramon, CA), neuron-specific enolase (Biogenex), vimentin (Biogenex), and Melan-A (Dako, Carpinteria, CA) in a streptavidin–biotin alkaline phosphatase system as previously described. 18 Paired sections were bleached with potassium permanganate before this immunostaining. Selected sections from these 3 cases were alternatively bleached with 10% hydrogen peroxide in tris-buffered saline overnight before immunostaining for Melan-A. Positive control tissues were well-pigmented chicken melanocytes in lesions of melanosis. For transmission electron microscopy of ultrathin sections, formalin-fixed tumor tissue (case No. 4) was postfixed in osmium tetroxide, dehydrated, and embedded in Epon. Ultrathin sections were stained with lead citrate and uranyl acetate, placed on grids, and examined in a transmission electron microscope.
Molecular detection of exogenous avian leukosis viruses was attempted by polymerase chain reaction amplification of the envelope gene utilizing previously described methods that target the envelope gene of all known exogenous avian leukosis viruses.17,20 DNA integrity and suitability for amplification in these tissue samples were verified in all cases by polymerase chain reaction amplification of the Gallus gallus α2(IV) collagen gene. 8 Isolation of exogenous avian leukosis viruses was also attempted by tissue culture of unfixed tissues (case No. 4 only) as previously reported. 21
All 4 cases were 6- to 8-week-old broiler chickens detected at slaughter. Case No. 1 was a small carcass with multiple 3- to 6-mm lesions scattered throughout the skin, muscle, kidney, liver, spleen, proventriculus, intestine, and heart. Case No. 2 involved a well-fleshed but apparently dehydrated broiler chicken with tumors up to 1.5 cm in diameter that affected the skin, muscle, liver, lung, heart, testis, proventriculus, intestine, kidney, air sac, and esophagus (Figs. 1–4). In case No. 3, tumors were noted in the skin, kidney, liver lung, spleen, heart, and intestine. In case No. 4, melanomas were noted in skin, liver, muscle, proventriculus, spleen, and heart. Tumors varied from flattened macule-like areas of pigmentation to distinct masses that multifocally replaced visceral organs.
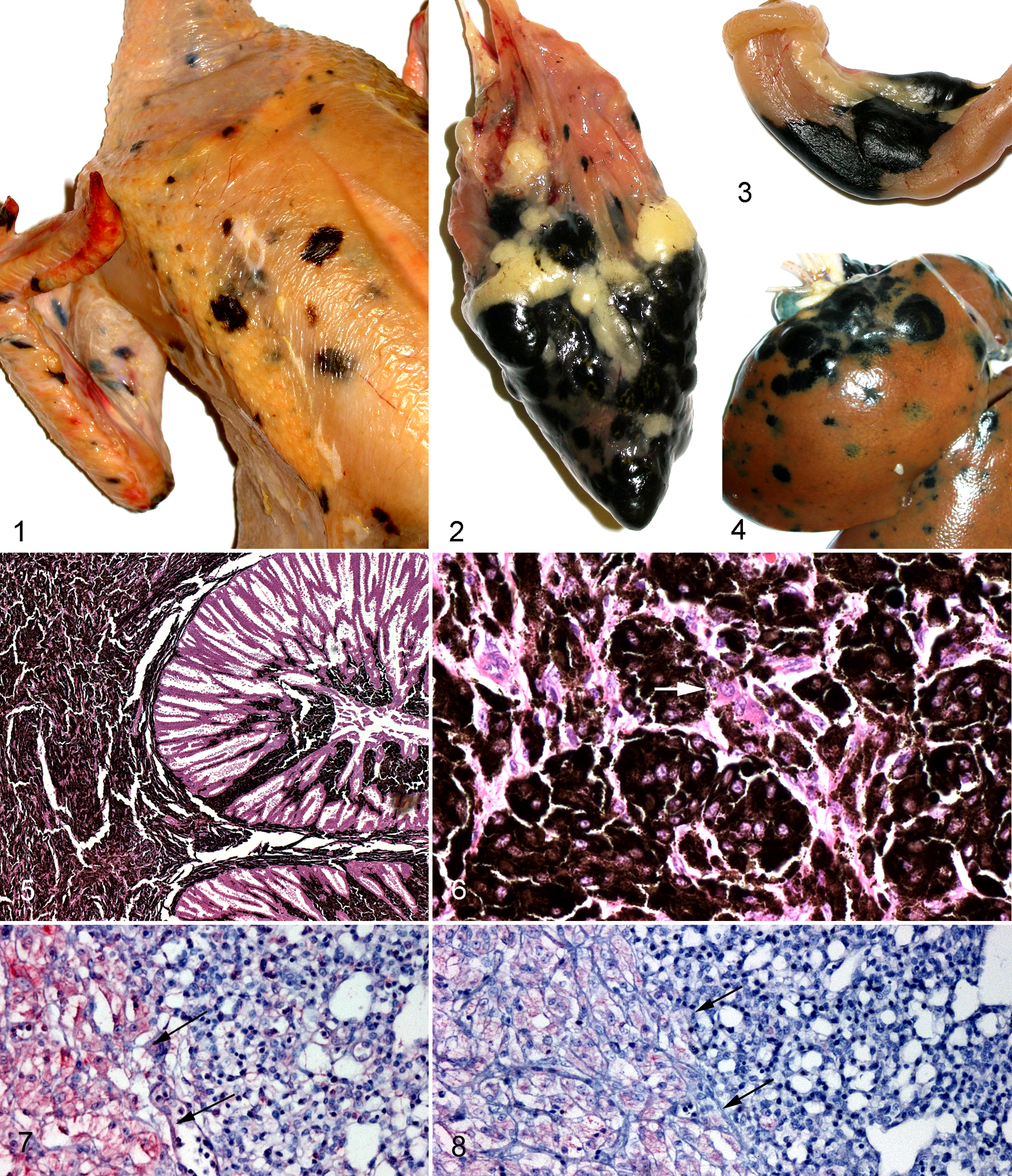
Skin; chicken. Multiple, variably sized melanomas multifocally replace the skin and underlying muscle.
Electron micrograph; chicken. Melanocytes filled with round to ovoid melanosomes extensively replace the liver; occasional premelanosomes are present (dark arrow). Inset: Some melanosomes contained longitudinal striations or electron lucent vacuoles (white arrows).
Microscopically, the melanocytes in the tumors of these young chickens appeared similar. Many tumor cells contained numerous cytoplasmic melanin granules, but in some areas, small aggregates of tumor cells contained only minimal numbers of melanin granules. Tumor cells ranged from spindloid to epithelioid and often extensively effaced the normal architecture of the affected organ (Fig. 5). Nuclei were irregularly round to ovoid, moderately anisokaryotic, and contained 1 to 3 prominent eosinophilic nucleoli within a predominantly euchromatic chromatin that was bounded by thin chromatinic rims. The mitotic rate was low, commonly ranging from 0 to 2 per 5 high-power fields (40×), but tumor cells were present within some vessels, and some masses extensively replaced visceral organs (Fig. 6).
In unbleached sections used for immunohistochemical staining, melanocytes were specifically immunostained by neuron-specific enolase. In sections bleached with potassium permanganate, there was moderate to marked immunostaining with neuron-specific enolase (Fig. 7), mild to moderate immunostaining with vimentin, and a mild specific immunostaining of melanocyte cytoplasm by Melan-A (Fig. 8). Sections bleached with hydrogen peroxide also demonstrated mild specific immunostaining of the cytoplasm of tumor cells with antibodies to Melan-A. Antibodies to S-100 proteins brightly immunostained normal peripheral nerves, some of which were within tumors, but did not stain tumor cells, even in bleached sections.
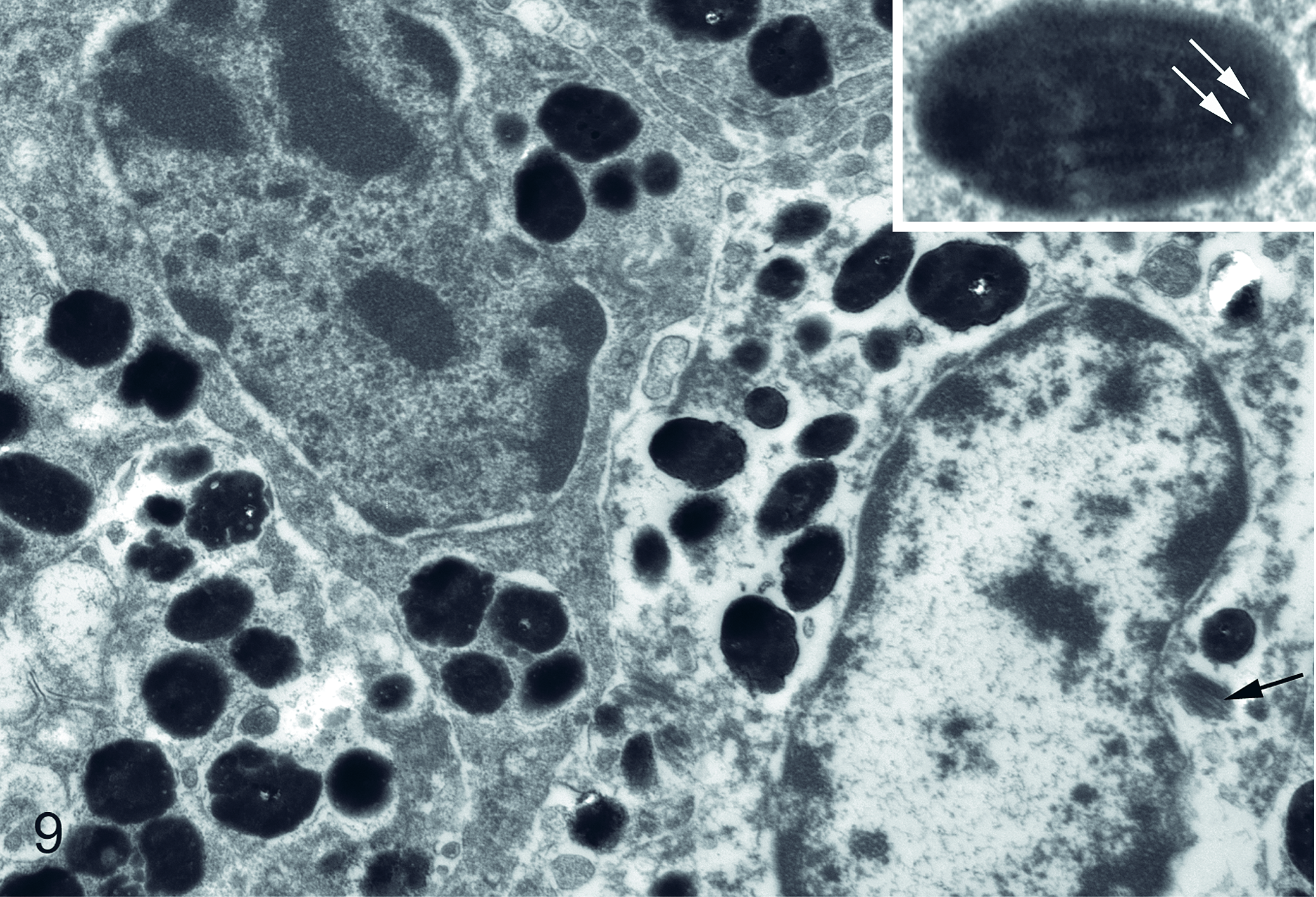
Ultrastructurally, melanocytes contained numerous well-melanized, round to ovoid melanosomes that were 200 to 500 nm in diameter (Fig. 9). Occasional premelanosomes were present. Melanosomes were often moderately electron dense, but many contained small electron-lucent vacuole-like spaces or longitudinal striations. No virions were identified.
All attempts to detect proviral genome sequences from exogenous avian leukosis viruses were unsuccessful. Exogenous avian leukosis viruses were also not detected in unfixed tissues from case No. 4 after 3 passages of filter-sterilized tissue homogenates in cell culture.
Melanomas in birds, except for a few affecting the eye, 2 have been nearly universally considered malignant.1,4,6,7,9,10,13–16,18 The primary sites of origin for avian melanomas have been variously cited as skin, adrenal gland, ovary, lung, or liver, but several tumors, particularly those affecting aged birds, have apparently originated on the facial skin near the beak.1,7,10,13,16 This may suggest, as some authors have alluded, induction of some avian melanomas by solar radiation, as this would be one of the few sites not well covered by feathers.13,16 Young broiler chickens, however, are not exposed to significant amounts of solar radiation, as they are raised within production houses during their short life span. Tumors in these young chickens are often induced by infection of the birds with avian leukosis viruses, generally subgroups A, B, or J, or myeloblastosis-associated viruses,20,21 but the unsuccessful attempts to identify those viruses in the young chickens affected by the described melanomas suggest that those viruses were not involved in the induction of these tumors.
Silkie chickens are chickens with melanosis present in many organs, such as trachea, lung, gonads, and spleen, in addition to having diffusely dark pigmented skin. In many avian species, melanocytes are present in only the feather bulb epithelium to pigment the feathers and not in the interfollicular epithelium. Silkies are the exception in that numerous melanocytes are present in the dermis, below the elastic and collagen fibers of the skin, and rarely in the epidermis. 12 The normal architecture of the organs is not disrupted in melanosis, while the neoplasia seen in this report does disrupt normal architecture of the affected organs.
While melanomas are relatively uncommon in birds and rare in chickens,3,15 the large numbers of young broiler chickens slaughtered often allow detection of otherwise rare tumors. A prevalence of 0.000194% (7 broiler chicken carcasses with multiple melanomas per 3.6 million chickens slaughtered) was calculated utilizing data from a broiler operation slaughtering 1.8 million chickens per week. A single breed cross was involved in the parentage of the chickens affected with these melanomas, lending credence to a possible genetic link (G. Zavala, personal communication).
Immunohistochemical evaluation of avian melanomas is often less than satisfactory. S100 has been cited by a few reports as not positive in avian tumors while positively staining normal avian neural tissues,6,9,18 and several reports have described a lack of staining of neoplastic avian melanocytes with Melan-A,6,18 while one report cites a poorly melanized melanoma that stained with both S100 and Melan-A. 4 In this study, unbleached tumor cells were evaluated as negative for Melan-A, but faint specific staining was evident in neoplastic melanocytes bleached with potassium permanganate or hydrogen peroxide before immunostaining. Potassium permanganate bleaching followed by immunohistochemical evaluation has resulted in nonspecific staining with some antibodies but not with Melan-A, while bleaching with dilute hydrogen peroxide has resulted in neither loss of specificity nor a reduction in sensitivity. 11 Antibodies to Melan-A (MART-1) detect a protein largely restricted to melanocytes, and this reactivity has been documented in chickens. 19 While neuron-specific enolase yielded the most distinctly positive immunostaining, this antibody is not specific to melanocytes or even to neural tissues.
The mitotic rate of the melanocytes in the tumors of these young chickens was often low when compared to that of many malignant neoplasms, and although there was a paucity of pigmentation in some tumor cells, melanocytes were often well pigmented. Some of the previous descriptions of avian melanomas that were deemed malignant also exhibited what the authors described as a low mitotic rates if tumors were bleached to allow this evaluation.7,10 Despite the low mitotic rate in some tumors, the appearance of the melanocytes and the extensive replacement and distortion of some visceral organs suggest that these tumors should be considered malignant.
Footnotes
Acknowledgements
We thank Alice Self and Brenda King for the immunohistochemical stains and Rebecca Patlan-Garcia for providing gross photographs.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
