Abstract
Survivin is a recently discovered protein belonging to the inhibitor of apoptosis (IAP) gene family. IAP molecules are characterized by both the presence of a zinc-binding fold termed the baculoviral IAP repeat and the ability to suppress apoptosis. In addition to inhibiting apoptosis, survivin is essential for proper cell division. Survivin is expressed during embryonal development but is absent in most normal, terminally differentiated tissues. Survivin is also upregulated in a variety of human cancers, and its expression in tumors is associated with a more aggressive phenotype, shorter survival times, and a decreased response to chemotherapy. The exact mechanism behind the ability of survivin to inhibit apoptosis is still unclear. Furthermore, it is not known why this protein is upregulated in cancer. The purpose of this article is to provide an overview of the current knowledge of survivin, including its role in cell division and its expression in normal and neoplastic tissues. Although much of the current research in this field is focused on human medicine, this area also has potential significance for veterinary species.
The inhibitor of apoptosis (IAP) proteins are a family of highly conserved cell death inhibitors that have been found in yeast, invertebrates, and vertebrates. IAPs were first discovered in baculoviruses, where they were shown to be involved in suppressing host cell death response to viral infection. 11,18 Survivin, a recently discovered IAP, is a bifunctional protein that regulates cell division and suppresses apoptosis. 5 Survivin is unique among the IAP proteins because it exhibits cell cycle–regulated expression that peaks at mitosis. 41 Survivin is abundantly expressed in fetal tissues, 2,5 yet undetectable in most normal, terminally differentiated adult tissues. 6 Survivin is also overexpressed in a variety of human neoplasms, suggesting that reactivation of the survivin gene frequently occurs in cancers. 6 Currently, survivin protein expression is being used as a prognostic factor in several human neoplasms. 6 High survivin expression by neoplasms correlates with more aggressive behavior, decreased response to chemotherapeutic agents, and shortened survival times, as compared with cancers that are survivin negative. Because its expression is among the most tumor-specific of all gene products, there is considerable biomedical interest in this protein. Manipulation of survivin regulation and expression may also lead to the development of new immunotherapy and gene therapy strategies for the treatment of cancer. The purpose of this review is to discuss the role of survivin in normal and neoplastic tissues and in the targeting of survivin as a potential therapy in cancer.
Molecular Organization and Structure of Survivin
IAP molecules are characterized by the presence of one or more copies of an approximately 70–amino acid zinc-binding fold, termed the baculoviral IAP repeat (BIR), as well as the ability to inhibit apoptosis. 19 Survivin is the smallest member of the IAP family of proteins and has only a single N-terminal BIR domain, a long C-terminal alpha-helix coiled region, and a dimeric arrangement. 75 The BIR domain is thought to be critical for antiapoptotic function, whereas the coil-coiled domain probably interacts with tubulin structures. The survivin molecule contains three separate and chemically distinct surfaces, including acidic and basic regions on the BIR domain and a hydrophobic helical surface on alpha 6. This arrangement is consistent with functionally relevant protein-protein interaction surfaces. 75
The murine survivin gene, located on chromosome 11E2, contains four exons, which gives rise to three separate gene products through alternative splicing. 16 The longest open reading frame, containing all four exons, produces a 140–amino acid protein that is similar to human full-length survivin. A second isoform, lacking sequence derived from exon 2, gives rise to a 40–amino acid protein that lacks the BIR domain and the C-terminal coil-coiled domain. The third gene product results from the retention of intron 3, subsequently acquiring a new in-frame stop codon. The 121–amino acid protein produced contains the BIR domain but lacks the coil-coiled domain.
The human survivin gene, spanning 14.7 kb on telomeric position of chromosome 17, produces a 16.5-kDa protein. 7 Similar to murine survivin, the human survivin gene, as a result of three alternatively spliced transcripts, can give rise to three different isoforms of the protein 44 (Fig. 1). These isoforms include full-length survivin, survivin-2B, and survivin-ΔEx-3. The full-length survivin gene consists of a three-intron, four-exon structure. The survivin-2B transcript results from the retention of a portion of intron 2, whereas the survivin-ΔEx-3 transcript results from the removal of exon 3. The sequence alterations produced from the splice variants result in marked changes in the corresponding protein structure and, subsequently, differences in their ability to inhibit apoptosis. Insertion of exon 2B in survivin-2B interrupts the essential BIR domain, which leads to a marked decrease in the apoptotic activity of this isoform. The removal of exon 3 in survivin-ΔEx-3 likewise interrupts the BIR domain. However, this isoform of the protein retains its ability to suppress apoptosis. In addition, loss of this exon results in a frame shift in exon 4, generating a novel COOH-terminal. There is also different subcellular localization of survivin and its splice variants. Survivin and survivin-2B are predominantly cytoplasmic, whereas survivin-ΔEx-3 is primarily nuclear. 43 These different isoforms of survivin and their varied locations in the cell may represent a regulatory balance between apoptosis and inhibition of apoptosis. 43
Survivin isoforms: survivin exists in three iso-forms as the result of alternatively spliced transcripts. Survivin-ΔEx-3 is primarily nuclear in location, whereas full-length survivin and survivin-2B are predominantly within the cytoplasm. These immunochemically distinct pools may explain the discrepancy regarding survivin's ability to play the roles as both a mitotic spindle–associated protein and a kinetochore-associated chromosomal passenger protein.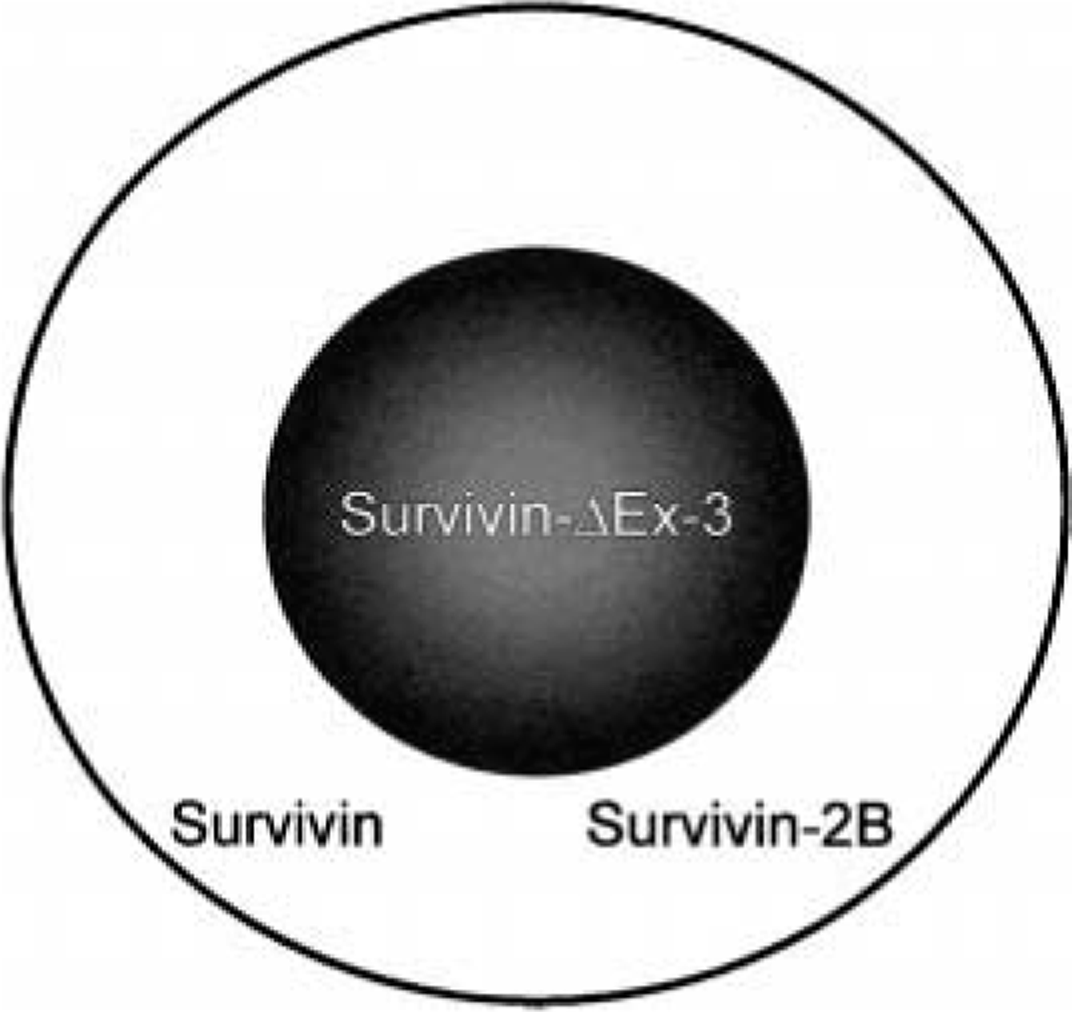
The Role of Survivin in Cell Division
Survivin is essential for proper execution of mitosis and cell division. 58 Its specific expression in G2/M is transcriptionally controlled, 40 as is typical of mitotic genes. 80 During mitosis, survivin binds to the micro-tubules of the mitotic spindle through its carboxy terminal alpha helices. Interference of survivin-microtubule interactions, by means of an antisense-mediated reduction in the expression of survivin, results in a failure of the antiapoptotic function of survivin and an increase in caspase-3 activity with subsequent apoptosis. 6 Disruption of survivin function has likewise been linked with cell division defects exemplified by supernumerary centrosomes, formation of multipolar mitotic spindles, and failure of cytokinesis, with cells becoming polyploid and multinucleated. 14,39 Additional evidence for the crucial role of survivin in mitosis has been demonstrated in knockout mice. Homozygous disruption of the survivin gene resulted in embryonic death at 4–5 days, with the null embryos revealing failed cytokinesis and disrupted microtubule formation. 74
In addition to its role in maintaining a normal bipolar mitotic apparatus, survivin is also a kinetochore-associated chromosomal passenger protein 66 in concert with Aurora B kinase and inner centromere protein. 12 Furthermore, Aurora B kinase activity is enhanced by survivin binding. 12 The mechanism behind survivin's ability to regulate Aurora B kinase activity and the exact interaction between these three chromosomal passenger proteins is speculative at this time. The discrepancy regarding survivin's cytoplasmic versus nucleoplasmic location lies in the fact that survivin exists in two immunochemically distinct subcellular pools, possibly as a result of alternatively spliced transcripts. 21
The Role of Survivin in Apoptosis
The IAP family of proteins is defined by the presence of at least one BIR domain and the ability to inhibit apoptosis. Using these criteria, humans have eight IAP family members: NAIP, cIAP1, cIAP2, XIAP, Ts-XIAP, ML-IAP, apollon, and survivin. Several IAPs in humans and Drosophila have been shown to directly bind and inhibit caspases via their BIR domains. 19,48 For example, XIAP binding and inhibition of caspase-9 is through its third BIR domain (BIR3), whereas caspase-3 and caspase-7 are suppressed via a region within the molecule between BIR1 and BIR2. 70 The precise mechanism by which survivin suppresses apoptosis, however, is still incompletely understood. Several mechanisms are theorized (Fig. 2). Direct suppression of caspase-3 by survivin has been speculated by some investigators; yet, survivin lacks structural components present in other IAPs that allow their direct binding to caspase-3. 59 There is also speculation that survivin binds to caspase-9 52 but there are likewise problems with this theory. Phosphorylation of survivin on the threonine at position 34 (Thr34) is critical for a functional survivin molecule. However, phosphorylation of survivin fails to explain why this would promote interaction with caspase-9. 65 Another possibility is that survivin requires the cofactor hepatitis B X-interacting protein to bind procaspase-9, thus preventing apoptosis via the intrinsic pathway. 45 Finally, survivin may indirectly inhibit caspases via intermediate proteins. Survivin binds to Smac/DIABLO, which is a proapoptotic protein that binds IAPs and thus prevents them from inhibiting caspases. 20,76 During interphase, survivin colocalizes with Smac/DIABLO in the cytosol. 69 A point mutation at amino acid Asp-71 in survivin results in the failure of survivin to complex with Smac/DIABLO. Consequently, this was shown to abolish survivin's ability to protect against taxol-induced apoptosis in HeLa cells. 69
Potential mechanisms by which survivin inhibits apoptosis: survivin may inhibit apoptosis by binding caspase-9 or it may block Smac, thus preventing this proapoptotic protein from blocking IAP proteins.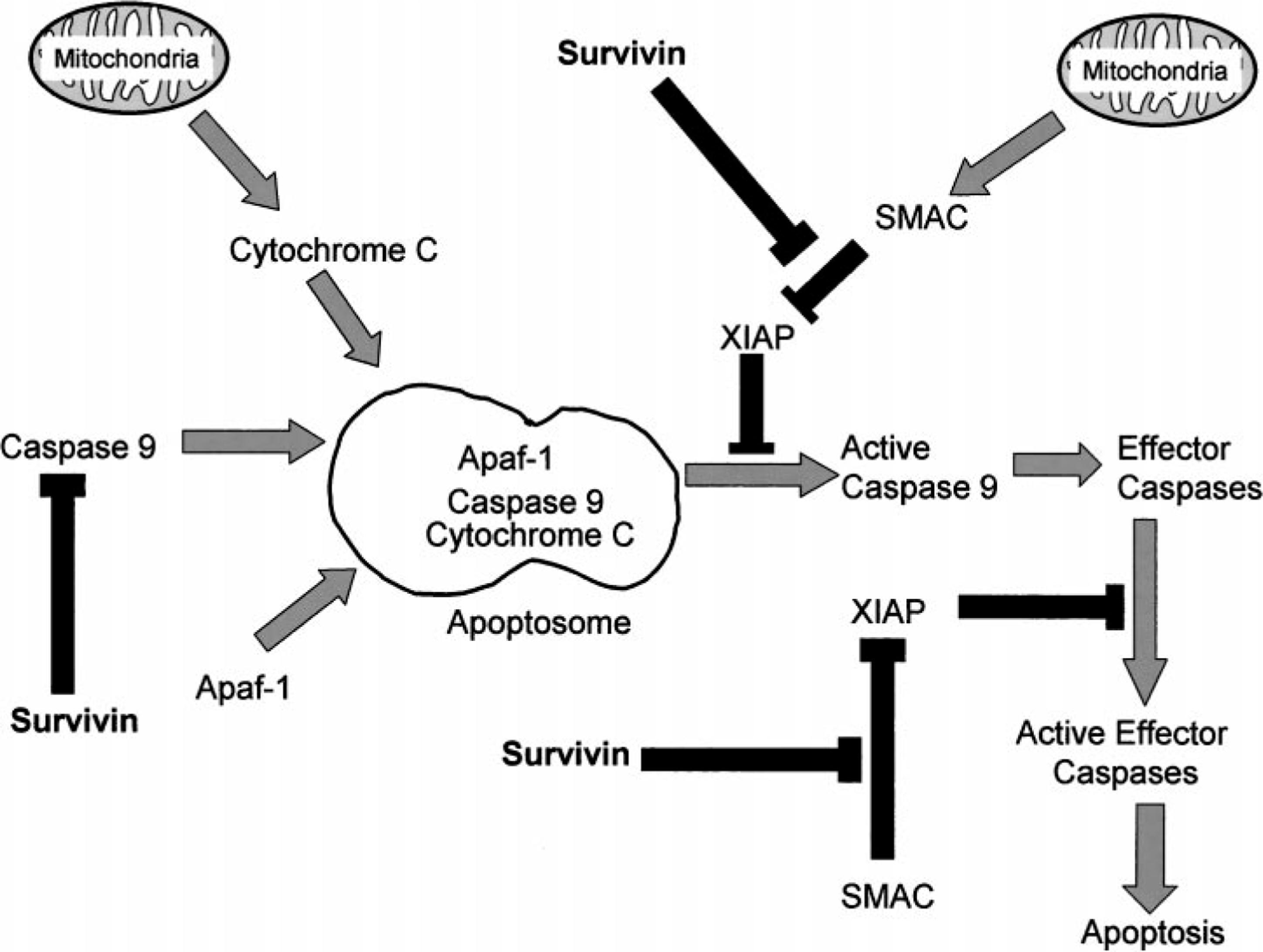
The role of survivin as an IAP versus its function in cell division has been explored in vivo through the use of transgenic (K14-survivin) mice, which constitutively express survivin in keratinocytes. 26 Overexpression of survivin in K14-survivin mice does not affect normal skin differentiation, neither is there an increase in basal keratinocyte proliferative rates between K14-survivin mice and controls on induction of keratinocyte proliferation. However, K14-survivin mice had a 60% reduction in apoptosis compared with the control group after UVB exposure. These results demonstrate that constitutive expression of survivin inhibits keratinocyte apoptosis; yet, cell proliferation remains unaffected. This implies that survivin expression in cancer does not simply reflect an increase in the number of mitotic tumor cells. In vitro, K14-survivin keratinocytes also have increased resistance to UVB-induced apoptosis. The transgenic cells, however, are more susceptible to Fas-mediated (extrinsic) apoptosis, indicating that the cytoprotection offered by survivin is ineffective against the Fas-induced apoptotic pathway. K14-survivin mice crossed with p53-deficient mice, creating K14-survivin mice lacking one p53 allele (p53/−), are equally resistant to UVB-induced apoptosis as p53−/− keratinocytes. Thus, it appears that survivin expression can substitute for the loss of one p53 allele in suppressing apoptosis.
Regulation of Survivin
Phosphorylation at Thr34 of survivin plays an essential role in its regulation. The cyclin-dependent kinase, p34cdc2-cyclinB1, is the only known kinase that is able to phosphorylate survivin and colocalizes with it on the mitotic apparatus. 51 Elevations in p34cdc2 kinase, induced by spindle checkpoint activation, result in increased survivin expression. 54 This increase in survivin does not result from a substantial change in RNA levels or promoter activity, suggesting post-transcriptional stability of survivin by p34cdc2 kinase. Indeed, nonphosphorylatable survivin, produced by a mutation of Thr34 → Ala (T34A), is more rapidly cleared than wild-type survivin 54 by the ubiquitin-proteosome pathway. 78 The Thr34 phosphorylation site appears to be in a position to stabilize antiapoptotic protein-protein interactions through the BIR domain of survivin. 75 Failure to phosphorylate Thr34, by the mutation T34A, leads to the dissociation of a survivin–caspase-9 complex and subsequent caspase-9–dependent apoptosis during cell division. 52 Apoptosis does not result from the inability to progress through the cell cycle because cells expressing survivin T34A do not exhibit G2/M arrest. Instead, apoptosis occurs on entry and progression through mitosis. These findings indicate that phosphorylation of Thr34 is critical for survivin in its role as a suppressor of apoptosis.
The mechanisms behind the differential expression of survivin in neoplastic tissues versus normal, terminally differentiated adult tissues are largely unknown at this time. There is mounting evidence that a multitude of tumorigenic pathways are potentially involved in the upregulation of survivin in cancer (Fig. 3). Wild-type p53 transcriptionally represses survivin, 30,49,79 and overexpression of survivin is able to counteract p53-dependent apoptosis induced by adriamycin 49 or ultraviolet light. 30 The means by which p53 inhibits survivin is still controversial. Although there is evidence supporting the direct binding of p53 to the survivin promoter, 30 other data indicate that alteration of the chromatin configuration within the survivin promoter, with subsequent decreased access to the promoter, is responsible for the suppression of survivin by p53. 46 Inhibition of signal transducer and activator of transcription-3 (STAT3) signaling pathways has also been shown to result in the reduction of survivin expression in primary effusion lymphoma 10 (PEL) and astrocytomas, 38 with subsequent induction of apoptosis. In addition, forced expression of survivin prevents apoptosis induced by STAT3 inhibition in PEL cells. 10 Thus, constitutive STAT3 activation, which is frequently observed in cancer, may lead to survivin up-regulation. Finally, survivin upregulation by c-H-Ras oncoprotein has been demonstrated, independent of cell cycle progression or cellular proliferation. 68 This supports the impression that elevated survivin levels are not simply the result of an increased fraction of G2/M cells secondary to Ras-mediated cell cycle alteration. 68 Furthermore, inhibitors of phosphatidylinositol 3-kinase (PI3-K) and MEK1 result in a reduction of survivin, suggesting that these intracellular Ras-pathways are involved in the regulation of survivin expression. 68
Regulation of survivin: multiple tumorigenic pathways are potentially involved in the overexpression of survivin in cancer. Wild-type p53 transcriptionally represses survivin, whereas mutant p53 may contribute to upregulation of survivin. Constitutive STAT3 activation and upregulation by c-H-Ras oncoprotein may also result in survivin over-expression. In nontransformed cell lines, upregulation of survivin in endothelial cells is observed after stimulation with VEGF, basic fibroblast growth factor, and angiopoietin-1. In CD34+ hematopoietic stem cells, there is upregulation of survivin after incubation with a combination of thrombopoietin, Flt3 ligand, and stem cell factor.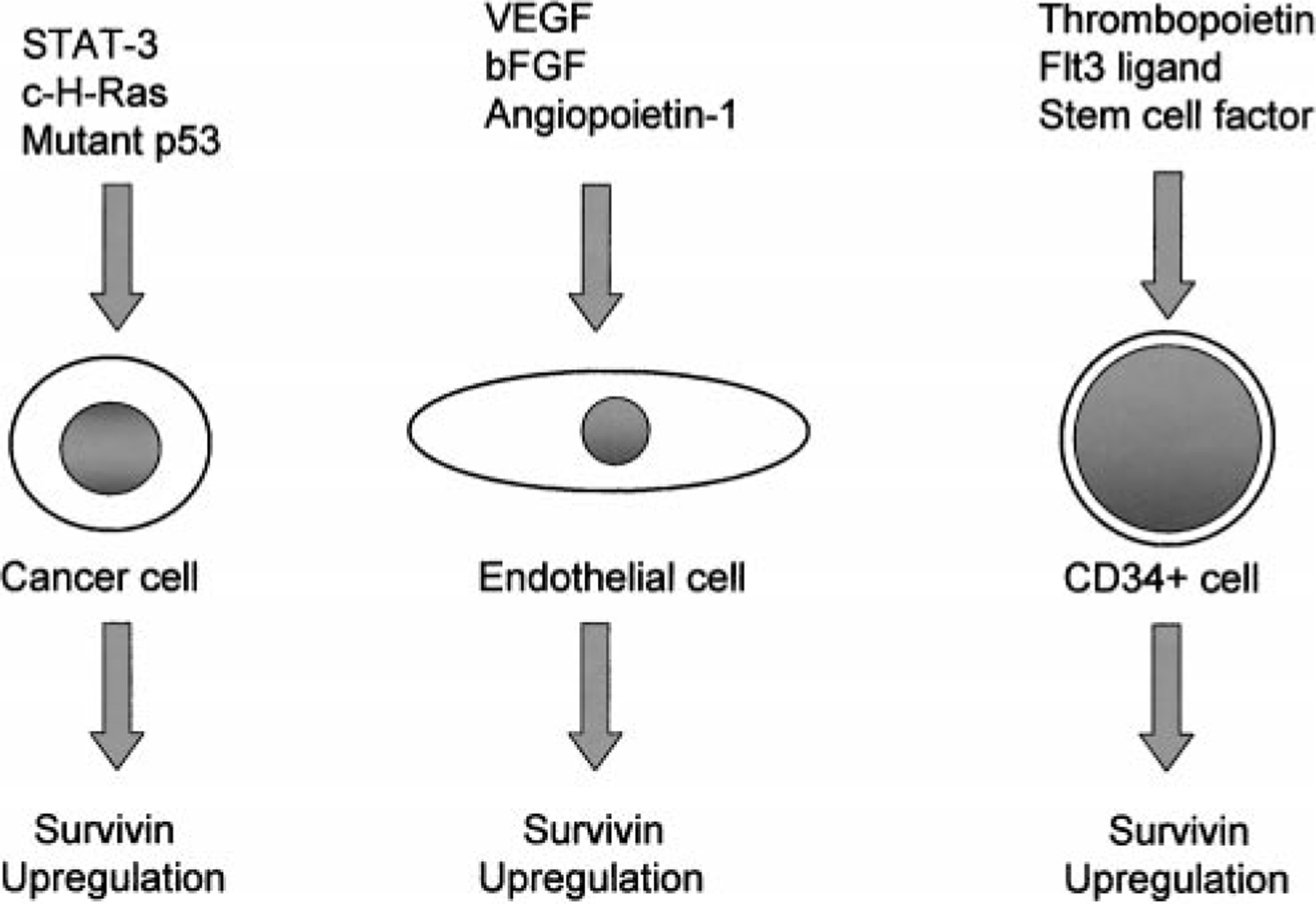
The regulation of survivin is also being investigated in nonneoplastic cell lines (Fig. 3). Survivin messenger RNA (mRNA) and protein expression increase in CD34+ bone marrow cells after incubation with a combination of the hematopoietic growth factors thrombopoietin, Flt3 ligand, and stem cell factor in a cell cycle–independent manner. 23,24 Subsequent removal of these growth factors results in a reduction of survivin mRNA and protein levels, cell cycle arrest, and an increase in the number of apoptotic cells. 24 In addition, inhibition of PI3-K/AKT and mitogen-activated protein kinase (MAPKp42/22) pathways results in reduced survivin expression in growth factor–stimulated CD34+ cells before arresting cell cycle progression. 23 Hence, it is likely that these pathways are involved in cytokine-regulated survivin expression of CD34+ cells and that the regulation by these growth factors is not simply a reflection of cell cycle progression.
Survivin is also induced in quiescent endothelial cells (EC) on mitogenic stimulation with vascular endothelial growth factor (VEGF) and basic fibroblast growth factor. 53 Antisense targeting of survivin results in the loss of cytoprotection afforded by VEGF against ceramide or tumor necrosis factor-α–induced apoptosis, suggesting that VEGF protects EC against apoptosis during angiogenesis by upregulating survivin. 46 VEGF-mediated cytoprotection of EC from the chemotherapeutic agents taxol and vinblastine (VBL), drugs that interfere with microtubule dynamics, results from the upregulation of survivin via activation of the PI3-K/PKB pathway. 73 Furthermore, induction of survivin by VEGF appears to protect cellular integrity against taxol and VBL by maintaining the microtubule integrity. Angiopoietin-1 has also been shown to upregulate survivin in EC, via a PI3-K/AKT pathway, subsequently protecting EC from apoptosis-inducing stimuli. 56 Interference by a dominant negative survivin, however, results in the loss of angiopoitein-1 to protect EC from undergoing apoptosis. Thus, targeting survivin, in addition to inhibiting tumor cell growth, may also prove to be beneficial in inducing apoptosis in proliferating EC of the tumor vasculature. Although upregulation of survivin in EC may be detrimental to the host by supporting tumor growth, survivin plays a crucial protective role in ischemic brain injury. 17 After middle cerebral artery occlusion in mice, survivin can be detected in microvessels of the infarcted areas, apparently both by VEGF-dependent and -independent mechanisms. In addition, heterozygous survivin-deficient mice (survivin ±/−) have significantly decreased vessel density in the infarcted areas as compared with the survivin +/+ mice. Thus, upregulating survivin in EC may prove to be beneficial in promoting angiogenesis in ischemic conditions.
Expression of Survivin in Normal Tissues
Survivin is highly expressed during embryonic development and may be important in tissue homeostasis and differentiation. The gene is quiescent in most terminally differentiated tissues. Among the IAP family members, survivin exhibits the most restricted expression in adult tissues but has been identified in several apoptosis-regulated fetal tissues. 2 In early murine embryogenesis, widespread distribution of survivin is observed, whereas, by late gestation, the protein is present in only limited locations. Normal adult human tissues that express survivin include thymus, 6 CD34+ bone marrow stem cells, 13 and basal colonic epithelium. 25 Survivin is also expressed in the gastric mucosa of adult humans and rats. 15
Of human thymocyte subsets, double-positive (CD4+/CD8+) thymocytes express the highest levels of survivin, demonstrating upregulation in this subset of T lymphocytes. 37 There is downregulation of survivin, however, in mature single-positive (CD4+ or CD8+) T cells, and peripheral T cells are negative for survivin. Thus, survivin may be important in T-cell development. Survivin is also normally expressed in CD34+ hematopoietic stem cells, which raises the concern that survivin-targeted therapy for cancer cells could disturb normal hematopoiesis. 24 CD34+ cells expressed survivin in all phases of the cell cycle, whereas cancer cells specifically expressed survivin in a G2/M-specific manner. Likewise, survivin upregulation in CD34+ cells was not as dramatic as in leukemic cells, implying dysregulation of survivin in leukemia. It is possible that CD34+ cells, under the regulation of hematopoietic growth factors, require only small amounts of survivin to undergo mitosis rather than apoptosis. Survivin expression can also be detected in normal colonic tissue and in hyperplastic, premalignant and malignant lesions of the colon. 25 This indicates that survivin cannot be used as a specific marker for colon cancer. Distribution of survivin within these tissues varies, however, with survivin expression restricted to the basal colonic crypts in normal colonic mucosa. Hyperplastic polyps have uniformly intense staining of all epithelial cells, whereas adenomas and adenocarcinomas are heterogeneous in their staining pattern. In humans and rats, there is strong nuclear expression of survivin in mucosal surface epithelial cells of the stomach. In addition, human chief and parietal cells show nuclear and cytoplasmic survivin expression. Thus, survivin may play a role in gastric mucosal integrity.
Expression of Survivin in Human Cancer
One of the clinically significant features of survivin is its differential distribution in many cancers compared with its limited expression in normal, terminally differentiated tissues. In human medicine, detection of survivin expression by immunohistochemical staining is becoming an important prognostic parameter in a variety of cancers including carcinomas, sarcomas, and hematologic neoplasms. High survivin expression in tumors correlates with a more aggressive and invasive clinical phenotype. Subsequently, a poorer prognosis and a decreased responsiveness to chemotherapeutic agents can be expected. 1,3,4,31,35,36,50,62,71,72,78
Several human carcinomas have been shown to express high levels of survivin: lung, 50 breast, 72 colon, 36 stomach, 42 esophagus, 35 pancreas, 63 bladder, 71 uterus, 61 ovary, 77 liver, 32 and nonmelanoma skin cancer. 29 The overexpression of survivin is consistently associated with more aggressive tumor types and a poorer prognosis than tumors that are negative for survivin. For example, normal oral mucosa and skin, including adnexal structures, are negative for survivin. In contrast, one study found that 56 and 64% of oral and cutaneous squamous cell carcinomas, respectively, were strongly positive for survivin. 51 Likewise, survivin expression correlated with larger, more poorly differentiated tumors, including those that had metastasized to lymph nodes. Detection of survivin in urine may also prove to be a diagnostic marker of bladder cancer. 67 Its presence in urine has been used to diagnose bladder cancer and to differentiate neoplastic lesions from inflammatory conditions with a sensitivity of 100% and specificity of 95%.
Overexpression of survivin has also been documented in leukemias, 4,33 neuroblastoma, 1,31 melanoma, 28 soft tissue sarcoma, 34 and high-grade non-Hodgkin's lymphoma. 6 Low-grade non-Hodgkin's lymphomas, however, do not express survivin. 6 Similar to the poor prognosis observed with its overexpression in carcinomas, high survivin expression in sarcomas is likewise a negative prognostic parameter. In a study involving patients with diffuse, large B-cell lymphoma, survivin was shown to be an independent prognostic factor for poor prognosis in this cancer. 3
Survivin expression associated with viral-induced neoplasms has also been observed, and its expression may be an early indicator of malignancy. 22 Immuno-histochemical evaluation of normal and abnormal human cervical squamous epithelium, including low- and high-grade squamous intraepithelial lesions (LSILs, HSILs) and squamous cell carcinomas, reveals nuclear staining in the normal epithelium, LSILs, and HSILs. Staining intensity is variable, however, with the greatest intensity of staining in the tissues containing human papillomavirus (HPV). In situ hybridization of these HPV-positive tissues reveals colocalization of HPV DNA and survivin, suggesting that survivin plays a role in HPV-mediated cervical dysplasia. Survivin expression is also present in juvenile-onset recurrent respiratory papillomatosis. 57 In comparison with normal laryngeal tissue that is negative for survivin, viral-induced papillomas have increased mRNA and protein expression of survivin. Likewise, the strongest survivin expression is present in papillomas that have undergone malignant transformation.
Targeting Survivin in Cancer
The observation that survivin is expressed in neoplastic tissues, yet undetectable in most normal differentiated tissues makes it a promising therapeutic target in cancer chemotherapy. Several experiments targeting survivin expression are currently under investigation. For example, a replication-deficient adenovirus encoding a survivin Thr34 → Ala (pAd-T34A) mutant to target tumor cells of breast, cervical, prostate, lung, and colorectal cancers causes the tumor cells to undergo apoptosis without affecting proliferation of normal fibroblasts, endothelium, or smooth muscle cells. 47 The combination of pAd-T34A with the chemotherapeutic agent, taxol, results in enhanced tumor cell death. Intratumor administration of pAd-T34A in mice tumors in vivo also inhibits tumor growth and induces apoptosis. Additional evidence that dysregulation of the survivin pathway leads to apoptosis in cancer is seen in melanoma cell lines transfected with T34A, which interferes with phosphorylation of endogenous survivin. This results in spontaneous apoptosis of the melanoma cell line and enhanced in vitro cell death by the chemotherapeutic agent cisplatin. 27 Finally, downregulation of survivin expression using an anti-sense oligonucleotide within a lung carcinoma cell line induces apoptosis and inhibits growth of the neoplastic cells. 55 Furthermore, a synergistic effect was noted when this cell line was treated with antisense oligonucleotide and the chemotherapeutic agent, etopside. This finding demonstrates that targeting of the survivin pathway in cancer, alone or in conjunction with chemotherapeutic agents, has potential as a novel therapeutic regimen.
Immunotherapy also appears to be a plausible approach to treating survivin-positive tumors. Autoanti-bodies against survivin have been detected in humans with lung and colorectal cancer, making this a potential diagnostic tool in certain neoplasms. 60 In some of the lung cancer patients, antisurvivin reactivity was found before clinical disease. Thus, antibodies against survivin may prove to be an early predictive marker of cancer. Spontaneous cytotoxic T lymphocyte response to survivin, in a major histocompatibility complex class I restricted manner, has been detected in patients with chronic lymphocytic leukemia, melanoma, and breast cancer. 8,9 In addition, in vitro cytolytic T-cell induction against a survivin epitope results in cytolytic activity against a wide variety of human tumors, including renal cell carcinomas, breast cancer, colon cancer, multiple myeloma, and leukemias. 64 Hence, survivin appears to be a universal tumor antigen and immunotherapy a conceivable approach to treating survivin-positive tumors.
Conclusions
Survivin is a recently discovered IAP that is expressed in embryonal development and a variety of neoplastic conditions, with minimal expression observed in normal, terminally differentiated adult tissues. Survivin-positive neoplasms are often more aggressive and less responsive to chemotherapeutic agents, making survivin an independent negative prognostic parameter in a variety of human cancers. Survivin is also a potential therapeutic target for cancer chemotherapy; however, survivin is also necessary for proper cell division. Therefore, a thorough understanding of its role in normal tissues must be determined before targeting survivin pathways in cancer. Mechanisms by which survivin suppresses apoptosis are still under considerable debate, and it is not known how survivin is upregulated in neoplastic cells.
Although survivin research is under intense investigation in human medicine, little is known regarding its expression in domestic animals. Information on survivin expression in both normal and abnormal tissues in domestic animals is needed and will facilitate subsequent investigations into its potential use in the diagnosis, prognosis, and therapy in animal neoplasms.
Footnotes
Acknowledgements
We thank Dr. Perry Bain for assistance with the scientific illustrations and Drs. Bruce LeRoy and Jaroslava Halper for critical review of the manuscript.
