Abstract
Oral lichen planus (OLP) is a chronic disease, with a central role to cell-mediated autoimmunity. Osteopontin promotes migration and recruitment of immune cells, CD44 is its receptor, and Survivin seems to be important in skin/mucosa homeostasis. The aim of this study was to investigate their expression in biopsy specimens of patients with different OLP clinical types and healthy controls.
Biopsy specimens from 30 patients with OLP (15 atrophic and 15 hyperplastic) and 15 healthy controls were subjected to immune-histochemical analysis, to detect the expression of osteopontin, CD44, and Survivin in oral epithelia. The distributions of positively stained cells were evaluated with a quantitative method, while the inflammation degree was evaluated with a semi-quantitative one.
Expression of osteopontin and CD44 was higher in OLP than controls, while Survivin expression was lower in OLP patients. There was a greater reduction of Survivin expression in atrophic OLP than hyperplastic OLP. A correlation between osteopontin expression and a high degree of inflammation was found. Furthermore, Survivin expression was higher in cases with low intensity of inflammation.
Osteopontin, CD44, and Survivin seem to be involved in OLP pathogenesis, and further investigations are needed for clarifying their role in this oral disease.
Introduction
Lichen planus is a chronic inflammatory auto-immune disorder mediated by T lymphocytes that involves the stratified squamous epithelial tissue.1,2 The oral mucosa is commonly affected with a variety of clinical presentations, and this area is usually affected before skin and genitals, and may be the only site of involvement.3,4 The oral manifestations are clinically referred as oral lichen planus (OLP), with a prevalence estimated to be 1.27% (0.96% in men and 1.57% in women). 5
The etiology of OLP is still unknown, since the nature of endogenous and/or exogenous agents responsible of T-cell mediated response against cellular surface of keratocytes remains unknown. Many factors have been proposed for etiology,6,7 although none of these is unique.
The pathogenesis of OLP is very complex, and Sugerman et al. proposed a pathogenetic model that implies the involvement of antigen-specific and non-specific mechanisms. 8
The clinical presentation of OLP includes six main different types that may occur singly or simultaneously: reticular, atrophic, papular, erosive (ulcerative), bullous, and plaque-like,5,9 although others have limited the classification to the reticular and erosive types. 10 The predominant clinical morphology of OLP can change over time, making it appear as a long protracted chronic disease with a dynamic evolution. 11 OLP lesions are often bilateral, differentiating them from contact lichenoid reactions of the oral mucosa, while unilateral lesions of OLP are rare and atypical. 12
Histopathological manifestation of OLP is characterized by dense subepithelial lympho-histiocytic infiltrate and intra-epithelial lymphocytes. There is the degeneration of the basal layer, where the keratinocytes forming numerous eosinophilic spherical bodies. 12 There are epithelial basement membrane changes, like breaks, branches, and duplications, 13 causing weakness at the epithelial-connective tissue interface. The World Health Organization classified OLP as a “potentially malignant disorder”, even if there is no definite malignant transformation mechanism identified. The current hypothesis is that chronic stimulation from the inflammatory and stromal cells, and the oxidative stress, provokes DNA damage, resulting in neoplastic changes. 14
Osteopontin (OPN) is a phosphorilated glycoprotein, member of sibling family protein, involved in several physiopathological processes. 15 This protein is synthesized by immune system cells, promoting migration and recruitment of macrophage and T-cells, acting like an inflammatory cytokine.16,17 Recently, an overexpression of OPN was found in OLP, suggesting an association between this protein and the clinical types of OLP. 18 Specific cell surface molecules control the migration and recruitment of T cell subpopulations into the inflammation site. Among multiple receptors for OPN, CD44 is the most characterized receptor that appears to mediate cell chemotaxis and attachment, 19 and plays important roles in lymphocyte activation, proliferation, and migration.20,21 Liu et al. showed that the elevated levels of plasmatic OPN act as a potent CD44 upregulating factor and may be responsible for the overexpression of CD44 in OLP patients. 22
Survivin, a member of inhibitor of apoptosis (IAP) family protein, 23 is a 16.5 kDa protein that plays a double role: on the one hand controls cell division, the other directly interacts with several caspases to inhibit apoptosis, playing a pivotal role in cell survival.24–27 There are many studies focused on the relationship between oral squamous cell carcinoma and Survivin,28–30 while the expression of this protein in the context of its relationship to chronic inflammatory diseases were limited, with only one study regarding the expression of survivin in OLP. 31 The aim of this study is to evaluate the expression of OPN, CD44, and Survivin in a group of patients affected by OLP, and assess if there are any differences in the expression levels of these proteins between erosive and reticular form of OLP.
Materials and methods
Selection of cases and classification criteria
A total of 30 patients (13 men, 17 women) with a mean age of 43.9 years (age range, 25–83 years) with a diagnosis of OLP were selected in this study. In addition, 15 healthy individuals were included as normal controls. The group of patients for this study considered only those patients with clinical diagnosis of OLP, confirmed with histopathological diagnosis. All lesions were bilateral, and all patient did not have either topical or systemic medications during the previous 3 months. All the controls approved the informed consent, and showed normal mucosa at the side of oral specimen.
The clinical diagnoses of OLP were classified as “hyperplastic” when the affected mucosa presented the clinical form of reticular OLP or cases with more clinical forms simultaneously, but with a predominance of reticular form and with the absence of clearly atrophic/erosive areas. On the other hand cases were classified as “atrophic” when they predominantly or exclusively presented mucosal lesions with atrophic/erosive OLP. Following these criteria, the sample of patients was divided in two groups: “hyperplastic OLP” (15 patients; mean age, 45.2 years), and “atrophic OLP” (15 patients; mean age, 42.6 years).
Histopathological diagnosis was performed on histological sections stained with hematoxylin-eosin to confirm clinical diagnosis. Furthermore, the entity of inflammation has been considered, classifying OLP cases through a semi-quantitative scale: mild inflammation (+), moderate inflammation (++), and intense inflammation (+++).
Immunohistochemical staining
Serial sections of 4-μm from formalin-fixed, paraffin embedded tissue blocks were cut and mounted on poly-L-lysine-coated glass slides. Histopathological diagnosis of OLP was evaluated independently by two of the authors (CR and AZ), confirming the clinical diagnosis. Only sections containing sufficient epithelium to assess the antibody reactivity with 1000 cells were considered for this study. The following primary monoclonal antibodies were used for immunohistochemical staining: rabbit anti-human OPN antibody (1:200; R&D Systems, Inc., Minneapolis, MN, USA), mouse anti-human CD44 antibody (1:150; Biolegend, San Diego, CA, USA), and mouse anti-human Survivin antibody (1:200; ab3258, Abcam plc, Cambridge, UK). The standard streptavidin-biotin-peroxidase complex technique was performed, using the L.V. Dako LSAB kit (DAKO A/S, Carpinteria, CA, USA) after antigen retrieval by pressure-cooking. Briefly, deparaffined sections were immersed in a 10–3 M sodium citrate buffer (pH 6.0) after bringing the solution to a boil in a pressure cooker and heated twice for 3 min each at a 10-min interval while keeping the pressure indicator valve rising. After quenching in 3% hydrogen peroxide and blocking, slides were incubated overnight at 4°C, with the most suitable concentration of each antibody, as previously reported. Biotinylated anti-rabbit immunoglobulin and streptavidin conjugated to horseradish peroxidase were subsequently applied. Finally, 3,3’-diaminobenzidine was used for color development, and hematoxylin was used for counterstaining. Negative control slides in the absence of primary antibody were included for each staining. Two observers evaluated the results of the immunohistochemical staining separately.
Immunohistochemical quantification
To evaluate the proteins expression, for each antibody, a mean percentage of positive cells was determined by two observer, examining 300 cells in at least five areas at ×400 magnification.
A semi-quantitative assessment of inflammation was performed assigning cases to one of the four following categories: score 0, when the stained cells comprised 0–10% of the total; score (+), when the stained cells comprised 10–29% of the total cell population; score (++), when the stained cells comprised 30–49% of the total; score (+++), when the stained cells comprised >50%.
Statistical analysis
Data were analyzed using GraphPad Prism software version 5.00 for Windows (GraphPad Software, San Diego, CA, USA, www.graphpad.com). The Student’s t-test and one-way ANOVA were used for statistical analysis between groups. A P value of less than 0.05 was accepted as statistically significant. Data were presented as mean ± standard deviation.
Results
Assessment of the immunohistochemical staining of biopsy specimens
The data concerning the expression of different markers are summarized in Table 1.
Summary of clinical characteristic and immunochemical staining.

The results showed that OPN was not detectable in the epithelium of normal mucosa, while the localization in OLP cases was mainly in the cytoplasm of keratinocytes (Figure 1a). The levels of OPN in OLP patients showed a slight difference of expression between atrophic OLP and hyperplastic OLP (49.00 ± 17.75 vs. 47.00 ± 22.42), without reaching statistical significance (Figure 2a).
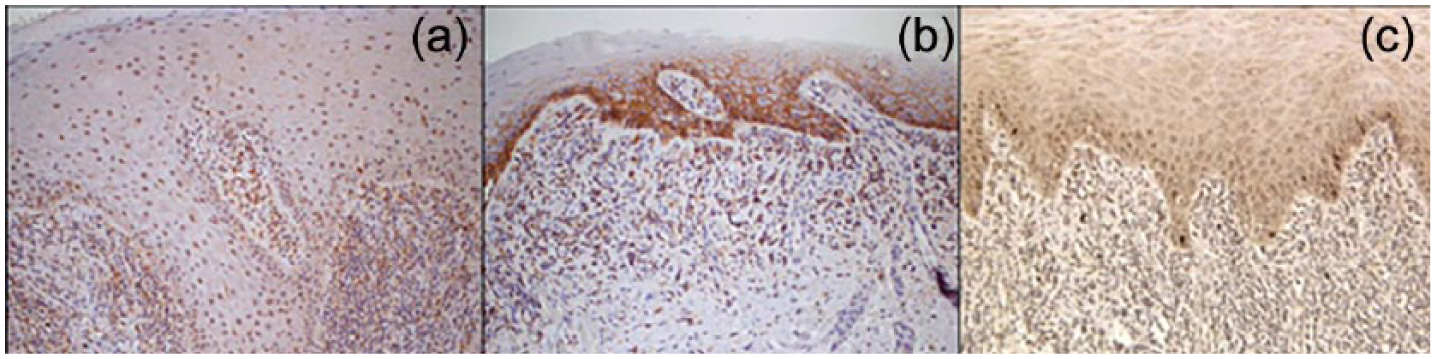
Immunohistochemical localization of OPN (a), CD44 (b), and Survivin (c) in OLP.

Comparison of expression levels of OPN, CD44, and Survivin between atrophic OLP and hyperplastic OLP.
The distribution of positively stained epithelial cells showed an increase of expression of CD44 in OLP cases compared to the keratinocytes of normal oral mucosa (Figure 1b). In contrast, there was found no statistically significant difference between atrophic and hyperplastic OLP (52.67 ± 16.78 vs. 57.33 ± 16.46) (Figure 2b).
The immunostaining for Survivin was evident in basal keratinocytes, with a reduction of expression in oral mucosa of OLP patients (Figure 1c). As regards the expression in the two groups of OLP patients, the expression level of Survivin was higher in hyperplastic OLP (14.13 ± 7.82) compared to atrophic OLP (3.53 ± 5.37), with a statistically significant difference (P = 0.0002) (Figure 2c).
Correlation between the expression of markers and inflammation
Taking into account the different degrees of inflammation as classification criteria, the percentage of cells expressing OPN in cases with intense inflammation (score

Correlation between OPN and Survivin expression with different degrees of inflammation.
As for CD44, no correlation between the expression and the various degrees of inflammation was found. Survivin expression was greater in cases of mild inflammation (score
Discussion
The OLP is a chronic inflammatory disease of the squamous epithelium of oral mucosa, representing the most common no infectious mucosal disease of the oral cavity, with a prevalence of 1.27%.5,6 Several factors have been proposed for the etiology, including: genetic background, dental materials, drugs, infections, association with other autoimmune diseases, immunodeficiency, food allergies, stress, habits, trauma, diabetes and hypertension, malignancies, and bowel disease, although none of these is unique. 6 The OLP is a chronic disease, with dynamic evolution in distribution, severity, and clinical type. In fact the clinical presentation includes six main different forms, which change in the course of time. 11
The complex pathogenesis of the OLP has been interpreted by Sugerman et al. with a model that involves antigen-specific and non-specific mechanisms. 8 The CD8+ lymphocytes bind to a specific antigen (AG1) expressed by MHC I of keratocytes. Their activation in CTL requires CD4+ lymphocytes, which are activated in T helper 1 (Th1) cells, before exercising their functions. The activation of CD4+ depends on the presentation of an antigen (AG2) with MHC II by an antigen presenting cell (AG1 and AG2 may be identical or different peptides). The recognition of antigen associated with MHC II, in combination with the presence of co-stimulatory molecules initiates Th1 differentiation of the CD4+ lymphocytes. The final effect is that Th1 secretes cytokines detected by CD8+ lymphocytes that become CTL. CTLs proceed with basal keratocyte lysis especially by TNF-α, but probably also through granzyme B and Fas. The activated cells clonally expand and secrete various cytokines that stimulate intra-lesional migration of mast cells and their degranulation. The mast cells release TNF-α, which increases the expression of receptors for the lymphocytes and the secretion of cytokines by the CTL. In addition, mast cells release chymase that damages the basal membrane, directly or indirectly. The destruction of the membrane allows the passage of lymphocytes, and removes the stimulus given to the keratocytes from the basal membrane to the survival, leading to their apoptosis.
It is evident how this pathogenetic model gives a central role to cell-mediated immunity. For this reason, we examined OPN and CD44, two proteins involved in immune cells activation and migration.16,20 OPN is a phosphorylated glycoprotein synthesized by the cells of immune system, that promotes migration and recruitment of macrophage and T lymphocyte, acting like an inflammatory cytokine.16,17 CD44 is a cell surface glycoprotein that acts as a receptor capable of mediating cells chemotaxis and attachment, playing important roles in lymphocyte activation, proliferation, and migration. 21 CD44 appears to be the most characterized receptor for OPN, and seems to be upregulated by elevated serum levels of OPN, which may be responsible for the overexpression of CD44 in OLP patients. 19 Furthermore, Liu et al. showed that the interaction between OPN and CD44 promotes the survival of CD8+ lymphocytes. 22
In agreement with other studies, immunohistochemical analysis of OPN showed no detection of this protein in the epithelium of normal mucosa, while the difference of expression between atrophic OLP and hyperplastic OLP was not statistically significant. 18 The differences of OPN levels between normal oral mucosa and OLP patients are consistent with the role that this protein seems to have in cell-mediated immunity. In fact this protein interact with macrophages, inducing the expression of IL-12, that leads to the differentiation of T lymphocytes in Th1, while the engagement with CD44 induces downregulation of macrophagic production of IL-10, with the consequent inhibition of the Th2 response. 16 Furthermore, the increased expression of OPN in oral mucosa of OLP patients is consistent with the higher levels of this protein in other autoimmune disorders,32,33 highlighting the involvement of this protein in diseases.
Immunostaining for CD44 showed an increase of expression in OLP cases compared to normal oral mucosa, without a statistically significant difference between atrophic OLP and hyperplastic OLP. The expression of CD44 showed a behavior similar to that of OPN regarding the different expression between normal and OLP mucosa. These results are in agreement with other studies, 22 confirming that interaction between CD44 and OPN has been implicated in regulation of inflammation in OLP.
The expression of OPN was higher in cases with intense inflammation (score
The alterations present in the mucosa of OLP patients may involve the homeostatic activities of oral epithelial cells, possibly resulting in an imbalance between cell proliferation and apoptosis.
31
Survivin, a member of IAP family protein, plays a pivotal role in cell survival with a double function: controlling cell division and interacting with several caspases to inhibit apoptosis.24,25 It is reasonable to think that Survivin is involved in OLP, although studies conducted to date were focused on oral squamous cell carcinoma,28–30 while only one study investigated the expression of Survivin in OLP.
31
The differences of Survivin expression between normal mucosa and OLP mucosa were in agreement with previous findings,
31
confirming that this protein is important in skin homeostasis, both for cell cycle progression to apoptosis resistance in human keratinocytes.
34
To the best of our knowledge, this is the first work that evaluated the differences of Survivin expression between different forms of OLP. Our results of immunohistochemical staining showed higher expression level in hyperplastic OLP compared to atrophic OLP, probably because the function of Survivin in OLP is the control of cell division, rather than inhibition of apoptosis. It should be considered that the cases classified as atrophic OLP also had erosive forms, where the destruction of the epithelium, and therefore also the basal layer, led to a lack of expression of Survivin. Survivin expression was greater in cases of mild inflammation; this is not surprising, considering that all but two cases with “score
The results of our immunohistochemical examination suggest that OPN, CD44, and Survivin are involved in the pathogenesis of OLP, and further investigations with a larger sample are useful for clarifying the role of these proteins involved in inflammatory pathways in OLP.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
