Abstract
A German Shepherd Dog was evaluated for clinical signs of multifocal, progressive brain disease. Despite supportive care, the dog died shortly after hospital admission. Granulomatous meningoencephalitis with intralesional fungal hyphae was diagnosed postmortem. The fungus was identified as Sporobolomyces roseus by polymerase chain reaction amplification and sequencing of the ITS2 region.
Fungal infection is an uncommon cause of granulomatous meningoencephalitis in dogs; most canine cases of mycotic meningoencephalitis have been attributed to Blastomyces dermatitidis, Histoplasma capsulatum, Coccidioides immitis, Cladophialophora bantiana, and species of Aspergillus and Fusarium.4,6,7 In the case reported here, fungal hyphae in lesions of granulomatous meningoencephalitis were identified as Sporobolomyces roseus.
Case History
A 9-year-old, neutered, female, German Shepherd Dog was presented for evaluation of a 3-day history of anorexia, lethargy, and signs of rapidly progressive central nervous system disease. The dog was unable to stand or walk without support and circled to the left. It had been treated for a 3-week history of presumptive back pain with nonsteroidal anti-inflammatory drugs. After the onset of therapy, the owner reported a decreased appetite and intermittent vomiting.
Upon presentation, the dog was obtunded with a left-sided head tilt and had nonambulatory tetraparesis, with reduced or absent postural reflexes in the forelimbs and hind limbs, respectively. Cranial nerve function was considered normal. The neurologic signs were consistent with a multifocal disease, affecting the left forebrain and brainstem.
Abnormal hematologic findings included mild leukocytosis and a mild normochromic, normocytic anemia. The chest radiographs were unremarkable, except for evidence of thoracic spondylosis deformans. Heterogeneous fluid was detected in the peritoneal cavity by ultrasound. An ultrasound-guided abdominocentesis yielded a turbid, bloody fluid. The differences between serum and the abdominal fluid in glucose and lactate concentrations were considered consistent with septic peritonitis; gastrointestinal perforation was suspected.
With magnetic resonance imaging of the brain, poorly defined foci of contrast enhancement were seen in the right frontal lobe and both parietal lobes of the cerebrum and in the left cerebellum. Cerebrospinal fluid (CSF), collected from the cerebellomedullary cistern, had mildly elevated total protein (35.5 mg/dl; reference value, <28 mg/dl) and a moderate increase in leukocytes (70/μl; reference value, <8/μl). Cytologic impression was mixed pleocytosis, with a predominance of monocytes and neutrophils. Some neutrophils had toxic changes, with less condensed chromatin. No organisms were observed. Imaging and CSF changes were consistent with meningoencephalitis. Despite administration of intravenous fluids and broad-spectrum antimicrobial therapy, the dog died 1 day after presentation.
Pathologic Findings
At necropsy, multiple variably sized (0.2–2 cm diameter), irregular, gray-yellow foci of malacia were at the junction of the diencephalon and mesencephalon. Additional findings included mild valvular endocardiosis with chronic passive congestion of the liver. Both kidneys had chronic infarcts. A perforating pyloric ulcer accounted for the peritonitis that had been diagnosed clinically.
Samples of the brain, kidney, spleen, liver, lung, heart, and stomach (pylorus) were fixed in neutral buffered 10% formalin and routinely processed for histologic evaluation. Paraffin sections, 5 µm thick, were stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), Fontana-Masson, and Grocott’s methenamine silver (GMS).
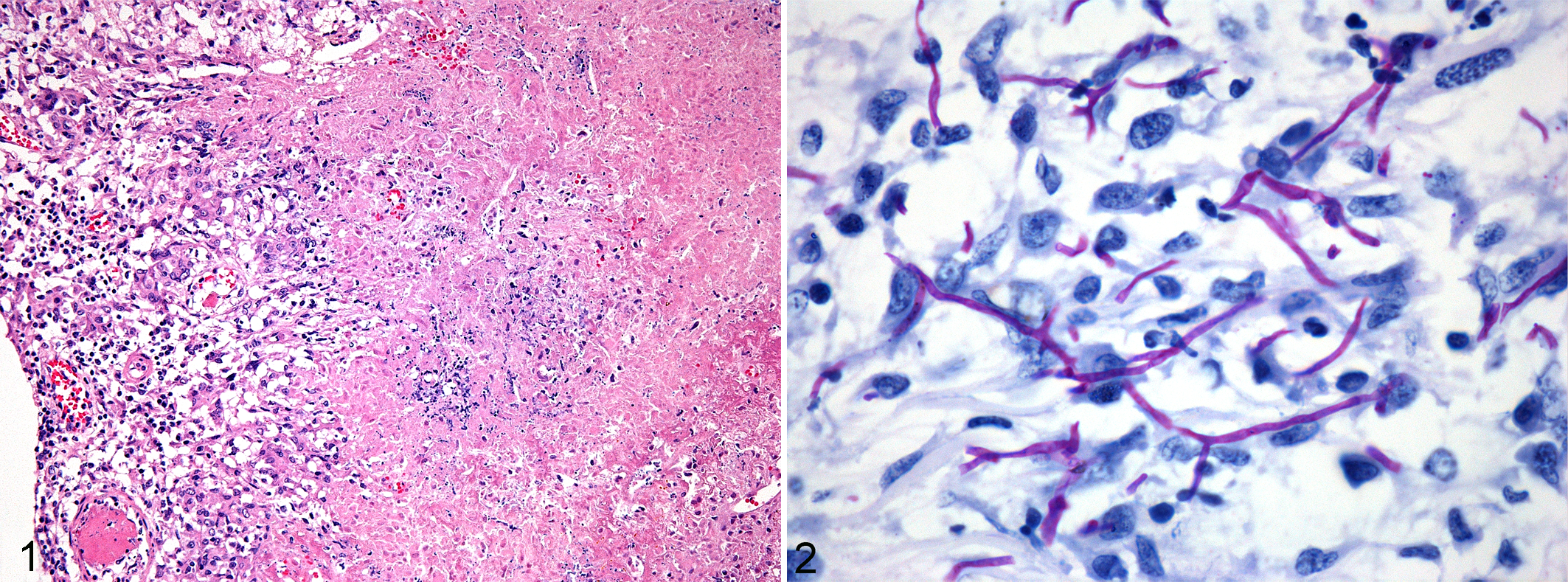
Histologically, granulomatous inflammation and fungi were seen only in the brain. The most severe lesions were in the medial part of the mesencephalon at the level of the rostral colliculi. A granuloma was also in the subarachnoid space and pia mater of the cerebellum at the level of the left facial nucleus. Another granuloma was in the myelencephalon, medial to the nucleus of the spinal tract of the trigeminal nerve. The granulomas (0.3–0.5 mm in diameter) consisted of epithelioid macrophages and multinucleated giant cells arranged around eosinophilic granular material, containing fibrin and cellular debris (Fig. 1). Reactive astrocytes were prominent at the borders of the granulomas in vacuolated (edematous) neuropil. With PAS and GMS, numerous irregularly branching (at 30°–90°), septate hyphae, 2 to 4 µm in diameter and up to 15 µm in length, were detected in the granulomas (Fig. 2). Although the hyphae were intensely positive with PAS and GMS, they were barely visible with HE and not reactive with Fontana-Masson technique for melanin. The fungi were not found in macrophages but were numerous in the lumen of cerebral and meningeal vessels; affected vessels had thrombosis with mural infiltration by macrophages and fibrinoid change.
Molecular Diagnostics
To speciate the fungus, DNA was extracted from formalin-fixed paraffin sections of brain using a commercially available kit (DNeasy Blood and Tissue Kit, Qiagen Sciences, Hilden, Germany). The ITS2 region was amplified and sequenced using the primers described by Turenne et al 3 and De Baere et al. 11 The base-pair length of the PCR product was 371 nucleotides. The ITS2 sequence thus obtained was compared with all sequences submitted to Genbank using the Basic Local Alignment Search Tool (BLAST) software (National Center for Biotechnology Information, Bethesda, MD) and had 96% homology with S roseus.
Discussion
S roseus is one of the most common phylloplane ballistoconidium–forming yeast species. 12 With the exception of the tropical zone, S roseus is geographical distributed worldwide. The yeast cells of S roseus multiply by budding but can also form ballistospores that are discharged by drop excretion. 12 Ballistospores are found only in culture and not in histologic sections. 5 Disseminated infection with Sporobolomyces sp has been reported with Sporobolomyces salmonicolor in 2 human patients with acquired immunodeficiency8,10 and in Chinook salmon. 9 The predisposition of German Shepherd Dogs to disseminated aspergillosis has been attributed to an inappropriate mucosal IgA response, 2 but no evidence of immune dysfunction was detected to explain susceptibility to mycotic meningoencephalitis in this case.
Biopsy may be necessary for antemortem diagnosis of intracranial mycosis, if organisms are not found in CSF. The spatial association of the fungal hyphae with the granulomas in the brain leaves little doubt about the diagnosis of mycotic meningoencephalitis. Cultures of the CSF or brain were not performed, so the fungus was speciated by sequence determination of the ITS2 region in formalin-fixed, paraffin-embedded tissue sections of brain. Although sporobolomycosis has been wrongly diagnosed from human CSF samples that were contaminated by airborne S salmonicolor spores, 1 contamination seems unlikely in this case; the obtained sequence was clear, which indicates that the polymerase chain reaction amplified only 1 species.
The brain may have been infected by direct extension from nasal cavity or sinuses. However, in this dog, the absence of lesions in the nasal cavity or paranasal sinuses at necropsy, the distribution of the lesions in the brain, and the presence of fungal hyphae in cerebral and meningeal vessels are more consistent with hematogenous dissemination.
Footnotes
Acknowledgements
We thank Sofie De Bruyckere for performing the DNA extraction and Christian Puttevils, Delphine Ameye, and Sarah Loomans for technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
