Abstract
Two llamas with pulmonary tumors were examined. Llama No. 1 had multiple nodules throughout the lung that consisted histologically of solid clusters of polygonal to spindle cells with rare glandular differentiation. Intravascular emboli were common. Similar neoplastic masses were present in the kidney, heart, and liver. Immunohistochemically, neoplastic cells were positive for broad-spectrum cytokeratins (CKs), high- molecular weight CKs, CKs 5/6, and vimentin. The diagnosis was pulmonary carcinoma. Llama No. 2 had pulmonary nodules without extrapulmonary involvement. Microscopically, neoplastic cells formed acini lined by simple epithelium and solid cords of squamous cells that sometimes surrounded acini. Neoplastic cells were strongly positive for broad-spectrum CKs and weakly positive for thyroid transcription factor-1. The diagnosis was adenosquamous carcinoma. Pulmonary tumors account for 23% of neoplasms in South American camelids in our laboratory, making this the second most common type of neoplasm after lymphosarcoma.
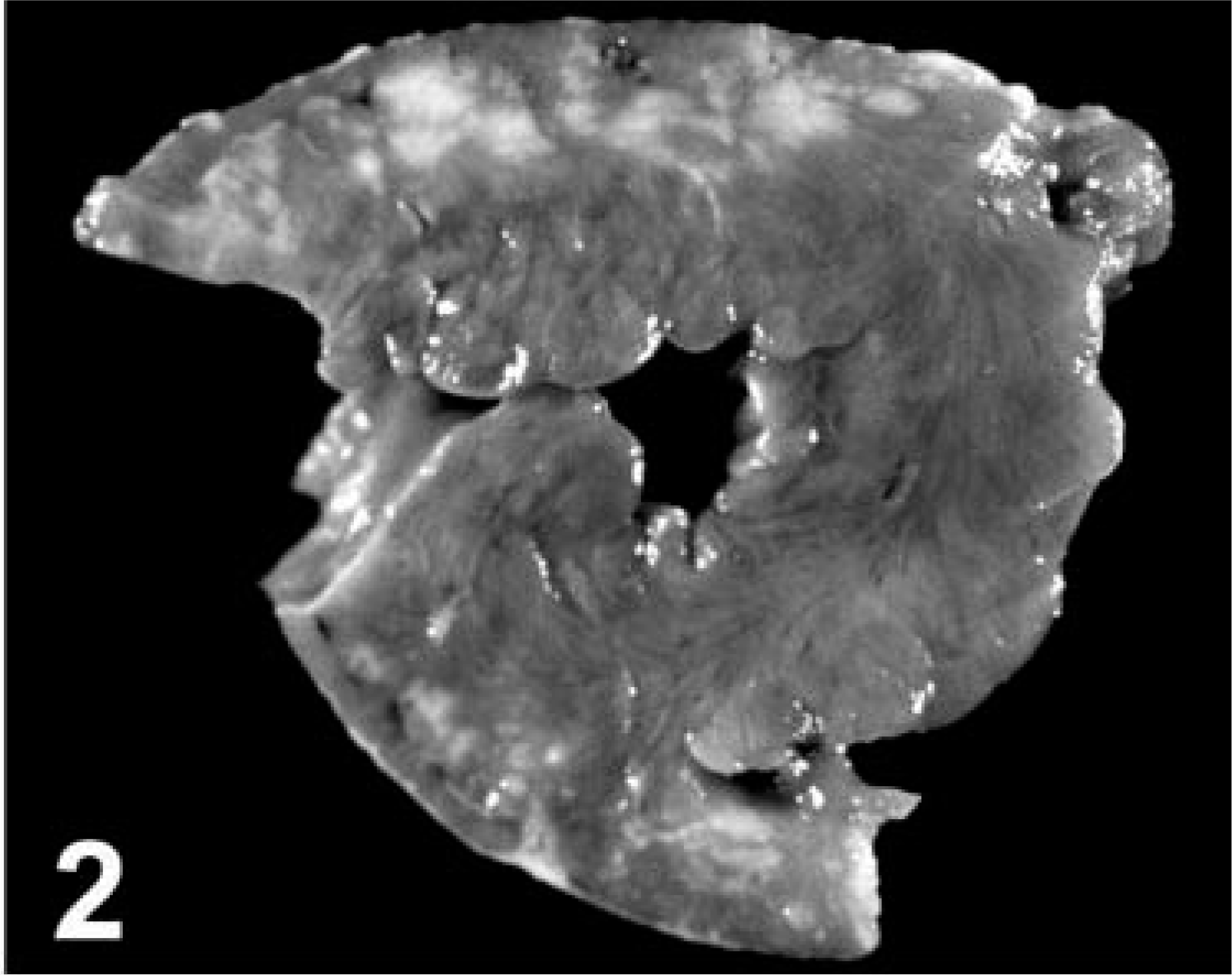
Only one case of pulmonary neoplasia has been reported in South American camelids. 18 We describe the clinical and pathologic findings in two llamas with pulmonary neoplasia. Llama 1 was a 12-year-old male with a 1-month history of progressive weight loss, anorexia, lethargy, and dyspnea. An ultrasound study revealed pleural and pericardial effusion and an apparent cardiac mass. The differential diagnosis for the cardiac mass included lymphoma, hemangiosarcoma, mesothelioma, and possible pleuropneumonia. Cytology of the pleural fluid revealed a round cell population suggestive of lymphoma. The owner elected euthanasia. Bilaterally, the parietal pleura was adhered multifocally to the visceral pleura that contained 5-mm to 10-cm-diameter tan pulmonary nodules (Fig. 1). The largest nodule was in the right caudal lobe. The lumen of one branch of the pulmonary vein was occluded by a tan mass. Fewer but similar nodules were observed in the liver and kidney. The myocardium of both ventricles had confluent and variable-sized tan areas (Fig. 2).

Lung; llama No. 1. Multiple subpleural nodules.
Llama No. 1. Coronary section of heart with confluent areas of neoplastic growth (pale areas).
Formalin-fixed tissues from multiple organs were embedded in paraffin and stained with hematoxylin and eosin. Neoplastic masses in the lung and elsewhere were not encapsulated and consisted of round to polygonal or spindle (pleomorphic) cells arranged in solid groups with rare glandular differentiation (Fig. 3). Neoplastic cells had distinct cell limits, moderate to abundant eosinophilic to amphophilic cytoplasm, and large round nuclei with one or multiple nucleoli. This tumor had an average of three mitotic figures per high-power field. There were multifocal necrosis and mixed leukocytic infiltrates within neoplastic nodules. Numerous intrapulmonary blood and lymphatic vessels were filled with neoplastic cells. Squamous differentiation (e.g., intracytoplasmic keratin and intercellular bridges) was particularly prominent in the renal nodules (Fig. 4). Neoplastic masses in the heart and liver were phenotypically similar to those in the lung.
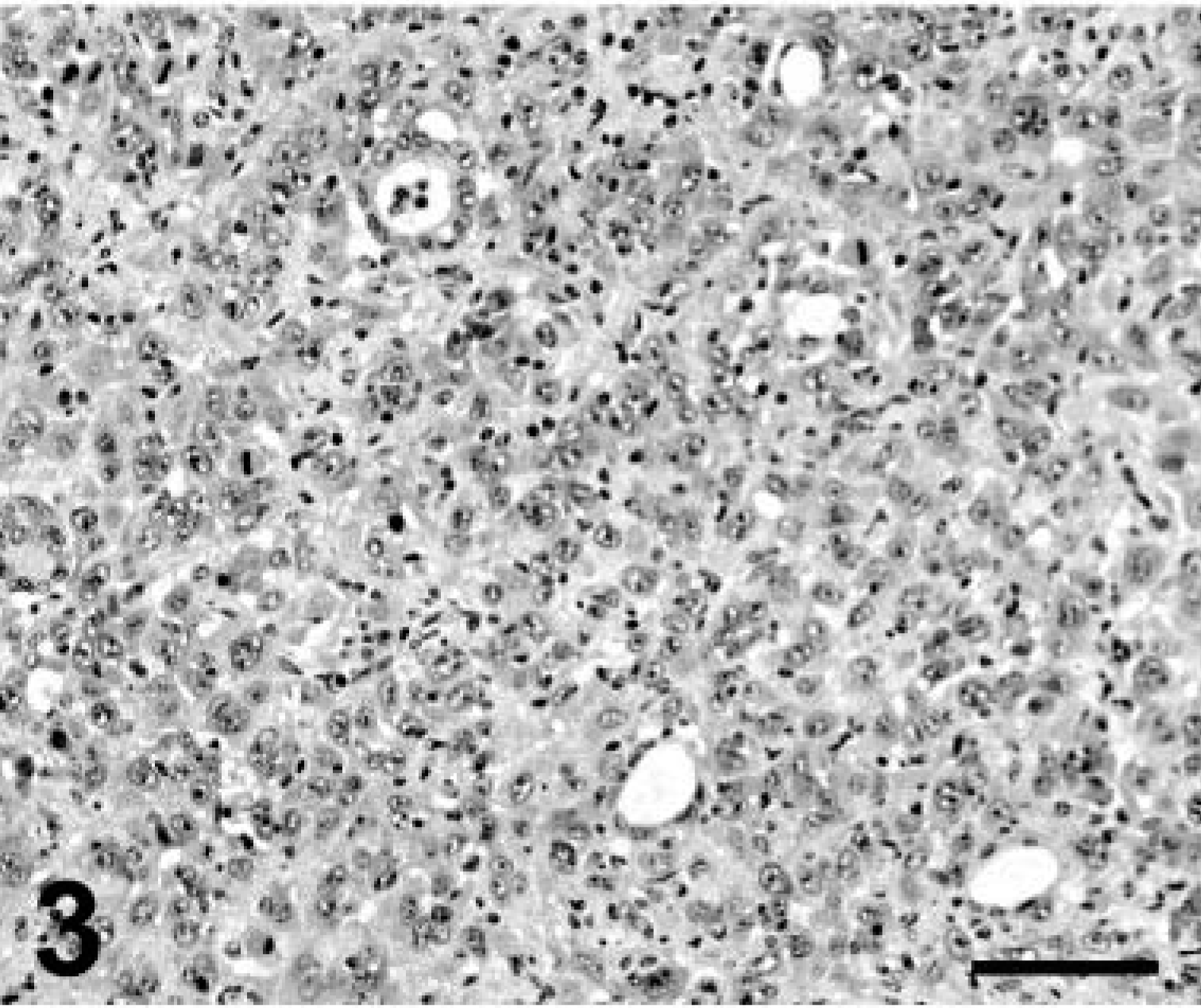
Lung; llama No. 1. Neoplastic cells in solid aggregates with occasional acinar formation. HE. Bar = 150 µm.
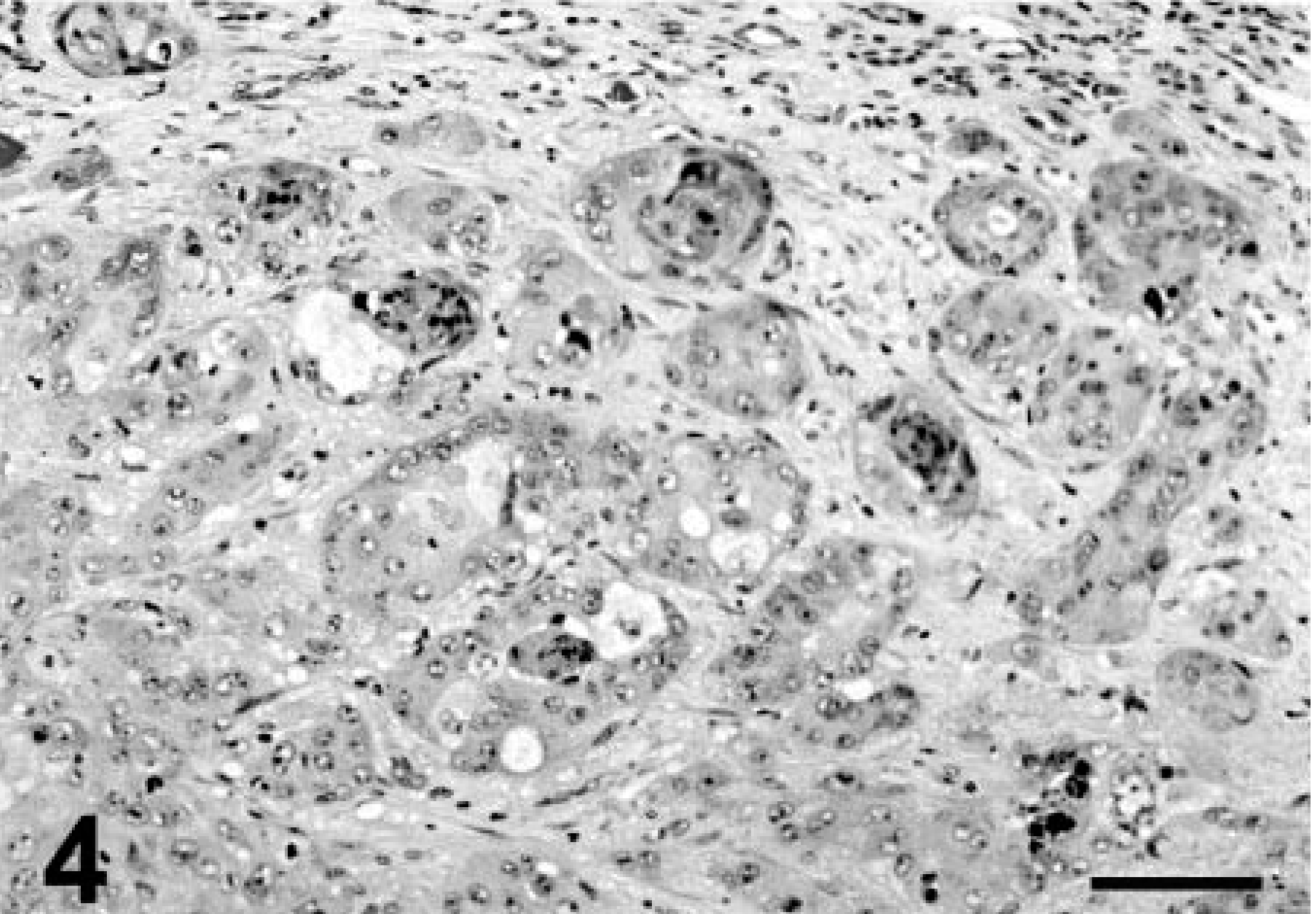
Kidney; llama No. 1. Carcinoma. Distinct squamous differentiation. HE. Bar = 100 µm.
Formalin-fixed, paraffin-embedded samples of the pulmonary and renal masses were examined immunohistochemically using an immunoperoxidase detection system and antibodies to broad-spectrum cytokeratins (CKs), low–molecular weight (LMW) CKs, high–molecular weight (HMW) CKs, CKs 5/6, CK 7, CKs 8/18, CK 20, calretinin, desmin, carcinoembryonic antigen (CEA), thyroid transcription factor–1 (TTF-1), uroplakin III, and vimentin following procedures reported previously. 17,19,20 Normal canine and llama lung samples were used as tissue controls. The pulmonary and renal masses had similar immunohistochemical staining although with slight variation in intensity. There was moderate to intense staining for broad-spectrum CKs, HMW CKs, CKs 5/6, and vimentin; rare neoplastic cells in the kidney were positive for CEA and CK 20 (Figs. 5, 6). Neoplastic cells in the organs examined were negative for CK 7, CKs 8/18, calretinin, LMW CKs, TTF-1, and uroplakin III.
Lung; llama No. 1. Numerous neoplastic cells are positive for broad-spectrum CKs. Streptavidin-peroxidase stain with Mayer's hematoxylin counterstain. Bar = 150 µm.
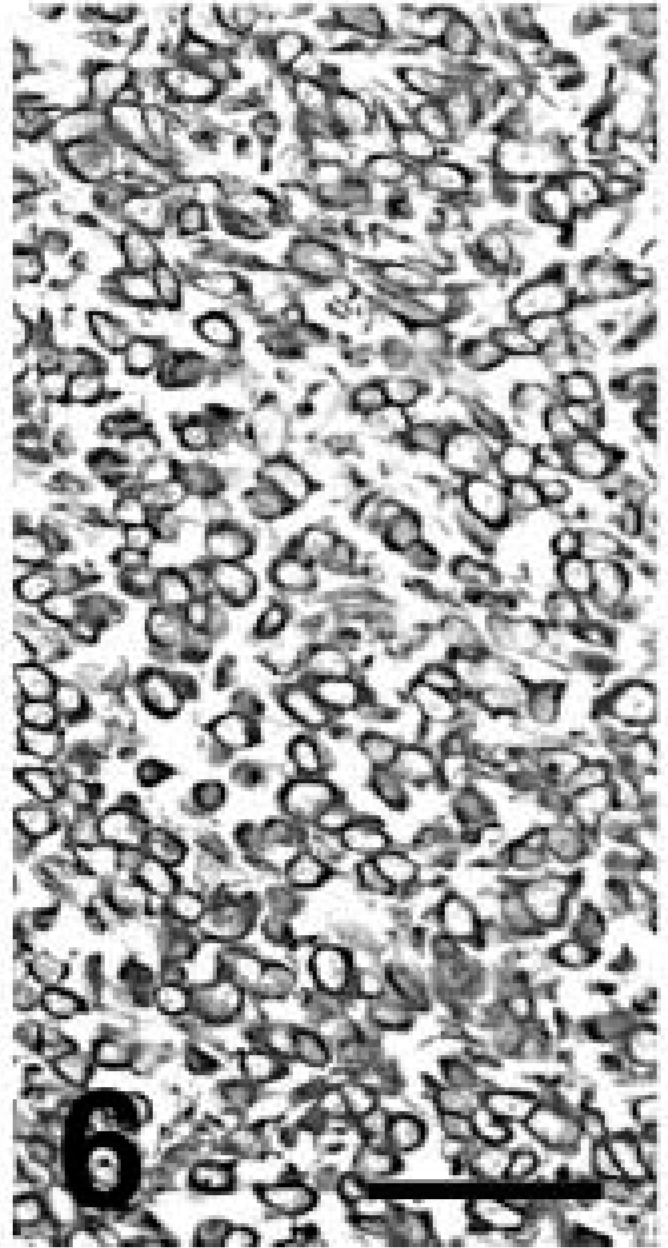
Lung; llama No. 1. The majority of neoplastic cells are also positive for vimentin. Streptavidin-peroxidase stain with Mayer's hematoxylin counterstain. Bar = 150 µm.
Because of the atypia of this tumor and to rule out mesothelioma, paraffin-embedded samples of the pulmonary mass were processed for electron microscopy. After dewaxing and dehydration, samples were embedded in epon-araldite and postfixed in osmium tetroxide. Neoplastic cells had variable numbers of organelles including mitochondria and endoplasmic reticulum. Intercellular junctions were not prominent. Neither microvilli nor glycogen granules was observed. The final diagnosis was pulmonary carcinoma.
Llama No. 2 was an adult female presented to the veterinary teaching hospital with a history of several days of anorexia. A gastrointestinal impaction was diagnosed based on radiographic findings, decreased gastrointestinal motility, and decreased fecal output. Treatment to improve this condition was unsuccessful, and because of deterioration of this animal's condition, it was euthanatized.
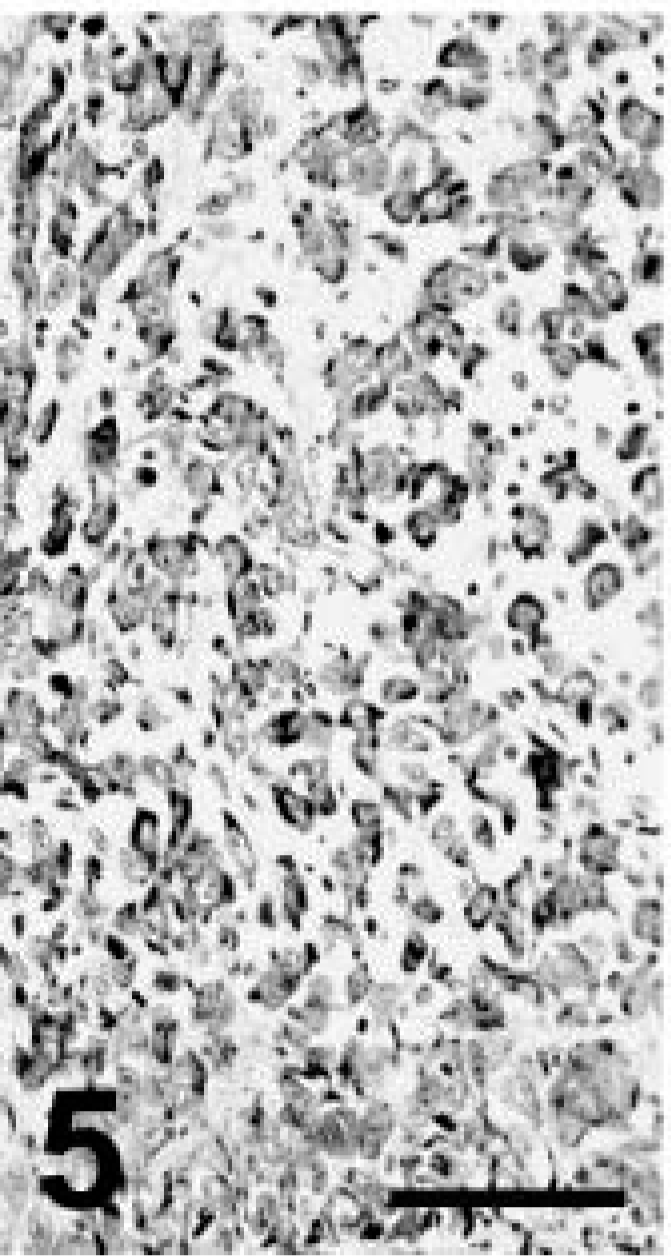
Grossly, this llama had depleted fat stores and multiple, less than 3-cm-diameter tan nodules along the dorsal aspect of both lungs, extending 1–2 cm into the pulmonary parenchyma. Microscopically, the pulmonary nodules were unencapsulated and consisted of acini lined by cuboidal to low columnar simple epithelium with occasional cilia. Frequently, the acini were surrounded by several layers of larger epithelial cells with hypochromic nuclei and pale cytoplasm (Fig. 7). A second growth pattern consisted of cords and streams of squamous epithelial cells with distinct cell limits and no apparent acinar formation. Some clusters of neoplastic cells had central keratin pearls. Stroma was abundant and poorly cellular consisting of plump spindle cells and eosinophilic to amphophilic extracellular matrix. Mitoses were not apparent. There was strong staining for broad CKs and occasional weak staining of neoplastic cells for TTF-1, particularly in the nuclei of cells lining acinar structures and less commonly in squamous cells (Fig. 8). Other markers were not used in this case. The diagnosis was pulmonary adenosquamous carcinoma.
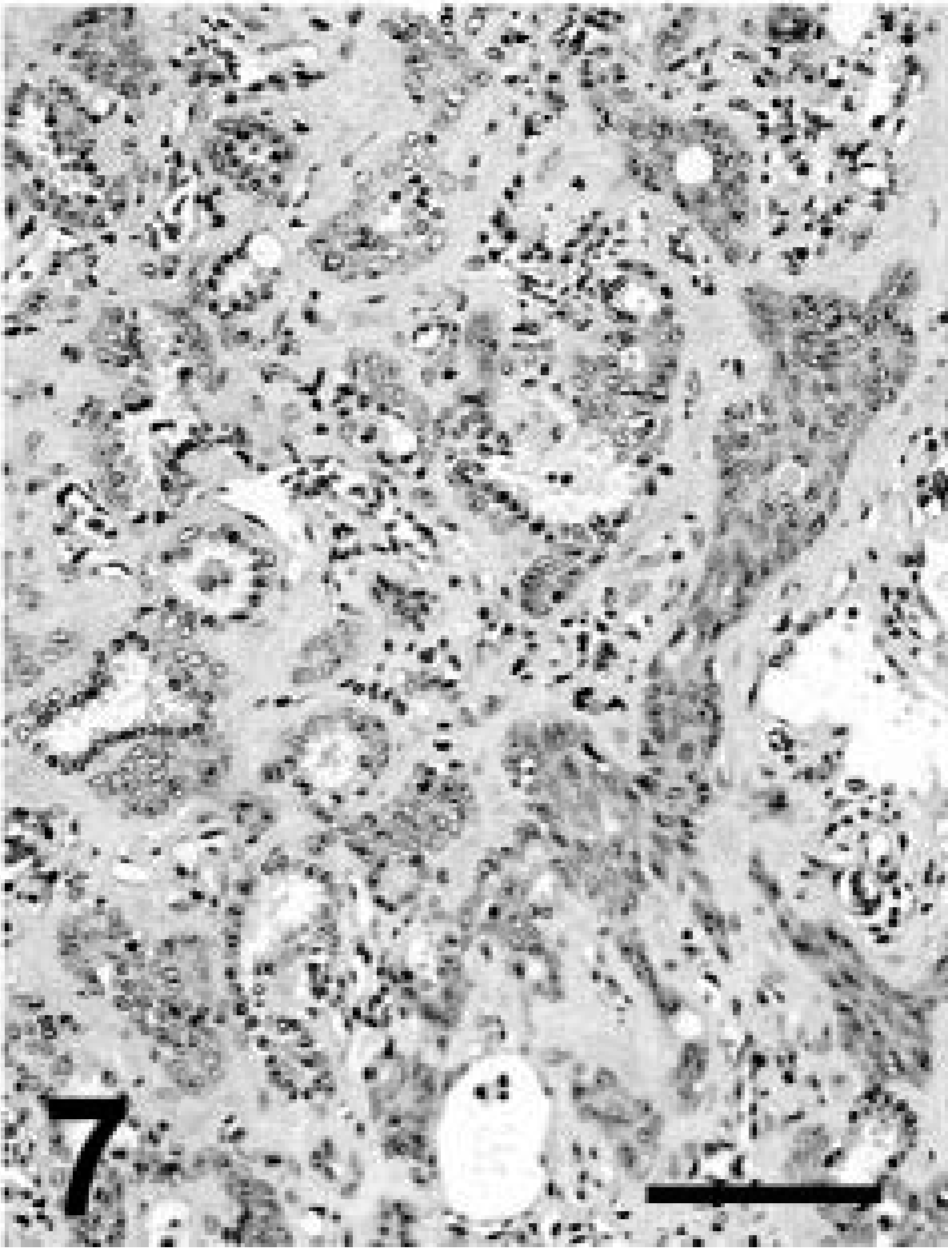
Llama No. 2. Pulmonary adenosquamous carcinoma. Streams of squamous cells are intermingled with acinar structures. HE. Bar = 150 µm.
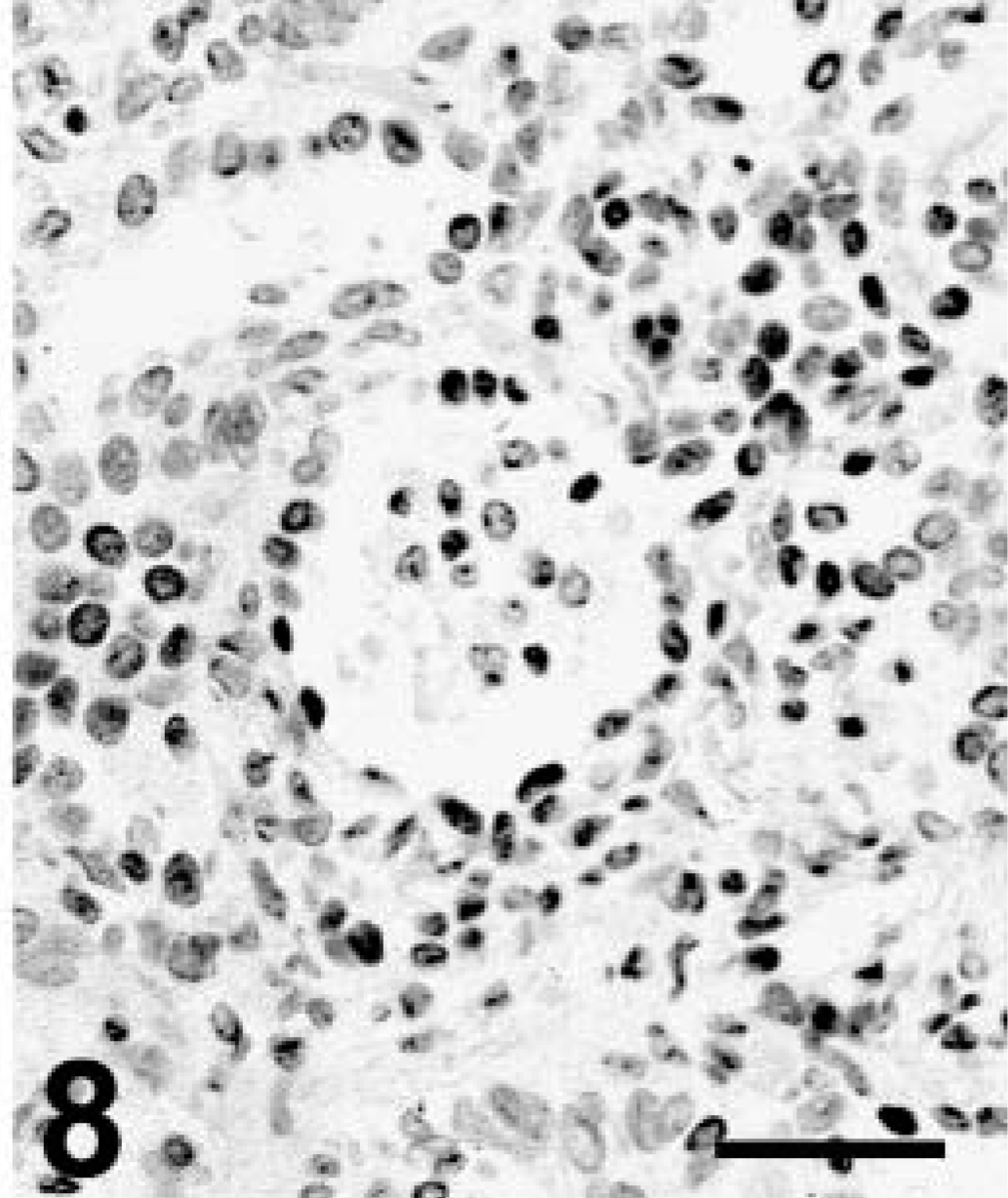
Lung; llama No. 2. Distinct nuclear staining (positive nuclei are black) in both the glandular and squamous epithelial components. EnVision+ peroxidase stain for TTF-1 with Mayer's hematoxylin counterstain. Bar = 100 µm.
This report describes clinical, morphologic, and immunohistochemical findings of pulmonary neoplasia in two llamas. According to the literature reviewed, pulmonary neoplasia in South American camelids is rare and only one case has been reported; 18 however, at the Veterinary Medical Diagnostic Laboratory, the lung is one of the most common sites of neoplasms in South American camelids. A search of our database revealed 13 of 185 (7%) South American camelids accessions with neoplasia. Lymphosarcoma (4 cases or 31%) was the most common neoplasia. Three cases (23%) were pulmonary carcinomas, two of these (including one in this report) had widespread metastases. In addition to pulmonary carcinoma, other tumors have been reported in South American camelids including lymphosarcoma, gastric squamous cell carcinoma, astrocytoma, hemangiosarcoma, medulloepithelioma, mammary carcinoma, anaplastic sarcoma, ossifying fibroma, transitional cell carcinoma, uterine carcinoma, and cutaneous squamous cell carcinoma. 1,2,6–8,10,12–16,21–23,26
Although the possibility of renal or hepatic carcinoma metastatic to the lung could not be completely ruled out in llama No. 1, the number of neoplastic masses in the lung and adjacent tissues suggests primary pulmonary neoplasia. Human lung cancer can metastasize to nearly every organ system. 24 In a previous report of pulmonary adenocarcinoma in a llama, distant metastases to the liver and rear limb were observed. 18 Immunohistochemistry is a valuable aid in the characterization of neoplastic diseases of numerous species. Diagnostic immunohistochemistry of neoplasia in South American camelids has rarely been reported; therefore, it is not known whether immunostaining patterns in this species correlate with those observed in other species such as dogs and cats. The pulmonary tumor reported recently in the literature 18 and in llama No. 1 were phenotypically and immunophenotypically different. In the current case, the immunophenotype of both the pulmonary and renal masses was consistent with carcinoma with squamous differentiation. The renal mass had also a distinct squamous phenotype that was less apparent in the pulmonary mass. TTF-1 is a marker present in most thyroid tumors, particularly follicular tumors, in the dog. 20 TTF-1 is also present in approximately 60–75% of human pulmonary adenocarcinomas and much less frequently in other pulmonary cancers. 9 We have observed a similar percentage of reactivity in canine pulmonary neoplasia (Ramos-Vara et al., manuscript in preparation). Llama No. 1 had reactivity for TTF-1 in normal pulmonary parenchyma, but tumor cells from various locations were negative for this marker. This negative result is not consistent with lack of cross-reactivity among species or processing of samples and may be the result of low reactivity of TTF-1 in pulmonary tumors without glandular differentiation (e.g., squamous cell carcinoma). Calretinin is a specific marker for mesothelioma in humans. 9 Immunoreactivity for vimentin in the pulmonary mass of llama No. 1 was unexpected. However, some carcinomas from different organs (lung, kidney, liver, genital tract) express both CKs and vimentin. 3 In particular, human pulmonary carcinomas, especially moderately or poorly differentiated tumors, can express vimentin. 25,27 The use of calretinin in domestic species or in camelids for the characterization of neoplasia has not been reported. Therefore, the significance of a negative result in our case is undetermined. However, typical characteristics of mesothelioma cells (e.g., long microvilli, prominent intercellular junctions, glycogen) 4 were not observed ultrastructurally.
The second llama did not have gross or microscopic extrapulmonary metastasis. The diagnosis was adenosquamous carcinoma; however, the tumor had some features of pulmonary blastoma (e.g., immature acini and stroma) 5 but glandular structures were lined by low columnar to cuboidal epithelium instead of tall columnar and a prominent spindle cell component was not apparent. Pulmonary blastomas did not typically show the distinct “cuffing” of squamous cells present around many acinar structures in our case. Histologic features also were not consistent with those reported in pulmonary blastomas of cattle. 11 This tumor had patchy but distinct staining for TTF-1, confirming that this antigen can be present in some pulmonary tumors. Attempts to determine the cross-reactivity of this antigen with other cell types of South American camelids were not made.
