Abstract
A unique form of pulmonary malignancy develops in cockatiels. This report describes the gross, histologic, electron microscopic, and immunohistochemical features of this tumor in 6 cockatiels. DNA in-situ hybridization for polyomavirus in the neoplasm was also performed. The tumor was comprised predominantly of compact sheets of anaplastic round to polygonal cells. All tumors had a high mitotic index, and had occasional large clear to slightly basophilic intranuclear inclusions that caused peripheral dispersal or complete masking of chromatin. Tumors were invasive but convincing metastases were not observed. Transmission electron microscopy identified intracytoplasmic intermediate filaments, desmosomes between cells, and intranuclear cytoplasmic invaginations corresponding to the intranuclear inclusions in light microscopic sections. Neoplastic cells stained positive for vimentin, lysozyme, and in 1 bird, pan cytokeratin. All 6 pulmonary neoplasms were negative for avian polyomavirus using the FN-19/FN-40 cocktail and the long VP-1 probe. We propose that these tumors may be poorly differentiated carcinomas of pulmonary or thymic origin.
Keywords
Cockatiels (Nymphicus hollandicus) are psittaciform birds indigenous to Australia,6 and are popular in the pet trade. Neoplasia is common in this species, especially tumors of the skin and associated soft tissues, including squamous cell carcinoma, fibrosarcoma, and myelolipoma.9, 17, 30 Some visceral tumors also are commonly seen in cockatiels, including oviduct and ovarian adenocarcinoma, seminoma, and pancreatic adenocarcinoma.9, 13, 30
Metastasis of various malignancies to the lung is common in birds, but spontaneously occurring primary pulmonary neoplasia is apparently uncommon to rare in birds.1, 11, 13, 21, 25, 32, 34 Only a few species trends have been reported and these include a possible trend in air sac adenocarcinomas in cockatoos,8, 24 adenocarcinomas of the lung in ducks,24 and fibromas and fibrosarcomas of the lung in chickens.27 Cockatiels also have a unique form of incompletely characterized pulmonary tumor,5, 9, 23, 26, 30 referred to as pulmonary bimorphic tumor,9 undifferentiated pulmonary tumor,23, 26, 30 or pulmonary sarcoma.5 This report describes the gross, histologic, electron microscopic, immunohistochemical, and DNA in-situ hybridization features of this tumor in 6 cockatiels.
Materials and Methods
The animals
A retrospective search of the case files at Norwest ZooPath identified 6 cases of unique pulmonary neoplasia. The 6 cockatiels in this study were pet birds that died or were euthanized due to respiratory disease, and were then necropsied by clinicians. Following necropsy, select tissues from each bird were submitted in 10% neutral-buffered formalin solution for histologic examination. Criteria for inclusion in the study included the presence of a pulmonary neoplasm with salient morphologic features in histologic section.
Histopathology
All tissues obtained by biopsy or at necropsy were preserved in 10% neutral-buffered formalin solution for up to 5 days before being processed routinely. Paraffin-embedded tissues were sectioned at 5 μm, mounted on frosted glass slides, and stained with HE. The neoplasm from bird No. 5 also was stained with Congo red.
Immunohistochemistry
Immunohistochemical staining was performed using an automated immunostainer (DAKO Autostainer Plus, Ft. Collins, CO) and markers for epithelial cells (pan cytokeratin and cytokeratin AE1/AE3) and mesenchymal cells (vimentin). Additional markers were used to detect thyroid epithelial cells (thyroglobulin), T-lymphocytes and B-lymphocytes (CD-3 and CD-79, respectively), and macrophages (lysozyme, Mac 387). Replicate paraffin-embedded tissue sections were cut at 4 μm and placed on charged glass slides (ProbeOn). The sections were dewaxed in xylene and endogenous peroxidase reactivity was quenched with 3% hydrogen peroxide application. Antigen retrieval was performed by steaming with citrate buffer (Target Retrieval S1700; DAKO, Carpenteria, CA). Nonspecific staining was minimized by the addition of a blocking reagent (Power Block, San Ramon, CA). The primary antibodies were added at the following dilutions: pan cytokeratin (1:100), cytokeratin AE1/AE3 (1:200), vimentin (1:200), CD-3 (1:400), CD-79 (1:200), lysozyme (1:600), Mac 387 (1:500), and thyroglobulin (1:50) (pan cytokeratin, cytokeratin AE1/AE3, and vimentin primary antibodies; BioGenex, San Ramon, CA; CD-3, CD-79, lysozyme, Mac-387, and thyroglobulin primary antibodies; DAKO, Carpenteria, CA). Following appropriate incubations ranging from 10 to 60 minutes, the sections were rinsed in buffer. Sites of primary antibody binding were identified by high affinity immunocytochemistry using a secondary antibody and strept-avidin-horseradish peroxidase. The chromagen was diaminobenzidine (DAB). The tissue sections were counterstained in Gill's hematoxylin, dehydrated, coverslipped, and examined microscopically. Sites of primary antibody localization were identified by the deposition of brown chromagen against a blue background. Appropriate mammalian and avian control tissues were used to validate the staining procedures. The mammalian tissues included sections of skin for pan cytokeratin and cytokeratin AE1/AE3 staining, intestine for vimentin staining, lymph node for T-lymphocytes and B-lymphocytes and macrophages (CD-3, CD-79, lysozyme, and Mac-387), and thyroid gland for thyroglobulin staining. Avian tissue sets were similar except spleen and bursa of Fabricius were used for lymphocyte and macrophage staining because birds lack lymph nodes.
Electron microscopy
Formalin-fixed tissue from the pulmonary neoplasm of bird No. 4 was subsequently retrimmed and placed into modified (50% strength) Karnovsky's fixative.14 After immersion in Karnovsky's fixative, the tissues were postfixed in 2% osmium tetroxide reduced with 2.5% potassium ferrocyanide.29 After osmification, the tissue was rinsed in 0.2 M sodium cacodylate, dehydrated through a graded ethanol series, transitioned through propylene oxide, and infiltrated and embedded in Eponate-12 epoxy formulation (Eponate-12; Ted Pella Inc., Redding, CA). Thick sections were cut, mounted on glass slides, stained with toluidine blue O, and examined by light microscopy. Thin sections were mounted on bare 150-mesh copper grids, stained in 4% uranyl acetate in 75% ethanol, followed by poststaining in Reynold's lead citrate.28 The grids were examined in a Zeiss 906E transmission electron microscope at 60 kv accelerating voltage (Carl Zeiss SMT, Peabody, MA).
DNA in-situ hybridization
DNA in-situ hybridization was performed using an oligonucleotide probe cocktail (FN-19/FN-40) to detect classic avian polyomavirus and polymerase chain reaction (PCR)-generated long probe (VP-1) to detect any variant avian polyomaviruses.7, 18 The probes were 3′-end labeled with digoxigenin Replicate paraffin-embedded tissue sections were cut at 4 μm and placed on charged glass slides (ProbeOn Plus slides, Hemo De, Microprobe Staining Station, and Automation Buffer; Fisher Scientific Company, Pittsburgh, PA). The tissue sections were paired, placed face-to-face in a slide holder, and processed using a MicroProbe Workstation to minimize reagent use. The sections were dewaxed in limonene and rehydrated through graded alcohols to Auto Buffer. The tissue sections were digested for 10 minutes at 37°C in 0.25% pepsin solution to promote probe penetration. Enzymatic activity subsequently was terminated by heating to 105°C for 8 minutes. After washes in Auto Buffer, the slides were prehybridized in formamide solution for 5 minutes at 105°C. After blotting, the hybridization solutions were applied at 105°C for 5 minutes and the temperature was then reduced to 37°C for 60 minutes. After hybridization, the tissue sections were blotted and subjected to low and high stringency washes using 0.5× standard sodium citrate (SSC) and 0.2× SSC, respectively. Sites of probe binding were localized by high affinity immunocytochemistry. The slides were incubated in Buffer 1 with 1% sheep serum to block nonspecific staining. The indicator system consisted of an antidigoxigenin antibody conjugated to alkaline phosphatase and a nitroblue tetrazolium dye solution (Antidigoxigenin labeling kit, Roche Molecular Biochemicals, Indianapolis, IN). After incubation and washing, the slides were counterstained briefly in fast green FCF dye solution, rinsed, dehydrated, coverslipped, and examined. Microscopically, sites of probe localization were visualized by the deposition of formazan, a dark blue, insoluble pigment. Avian positive and negative control tissues were used to validate the hybridization procedures.
Results
The animals
From 1994 to 2007, 667 cockatiels were submitted to Northwest ZooPath and the relative prevalence of this pulmonary tumor in this species was 0.9%. The characteristics, history, gross lesions, and additional histologic lesions for each bird are summarized in Table 1. Cockatiel No. 1 was a 10-year-old female that had a 6-month history of respiratory distress. Treatment included placement of air sac tubes and nebulization. The bird died and necropsy revealed a yellow nodular mass in the right lung field that measured 2.0 cm in greatest dimension (Fig. 1). Cockatiel No. 2 was a 6-year-old female with a history of respiratory difficulty. The bird died and necropsy revealed a nodular mass in the syringeal region, 1.6 cm in greatest dimension. Cockatiel No. 3 was a 10-year-old male with a history of respiratory difficulty and radiographic evidence of a mass in the cranial region of the coelomic cavity. Euthanasia was performed and necropsy revealed a white to tan mass 3 cm in greatest dimension in the cranial coelomic cavity. Cockatiel No. 4 was an adult male of unknown age that presented with open-mouth breathing and harsh lung sounds of several weeks' duration. The bird had good body condition but had radiographic evidence of opacity in the left lung. Necropsy revealed a fatty appearing mass in the left lung lobe. Cockatiel No. 5 was an approximately 4-year-old, male cockatiel that had a history of bilateral paresis and thin body condition. Radiographs revealed a mass effect in the caudal dorsal thoracic region. A yellow cavitated-appearing lesion was visualized endoscopically in the right lung field and biopsies were obtained. Necropsy revealed a cream-colored mass 1.5 cm in greatest dimension ventral to the lungs on the right side with extension to the left side of the lung. Cockatiel No. 6 was an adult of undetermined age and sex. This bird had a 1-month history of respiratory difficulty. Necropsy revealed a white–tan mass 2 cm in greatest dimension surrounding the trachea near the base of the heart.
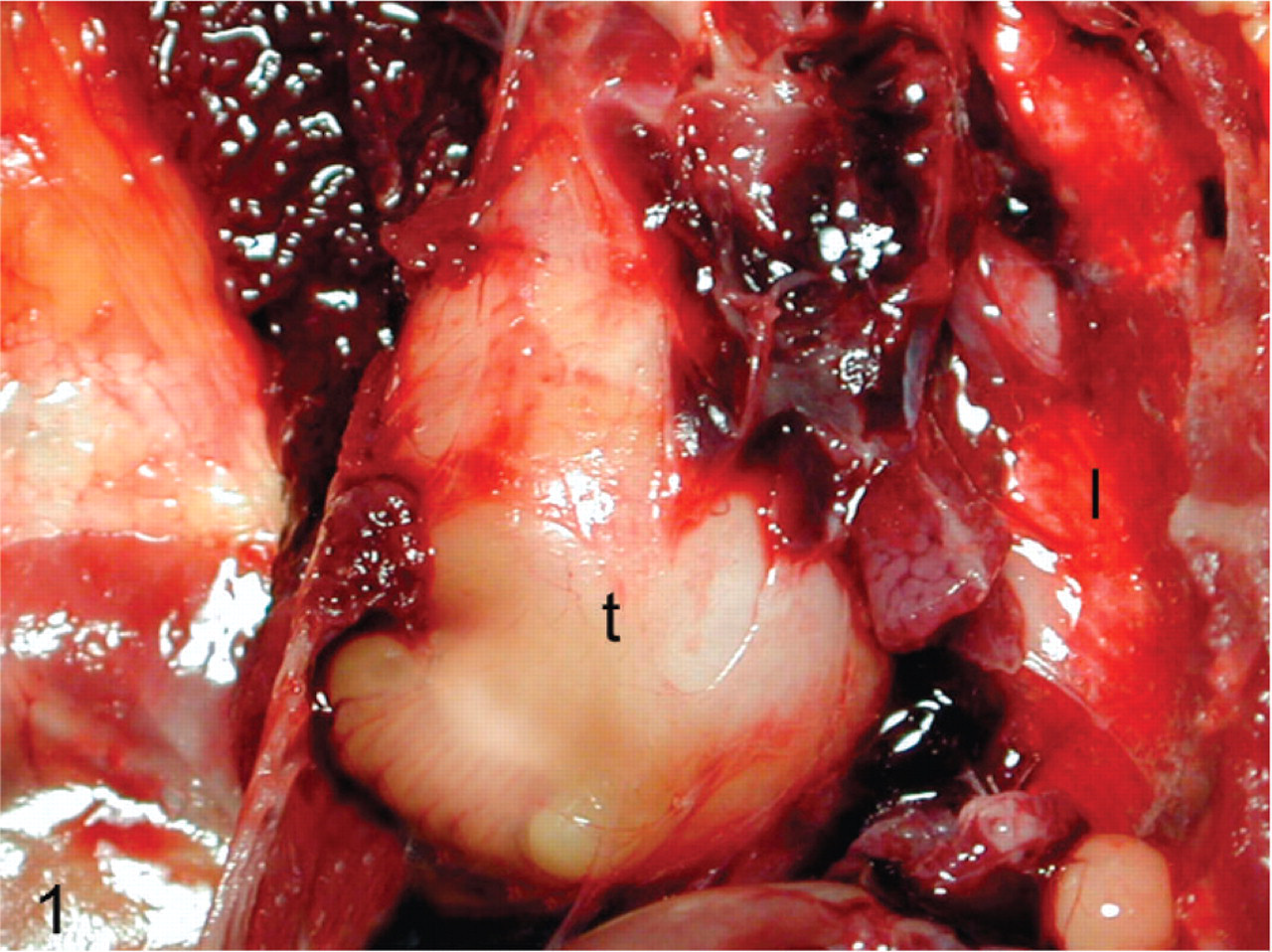
Cockatiel No. 1. Pulmonary tumor (t), effacing the entire right lung. Note unaffected left lung (l). Photo courtesy Dr. Robert Monaco.
Characteristics, history, gross lesions, and concurrent disease processes in cockatiels with pulmonary neoplasia.

Histopathology
All specimens had similar histologic features (Figs. 2–6). The tumor was comprised predominantly of compact sheets of anaplastic round to polygonal cells, sometimes interspersed with a few fusiform cells, clusters of lymphocytes, scattered granulocytes or necrotic cell debris. The neoplastic cells had scant eosinophilic to occasionally vacuolated cytoplasm and moderately anisokaryotic, vesicular nuclei with 1 or 2 prominent nucleoli. All tumors had a high mitotic index, with up to 10 mitotic figures per ×45 field. All tumors had occasional to frequent large clear to basophilic intranuclear inclusions that caused peripheral dispersal or complete masking of chromatin. The tumor in bird No. 5 had a small amount of homogenous, extracellular eosinophilic, congophilic material interpreted as amyloid interspersed with the neoplastic cells. In all cockatiels, the tumor effaced pulmonary parenchyma, compressed or obliterated bronchi and parabronchi, and extended into thoracocoelomic air sacs. Cockatiel No. 1 also had neoplastic cells on the endocardial surface of the left ventricle that extended a short distance along a fascial plane into the myocardium, incorporating a Purkinje fiber. The tumor in cockatiel No. 5 extended into the vertebral bodies, vertebral canal, and intervertebral joint with associated disarticulation of the fused joint. This neoplasm was associated with displacement, compression and neuraxonal degeneration of the spinal cord. The tumor in cockatiel No. 6 also had rare papillary or ductlike formations of neoplastic cells.
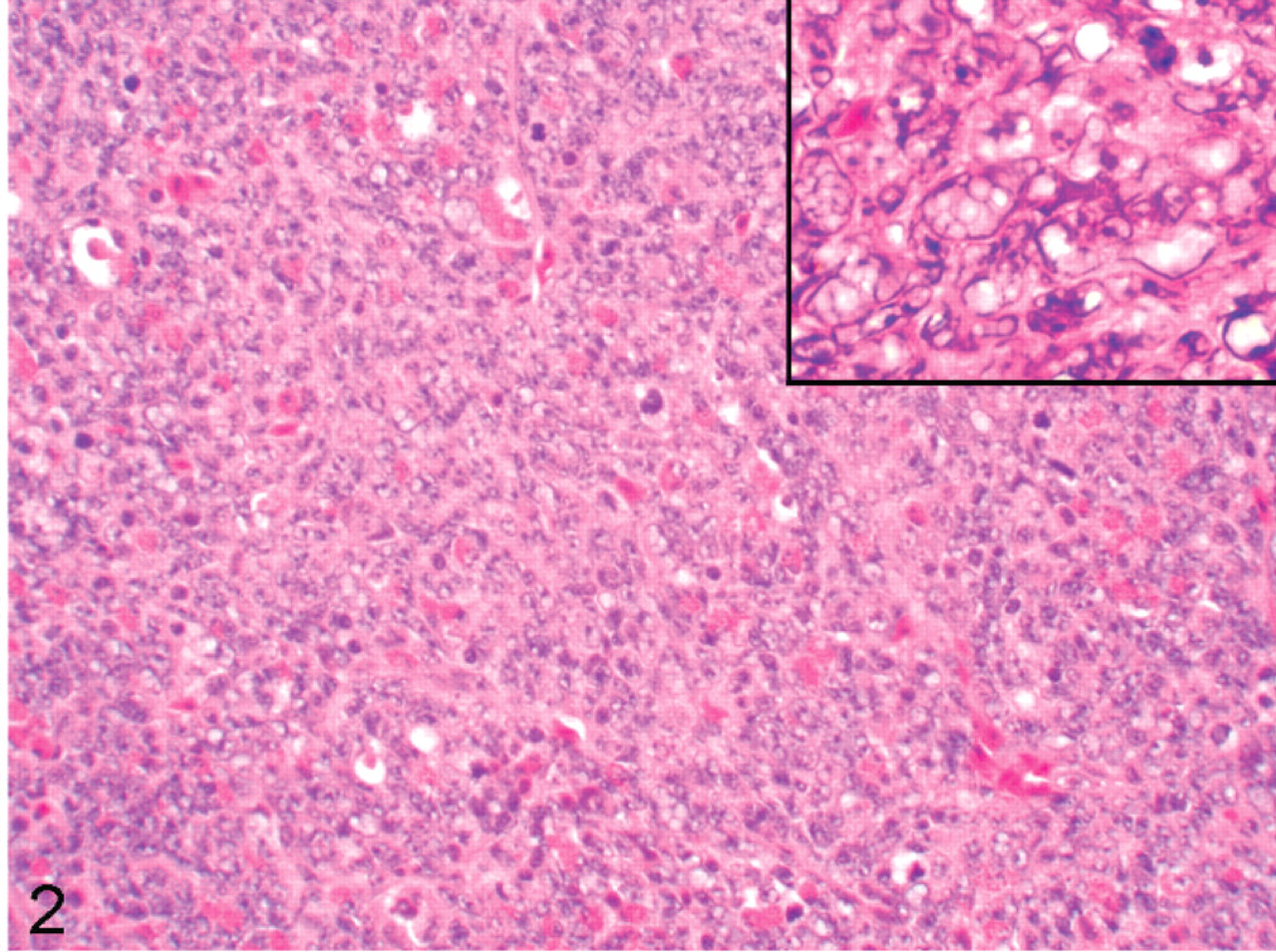
Cockatiel No. 2. The pulmonary tumor is comprised of sheets of round cells with high mitotic index. Inset: Cluster of neoplastic cells with large intranuclear clear inclusions that efface normal nuclear internal morphology. HE.
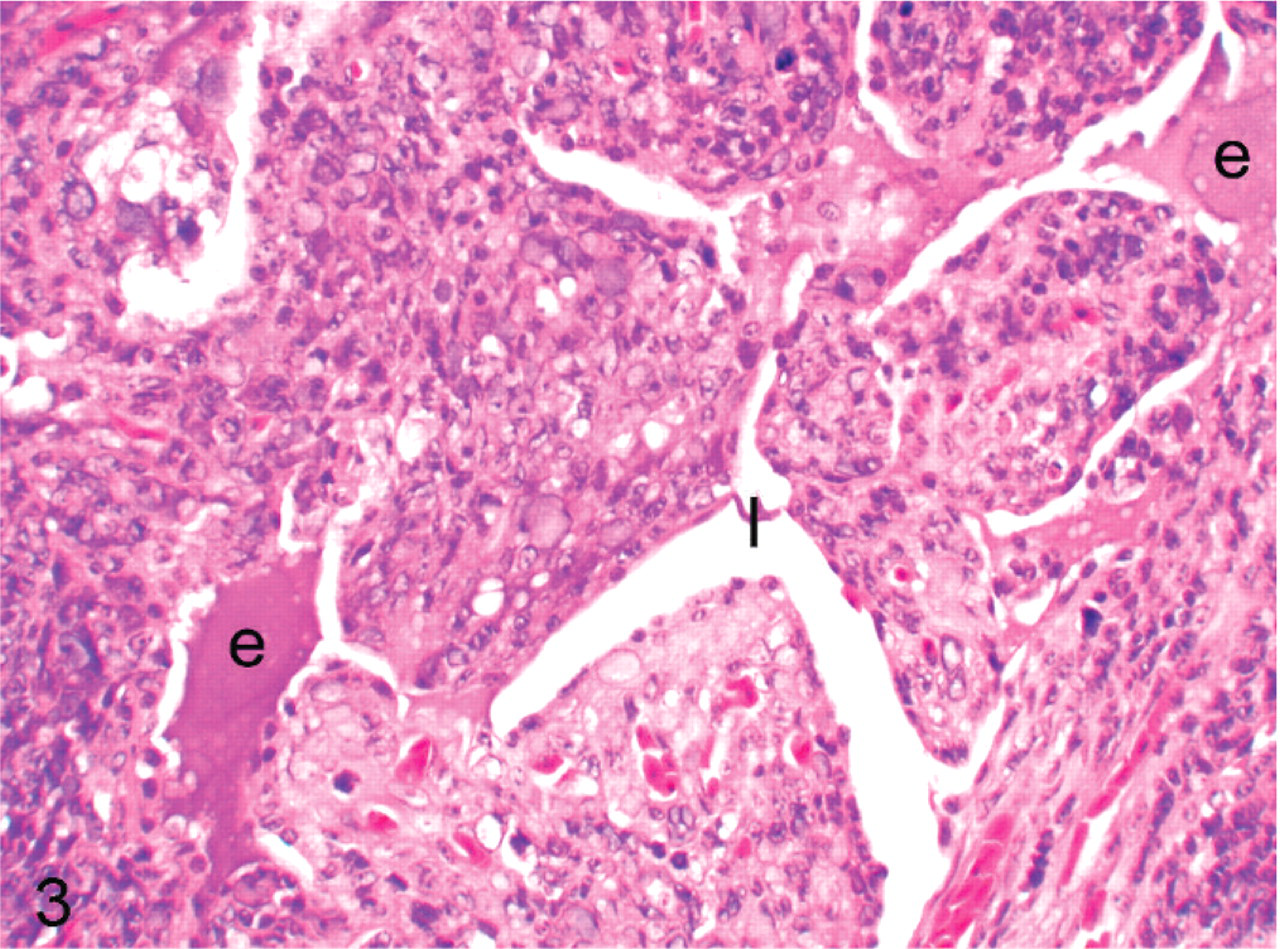
Cockatiel No. 4. The tumor is impinging on and distorting the parabronchial lumen (l) and some edema (e) is in the lumen. HE.
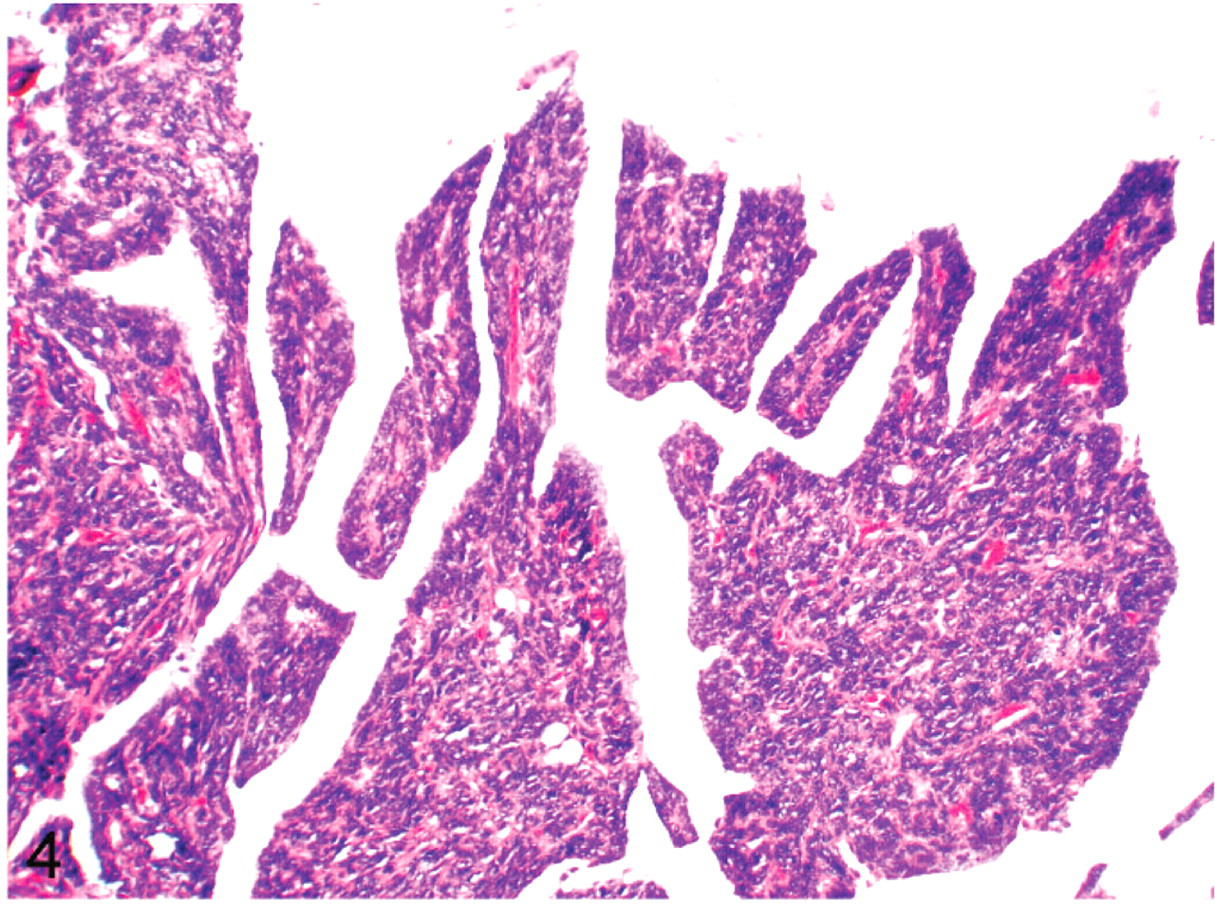
Cockatiel No. 4. Tumor is infiltrating the interstitium of the thoracocoelomic air sac, and forming papillary deformities in the air sac. HE.
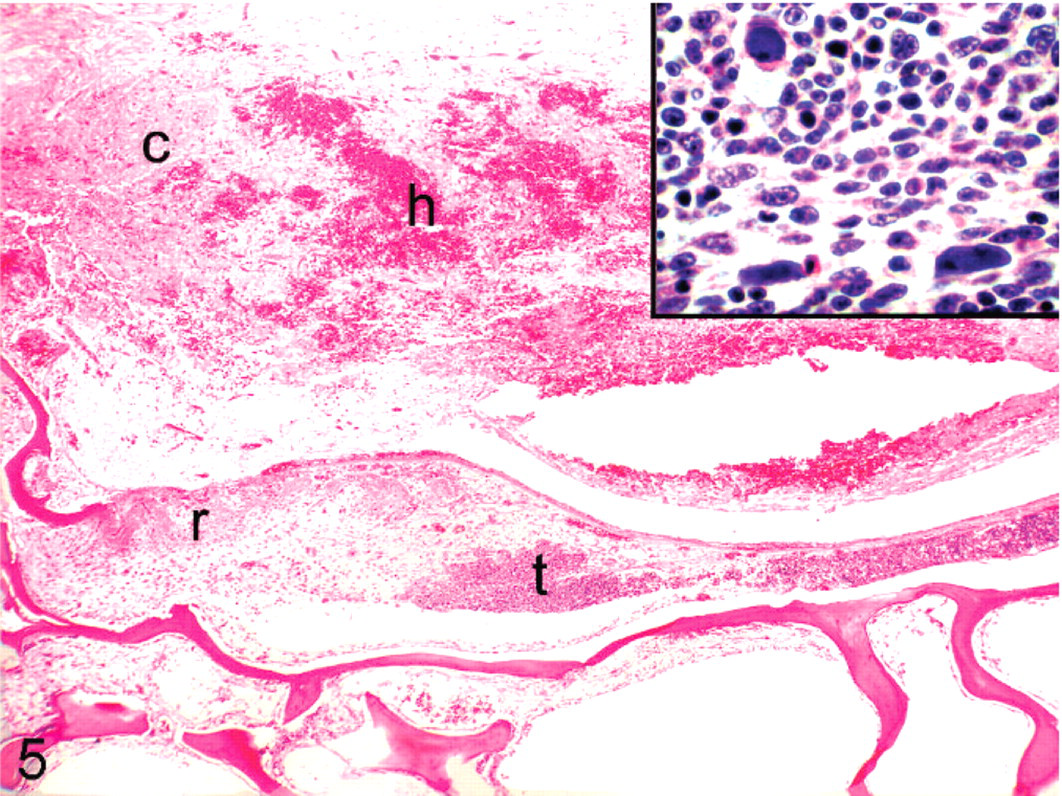
Cockatiel No. 5. Tumor (t) is infiltrating cortical vertebral bone, with associated reactive bone formation (r) and hemorrhage (h) in the spinal cord (c). Inset: Large basophilic intranuclear inclusions in the neoplastic cells. HE.
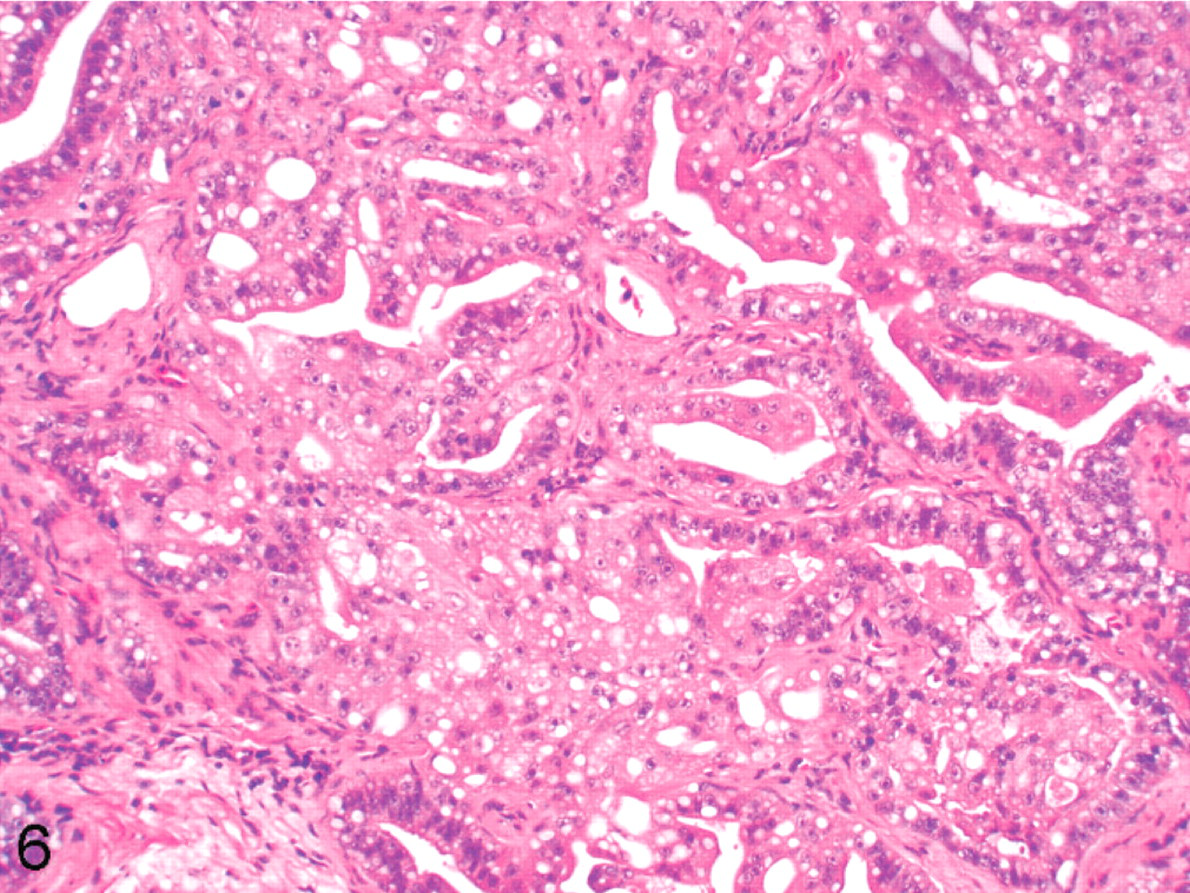
Cockatiel No. 6. Tumor is forming papillary and alveolar structures within the lung. HE.
Immunohistochemistry
Results of immunohistochemistry are summarized in Table 2. Five of six neoplasms were negative for pan cytokeratin, but the tumor from cockatiel No. 6 had trace positive reactivity. The normal epithelium lining the terminal airways adjacent to the neoplasm had positive reactivity for pan cytokeratin in all birds. All neoplasms also had trace to moderate immunoreactivity for vimentin. All neoplastic cells were devoid of reactivity for CD-3 and CD-79, although all tumors had scattered small round cells interpreted as non-neoplastic lymphocytes that had positive reactivity for CD-3. Neoplasms from cockatiels No. 1 and No. 3 lacked reactivity for lysozyme. However, neoplasms from cockatiels No. 2 and No. 5 had trace reactivity and the neoplasm from No. 6 had moderate reactivity for lysozyme. All neoplasms were devoid of thyroglobulin reactivity. All of the mammalian and avian control tissues stained appropriately for the primary antibodies (Figs. 7–10.
Immunohistochemical staining profiles of neoplastic cells in cockatiels with pulmonary tumors.

Scattered CD3-positive lymphocytes throughout the neoplasm.
− = negative; −/+ = trace; + = mild; ++ = moderate.
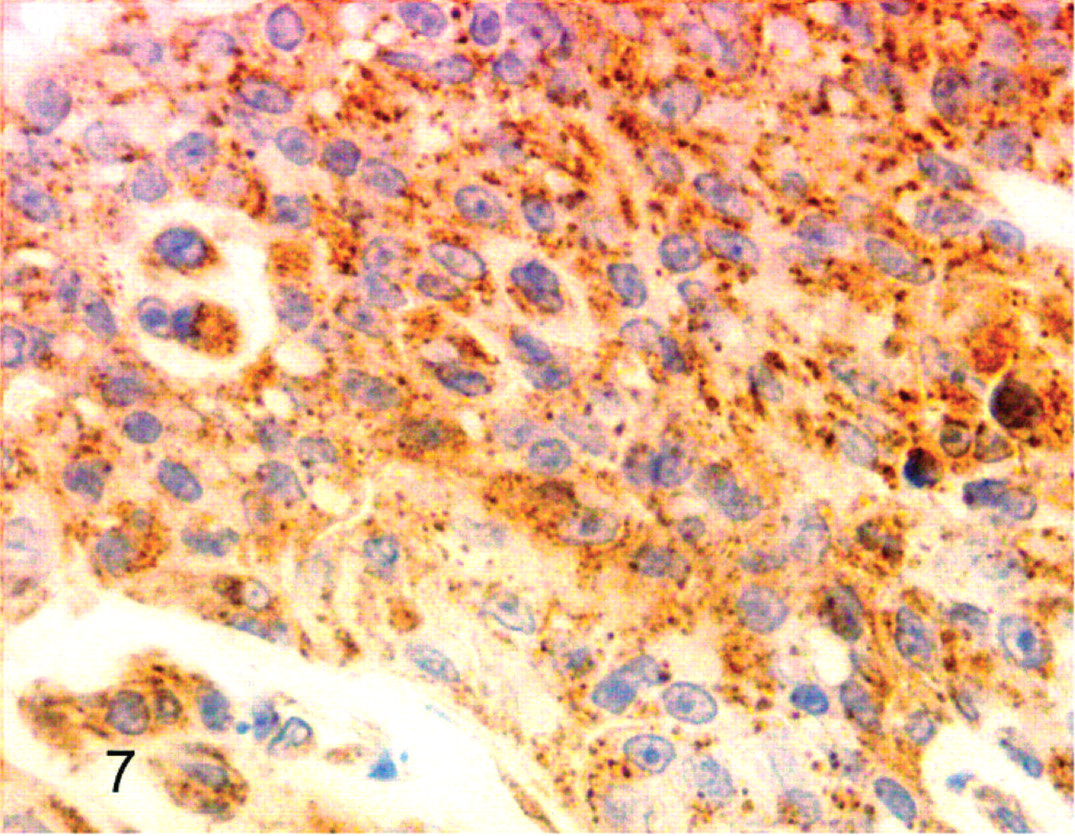
Cockatiel No. 6. Neoplastic cells have reactivity for lysozyme. Streptavidin-biotin, hematoxylin counterstain.
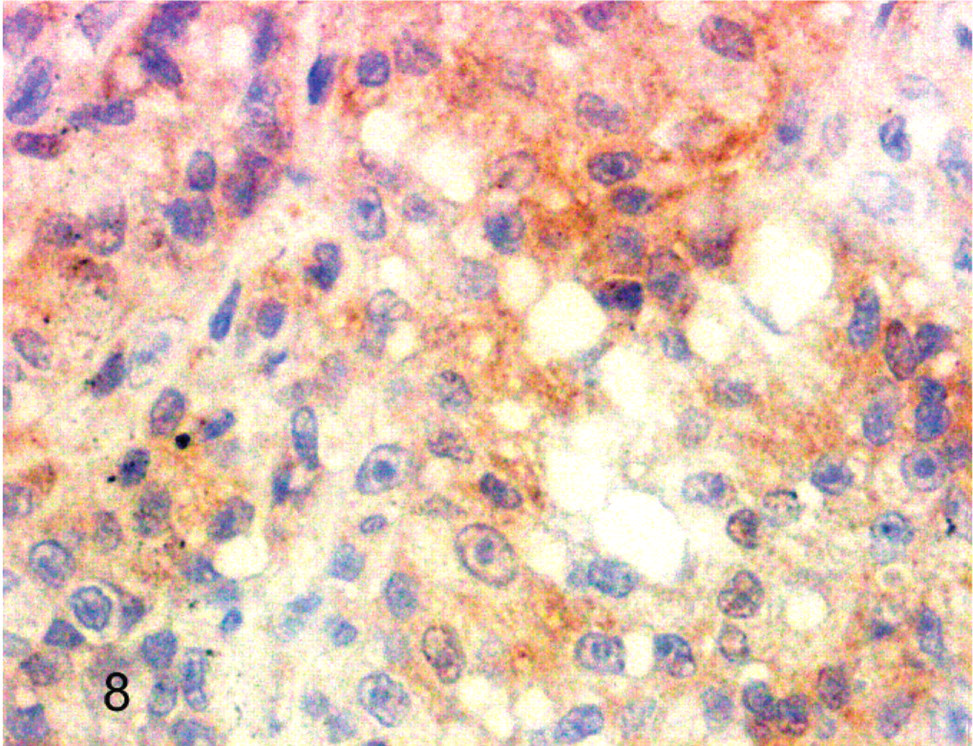
Cockatiel No. 6. Neoplastic cells have reactivity for vimentin. Streptavidin-biotin, hematoxylin counterstain.
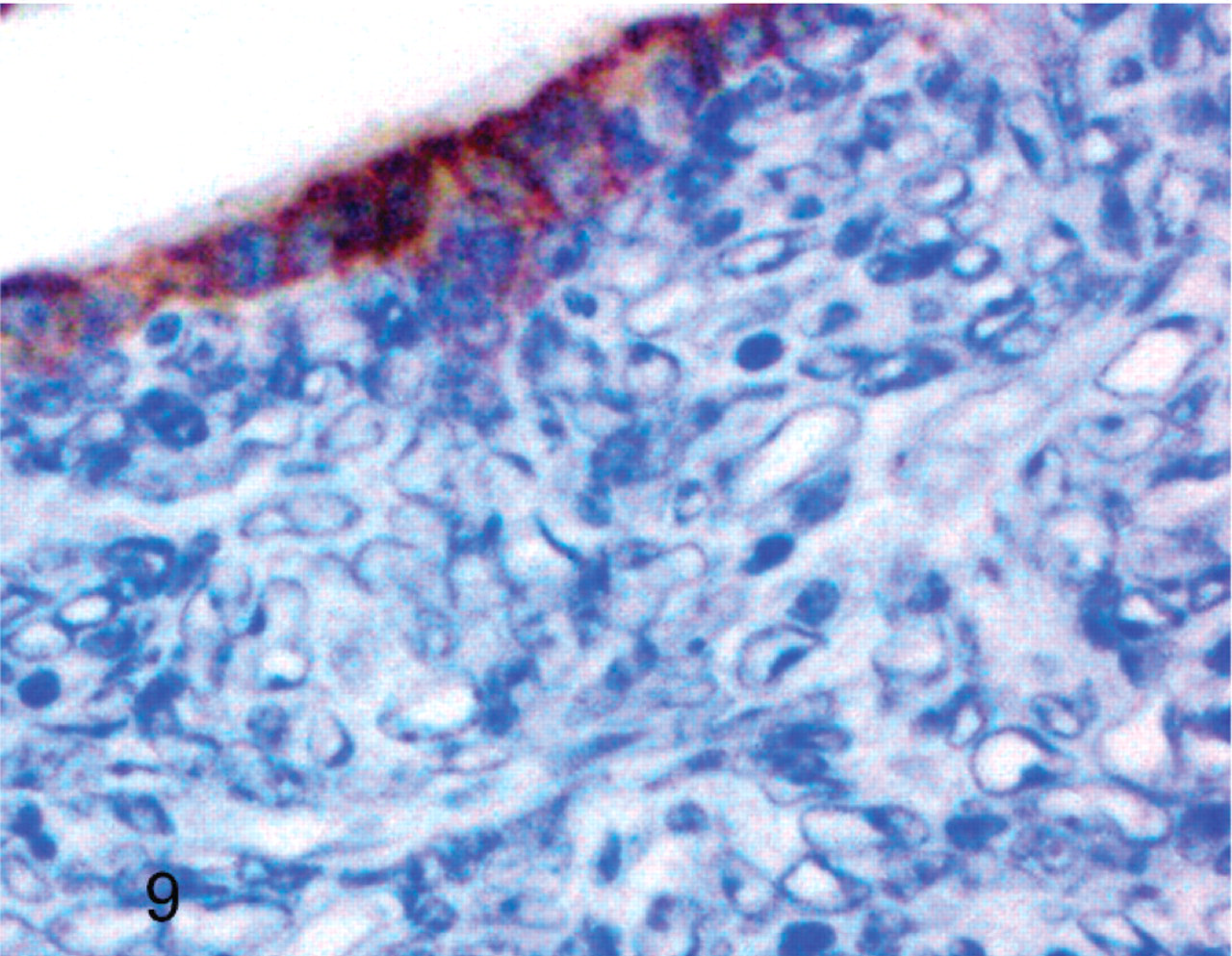
Cockatiel No. 2. Bronchial epithelial cells have reactivity for pan cytokeratin, but neoplastic cells do not. Streptavidin-biotin, hematoxylin counterstain.
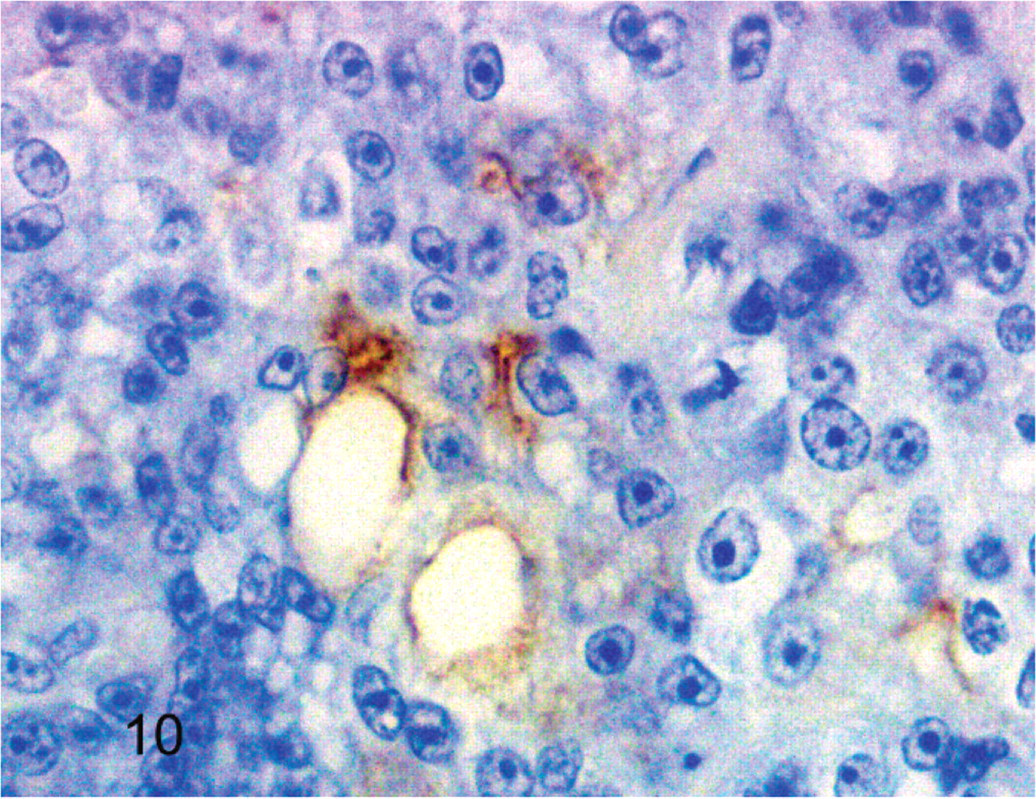
Cockatiel No. 6. Few neoplastic cells have reactivity for pan cytokeratin. Streptavidin-biotin, hematoxylin counterstain.
Electron microscopy
Electron microscopy revealed no evidence of viral particles in the inclusions from cockatiel No. 4. The inclusions were comprised of loosely aggregated granular material and the inclusions sometimes appeared to be membrane bound. Rarely, neoplastic cells had intracytoplasmic filaments approximately 30 nm in diameter that resembled intermediate filaments. Desmosomes occasionally were noted between adjacent tumor cells. Basement membranes were not identified on the external surfaces of cells or between adjacent cells (Figs. 11, 12).
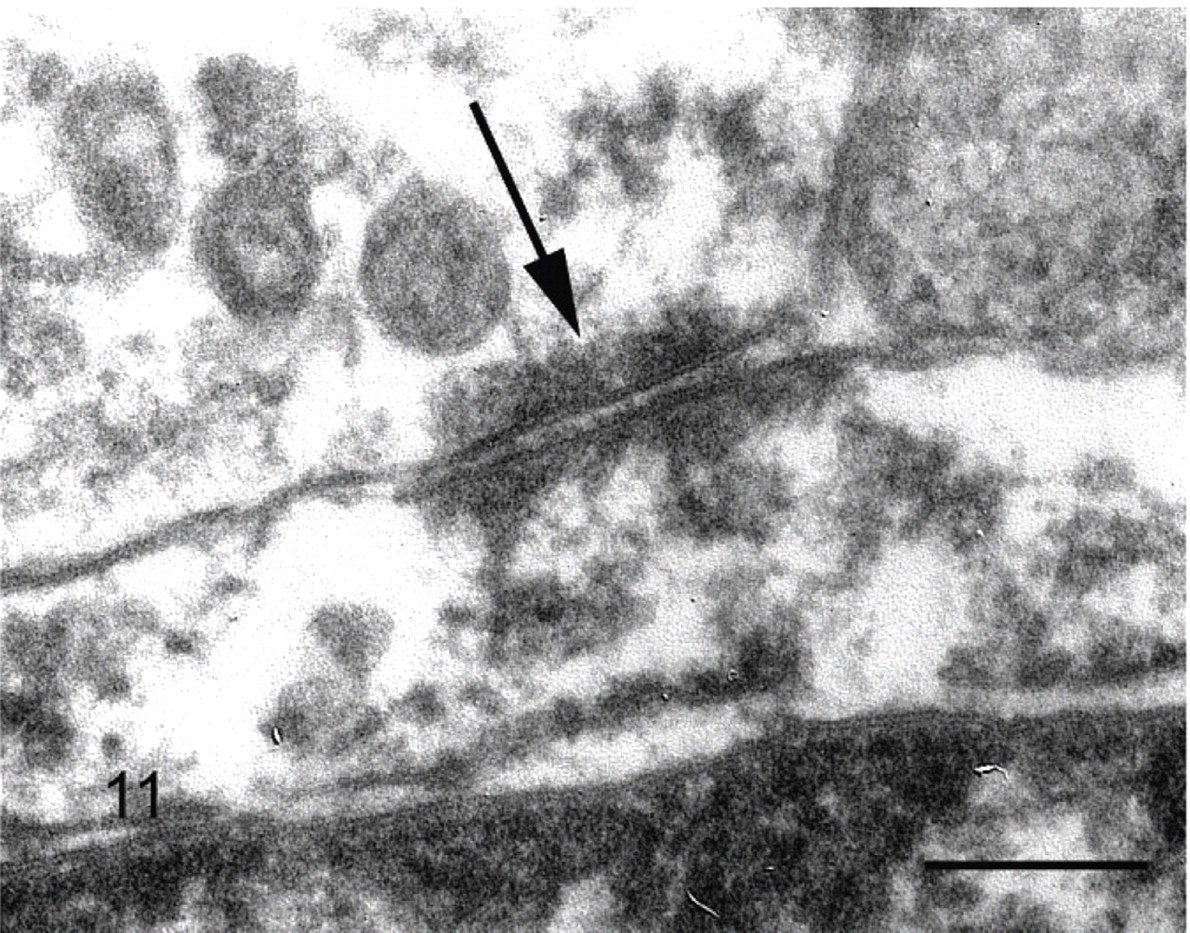
Cockatiel No. 4. Intercellular junction (arrow) resembling a desmosome between two neoplastic cells. Uranyl acetate and lead citrate. Bar = 100 nm.
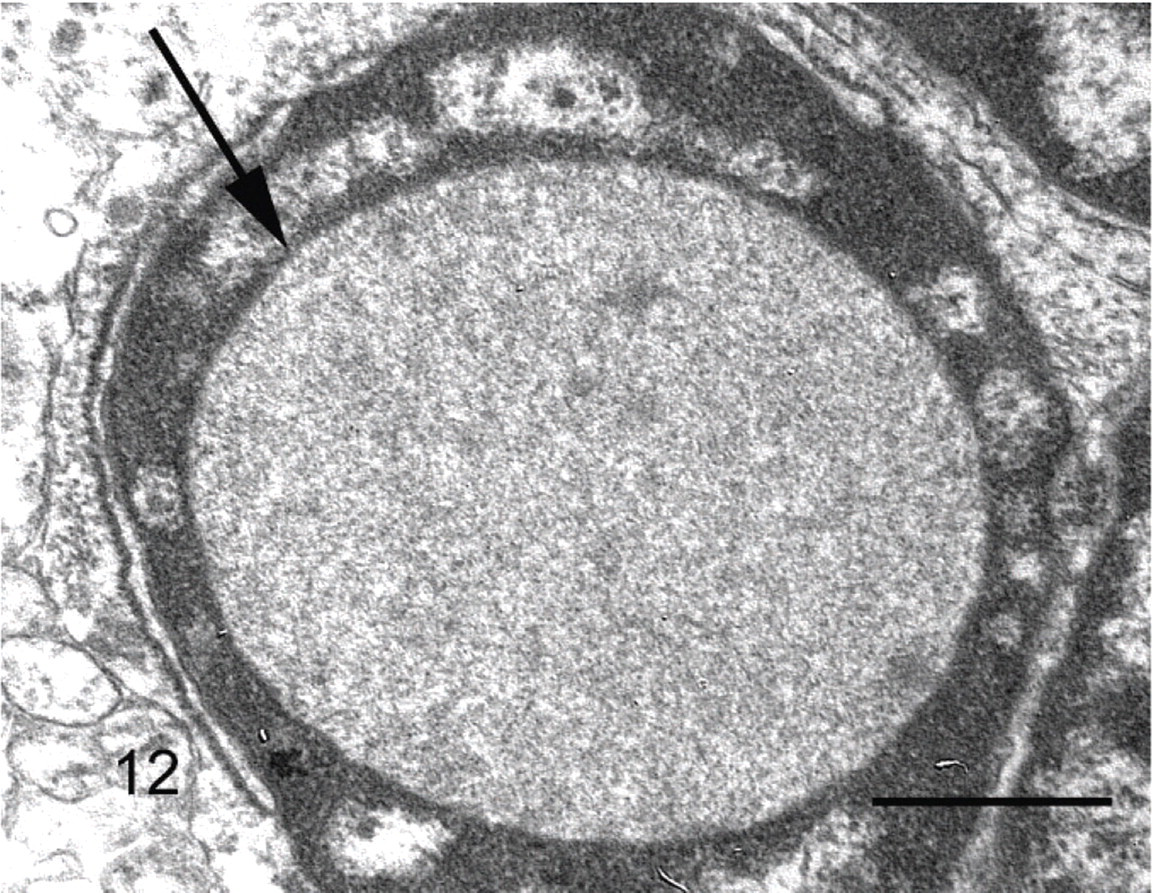
Cockatiel No. 4. Membrane-bound intranuclear inclusion (arrow) containing mildly radiodense granular material, resembling a cytoplasmic invagination. Uranyl acetate and lead citrate. Bar = 4 μm.
DNA in-situ hybridization
All six pulmonary neoplasms were negative for avian polyomavirus using the FN-19/FN-40 cocktail and the long VP-1 probe.
Discussion
Primary pulmonary neoplasia is uncommonly reported in birds, and spontaneously occurring primary pulmonary neoplasia does not appear to be common in any avian species.8, 9, 11, 17, 24, 25, 27, 30 In a retrospective survey of avian tumors submitted to Northwest ZooPath, a few individual cases of bronchogenic carcinoma were noted across species lines, and a possible trend was noted for air sac adenocarcinoma in cockatoos as previously reported,24 but no other species trends in tumor types were discerned with the exception of the unique pulmonary tumors in cockatiels, which are described in this report.9 Furthermore, the primary pulmonary tumors described in the literature do not have histologic features resembling the tumors of these cockatiels. For these reasons, we believe that this pulmonary neoplasm may be overrepresented in cockatiels and possibly is an entity unique to this species.
There did not appear to be any sex predisposition from the limited number of birds in our study, and all of the birds were adults, with the similar clinical sign of respiratory difficulty and the gross finding of a solitary mass in the lung. All tumors were clearly malignant. In each bird, the tumors were locally invasive within the lung and extended into the air sacs. As previously reported,23, 30 the neoplasm in cockatiel No. 5 also invaded bone and infiltrated the adjacent spinal column, compressing and partially destroying the spinal cord resulting in paresis. The neoplasm from cockatiel No. 1 also was present on the endocardial surface of the left ventricle. Presumably, this was a metastatic lesion, although other metastases were not observed in this bird or in any of the other cockatiels. Multicentric distribution was considered possible as has been described,30 although this phenomenon was not seen in the other cockatiels of this report. Direct extension of neoplastic cells from the lung through the cardiac wall was unlikely because neoplastic cells were not observed in other areas of the heart and the bird probably would have died before the neoplastic cells reached the endocardial surface by this route.
These cockatiel tumors have previously been referred to as bimorphic,9 undifferentiated,23, 26, 30 or sarcomas,5 based primarily on their histologic appearance. Tumors in the literature have been described as bimorphic based on histologic or immunohistochemical features. This term has been used for tumors that show different patterns of the same cell type such as solid formations of epithelium that are juxtaposed with acinar structures in adenocarcinoma of the aprocrine sac.19 Bimorphic tumors have also been described in the thymus, where the neoplastic population is comprised of more than one cell type.15, 31 The term also has been used for tumors that appear to have loss of differentiation based on histologic or immunohistochemical features, especially regarding the mesenchymal or neurogenic tumors.3, 4, 31 The term bimorphic is perhaps best used as a descriptive term rather than a term for cell or tissue of origin.
Although the pulmonary neoplasms of this report were considered anaplastic and poorly differentiated malignancies, their histologic appearance was distinctive and their immunohistochemical profile was fairly consistent. All of the tumors were densely cellular. With the exception of rare papillary or ductlike formations in the neoplasm from cockatiel No. 6, these tumors lacked histologic features that resembled normal avian respiratory tract or other normal structures. The presence of these glandlike formations in cockatiel No. 6, and the transmission electron microscopic evidence of cell to cell attachments morphologically consistent with desmosomes indicate that some of the cells in these neoplasms have epithelial differentiation.10 Aside from some trace reactivity in the neoplasm from cockatiel No. 6, the remaining tumors were negative for pan cytokeratin reactivity. In contrast, the bronchial epithelium of adjacent lung tissue did have pan cytokeratin reactivity. Although considered a specific marker for epithelial cells, not all epithelial neoplasms express pan cytokeratin reactivity, especially as cells become more anaplastic or lose features of differentiation.16 Therefore, a lack of pan cytokeratin expression by the neoplastic cells of these neoplasms does not exclude epithelial origin or confound the transmission electron microscopic findings of structures associated with epithelial differentiation. It is considered possible that the cockatiel lung tumors represent a poorly differentiated carcinoma arising from epithelial structures of the lung, rather than a true “bimorphic” tumor with two distinct morphologic patterns of differentiation.
Thymus, thyroid gland, and parathyroid gland were considered as possible origins of these neoplasms because of the close anatomic proximity of these structures to the lung in birds. The presence of CD-3–positive lymphocytes within the tumors suggests the possibility of a residual population of thymic T cells or recruitment of inflammatory T cells or natural killer cells,12 rather than a neoplastic population of T cells from the thymus. Neoplastic cells with light or transmission electron microscopic features of thyroid or parathyroid morphology, such as cytoplasmic secretory granules or organelles active in protein synthesis, were not observed in these neoplasms.2 In addition, none of the pulmonary neoplasms had positive reactivity for thyroglobulin, although avian thyroid control tissue did react strongly with this antibody. Our findings suggest that these tumors are not of thyroid or parathyroid origin, although epithelial cell origin from thymus could not be completely excluded as possible.
All neoplasms had reactivity for vimentin, an intermediate filament typically expressed in differentiated mesenchymal cells and in sarcomas; however, as neoplastic epithelial cells dedifferentiate, they also may express vimentin reactivity.16 In addition, this transition in intermediate filament status of neoplastic cells has been associated with the acquisition of aggressive biologic behavior,16 a feature of the cockatiel tumors. Lysozyme is expressed in a variety of mammalian cell types, and is a particularly useful marker of histiocyte differentiation in mammalian tumors. Lysozyme reactivity is seen in avian histiocytes, but also can be found in avian epithelial cells.20, 22 The findings of desmosomes, papillary or ductlike formation in one of the tumors, and lysozyme reactivity suggest that that these neoplastic cells are more likely of epithelial than mesenchymal origin.
An interesting histologic feature of these tumors is the presence of large intranuclear clear to amphophilic inclusions in some of the neoplastic cells that resemble avian polyomavirus inclusions by light microscopy. This finding suggested that these tumors may be caused by an oncogenic virus. Some polyomaviruses in mammalian species have potential to cause neoplastic transformation of infected cells, but this has not been documented in birds probably because avian polyomavirus large T (or transforming) antigen has a considerably different molecular sequence from that of other polyomavirus large T antigens.33 The presence of intranuclear inclusions and an early observation of polyomavirus-like particles in the inclusions of these cockatiel tumors30 suggested the possibility of viral oncogenesis. However, we were unable to document polyomavirus infection using transmission electron microscopy for cockatiel No. 4, or with DNA in-situ hybridization using the FN-19/40 cocktail or the long VP-1 probes7, 18 for any of the neoplasms. Electron microscopic examination of these inclusions identified dispersed granular material within membrane-bound structures consistent with cytoplasmic invaginations. There was no evidence of nucleocapsid assembly, and we were unable to confirm the presence of a viral component to these tumors.
In summary, a form of primary invasive and anaplastic lung tumor occurs in cockatiels that possibly originates from epithelial cells of the lungs or thymus. Although these neoplasms are highly invasive, their metastatic potential is questionable. Additional studies to further characterize the tumor might include a broader application of immunohistochemistry to include more cell markers, attempts to culture virus from tumor cells, and use of molecular methods to screen for known avian oncogenic viruses in tumor tissue.
Footnotes
Acknowledgements
We thank the following clinicians for case submission: Dr. Robert Monaco, Old Country Animal Clinic, Plainview, New York; Dr. David Barno, Rock Creek Veterinary Hospital, Aloha, Oregon; Dr. Katie Phillips, Dublin Animal Hospital, Colorado Springs, Colorado. We also thank Jamie Kinion for data retrieval, Christie Buie for photo editing, illustration layout, and manuscript submission, and Llewely B. Sellers for assistance with the DNA in situ hybridization. This study was funded by Northwest ZooPath, Angell Animal Hospital, and the DNA in-situ Hybridization Laboratory, Department of Pathology, The University of Georgia.
