Abstract
A 6-year-old, female llama (Lama glama) presented with progressive anorexia and ascites. Postmortem examination revealed an infiltrative uterine adenocarcinoma with widespread metastases. The neoplasm completely replaced and infiltrated the myometrium of the uterine body and cervix and metastasized largely to the serosal surfaces of the peritoneal cavity. Histopathology identified a highly invasive growth of neoplastic cells in solid packets or tubular arrangements and marked fibroplasia. No bovine or llama papillomavirus DNA was detected intralesionally by polymerase chain reaction.
Keywords
In recent years, increasing numbers of neoplasms have been reported in llamas (Lama glama). Obviously, this is related to the increasing popularity of New World camelids as farm and companion animals. Recent reports found cutaneous and mucocutaneous fibroma and fibropapilloma, cutaneous and mucocutaneous squamous cell carcinoma, and disseminated lymphoma to be the most prevalent neoplasms in the llama and alpaca (Lama pacos). 1,9 To date, reports on uterine neoplasms in llamas are restricted to 1 leiomyosarcoma and 1 nonmetastasizing adenocarcinoma. 4,9 Gross and microscopic findings and the results of a bovine and llama papillomavirus (BPV and LPV, respectively)–specific polymerase chain reaction (PCR) of a scirrhous uterine adenocarcinoma with marked hepatic lipomatosis in a llama is presented here.
A 6-year-old, female llama presented with a history of progressive anorexia and emaciation. Because of the progression of disease, the animal was euthanatized and submitted for postmortem examination. At postmortem examination, the llama had severe ascites and a mild hydrothorax, with 6 liters of a serous fluid admixed with a few fibrin clots. The epithelial lining of the cervix and the uterine body were effaced and diffusely replaced by a white, pasty to granular material (Fig. 1A). The uterine wall was moderately thickened, firm, and diffusely white. The peritoneal, and to a minor extent, the pleural serosal surfaces had multifocal, sharply demarcated, slightly elevated, white, partly coalescing nodules (Fig. 1B) and a few multifocal, adherent, fibrinous plaques. Multifocal nodules of similar appearance but varying sizes were found within the lungs. The diaphragm had large (up to 0.5 cm in diameter) multifocal white nodules in a linear arrangement. Numerous multifocal, randomly distributed, sharply demarcated, white, soft nodules also were present in the liver and widely replaced the normal hepatic parenchyma. Moderate to severe intimal mineralization was present in the aortic arch and the descending aorta.
Representative tissue samples were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Four-micrometer sections were cut and stained with hematoxylin and eosin. Histopathology of the uterus revealed a complete replacement and infiltration of the entire uterine wall by a nondemarcated, nonencapsulated, highly cellular neoplasm. Neoplastic epithelial cells were arranged in nests and packets and occasionally in primitive crypt-like structures (Fig. 2A). These neoplastic cells were medium-sized and mildly pleomorphic. Individual cells had a round nucleus with scant, coarsely stippled heterochromatin and a moderate amount of pale, homogeneous, eosinophilic cytoplasm. Less than 1 mitotic figure was observed per 400× microscopic field of view. The neoplastic epithelial cells were surrounded by a dense fibrous stroma with numerous proliferating fibroblasts and dense deposits of collagen fibers. Aggregates of neoplastic cells were occasionally found in lymphatic vessels. Histopathology of the multifocal to coalescing nodules on the pleural and peritoneal surfaces and the nodules in the lung consisted of similar aggregates of neoplastic cells, a scirrhous reaction, and multifocal, firmly adherent fibrinous tags. In addition, the hepatic parenchyma was replaced by moderately well demarcated, nodular aggregates of well-differentiated adipocytes, which compressed and replaced the surrounding hepatocytes (Fig. 3B). Sudan staining of the hepatic lesions demonstrated abundant lipid within the adipocytes (Fig. 3C). Immunohistochemistry, with use of the avidin–biotin–peroxidase complex method with 3-amino-9-ethylcarbazole chromogen, was performed to confirm the epithelial origin of the neoplastic cells within the scirrhous tumor masses. Antibodies specific for cytokeratin a reacted positively with neoplastic cells in the primary tumor and the metastases, whereas vimentin stain b failed to label the neoplastic cells (Fig. 2B). Furthermore, PCR testing was performed on formalin-fixed, paraffin-embedded tissue sections of the uterine adenocarcinoma as previously described in an attempt to discover a viral etiology. 8 Paraffin-embedded tissue sections from BPV-positive equine sarcoids were used as positive controls. Llama papillomavirus E1 gene–specific primers LPV forward (5′-GCCTCCAAATAGAGGCAAGT-3′)and LPV reverse (5′-GTGTCTGATAGAGGGGCAAG-3′) were designed according to recently published sequences. 8 The presence of intact DNA in the llama specimens was confirmed with the use of primers for camelid cytochrome b. 8 After PCR amplification, amplicon sequencing, and a BLAST search (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi), LPV DNA was not detected in tissue samples of the uterine adenocarcinoma. In contrast, highly homologous BPV DNA was detected in the equine sarcoid.
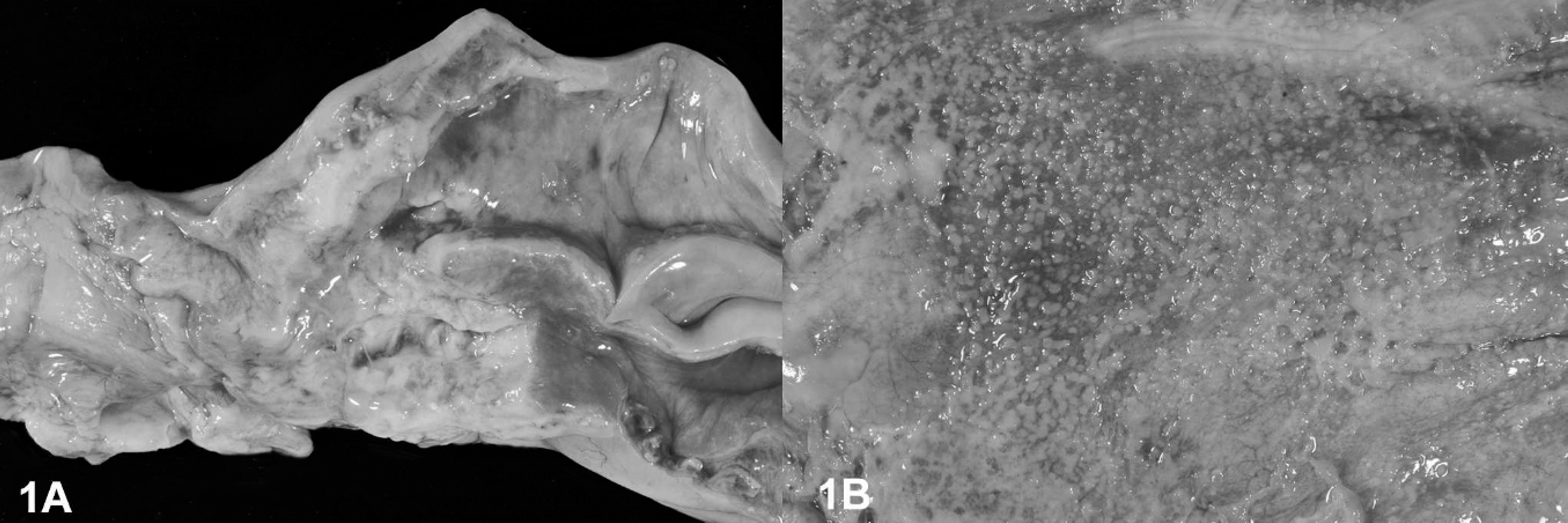
Uterus, diaphragm, llama.
On the basis of the gross, histopathological, and immunohistochemical findings, the neoplasm was diagnosed as a scirrhous uterine adenocarcinoma with widespread metastases by direct seeding and hematogenic and lymphatic spread. The nodular aggregates of mature adipocytes in the liver were diagnosed as hepatic lipomatosis with unknown relevance to the neoplastic lesion.
Uterine adenocarcinomas are common malignancies in cows and rabbits. Bovine uterine adenocarcinomas are usually found in the uterine horns, evoke a scirrhous response, and metastasize rapidly. Thus, the adenocarcinoma presented in this report displays properties similar to the bovine malignancy. In humans, cervical cancer can be caused by several strains of human papillomavirus, 10 but a similar association of uterine adenocarcinoma with papillomavirus infection has not been observed in animals. Nevertheless, BPVs have been detected in vasoformative urinary bladder neoplasms in cows. Recently, a unique LPV that is highly homologous to BPV has been identified as the causative agent of fibropapillomas in llamas. 8 In this case, PCR testing for llama- and bovine-specific papillomaviral DNA failed to detect virus-specific DNA in the uterine neoplasm.
Lipomatosis is defined as an abnormal deposition and replacement of the original tissue by otherwise apparently normal adipocytes. Here, the macroscopic findings and the cellular morphology indicated a replacement of hepatic tissue by well-differentiated adipocytes rather than a multifocal fatty change of hepatocytes. The etiology of similar human lipomatous lesions is commonly associated with obesity, replacement of lost tissue, or hereditary diseases. 2,3,7 The exact cause and pathogenesis of this lesion in this llama remains unclear. However, the hepatic lipomatosis observed seems to be unique in that lipomatosis has been described in several organs, mainly skin, pancreas, and heart, but not in the liver. 5,6,11 The association, if any, between the uterine adenocarcinoma and the hepatic lipomatosis remains to be established.
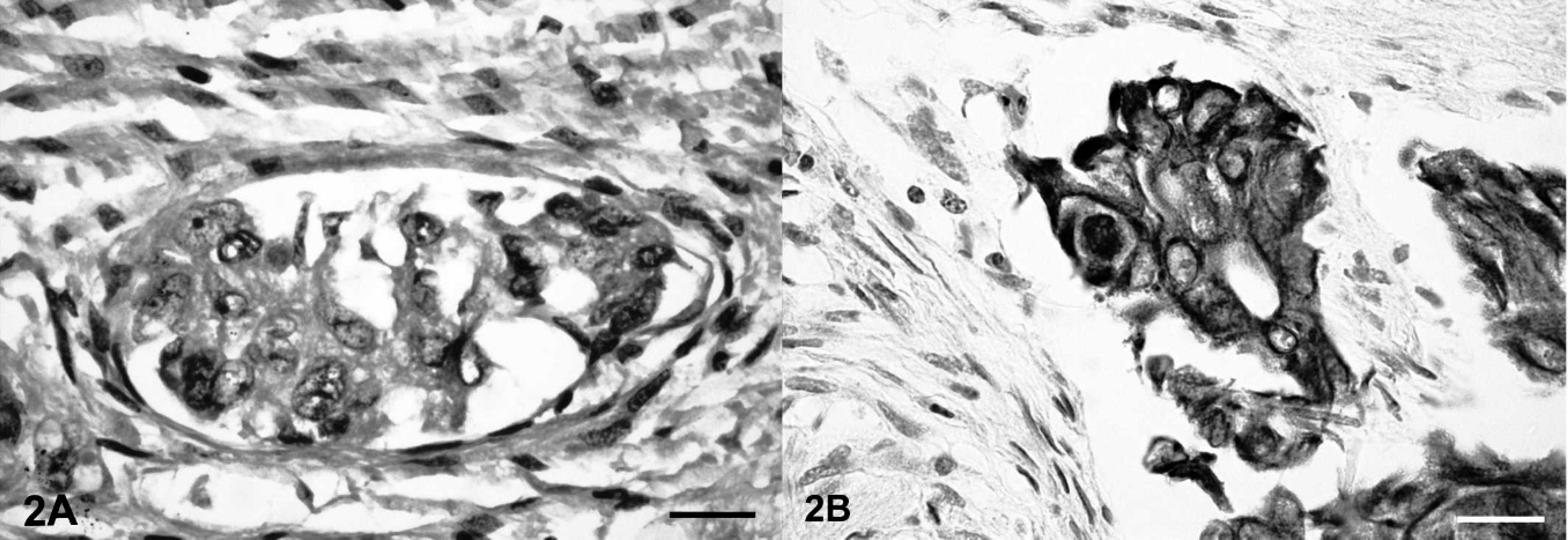
Uterus, llama.
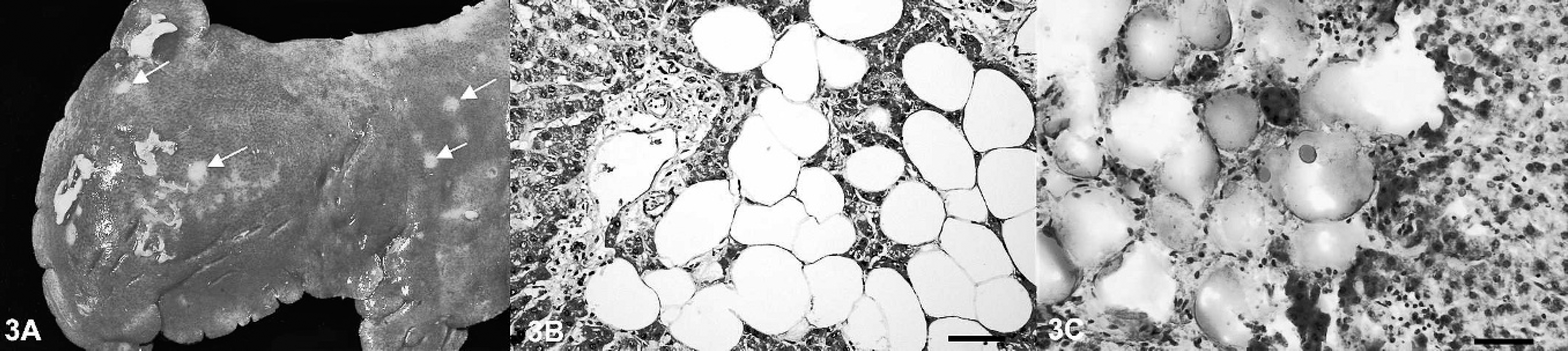
Liver, llama.
Acknowledgements. The authors thank Prof. K. Müller for her help and M. Schaerig for excellent technical assistance.
Footnotes
a.
Rabbit-anti-cytokeratin antibody, 1:150; Dako Deutschland GmbH, Hamburg, Germany.
b.
Goat-anti-rabbit IgG antiserum, 1:40; Dako Deutschland GmbH, Hamburg, Germany.
