Abstract
Neoplastic diseases associated with retroviruses were diagnosed in four Burmese pythons (
Neoplastic or proliferative diseases associated with retroviruses have been well documented in fish.
9
In contrast to feral and farm-raised fish, the majority of neoplastic diseases associated with retroviral particles in reptiles represent isolated cases. Retroviruses were first identified in poikilothermic animals during electron microscopic examination of a spleen cell line from a Russell's viper (
Here we describe retroviral particles in neoplastic tissues of four Burmese pythons (
Snake No. 1 was euthanatized after a second recurrence of an intermandibular undifferentiated malignant round cell tumor. Necropsy revealed tan nodular masses in the lower left distal mandible, soft palate, ovaries, and oviduct and diffuse replacement of the spleen by similar neoplastic tissue. The primary site could not be determined but was assumed to be the spleen. HE-stained sections revealed sheets and packets of round cells occasionally separated by thin fibrovascular septa. The cells were individualized, with moderate amounts of eosinophilic cytoplasm and round nuclei with a prominent nucleolus (Fig. 1); mitosis was common. The histologic origin of the neoplasm could not be ascertained, but the features were suggestive of lymphosarcoma.
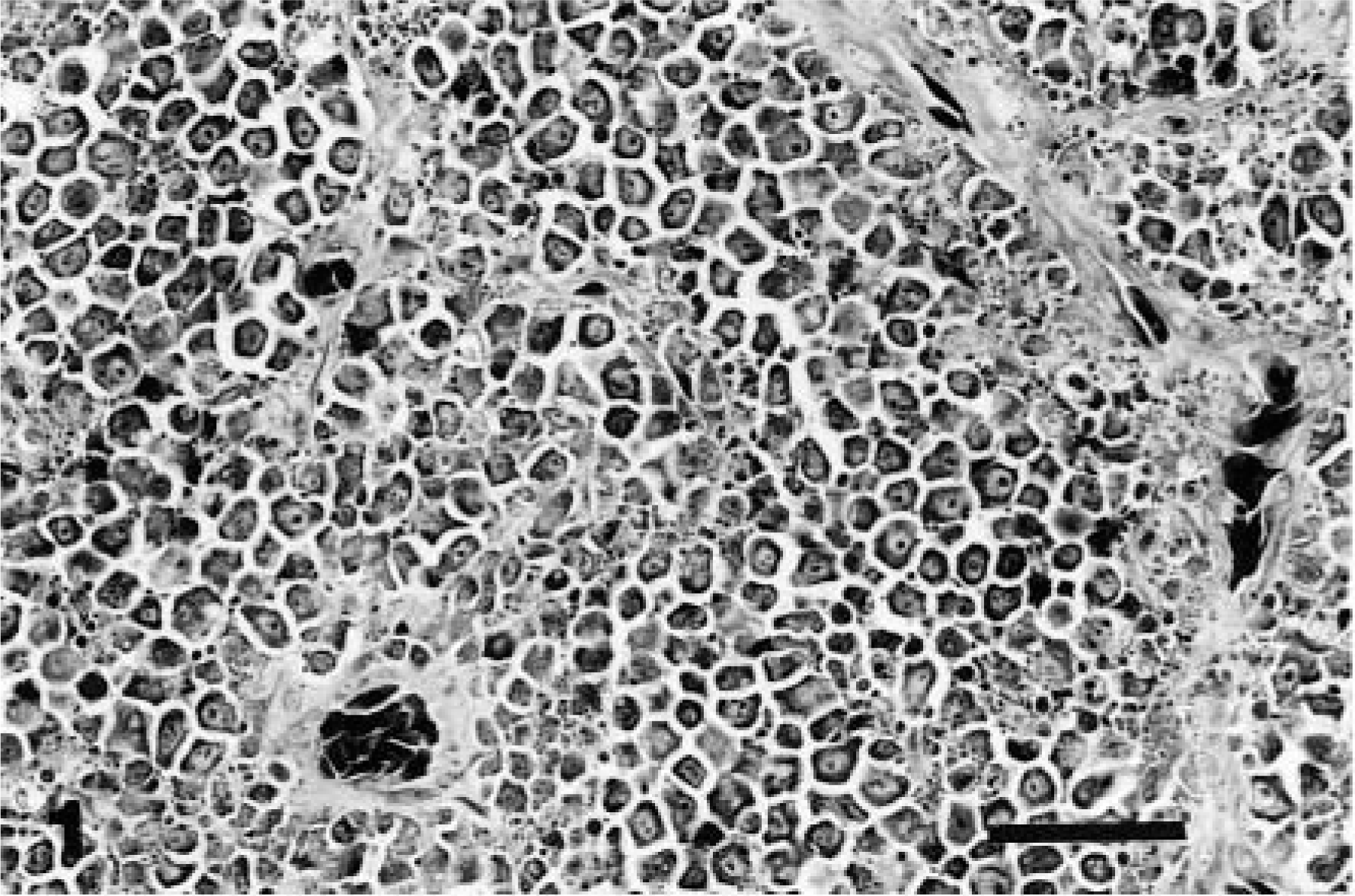
Spleen; snake No. 1. Undifferentiated round cell tumor. The neoplasm is composed of individualized round cells supported by thin fibrovascular septa. HE. Bar = 30 µm.
Snake No. 2 was euthanatized because of complications following resection of an intramural multinodular mass within the wall of the colon. HE-stained sections of the resected mass revealed neoplastic epithelial cells forming glandular acini and nests embedded within a myxomatous fibrous stroma. The interpretation was mucinous colonic adenocarcinoma (Fig. 2). Necropsy revealed a fulminant fibrino-suppurative coelomitis with no evidence of metastasis.
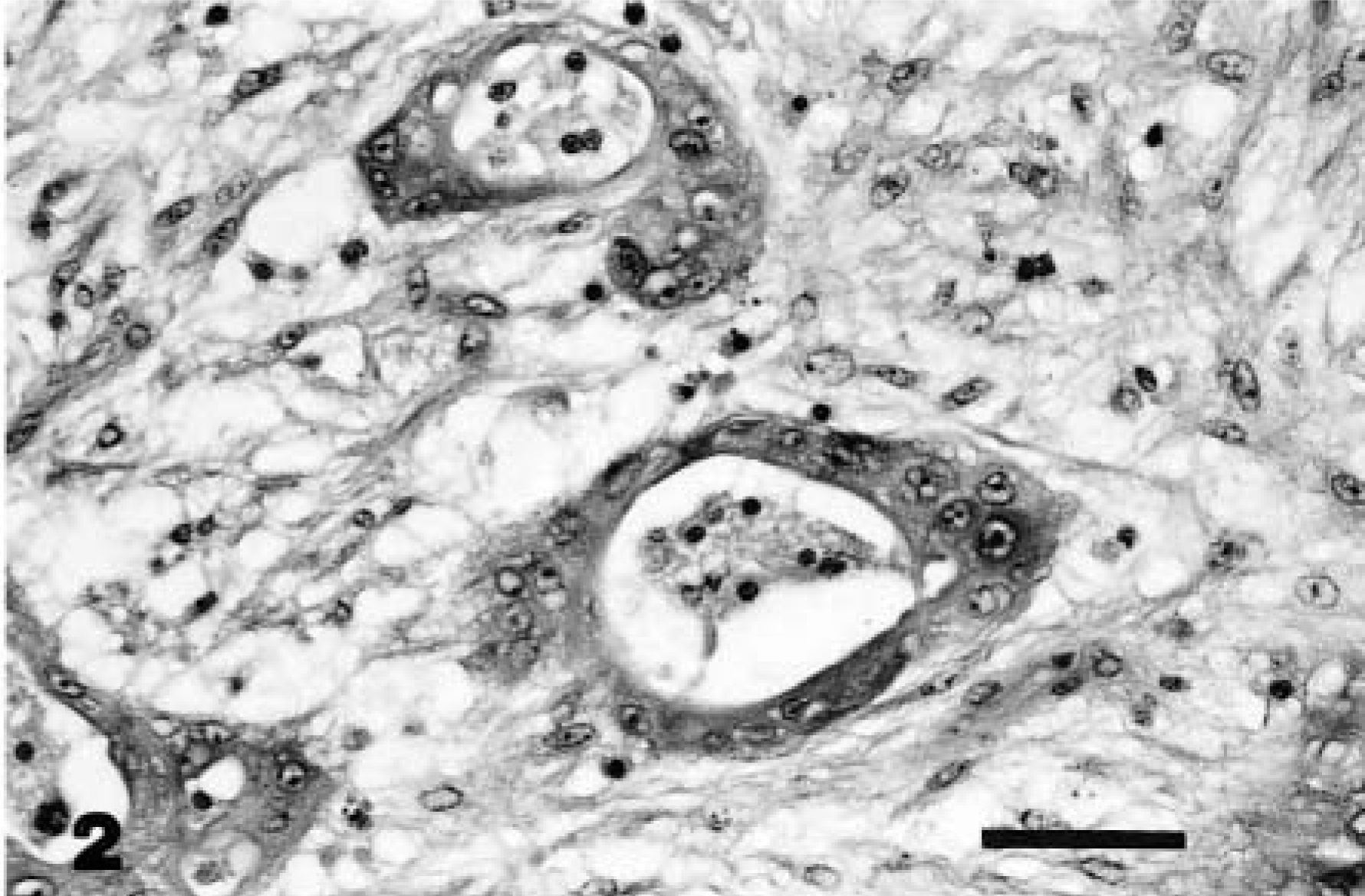
Colon; snake No. 2. Adenocarcinoma. Neoplastic acini embedded within a background of fibrous stroma in the submucosa. HE. Bar = 30 µm.
Snake No. 3 was presented with a palpable coelomic mass. Exploratory surgery revealed a highly vascular mass in the region of the right kidney. The snake was euthanatized because of involvement of vital organs. Necropsy revealed a 24- × 19- × 9-cm multinodular smooth firm mass (2.5 kg) originating in the pelvis, with subtotal replacement of the right kidney. Metastatic foci were not evident in any of the organs. HE-stained sections of the mass revealed invasive nests and lobular masses of closely packed vacuolated epithelial cells with abundant (disproportionate) intervening loose fibrous stroma (Fig. 3). The neoplasm was consistent with transitional cell carcinoma arising from the renal pelvis.
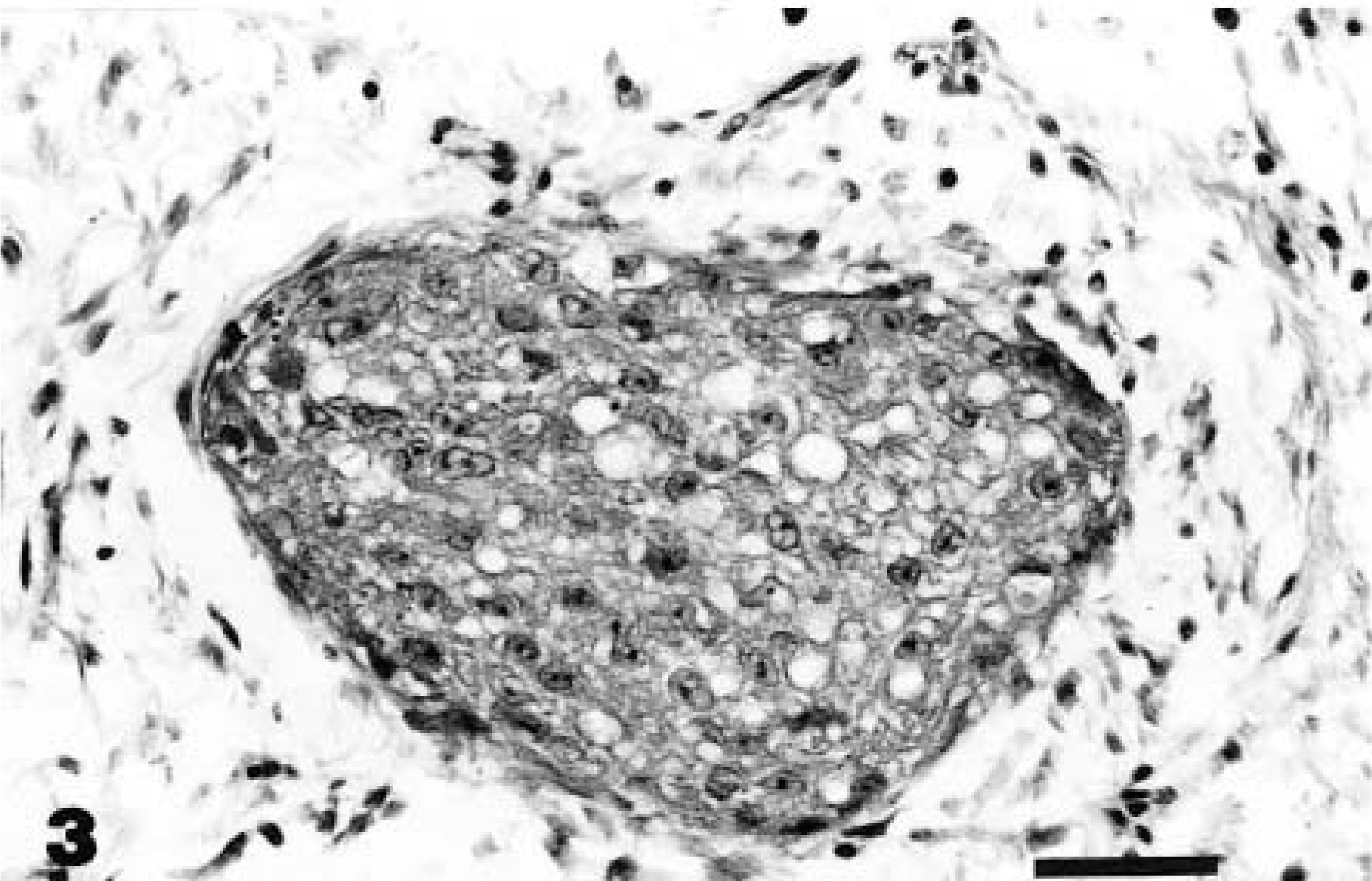
Kidney; snake No. 3. Transitional cell carcinoma. A lobule of closely packed vacuolated epithelial cells surrounded by loose fibrous stroma in the renal medulla. HE. Bar = 30 µm.
Snake No. 4 was euthanatized after a third recurrence of an inoperable intermandibular fibrosarcoma. Metastatic foci were not evident in any of the organs. HE-stained sections of the mass revealed a moderately cellular mass composed of spindle cells (Fig. 4) embedded within a background of collagenous stroma with osseous and cartilagenous differentiation.
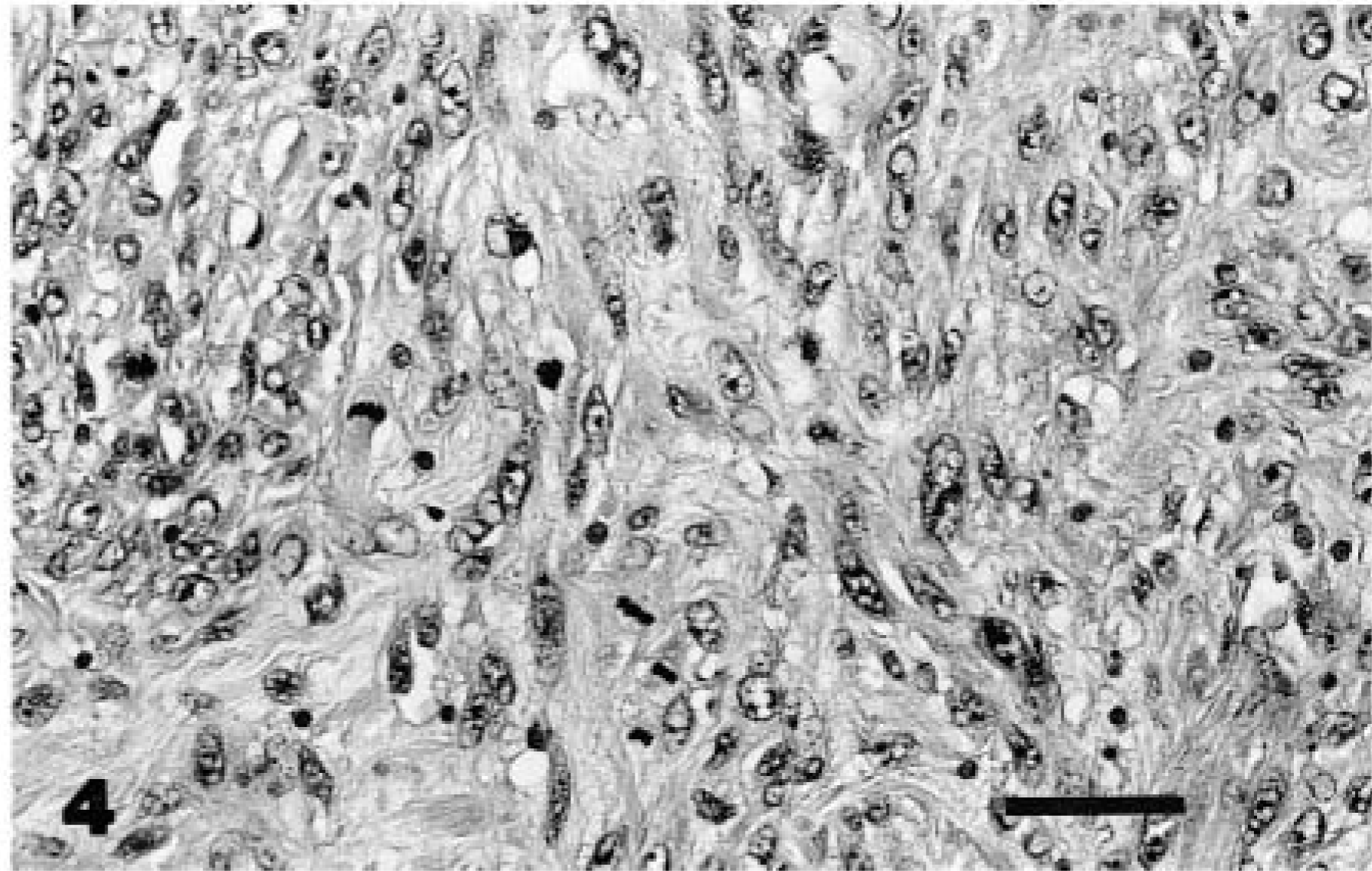
Mandibular mass; snake No. 4. Fibrosarcoma. The neoplasm is composed of closely packed spindle-shaped fibroblasts. Note the numerous mitotic figures. HE. Bar = 30 µm.
Ultrastructural examination of all neoplastic masses revealed few to numerous aggregates of extracellular and intracytoplasmic viral particles. These particles were present within cytoplasmic vacuoles, budding into vacuoles, free within cytoplasmic matrix, and as compact clusters within phagolysosomes (Fig. 5). Particles were about 93–96 nm in diameter and had a central electron-dense nucleoid of 55–60 nm surrounded by an electron-lucent halo with a distinct bilaminar external membrane (Fig. 6). The particles exhibited mild pleomorphism and were budding from the plasmalemma (Fig. 7). The number of viral particles observed in the tissues varied among the snakes, with numerous particles in neoplastic tissues from snake Nos. 1 and 3 and few particles in snake Nos. 2 and 4. The morphologic features and location of the observed virions were consistent with those of members of the family Retroviridae. Ultrastructural examination of nonneoplastic tissues (liver, kidney, and heart) from these snakes revealed rare viral particles after a thorough search, unlike the overt presence in the neoplastic tissues.
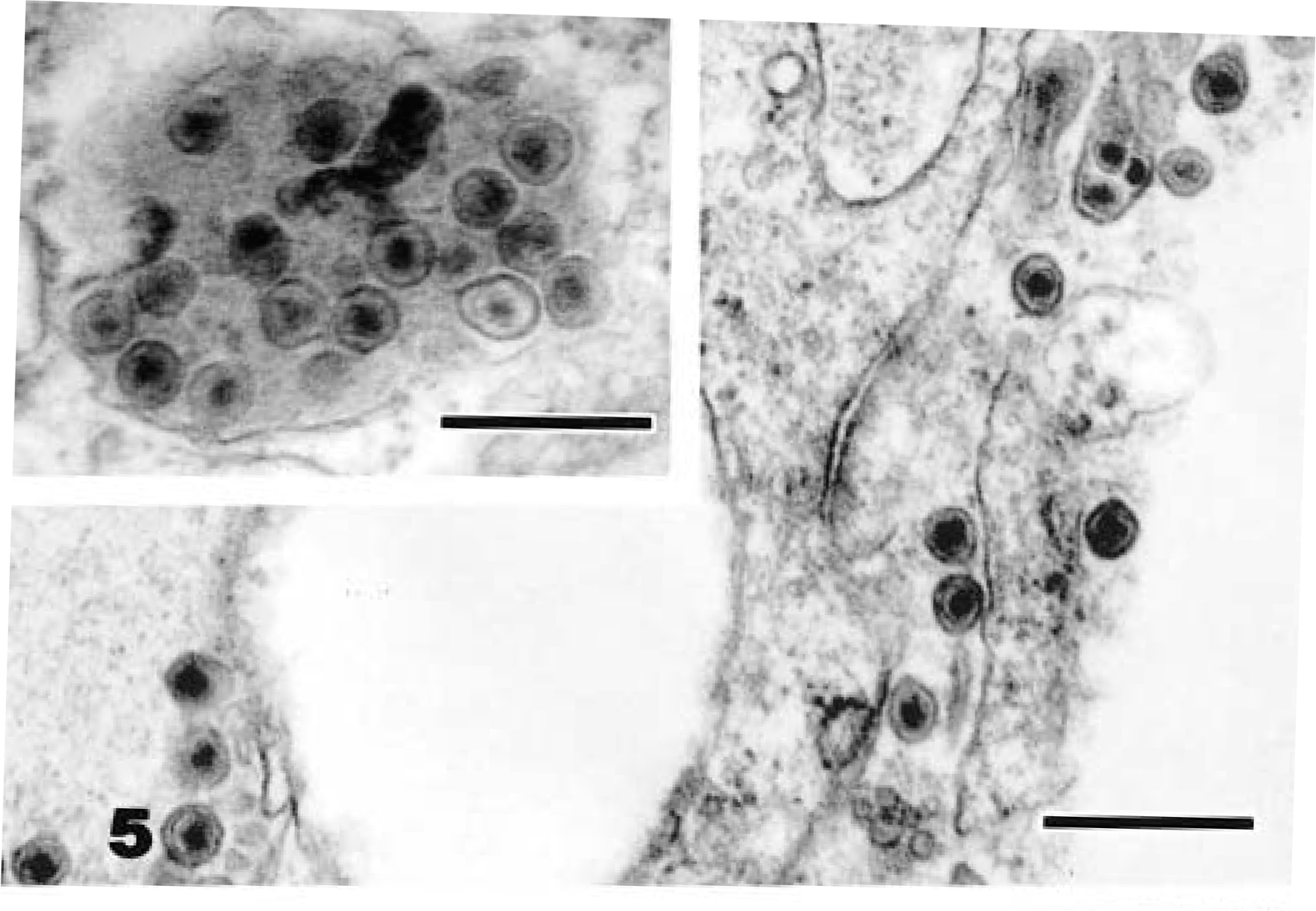
Electron micrograph. Kidney; snake No. 3. Intracellular type C-like particles in a neoplastic epithelial cell. Uranyl acetate and lead citrate. Bar = 0.4 µm.
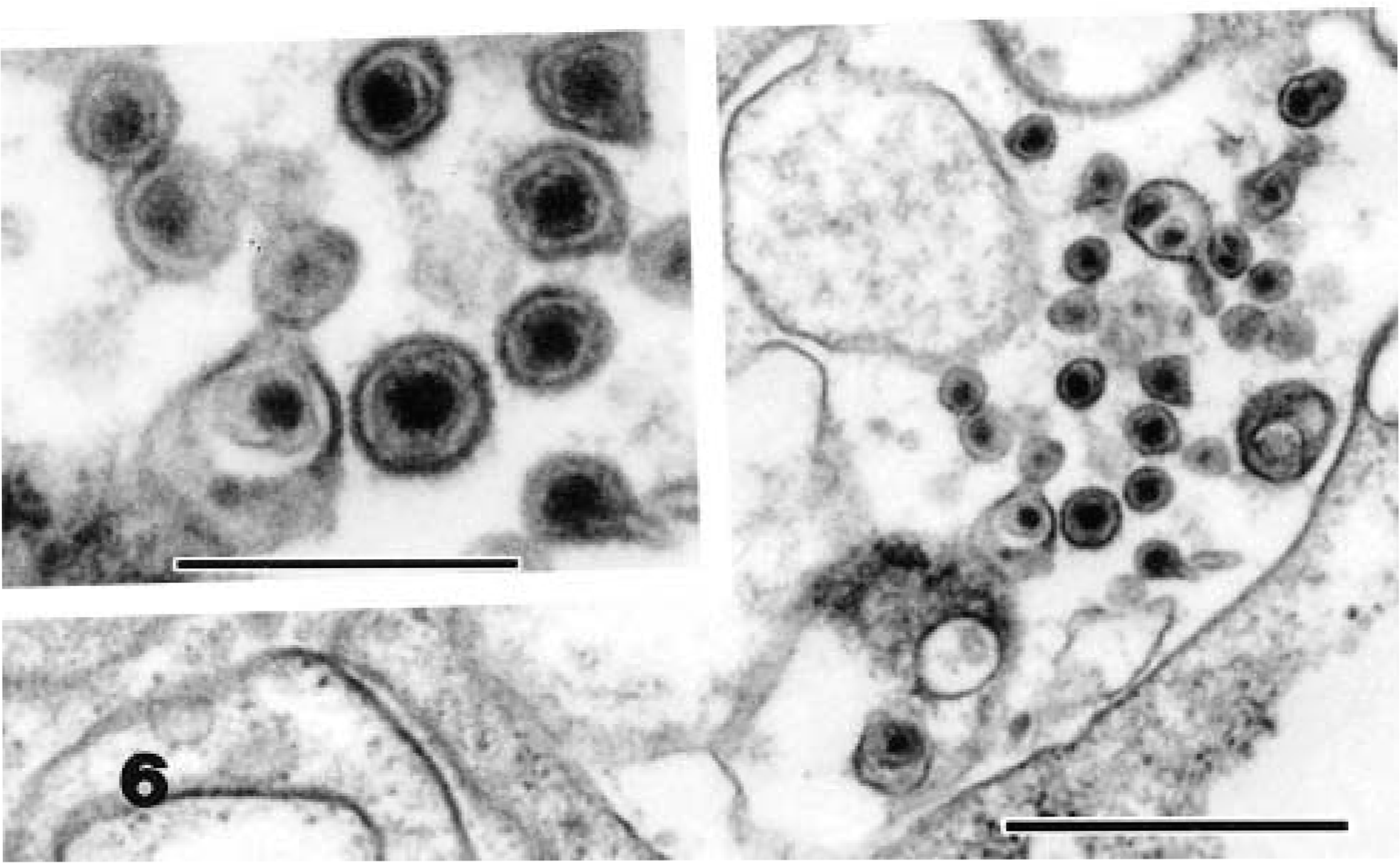
Electron micrograph. Spleen; snake No. 1. Intracellular type C-like particles in a neoplastic cell. Uranyl acetate and lead citrate. Bar = 0.7 µm.
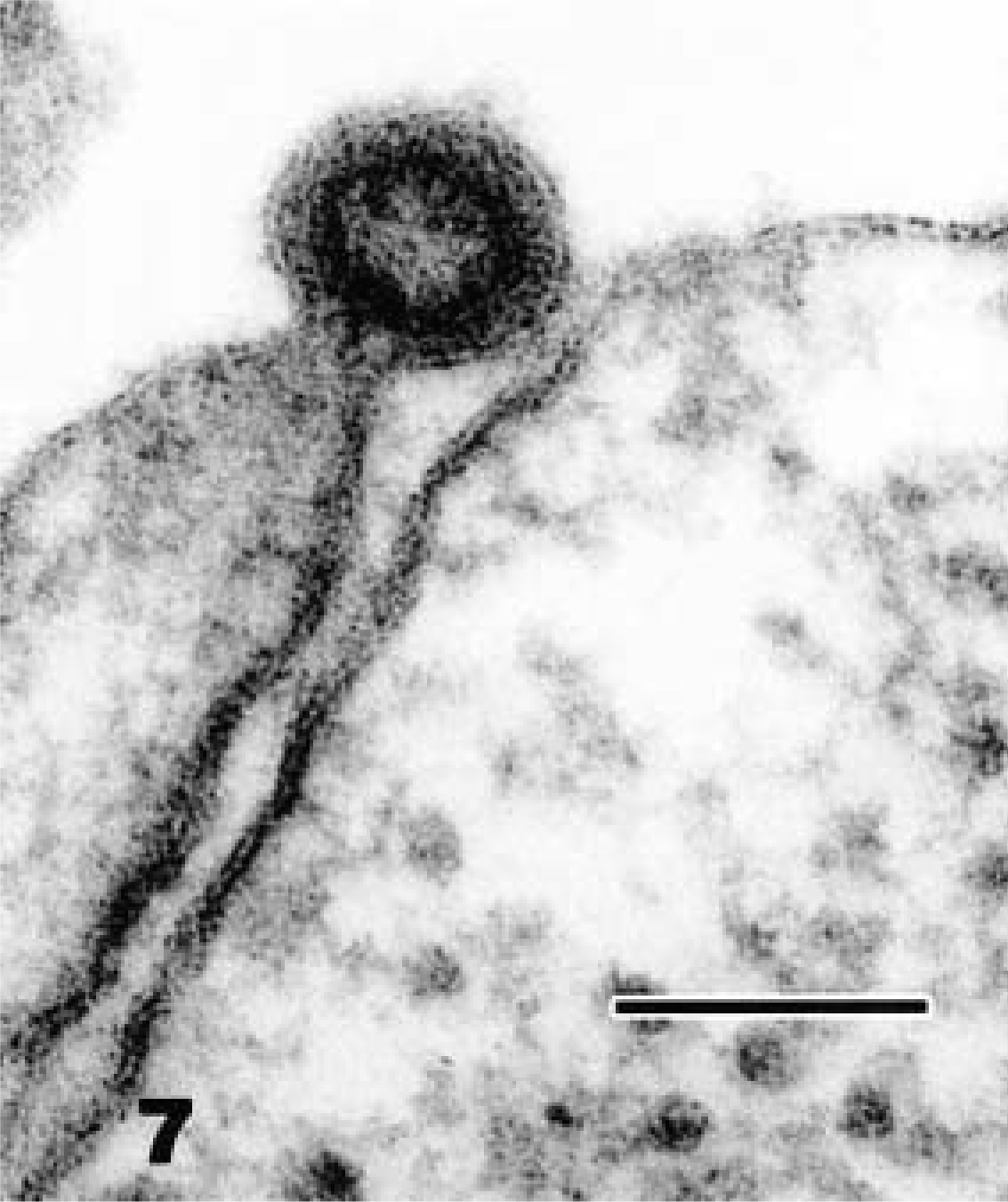
Electron micrograph. Spleen; snake No. 1. Budding of type C-like particle with a ring-shaped nucleoid. Uranyl acetate and lead citrate. Bar = 100 nm.
The particles observed in these snakes most closely resemble type C oncovirus particles, with a centrally located electron-dense core. Mature C particles are 80–110 nm in diameter, and they form by the condensation of viral nuclei acid and core proteins beneath the plasmalemma, with subsequent evagination, often forming a crescent-shaped bud. Intranuclear particles were not observed in any of the tissues. Type C particles lack precursor intracytoplasmic (type A) particles, which help to distinguish them from type B and type D retroviruses. 6,8
Several retroviruses have been reported in reptiles.
3
Almost all the retroviruses identified in snakes were from snakes with neoplasms. Among the reptilian retroviruses, the two well-documented examples are Russell's viper retrovirus (VRV)
11
and corn snake retrovirus (CSRV).
7
VRV originated from a late passage of cultured spleen cells from a viper that had a myxofibroma. Electron microscopic examination of the spleen-derived cell line revealed spontaneous production of 108-nm type C retrovirus particles. No virus was found either within the neoplasm or within tissues of the tumor-bearing viper.
10,11
CSRV was first observed on electron micrographs of an embryonal rhabdomyosarcoma from a female corn snake (
In addition to the CSRV and VRV, type C particles have been identified in neoplastic tissues from various snakes, including a boa constrictor (
The identification of type C-like particles in diverse species of snakes suggests an etiologic relationship between these viral particles and concurrent neoplasia. The role of the viral particles in the etiology of the tumors in these pythons is uncertain. All snakes in the present report were from a single collection, and viral particles were readily observed in the neoplastic tissues. Horizontal transmission of the virus in the closed collection is possible. Snake No. 1 was the offspring of snake No. 4; hence, vertical transmission of the virus is another possibility. In addition to the four snakes reported here, three other snakes from the same collection had been euthanatized previously because of neoplastic processes (two Burmese pythons with fibrosarcoma involving the subcutaneous tissue and mandible and a ball python [
Retrovirus-like particles also have been identified in various cellular and intercellular compartments in venom glands of apparently healthy vipers (
Type C-like viral particles were detected in neoplastic and nonneoplastic tissues of Burmese pythons from a captive collection. The role of the viral particles in the etiology of the tumors is uncertain. Additional studies including virus isolation and transforming ability of these viruses are needed.
Footnotes
Acknowledgements
We acknowledge the Electron Microscopy Core Laboratory of the Interdisciplinary Center for Biotechnology Research at the University of Florida for technical assistance with the electron micrographs. The help of Ms. R. Davis also is also greatly appreciated.
