Abstract
The pathogenicity of a serotype-1 fowl adenovirus (FAV-99ZH), which causes adenoviral gizzard erosion by oral inoculation in chickens, was investigated in specific pathogen-free white leghorn chickens. In trial 1, 14 chickens were inoculated intravenously with the virus at 21 days of age and euthanatized for necropsy within 1–14 days of inoculation. Gizzard erosion was grossly observed from day 7 postinoculation (PI), and histologically, FAV-99ZH antigen-positive, basophilic intranuclear inclusion bodies were seen in the gizzard lesions from day 7 to 11 PI. Necrotizing pancreatitis, and cholecystitis and cholangitis associated with the inclusions were observed from day 3 to 14 PI (pancreatitis) and from day 5 to 9 PI (cholecystitis and cholangitis), respectively. The inclusions were also observed in the epithelial cells of the cecal tonsils from day 3 to 5 PI. The virus was recovered from samples of the lesions. It was revealed that FAV-99ZH causes not only gizzard erosion but also pancreatitis, cholecystitis, and cholangitis by intravenous inoculation in chickens. In trial 2, 10 chickens were inoculated orally with the virus twice, at 13 and 36 days of age, and euthanatized for necropsy within 4–17 days after reinfection. Macroscopically, focal gizzard lesions were observed; however, neither necrosis nor inclusions were observed by microscopy. Moreover, FAV was not recovered from the gizzard or rectum of any of the chickens at necropsy. This suggests that the gizzard lesions occurred as a result of the primary infection, and that the chickens were able to resist reinfection.
Group-I fowl adenovirus (FAV) is associated with naturally occurring outbreaks of inclusion body hepatitis, 14 hydropericardium syndrome, 1 respiratory disease, 5 necrotizing pancreatitis, 22 or adenoviral gizzard erosion (AGE). Natural cases of AGE in chickens were first reported in 1993. 3,7,27 In Japan, AGE in broiler chickens has been reported 2,21 and is frequently found in slaughtered broiler chickens. 20 In our previous studies, AGE was experimentally reproduced in specific pathogen–free (SPF) white leghorn chickens or in commercial broiler chickens inoculated by both oral and ocular routes with the FAV serotype-1 isolated from gizzard lesions; 17,18 the histopathologic and immunohistochemical findings of the affected birds have been described in a previous report. 19 Nakamura et al. 15 successfully reproduced gizzard erosion in SPF chickens after oral inoculation with three strains of FAV serotype-1, including the FAV-99ZH strain used in our present and previous studies. However, the details of the pathogenicity of the FAV strain that causes gizzard erosion were unclear.
In the present study, two experiments were conducted with SPF chickens. In the first, chickens were subjected to intravenous inoculation (trial 1). Many FAVs fail to cause disease when given by natural routes but are highly pathogenic when given by parenteral injection. For example, a representative strain of FAV serotype-1 in Japan, the Ote strain, does not cause lesions by oral inoculation; 15 however, it causes inclusion body hepatitis in SPF chicken by intravenous inoculation. 11 It is possible that FAV strains that cause gizzard erosion may also cause other lesions such as inclusion body hepatitis by intravenous inoculation. In our second experiment, chickens were reinfected orally (trial 2). It has been reported that chickens are resistant to reinfection with the same serotype of FAV 45 days after primary infection. 4 However, a different study found that it was possible to reinfect chickens with the same strain after 8 weeks. 29 To be able to make appropriate decisions, it is important to determine whether chickens naturally invaded by a primary FAV infection develop resistance to reinfection.
Materials and Methods
Virus
In the present study, we used FAV-99ZH, a strain belonging to group-I FAV serotype-1 and originally isolated from an outbreak of AGE in a commercial broiler chicken flock in Japan. 21 In both trials, a 106 median tissue culture infectious dose (TCID50) was used per 0.05 ml/chicken.
Chickens
White leghorn chickens hatched from SPF eggs were used for these experiments to ensure the absence of maternal antibodies against FAV. Throughout the experimental period, chickens in each of the two groups were kept in isolated rooms in a negatively pressured house to prevent crosscontamination between groups. Control chickens were also maintained in another room under the same conditions.
Experimental design
In trial 1, 14 chickens were inoculated intravenously with FAV-99ZH in 0.05 ml of growth medium at 21 days of age. On days 1, 3, 5, 7, 9, 11, and 14 postinoculation (PI), two chickens were euthanatized by cervical dislocation for necropsy. Two control chickens were also inoculated with growth medium alone and necropsied on days 3 and 9 PI. In trial 2, 12 chickens were inoculated orally with FAV-99ZH in 0.1 ml of growth medium at 13 days of age. Ten of 12 chickens were reinfected at 36 days of age (23 days after primary inoculation). On day 0 (preinoculation) and on days 4, 7, 11, 14, and 17 after reinfection, two chickens were euthanatized for necropsy. Three control chickens were inoculated with growth medium alone at 13 and 36 days of age and necropsied on days 0, 4, and 14 after reinfection.
Histology
After postmortem examination of the chickens, in trial 1, sections of the esophagus, proventriculus, gizzard, duodenum, jejunum, ileum, distal cecum, cecal tonsils, bursa of Fabricius, liver, pancreas, spleen, kidney, heart, lung, trachea, thymus, and brain were removed. In trial 2, sections of gizzard (main organ affected by oral inoculation), pancreas (affected organ by intravenous inoculation, from the result of trial 1 in this study), and duodenum were removed. The tissues were fixed in 20% neutral buffered formalin. The paraffin-embedded tissues were sectioned at 4 µm and stained with hematoxylin and eosin (HE).
Immunohistochemistry
As in our previous study, 19 the FAV-99ZH antigen was detected by immunohistochemical staining in formalin-fixed, paraffin-embedded tissues from all sections from both trial groups. In brief, rabbit anti–FAV-99ZH serum (prepared in the Zennoh Institute of Animal Health) was used as a primary antibody at a dilution of 1 : 12,000 in 0.01 M phosphate-buffered saline (pH 7.2). The presence of the FAV-99ZH antigen was verified by the EnVision+ Rabbit/HRP system (Dako, Copenhagen, Denmark), following a previously published procedure. 28 3,3′-Diaminobenzidine tetrahydrochloride served as the chromogen, and the sections were counterstained with methyl green.
Virus isolation
At necropsy, virus isolation was attempted from several tissues including the gizzard, kidney, liver, lung, pancreas, rectum (including feces), and bile in trail 1 and from the gizzard and rectum (including feces) of all chickens in trial 2. For virus isolation, 10% tissue homogenates were inoculated onto chicken kidney (CK) cells, cultured for 7 days, and observed for a cytopathic effect. FAV was identified by the indirect immunofluorescence test and the polymerase chain reaction restriction fragment length polymorphism (PCR-RFLP) method as follows. The TCID50 of the virus was determined on microculture plates with CK cells, and titers were calculated as described previously. 18
Polymerase chain reaction
Forward primers H1 and H3, reverse primers H2 and H4, and restriction enzymes HaeII and HpaII were used for PCR-RFLP as described previously. 24 Serotype-1 FAV-specific restriction profiles could be found in digests of the H1/H2 PCR products by HaeII and in digest of the H3/H4 PCR products by HpaII. PCR was also performed in the cloacal swabs for the detection of FAV from preinoculated chickens in both trials and on days 7, 14, and 21 after primary inoculation in trial 2.
Serology
In trial 1, serum antibodies against FAV serotype-1 were examined by a virus neutralization test in all chickens at both preinoculation and necropsy. Virus neutralization tests were performed on microculture plates with CK cell monolayers. 9 In trial 2, the antibodies were examined as in trial 1 on days 7, 14, and 21 after primary inoculation and again at necropsy.
Results
Trial 1 (intravenous inoculation)
Two chickens that were necropsied on day 9 PI showed clinical signs such as depression and anorexia from day 4 PI. Gizzard erosion was grossly observed from day 7 to 14 PI. Significant gross lesions were not present in other organs or in the uninoculated control chickens.
Histologically, gizzard lesions were observed from day 7 PI (Table 1, Fig. 1). The koilin layer was widened, stained somewhat poorly with eosin, and loosened, and it contained desquamated cells. Epithelial cells of affected glands were degenerative, and basophilic intranuclear inclusion bodies were occasionally present in the enlarged nuclei. Heterophils, macrophages, lymphocytes, and plasma cells were observed in the edematous lamina propria. There was mononuclear cell infiltration in the submucosa and muscle layer under the erosive lesion. These gizzard lesions were similar to lesions found in chickens experimentally inoculated orally with FAVs isolated from gizzard erosion. 15 The inclusions were also found in the epithelial cells of the extrahepatic (rarely intrahepatic) biliary passages, the cecal tonsils, and the acinar cells of the pancreas from day 3 PI. Multifocal necrosis in pancreata associated with the inclusions was observed with infiltration of mononuclear cells and heterophils (Fig. 2). The pancreatic acinar cells in the necrotic lesions had fewer eosinophilic zymogen granules. Multifocal cholangitis and cholecystitis were observed with or without inclusions from day 5 to 9 PI. The epithelial cells of biliary passages were necrotic and desquamative, and inflammatory cells were observed in the lumen and lamina propria (Fig. 3). Multifocal necrosis of the epithelium was observed in the cecal tonsils of chickens necropsied on days 7 and 14 PI (Fig. 4). The inclusions were found by immunohistochemical staining to be positive for FAV-99ZH antigen (Figs. 5–7).
Histologic lesions in various organs of chickens inoculated intravenously with FAV-99ZH.

No. of birds positive/no. of birds examined.
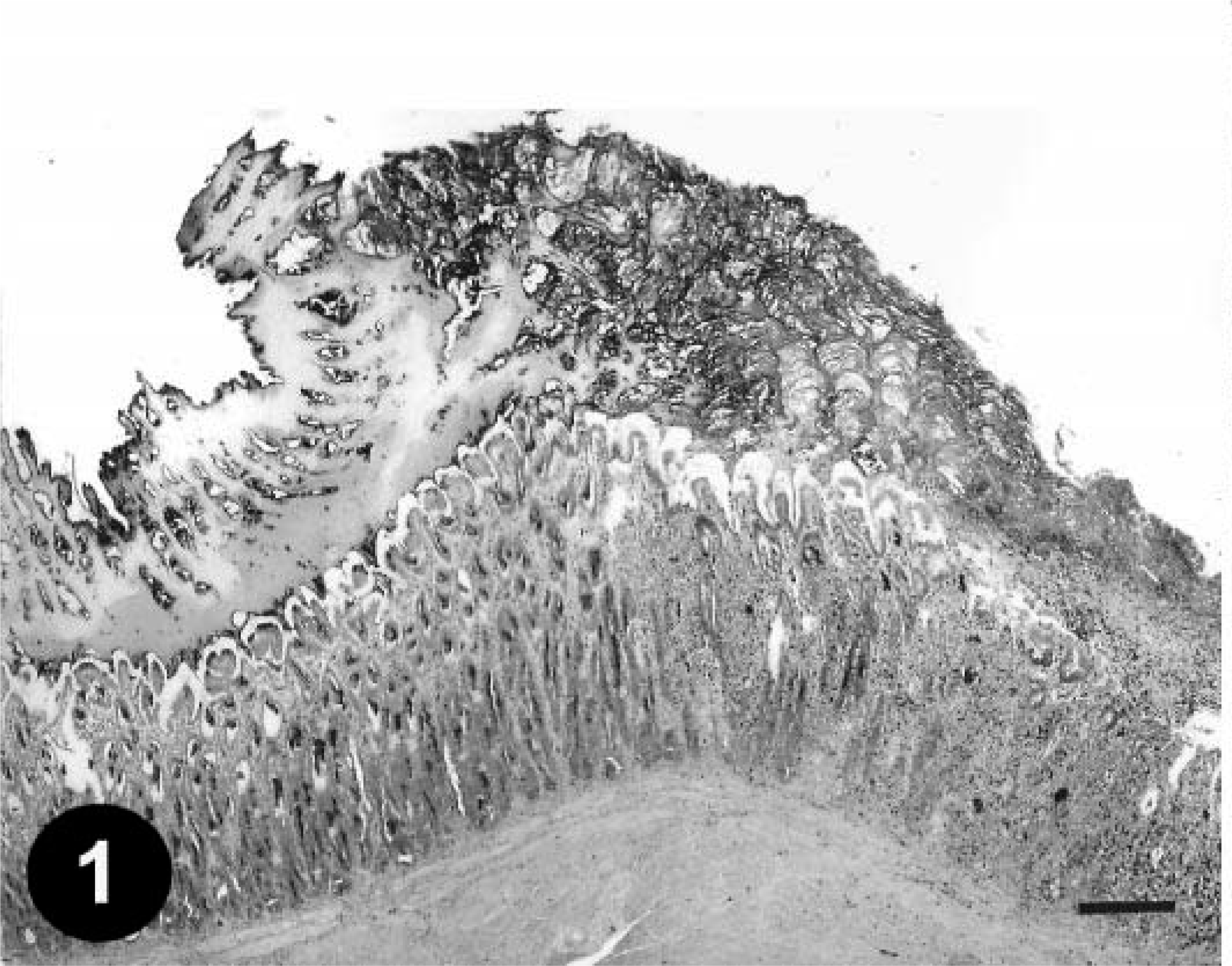
Gizzard; a chicken from trial 1 on day 7 PI. Degeneration of the koilin layer, disappearance of epithelial cells, and inflammatory cell infiltration in the focal necrotic mucosal membrane. HE. Bar = 250 µm.
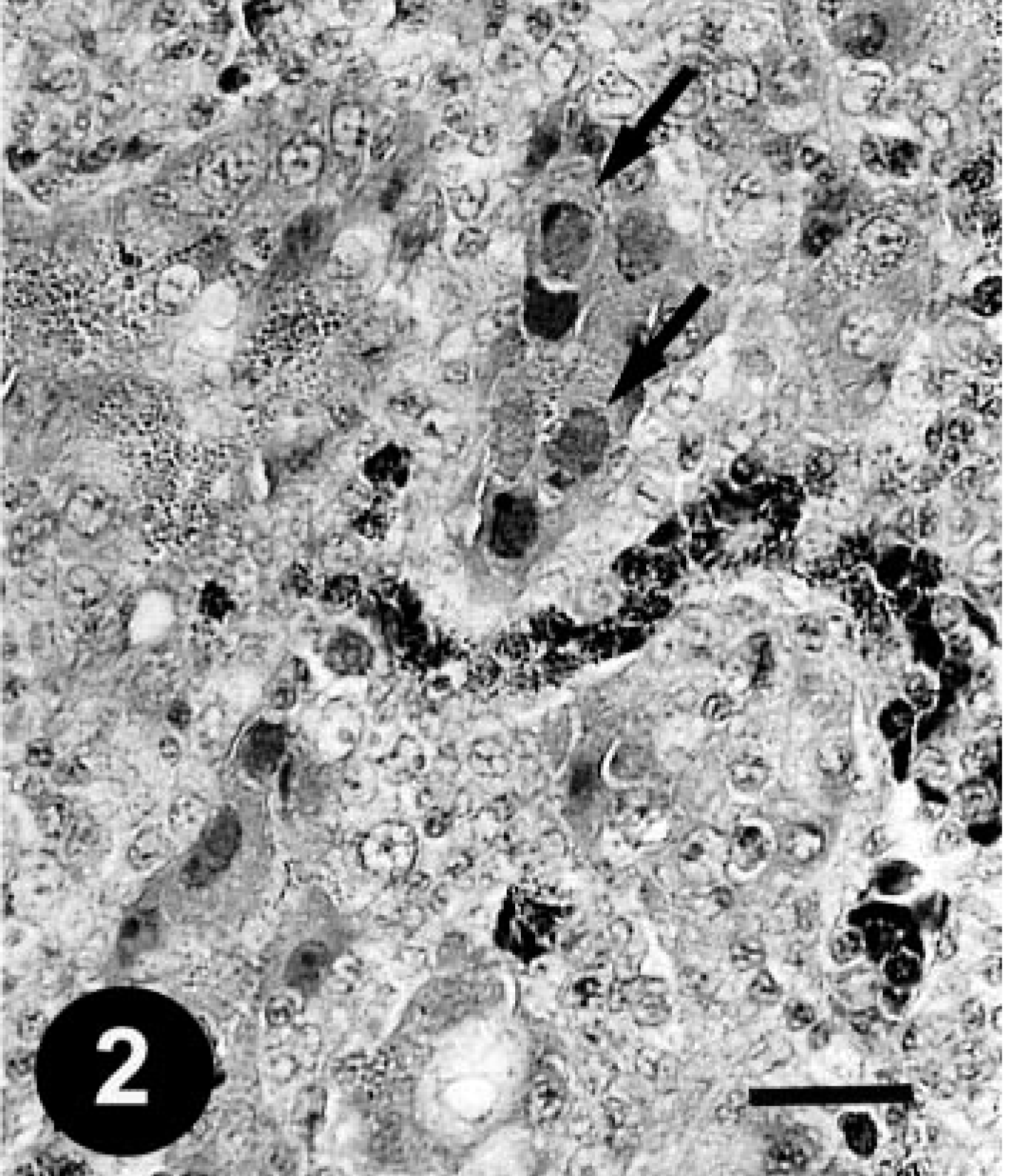
Pancreas; a chicken from trial 1 on day 3 PI. Large intranuclear inclusion bodies (arrows), which completely fill the enlarged nuclei of the acinar cells associated with the focal necrotic lesions. Mononuclear cells and heterophils are seen in the interstitial tissue. HE. Bar = 25 µm.
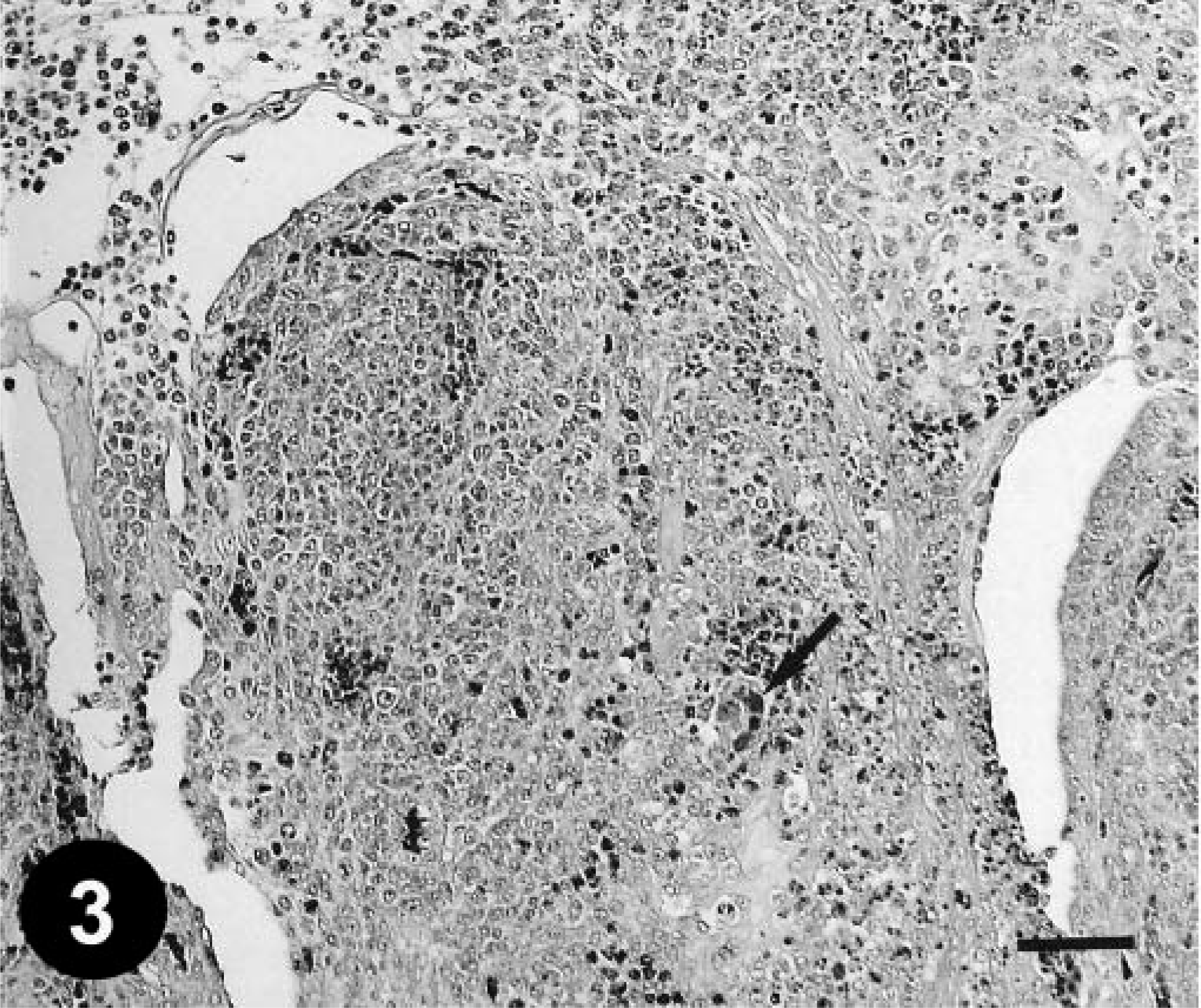
Gallbladder; a chicken from trial 1 on day 5 PI. Cholecystitis. Numerous inflammatory cells are seen in the lumen and lamina propria, and most epithelial cells are desquamated. The arrow indicates the inclusions in the desquamated epithelial cells. HE. Bar = 50 µm.
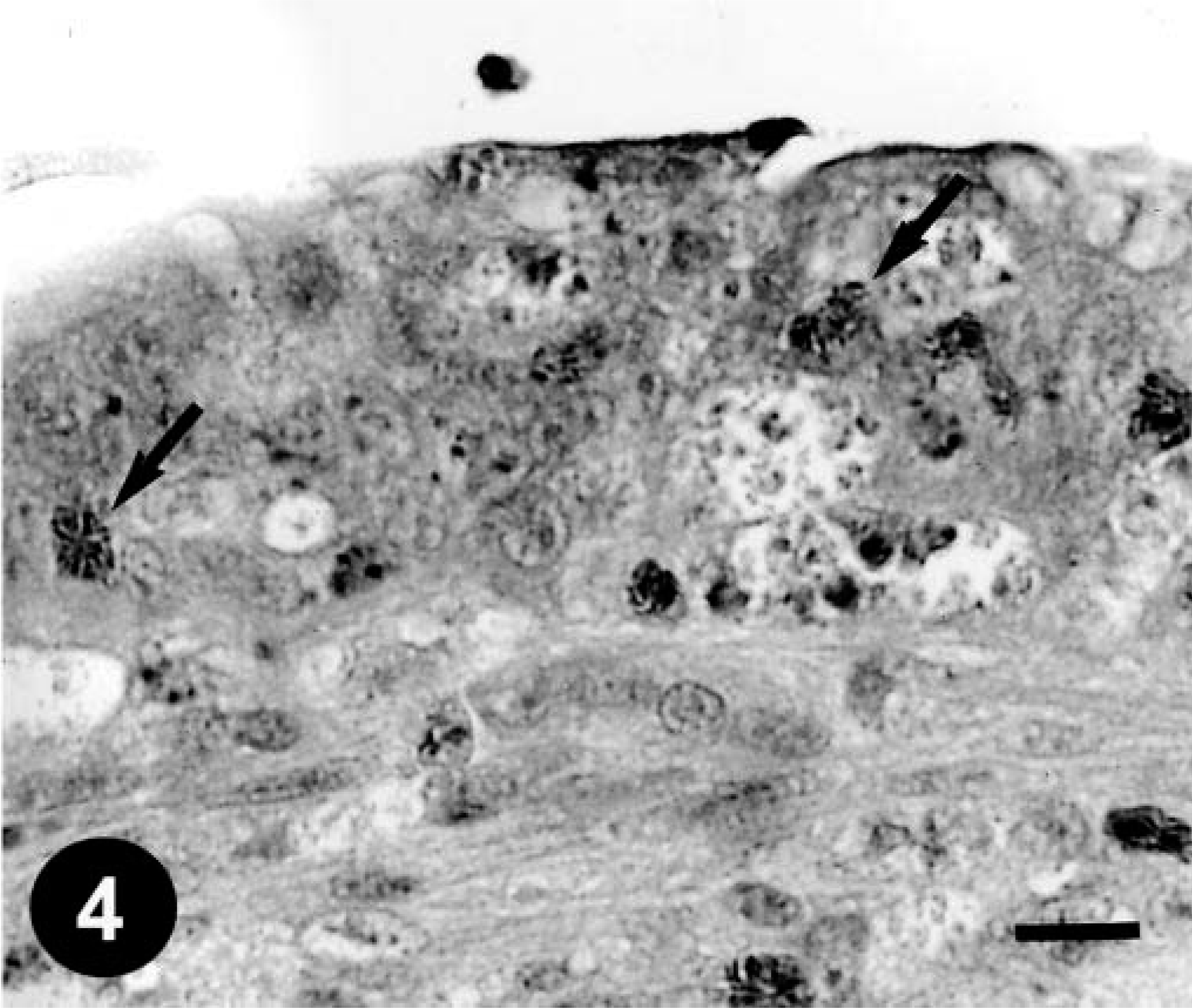
Cecal tonsil; a chicken from trial 1 on day 7 PI. Necrotic epithelium. Heterophils are seen in the lesion (arrows). HE. Bar = 10 µm.
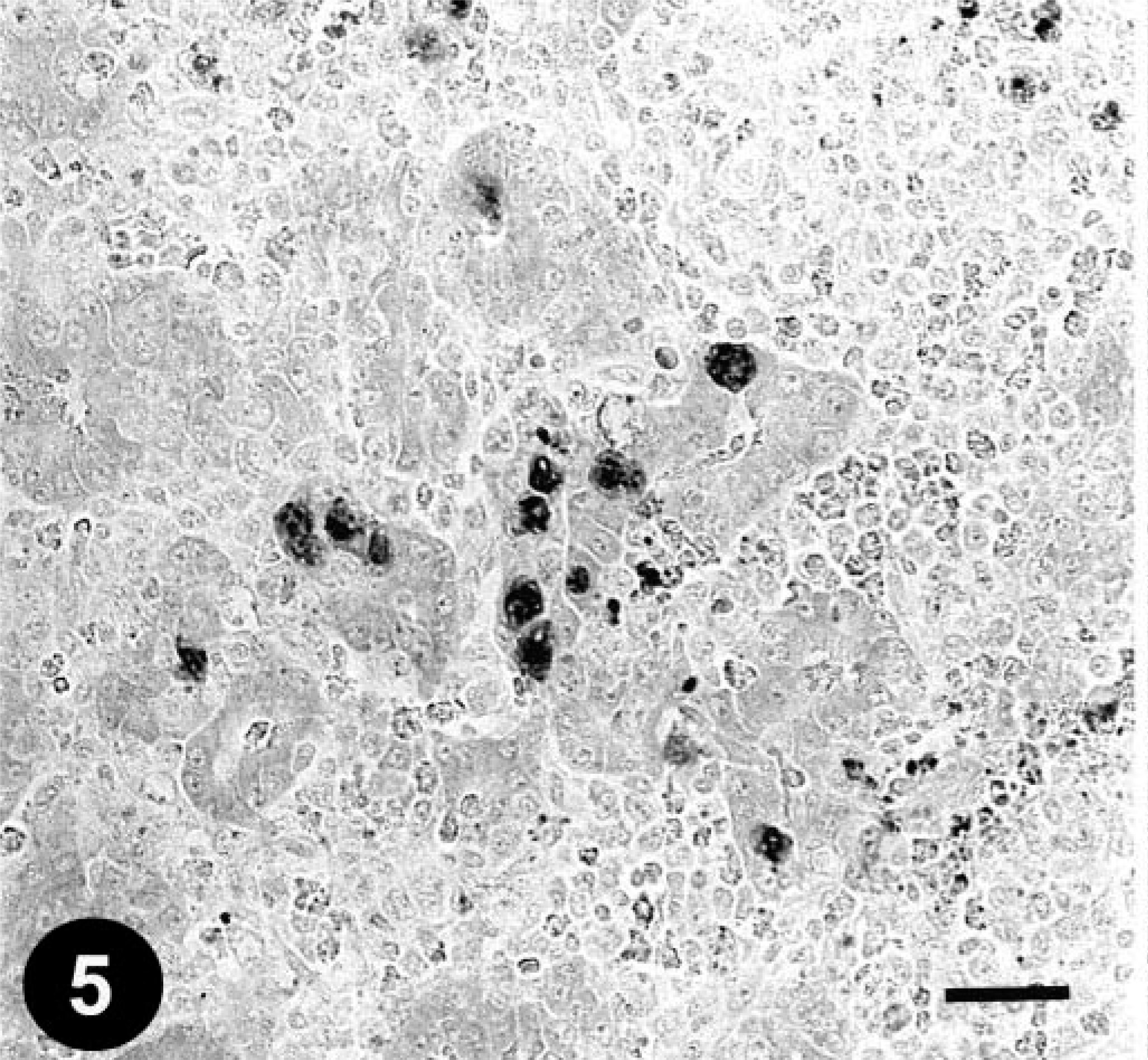
Pancreas; a chicken from trial 1 on day 11 PI. The FAV antigen–positive inclusions in the acinar cells. Immunohistochemical staining for the FAV antigen, methyl green counterstain. Bar = 25 µm.
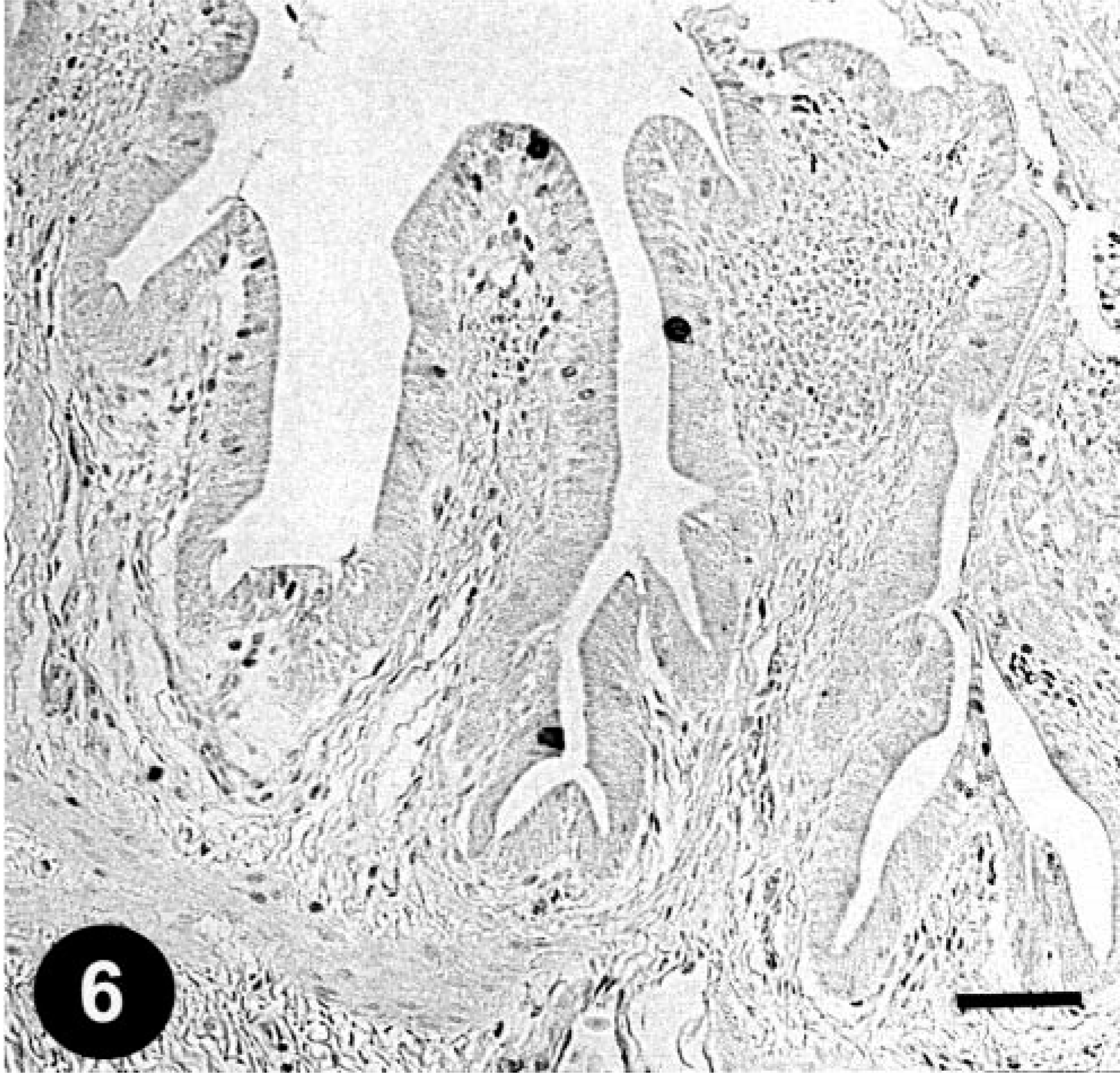
Gallbladder; a chicken from trial 1 on day 3 PI. FAV antigen–positive inclusions in the epithelial cells. Immunohistochemical staining for the FAV antigen, methyl green counterstain. Bar = 50 µm.
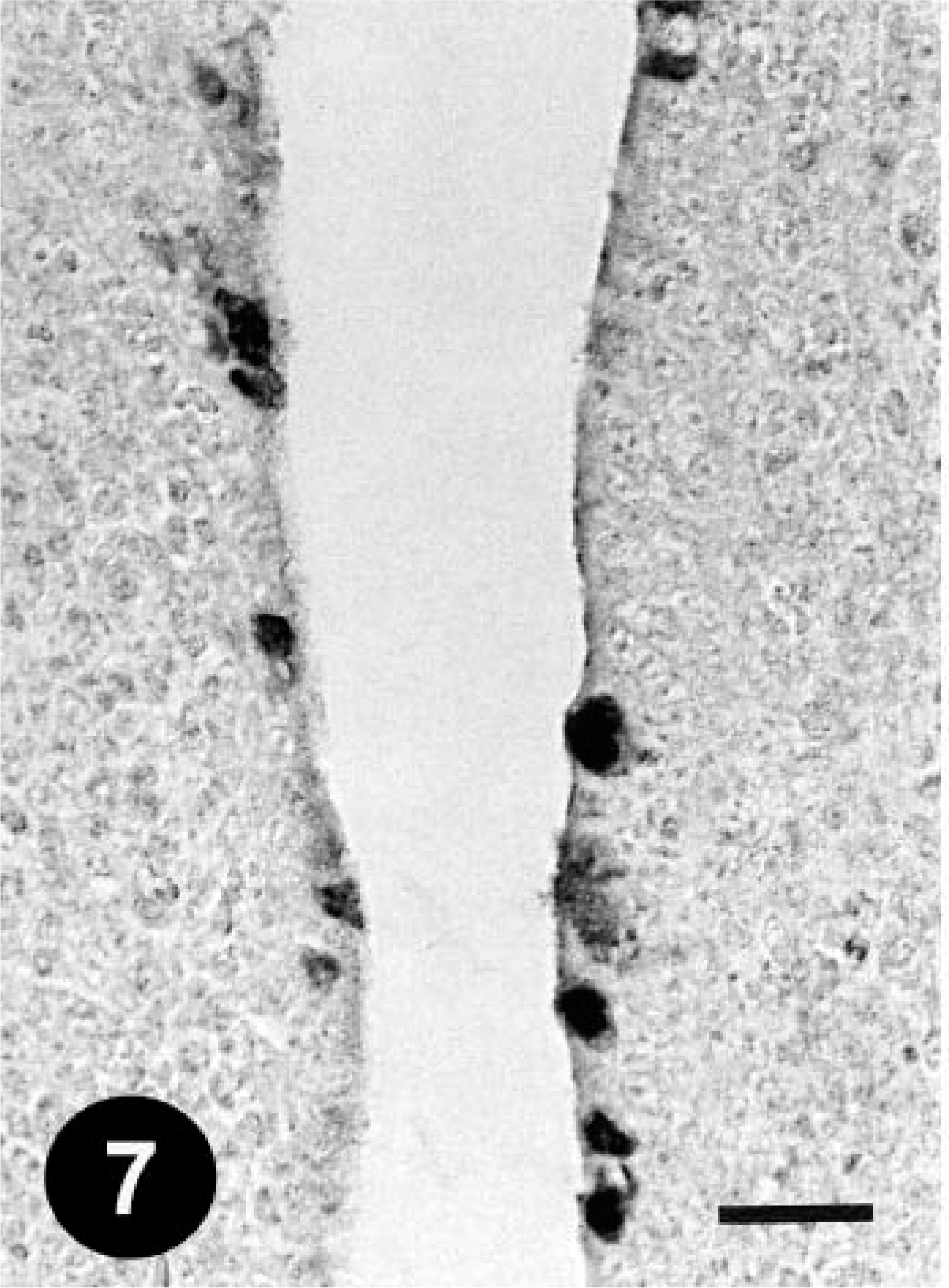
Cecal tonsil; a chicken from trial 1 on day 5 PI. FAV antigen–positive inclusions in the epithelial cells. Immunohistochemical staining for the FAV antigen, methyl green counterstain. Bar = 25 µm.
Virologic and serologic results are summarized in Table 2. FAV was recovered from the gizzard, liver, pancreas, kidney, rectum, and bile samples of inoculated chickens. FAV was not recovered from samples of uninoculated chickens. PCR for the detection of FAV from cloacal swabs of preinoculated chickens was negative in all chickens. Serum neutralizing antibodies to FAV serotype-1 were present in inoculated chickens from day 5 PI. Sera from preinoculated or uninoculated chickens were serologically negative for FAV serotype-1.
The virologic and serologic results in samples of chickens inoculated intravenously with FAV-99ZH.
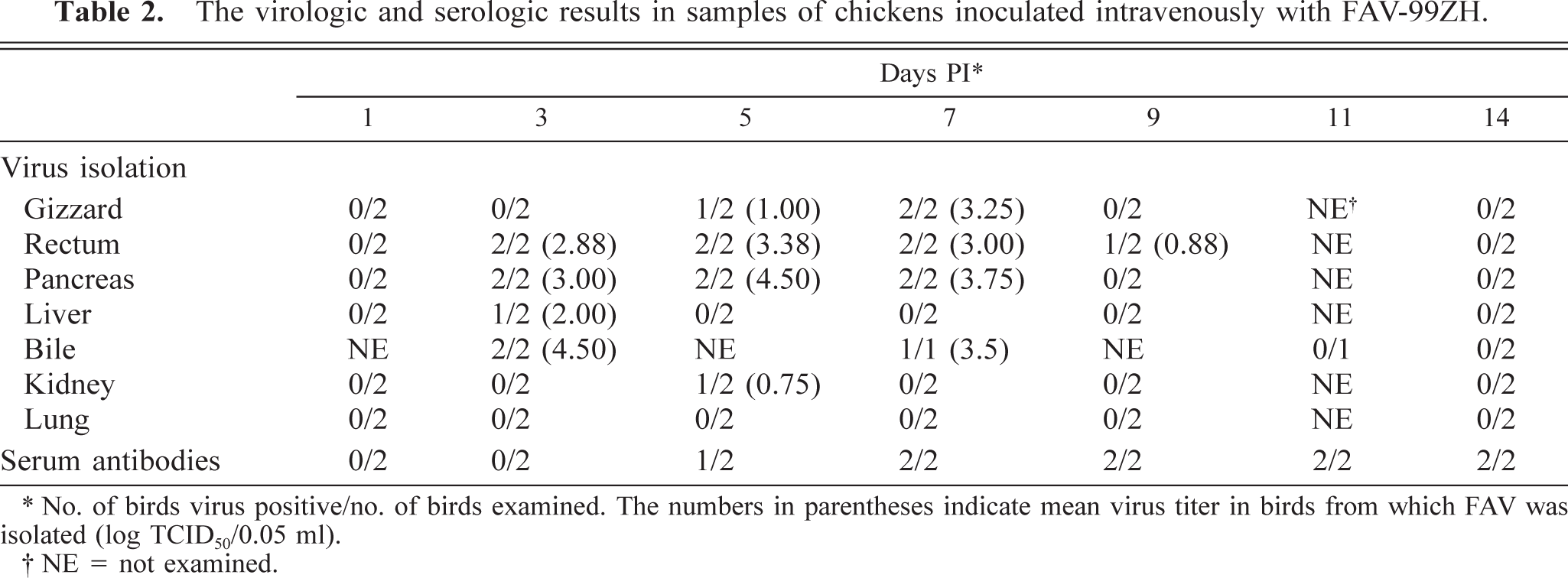
No. of birds virus positive/no. of birds examined. The numbers in parentheses indicate mean virus titer in birds from which FAV was isolated (log TCID50/0.05 ml).
NE = not examined.
Trial 2 (reinfection orally)
Five of 12 chickens inoculated orally with FAV showed slight depression and anorexia for a few days, beginning at day 4 after primary inoculation. However, after reinfection, no clinical signs were observed in any of the chickens. Macroscopically, focal gizzard lesions such as a rough or cloudy koilin layer were observed in one of two chickens at 4, 7, and 11 days PI. Histologically, the degenerative koilin layer was thickened and loosened with desquamated cells at the time of reinfection and at all times thereafter (Table 3). However, neither necrosis nor inclusions were observed in the lamina propria (Fig. 8). No significant histologic lesions were present in other organs or in the uninoculated control chickens. No FAV antigen was immunohistochemically detected in any examined tissues. FAV was detected by PCR in the cloacal swabs from 10 of the 12 inoculated chickens on day 7 after primary inoculation. FAV was not recovered from the gizzard or rectum of any of the chickens at necropsy. All inoculated chickens were serologically positive for FAV serotype-1 from day 14 after primary inoculation.
Histologic lesions of chickens inoculated orally twice with FAV-99ZH.

No. of birds positive/no. of birds examined.
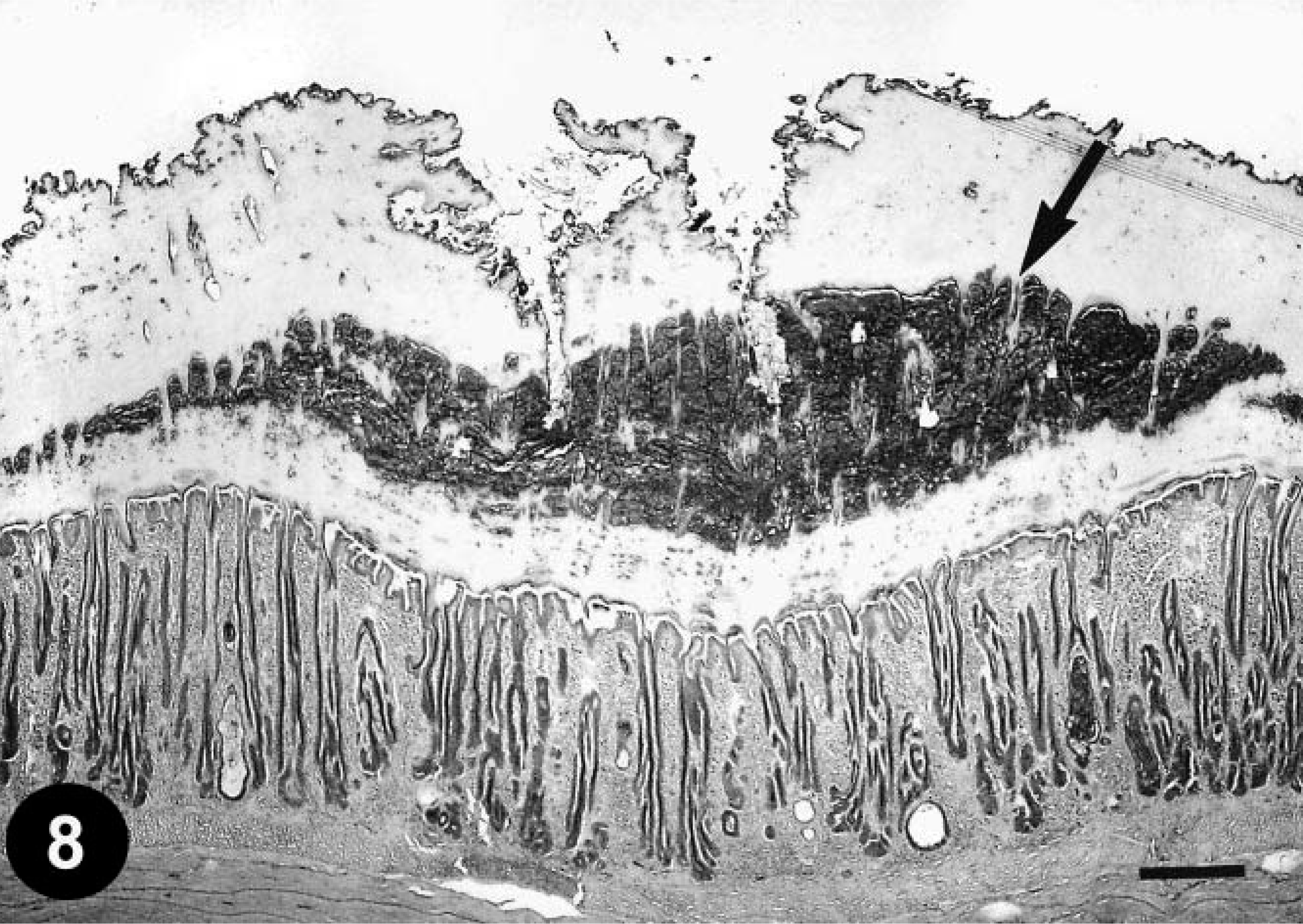
Gizzard; a chicken from trial 2 on day 7 after reinfection (day 30 after primary inoculation). Degenerative koilin is observed in the middle of the layer (arrow); however, normal koilin is newly formed under the degenerative layer. Hyperplasticity of the epithelium is sparsely observed in the lamina propria, compare with Fig. 1. HE. Bar = 250 µm.
Discussion
In trial 1, basophilic intranuclear inclusion bodies, which were immunohistochemically FAV antigen positive, formed in the epithelial cells of the gizzard, biliary passages, cecal tonsils, and acinar cells of the pancreas of chickens inoculated intravenously with FAV-99ZH, and FAV was recovered from these organs. It has been reported that lesions are observed only in the gizzards of SPF chickens inoculated by natural routes; 15,18 we suggest here that lesions in the pancreas and biliary passages are the result of hematogenous dissemination. In addition, it has been reported that FAV (strain 93) is rarely recovered from the pancreas and bile of chickens experimentally inoculated with the virus. 4 In our previous study, inclusions in the cecal tonsils were observed in broiler chickens inoculated by natural routes. 19 Nevertheless, in the latest natural outbreaks of AGE in broiler chickens in Japan, no lesions were observed except in the gizzard. 2,21 This suggests the possibility that the naturally affected chickens were infected with FAV not by trauma but by oral routes.
Necrotizing pancreatitis associated with inclusion body hepatitis has been reported in natural outbreaks 8,16,25,27 as well as in experimental avian adeno-virus disease of chickens. 6,10 However, in trial 1, no lesions of these diseases were observed. It has been reported that two strains of FAV cause gizzard erosion and necrotizing pancreatitis without hepatic or intestinal lesions when FAV is administered experimentally by oral inoculation. 12 The observed pancreatic lesions were clearly different from the lesions of malabsorption syndrome (runting and stunting syndrome) such as atrophy, occlusion of the pancreatic ducts, interstitial fibrosis, and dilation of the acinar lumens. 13,23
Inclusions in the epithelial cells of the biliary passages were observed in four chickens in trial 1. Cholangitis and cholecystitis associated with FAV infection have not been previously reported in chickens although both a different type of cholangitis and cholangiohepatitis have been reported, and Clostridium perfringens has been isolated from the lesions. 26
The difference in time of the appearance of inclusions among organs is interesting. At first, the inclusions were formed in the biliary passages, cecal tonsil, and pancreas from day 3 PI. In inclusion body hepatitis, inclusions were also observed in the hepatocytes during the same period. 10,11 On the other hand, in the gizzard, the appearance of inclusions and FAV recovery occurred later than in other affected organs as well as later than in SPF chickens inoculated with FAV-99ZH by natural routes. In our previous study, inclusions were observed from day 5 to 7 PI, and FAV was recovered from day 3 to 7 PI from the gizzards of chickens inoculated with the same dose of FAV by natural routes at 53 days of age. 18 The exact mechanism of this difference in time remains unclear; however, we suggest first that the epithelial cells of the gizzard suffered less infection because the gizzard lesions appeared later in the inoculated chickens, which received a lower dose of the virus, 19 and second, that FAV replicated in other affected organs and infected the gizzard secondarily. It is possible that it replicated in the biliary passages and pancreas, was excreted to the duodenum on day 3 PI, and then reached the mucosal surface of the gizzard by reversed peristalsis or deglutition of feces. It is nevertheless apparent that replication of the virus is slower in the gizzard than in the biliary passages or pancreas.
In trial 2, oral reinfection with FAV-99ZH at 23 days after primary infection was unsuccessful in the present study. We suggest that FAV cannot repeatedly form the lesions of AGE in commercial broiler chickens because chickens of the affected flock live on litter contaminated with FAV and always acquire FAV from the litter. The combined results of this and our previous studies suggest that slaughtered broiler chickens that have AGE lesions may have been infected with FAV at approximately 4–18 days before the date of slaughter (at 35 or more days of age). 19 This suggests that interception of the horizontal spread of the virus by direct fecal contact or aerial contact over short distances is important for reduction of the damage caused by AGE at slaughter.
