Abstract
Seven 19-day-old broiler chickens affected with hydropericardium syndrome (HPS) with pancreatic necrosis and gizzard erosions were investigated pathologically and virologically. Mortality increased after 13 days of age in a flock on a broiler farm. The mortality rate of the flock reached 10% by 19 days of age. Macroscopically, the chickens had hydropericardium (the characteristic gross change of HPS), pinpoint white foci in the pancreas, and ventricular erosions. Histologically, the chickens had multifocal hepatic necrosis with intranuclear inclusions in hepatocytes, a marked increase of macrophages in the spleen and lung, mild epicardial edema, multifocal necrosis of pancreatic acinar cells with intranuclear inclusions, focal necrosis of the ventricular koilin layer, and degeneration of the ventricular glandular epithelium with intranuclear inclusions. Immunohistochemically, intranuclear inclusions in the liver, pancreas, and ventriculi were stained positively against group I avian adenovirus (GIAAV) antigens. Ultrastructurally, 67-nm diameter viral particles were present in intranuclear inclusions. Virologically, serotype 4 of GIAAV was isolated from the liver, heart, and kidney of affected chickens. The pathologic changes of the present cases differ from previous cases of HPS; therefore, the present strain of GIAAV may have different pathogenicity for chickens than the previous virus strain of HPS.
Keywords
Hydropericardium syndrome (HPS) in broiler chicks was first reported in Pakistan.3 Affected chicks died suddenly without apparent clinical signs. The most characteristic gross change was hydropericardium. This disease was reported subsequently in Iraq,2 India,12 Mexico,8 Ecuador,5 Japan,1 and Chile.14 Chicks affected with HPS had inclusion body hepatitis and serotype 4 of group I avian adenovirus (GIAAV) was isolated from livers.1,5 GIAAV could experimentally produce mortality with HPS in broilers3 and specific-pathogen–free (SPF) chickens.5,10 The pathologic changes of natural HPS are hydropericardium with acute hepatocytic necrosis with intranuclear inclusion bodies in hepatocytes.1–3,10 No reports have been made of pancreatic or ventricular (gizzard) lesions by GIAAV in natural HPS, although intranuclear inclusions of GIAAV formed in the pancreas and ventriculi of the chickens experimentally inoculated with GIAAV.10 This paper describes the pathology of HPS with pancreatic necrosis and ventricular erosions in broiler chickens.
The disease occurred in Kagawa, Japan, in 2000. In a flock (4,000 birds) of a broiler farm rearing 13,000 birds, mortality increased after 13 days of age. By 19 days of age, 400 birds had died and the mortality rate of the flock had reached 10% cumulatively. Seven 19-day-old chicks, including four live and three dead ones, were examined pathologically and virologically.
Portions of the liver, heart, spleen, kidney, lung, trachea, cloacal bursa, ventriculus, proventriculus, pancreas, duodenum, jejunum, ileum, cecum, peripheral nerve, and brain were removed each chick after a postmortem examination and were fixed with 10% buffered formalin. Sections of organs were stained with hematoxylin and eosin. The streptoavidin–biotin–peroxidase (SAB) immunoperoxidase technique was used to detect GIAAV antigen in the formalin-fixed, paraffin-embedded organs. The SAB kit (Dako, Japan) was used according to the manufacturer's instructions. The primary antibody was rabbit antiserum against the mixed antigens containing serotypes 1, 4, and 8 of GIAAV.10 The serum was used at a 1:4,000 dilution in 0.01 M phosphate-buffered saline solution (pH 7.6). After SAB staining, the sections were counterstained with methyl green. Livers treated with phosphate-buffered formalin were fixed with 1% osmium tetroxide, dehydrated in alcohol, and embedded in epoxy resin. Ultrathin sections were stained with saturated alcoholic uranyl acetate and Reynold's lead citrate and examined with a transmission electron microscope. Homogenates10 of the samples (hearts, livers, and kidneys) were inoculated into a cell culture of chicken kidney (CK) cells4 and MSB1 cells.10 After cloning,4 GIAAV isolates were serotyped by a cross-neutralization test.7
Hydropericardium (Fig. 1), the characteristic gross lesion of HPS, was present in four chickens. The volume of pericardial fluid was 2–15 ml. Pinpoint white foci were scattered in the pancreas of four chickens. Ventricular erosions were present in four chickens. The cloacal bursa was normal in size (10–13 cm in diameter) except in one bird (7 cm in diameter). The bone marrow was red in color. Multifocal necrosis of the hepatocytes was scattered in the livers. Histologically, intranuclear inclusions were present in the degenerative hepatocytes around the necrotic foci or in the degenerative hepatocytes scattered diffusely. The inclusions were mostly basophilic type, and some were basophilic or eosinophilic in appearance with a clear halo. Some of the hepatocytes with inclusions were swollen to several times the size of unaffected hepatocytes. Many macrophages with yellow pigments were observed in the sinusoids of the livers of a few chickens. Multifocal necrosis of the pancreatic acinar cells (Fig. 2) and infiltration of macrophages and lymphocytes in the necrotic foci had occurred. Some of the degenerative cells around necrotic foci had intranuclear inclusions (Fig. 2). Glandular epithelium in the ventriculi showed degeneration, with intranuclear inclusions and necrosis. Subsequent reduction and loss of koilin (keratinoid) substance were followed by erosion with an associated bacterial infection (Fig. 3). Desquamated epithelial cells that had intranuclear inclusions were seen in the degenerative koilin layer. Spleens had a marked increase of macrophages, fragmentation of erythrocytes, deposition of yellow pigment in splenic sinuses, proliferation of the reticular cells around sheathed arteries, and lymphocyte depletion from lymphoid follicles. Rarely, intranuclear inclusions in the tubular epithelium without inflammation changes were seen in the kidneys. Mild serous exudation was seen in the epicardium in the hearts of all chickens. No lesions were present in the myocardium. Many macrophages with yellow pigment were present between the air capillaries in the lungs, and consequently, the lumens of the air capillaries were narrow. Pigment-laden macrophages were seen in the blood vessels. Intranuclear inclusions were observed in intestinal epithelial cells, and rarely in the goblet cells of the duodenum, jejunum, ileum, and cecum. The inclusions were observed in the epithelium from the glands to villous tips. Desquamated cells with intranuclear inclusions occasionally were seen in the lumens. No significant lesions were found in the cloacal bursa except for mild depletion of lymphocytes in the follicles. The bone marrow had typical hematopoiesis.
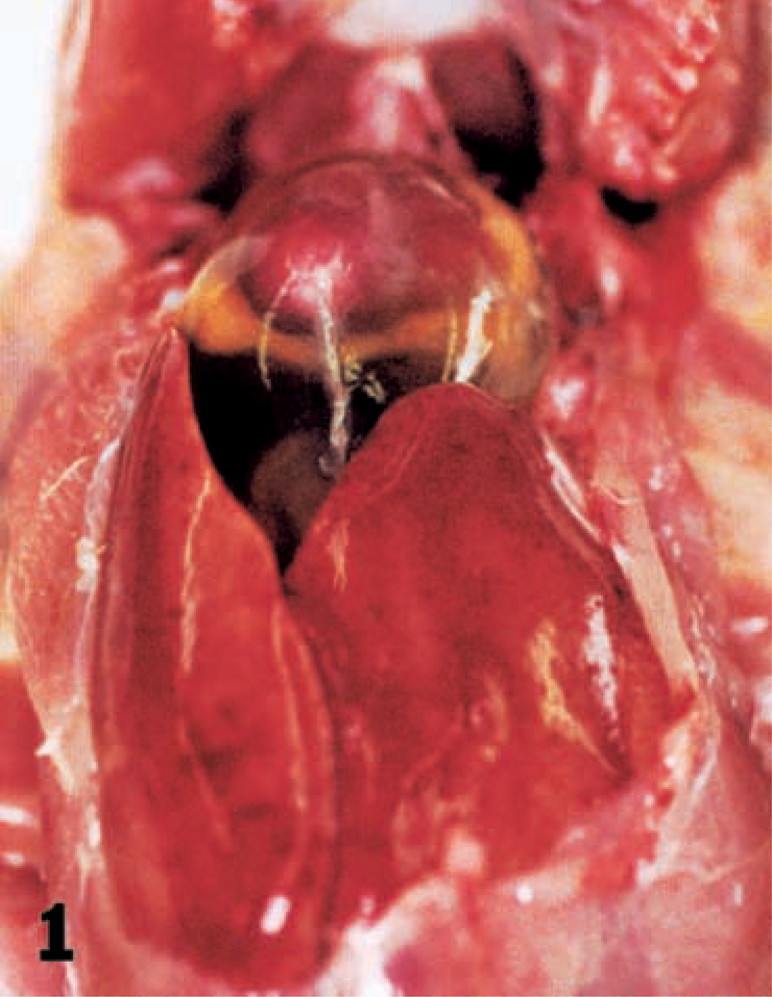
Hydropericardium in a chicken. Excessive fluid is seen in the pericardial sac. Liver is swollen.
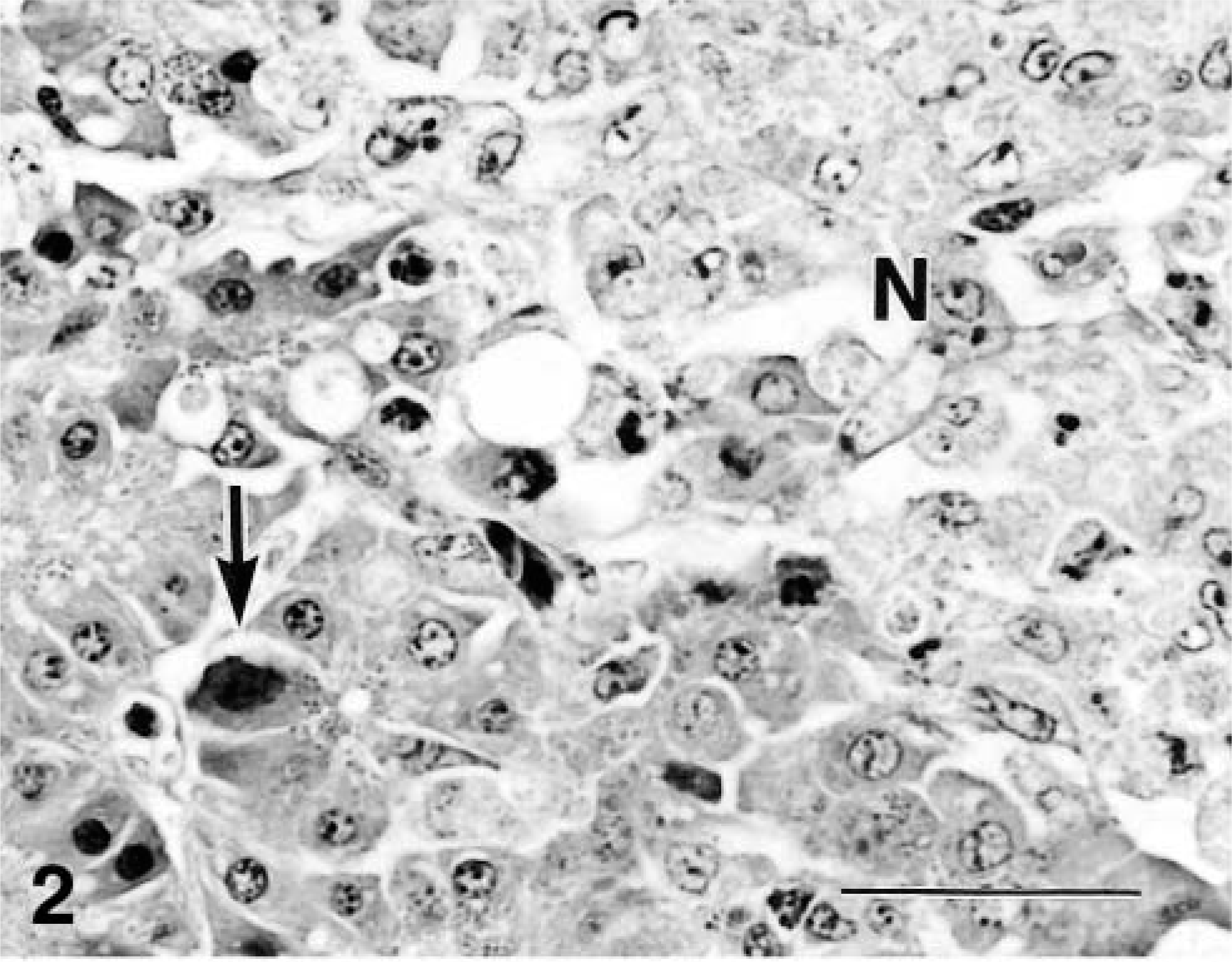
Pancreas; chicken. Intranuclear inclusions (arrow) of degenerative acinar cells of pancreas is seen around necrotic area of glands (N). Bar = 33 μm.
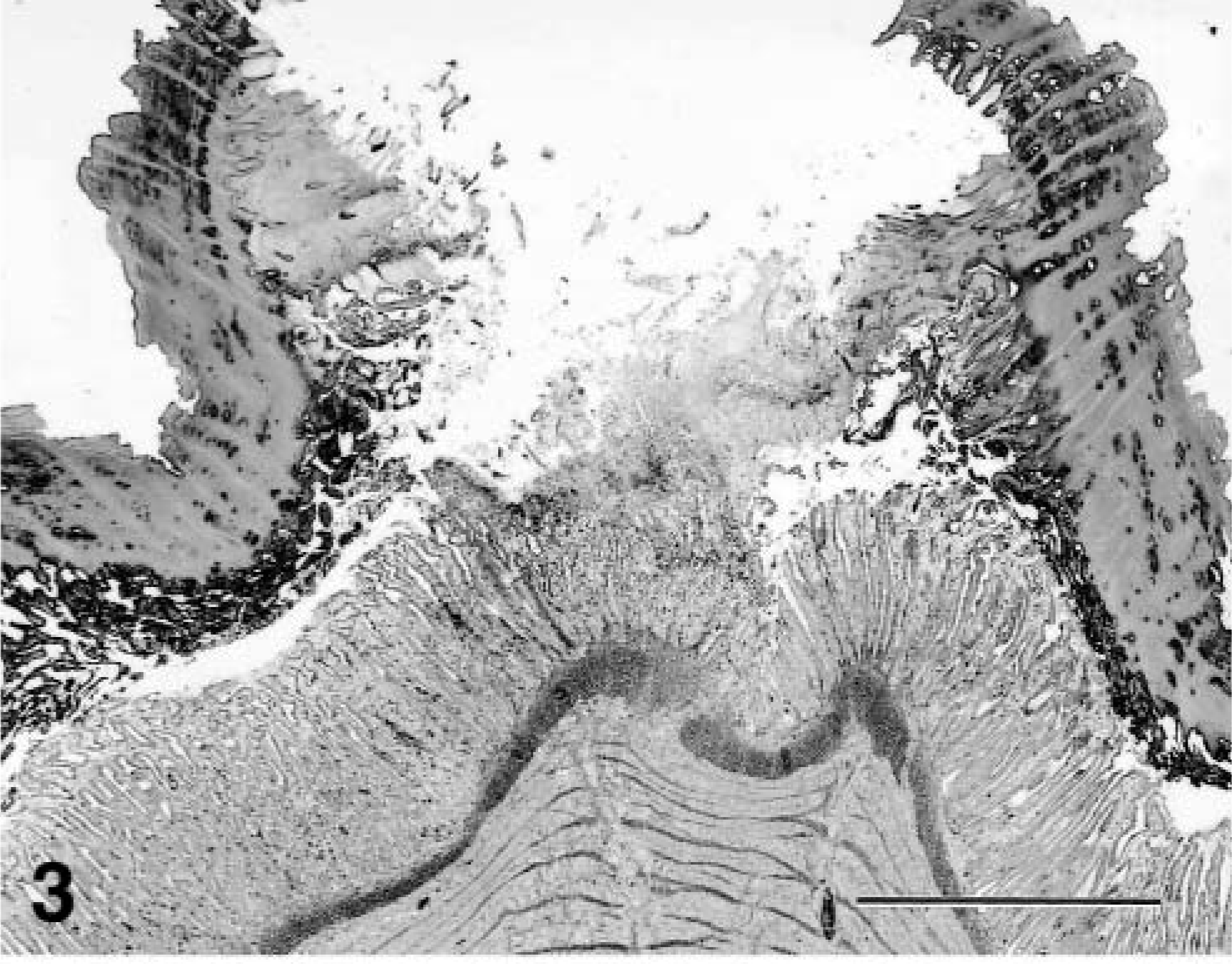
Gizzard; chicken. Erosion of gizzard. Necrosis and depletion of koilin layer with necrosis of glandular epithelium in ventriculus. HE. Bar = 800 μm.
Immunohistochemically, the GIAAV antigens were distributed in the intranuclear inclusions of the affected cells in the liver (Fig. 4), pancreas, ventriculi, duodenum, jejunum, ileum, cecum, and kidney. The amount of antigen paralleled the number of inclusions. The most prominent reactions for GIAAV by immunoperoxidase staining were seen in the liver. Nuclei of pulmonary air capillary epithelial cells were rarely positive for GIAAV. Ultrastracturally, swollen nuclei of hepatocytes with intranuclear inclusions were filled with many viral particles of hepatocytes (Fig. 5). Mature and immature viral particles (mean 67 nm in diameter) were present in hepatocyte nuclei. Virologically, in CK cell culture of homogenized samples of livers, hearts, and kidneys, GIAAV was isolated from all chickens. The cloned isolate (Kagawa/2000HPS) was identified as serotype 4 by a cross-neutralizing test. Neither infectious bursal disease virus (IBDV) nor chicken anemia virus (CAV) were isolated from these organs by CK or MSB1 cell culture.
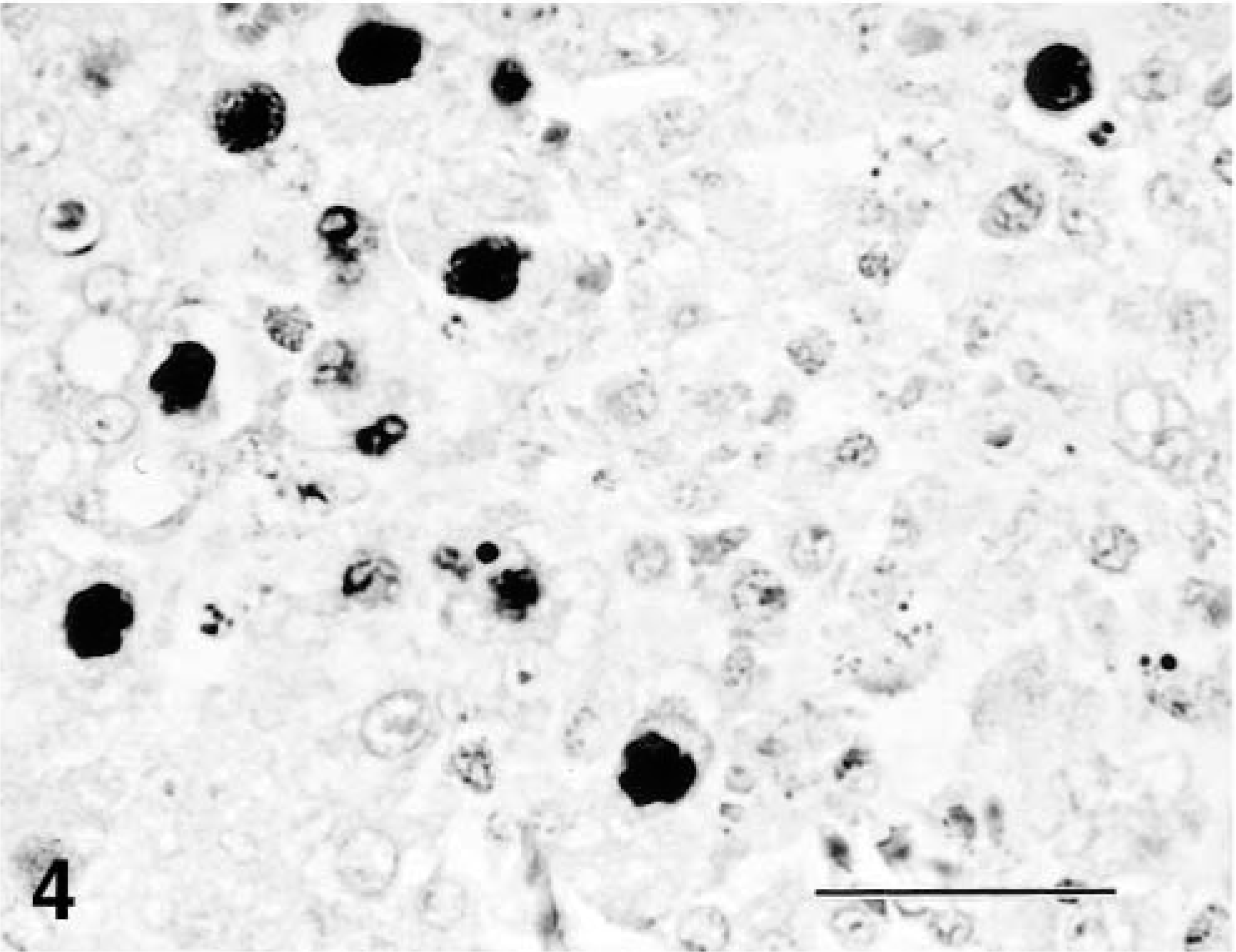
Immunohistochemistry of intranuclear inclusions of hepatocytes from a chicken. Many intranuclear inclusion bodies of hepatocytes exhibit a positive reaction for GIAAV. SAB immunoperoxidase and hematoxylin staining. Bar = 33 μm.
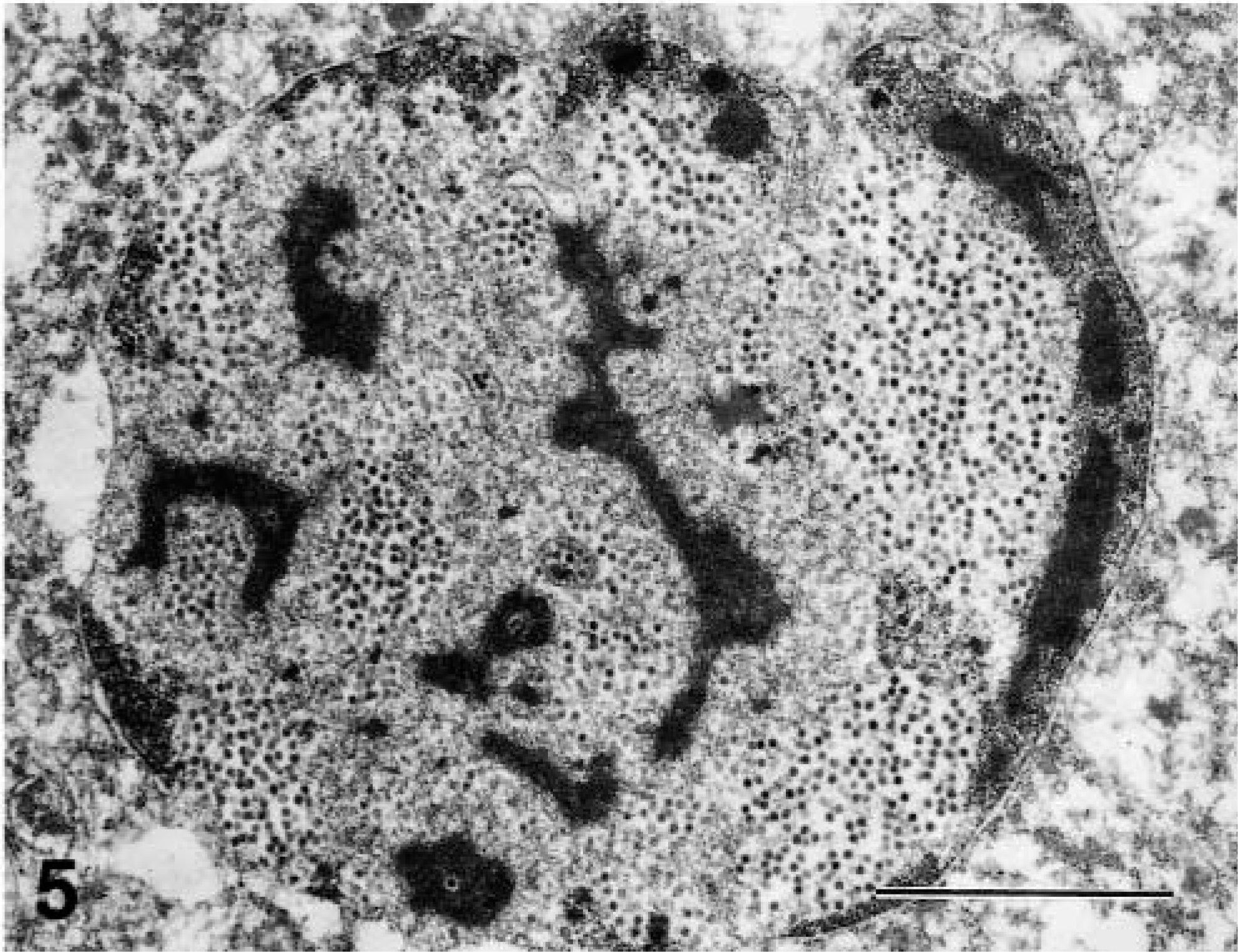
Ultrastructure of intranuclear inclusions of hepatocyte of a chicken. Swollen nucleus is filled with many viral particles. Bar = 2.5 μm.
Because hydropericardium is the most characteristic gross lesion, this disease has been designated as HPS. However, histologic changes in the heart of reported cases were scarce, except for mild to moderate edema of the epicardium. No GIAAV antigens could be detected in our study in the heart by immunohistochemistry. The most prominent change was acute hepatic necrosis with intranuclear inclusions of hepatocytes. A close correlation was found between hydropericardium and hepatic necrosis in experimental10 and natural cases.1 Acute hepatic necrosis may cause marked hepatic circulatory failure. This hepatic circulatory failure may predispose to heart circulatory failure and then hydropericardium. The acute hepatic necrosis results in acute heart failure, and ultimately death. This hypothesis may be supported by the pathologic findings in ascites syndrome in broilers.9 The chickens affected with hydropericardium consistently had hepatic necrosis.9 Immunohistochemically, large amounts of antigens were stained in the livers, pancreas, and ventriculi with marked lesions, but not in the hearts.
In GIAAV infections of chickens, classical inclusion body hepatitis (IBH) with hypoplastic anemia,6 HPS, 10 pancreatitis,13 and ventricular erosions are present.13 GIAAV from classical IBH belongs to serotypes 4, 5, and 8.6 HPS was induced by serotype 4 GIAAV.5,10,14 The present virus isolate is also serotype 4 of GIAAV. Although serotypes of GIAAV are not always consistent with virus virulence, the prevalent GIAAV strain causing HPS may be the same serotype, serotype 4. Necrotizing pancreatitis and ventricular erosions were reported in young layers infected with IBDV or CAV.13 Serotype 8 GIAAV was isolated from these cases. These cases did not have hepatic necrosis or hydropericardium, although the main lesions of the cases present here were hepatic necrosis and hydropericardium. The HPS strain can experimentally induce mortality and hydropericardium not only in SPF chicks but also in SPF hens.10 GIAAV from IBH can also induce mortality and hydropericardium in 1-day-old SPF chickens, but cannot produce mortality or hydropericardium in 3-week-old SPF chickens.11 The lesions caused by HPS strains and IBH strains are essentially the same, although the virulence of HPS and IBH strains differ. In IBH, infectious agents such as IBDV or CAV enhance the pathogenicity of the IBH adenovirus.6 However, in HPS no evidence was found of any association of IBDV or CAV. This suggests that GIAAV from HPS is more virulent than IBH. The previous reports of natural HPS1–3,12,14 referred to hepatocytic necrosis with intranuclear inclusion bodies of hepatocytes, but did not report pancreatic necrosis or gizzard erosion. This virus strain may have different pathogenicity from previous virus strains of HPS.6 A reproductive experiment with the HPS strain of the present case and a molecular epidemiologic investigation of the virus strain are necessary for clarification.
Footnotes
Acknowledgements
We thank Dr. Y. Ando and Mr. T. Fujisawa for providing photographs, Mr. M. Kobayashi and Miss Megumi Shimada for histologic and immunohistochemical assistance, and Miss Asuka Saito for various technical assistances.
