Abstract
Five cases of exfoliative dermatitis in cats were presented from 1996 to 2002 in which a feline thymoma was found by postmortem or postsurgical examination. Besides abundant exfoliation of keratin squames and layers, the histologic picture of the skin revealed a similar pattern of interface dermatitis with predominantly CD3+ lymphocytes and fewer mast cells and plasma cells. In the epidermal basal layer a hydropic degeneration of keratinocytes was present. In all cases an infundibular lymphocytic mural folliculitis and absence of or drastic decrease in the number of sebaceous glands occurred. In addition to the so far described cell-poor type, we also found examples of a cell-rich skin lesion. Together with the clinical observation of generalized exfoliative dermatitis, the histologic pattern of this dermatitis was suggestive of an underlying thymoma. The pathogenesis of this skin disease in association with thymic neoplasia remains obscure, and our results contradict the hypothesis of production of autoantibodies that cross-react with epithelial antigens. The morphology of the thymomas and CD3 expression of the thymocytes varied and did not seem to have an impact on the dermal lesions.
Exfoliative dermatitis is a clinical entity characterized by generalized severe desquamation with or without erythema. In cats the differential diagnosis of this rare finding includes systemic lupus erythematosus, drug eruptions, epitheliotropic T-cell lymphoma, erythema multiforme, cheyletiellosis, demodicosis, Malassezia dermatitis, dermatophytosis, feline immunodeficiency virus (FIV) or feline leukemia virus (FeLV) dermatitis, parapsoriasis, sebaceous adenitis, and paraneoplastic syndrome caused by thymoma. 15,18
Little is known regarding the latter, and the condition lacks a detailed histologic characterization. 1,3–5,7,13,16,18 The histologic changes that are described in the literature focus on parakeratosis and a cell-poor interface dermatitis. 18
In humans exfoliative erythroderma is described as a cutaneous manifestation of a paraneoplastic syndrome, but it is also associated with a number of different cutaneous diseases. 6 So far, there is no accurate explanation for the coexistence of malignancies and cutaneous lesions, but a number of mechanisms have been suggested. 8 These include production or depletion of a specific substance by the tumor 11 or an immune-mediated mechanism that leads to the activation of autoreactive cytotoxic T cells, a process similar to that found in graft-versus-host disease. 18 Other theories imply an aberrant antitumor response with production of autoantibodies that cross-react with epithelial antigens that might be induced by dysregulated cytokine production of tumor cells, known to promote B-cell differentiation and immunoglobulin production in vitro. 14 In some cases of paraneoplastic syndrome in humans, an association between a specific cancer and a specific dermatitis is described. 2 In cats an example of paraneoplastic dermatopathy is ventral alopecia and “glistening skin,” which has been observed in association with pancreatic carcinoma or bile duct adenocarcinoma. 12,17
A similar correlation seems to occur in cats, where thymomas are typically associated with a nonpruritic, generalized exfoliative dermatitis.
In this study five cases of feline exfoliative dermatitis are reviewed in which necropsy (cat Nos. 1–4) or thymectomy (cat No. 5) confirmed the presumptive diagnosis of thymoma. Skin biopsy samples and thymic samples from routine diagnostic service were formalin fixed, paraffin embedded, and characterized by histochemistry, immunohistochemistry, and immunofluorescence. Clinically, all cats showed generalized desquamation, alopecia on the body, and multifocal crusts (especially on the head, as demonstrated in Fig. 1). In addition, lethargy, anorexia, and weight loss for several weeks had been reported. The cats were nonpuritic or only mildly pruritic, and axillary (cat No. 2) or ventral (cat No. 3) erythema could be found. Because skin scrapings and Wood's lamp examination were negative in all cases and prednisolone (cat Nos. 2, 4 and 5), ivermectin (cat No. 2), enrofloxacin (cat No. 4), or amoxicillin (cat No. 5) treatment did not improve the animals' condition, randomly distributed punch biopsy samples (4–6 mm) of skin lesions were taken for histopathologic examination. On comparing these cases similar morphologic and immunohistochemical results could be detected (Table 1). In the epidermis an orthokeratotic and parakeratotic hyperkeratosis with extensive desquamation was visible (Fig. 2). Additional findings in all cases included bacterial colonization, mild to moderate acanthosis, exocytosis of CD3+ lymphocytes (anti-CD3, M7254, Dako, Glostrup, Denmark), hydropic degeneration of basal keratinocytes, and small numbers of dying (“apoptotic”) cells. In cat No. 5 a focal intraepidermal pustule was found.
Histopathologic characterization of skin lesions from cat Nos. 1–5.
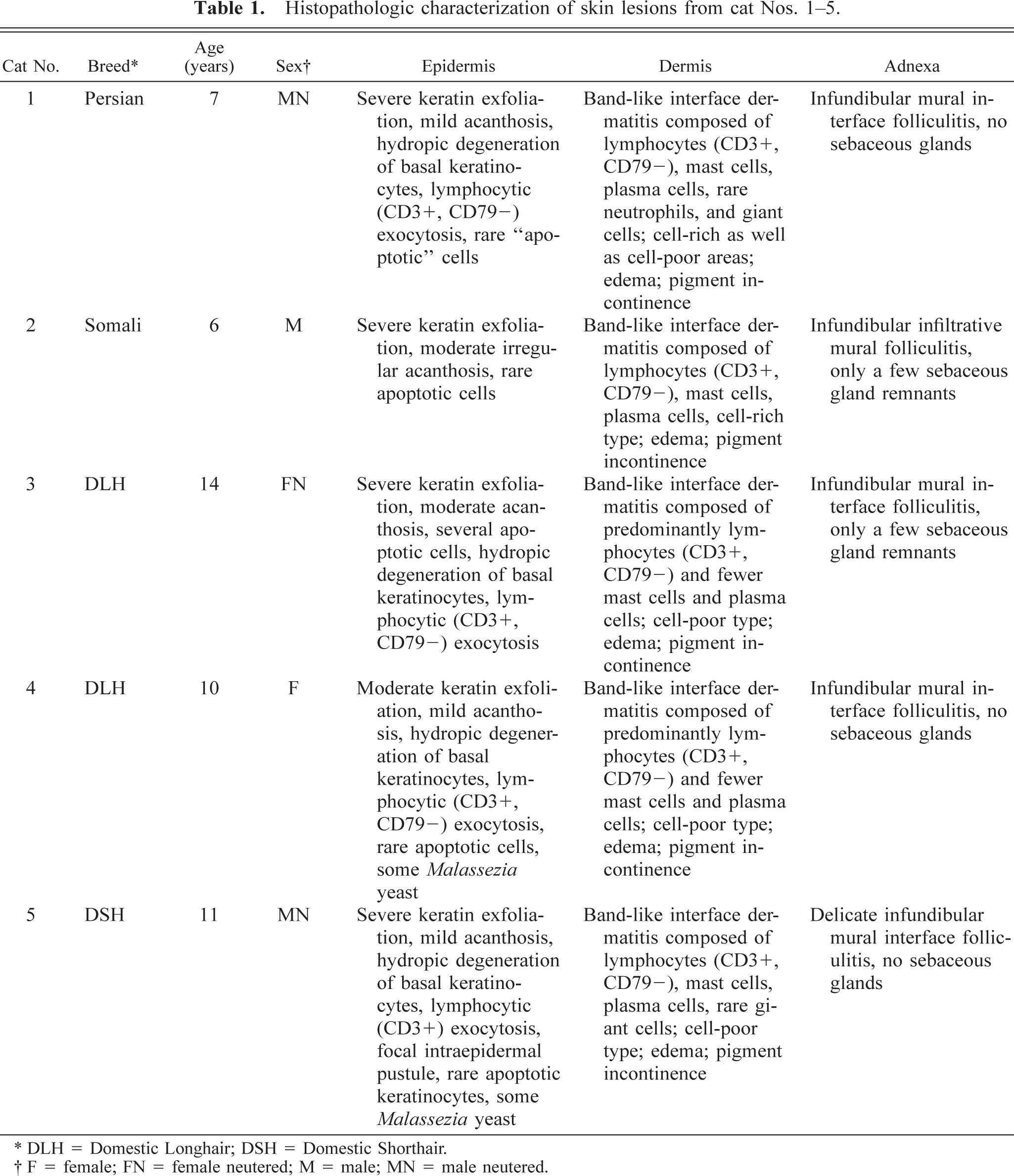
∗ DLH = Domestic Longhair; DSH = Domestic Shorthair.
† F = female; FN = female neutered; M = male; MN = male neutered.
Head; cat No. 5. Note the typical large adherent scales. Bar = 1.5 cm.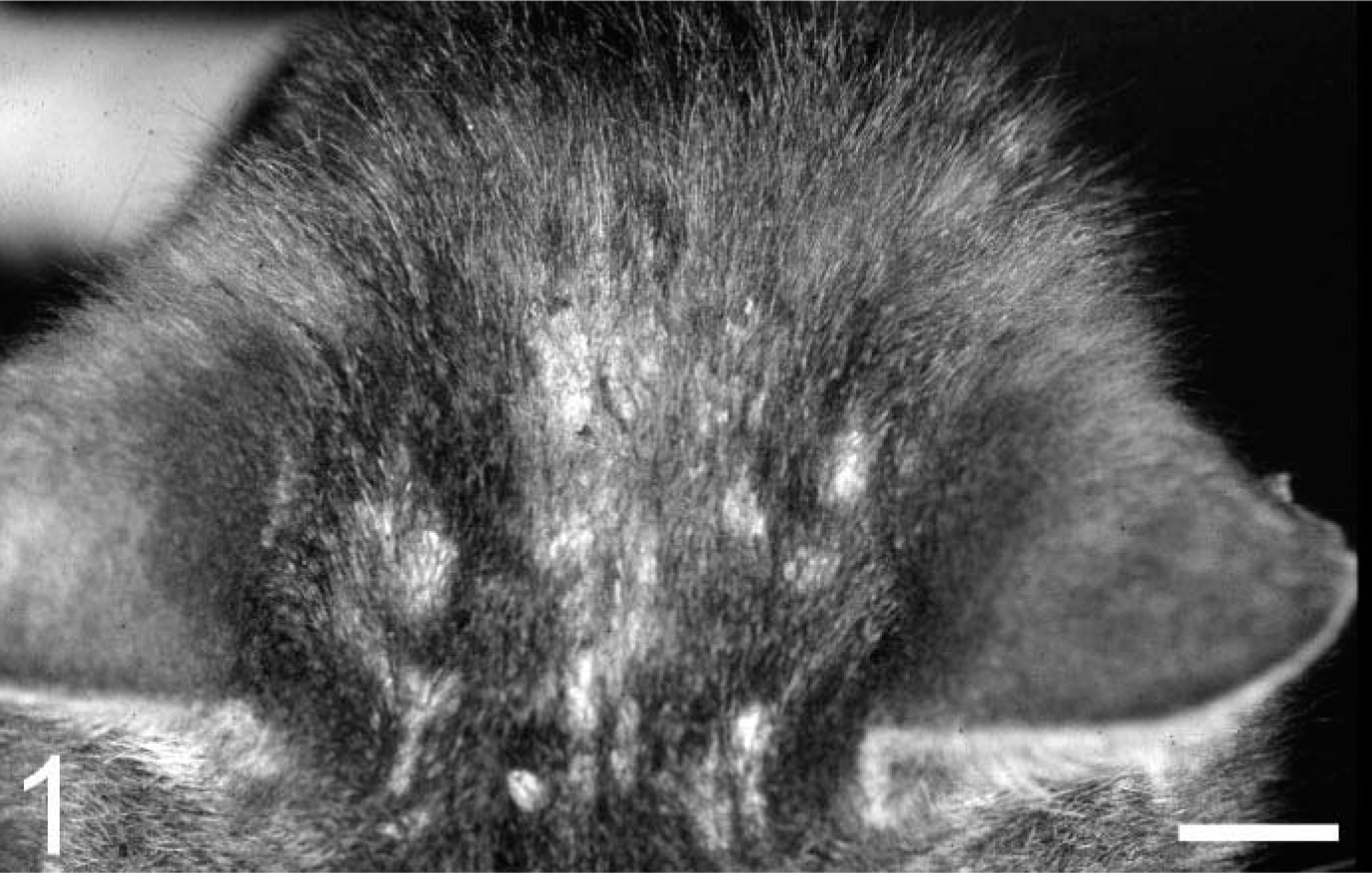
Skin; cat No. 1. Note the hyperkeratosis with extensive desquamation and cell-poor interface dermatitis. HE. Bar = 150 µm.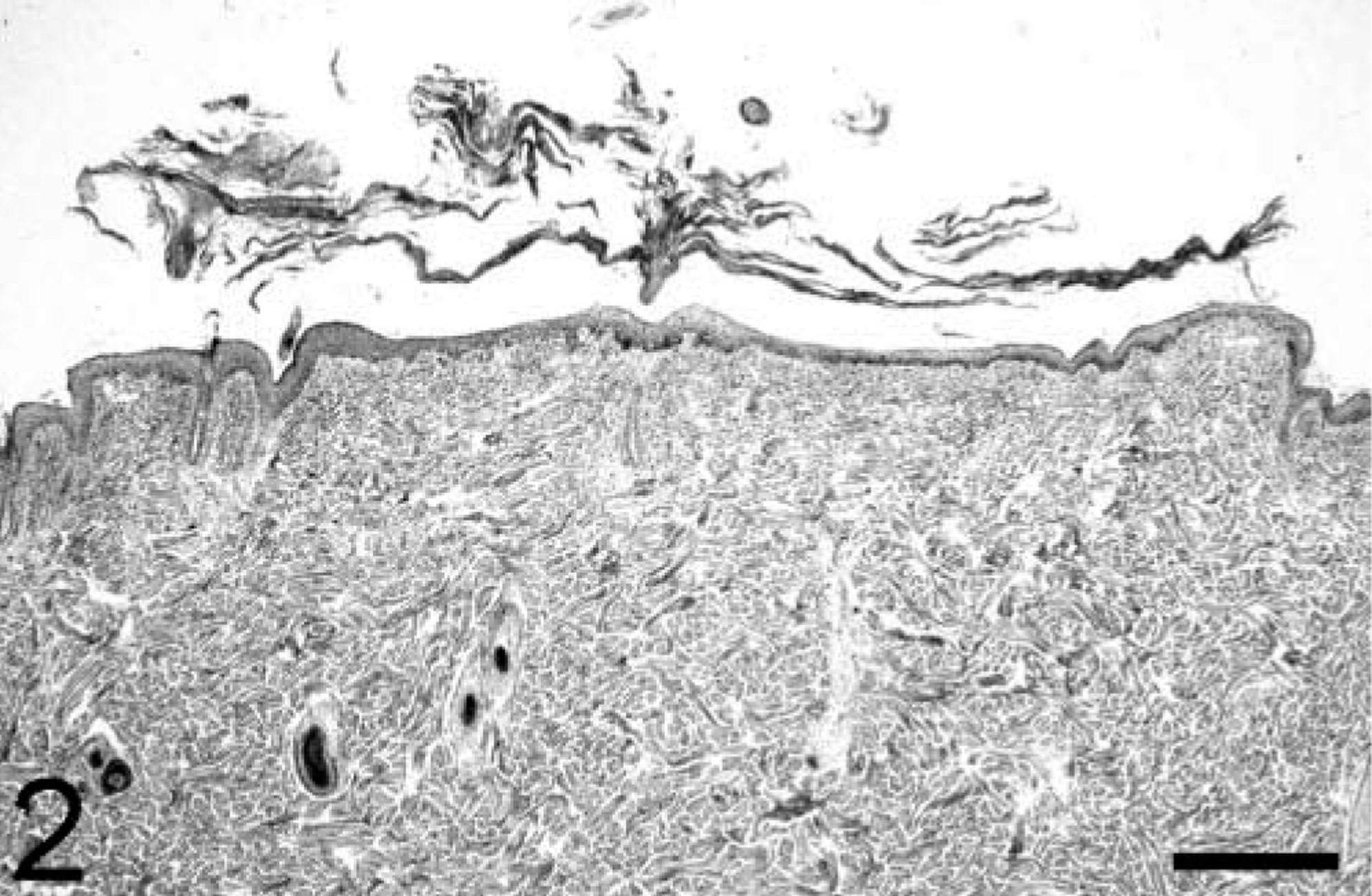
In the dermis the most striking lesion was the interface dermatitis composed of mainly CD3+ lymphocytes and varying amounts of mast cells and plasma cells. Interestingly, in addition to an expected cell-poor infiltration (cat Nos. 1, 3–5), a cell-rich interface dermatitis could be observed in cat Nos. 1 and 2 (Fig. 3). Further findings included pigment incontinence, edema, and rare giant cells (cat Nos. 1 and 4) or neutrophils (cat No. 1).
Skin; cat No. 2. Note the cell-rich interface dermatitis composed of lymphocytes, mast cells, and plasma cells. HE. Bar = 75 µm.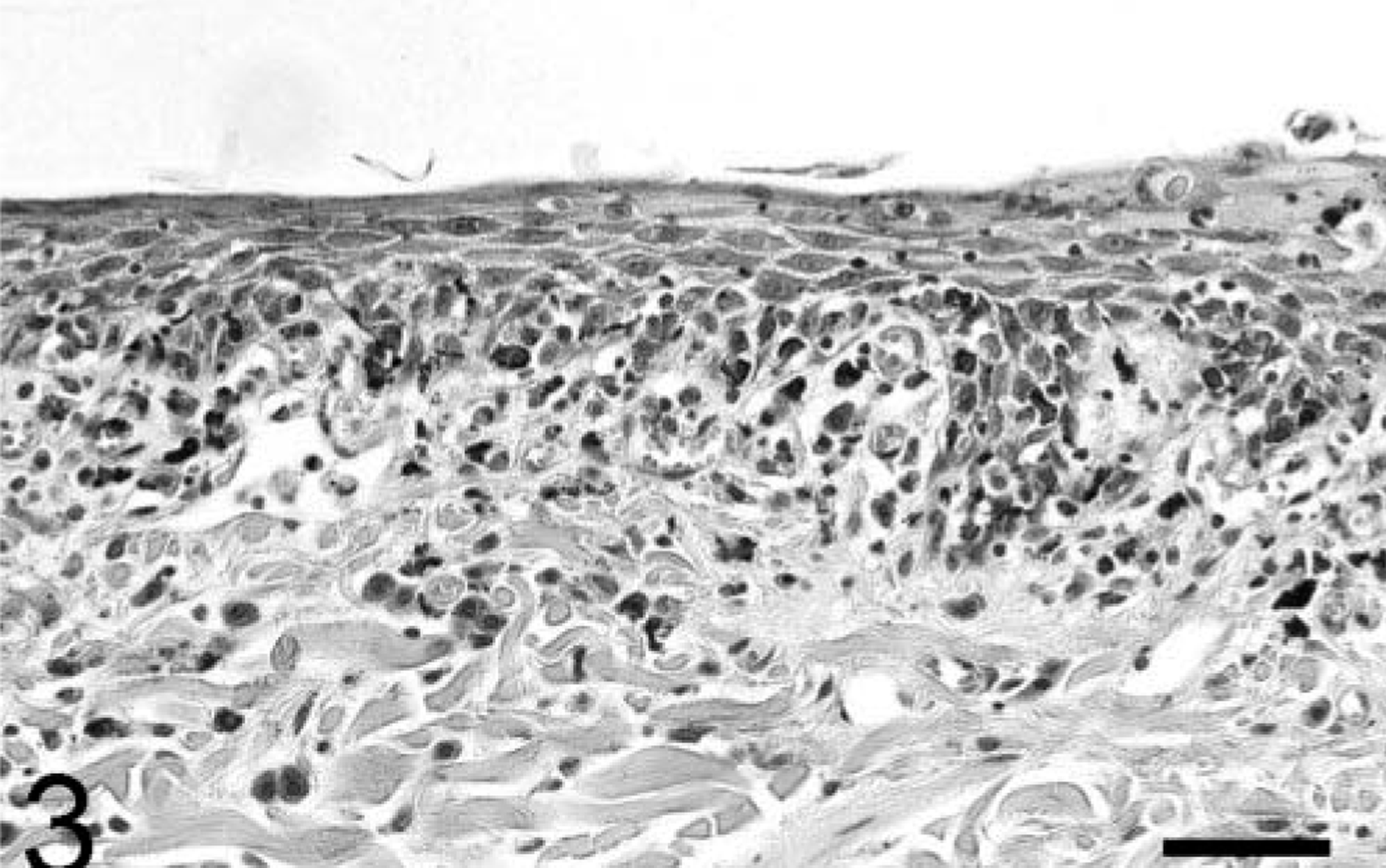
Another common feature of the cases investigated was the presence of an infundibular lymphocytic interface or infiltrative mural folliculitis in many hair follicles. Furthermore, no sebaceous glands or only a few remnants were detectable (Fig. 4).
Skin; cat No. 4. Mural folliculitis with loss of sebaceous glands. HE. Bar = 100 µm.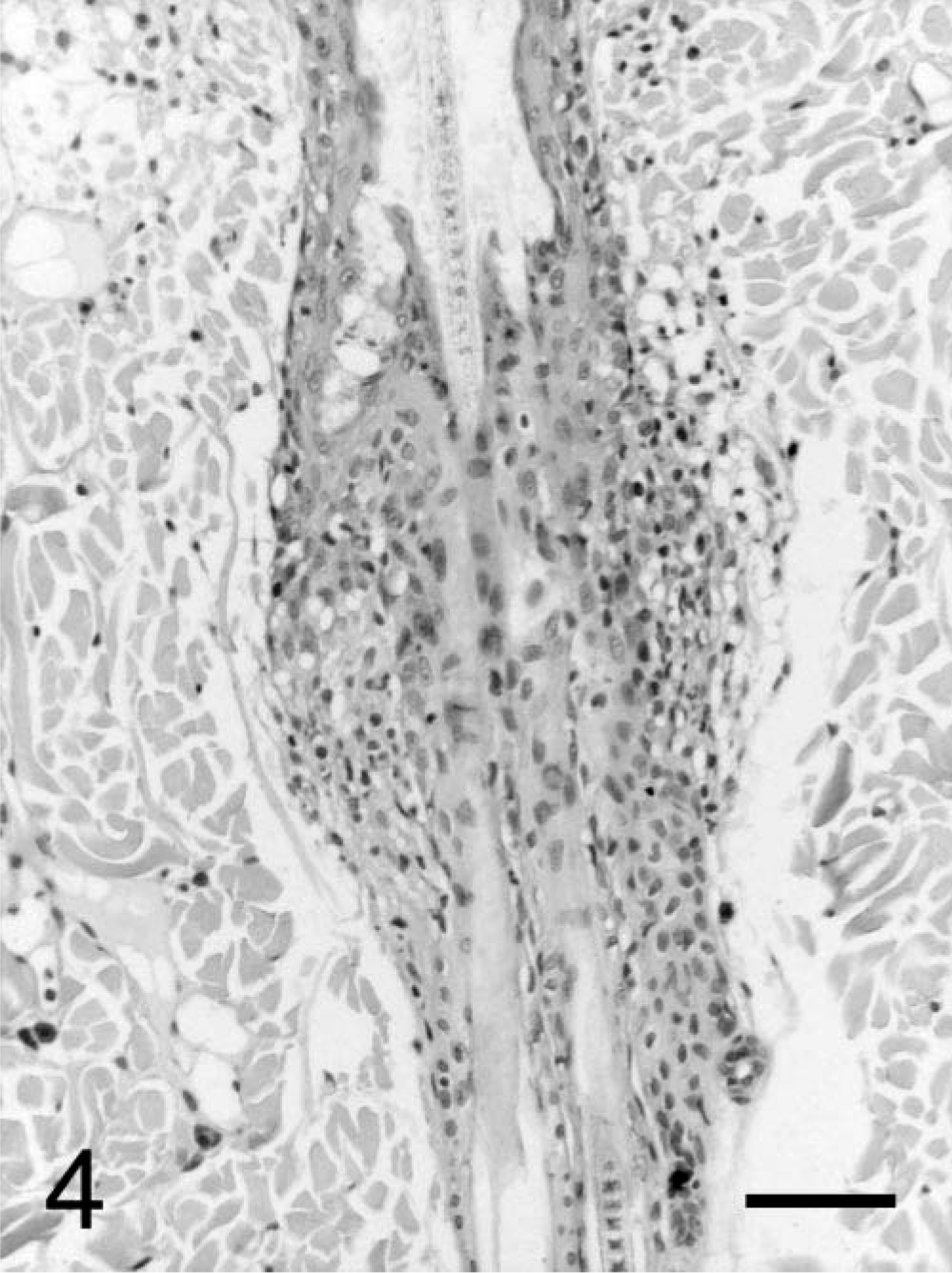
In all cases immunofluorescence studies using a fluorescein isothiocyanate–labeled antibody against feline IgG (F4262, Sigma-Aldrich, Division of Fluka Chemie, Buchs, Switzerland) did not reveal the presence of cross-reacting IgG autoantibodies in the skin (data not shown).
Because of constant aggravation of the cats' condition and in accordance with the owner's request, four cats were euthanatized and further examined by necropsy. In cat No. 5 surgical removal of a mediastinal mass was performed. In all cases a single precardial mass, which was histopathologically diagnosed as thymoma, was identified. The FeLV and FIV status was tested in cat Nos. 4 and 5, with a negative result. To investigate an underlying virus infection in cat Nos. 1–3, we isolated DNA from formalin-fixed, paraffin-embedded thymoma tissue using standard phenol-chloroform extraction (Fluka Chemie, Buchs, Switzerland). The purified DNA was analyzed by quantitative polymerase chain reaction (PCR) for incorporated gag (FIV) and U3 long-terminal repeat (FeLV) sequences at the Veterinärmedizinisches Labor (Vet-Suisse Faculty, Zurich, Switzerland), but no viral sequence could be amplified in any of the three cases. Necropsy of cat No. 4 was performed in a different laboratory, and, unfortunately, we did not have access to the thymoma slides of this case.
The thymic neoplasms revealed a proliferation of epithelial cells (cytokeratin positive, Z0622, Dako) with varying amounts of lymphocytes (both CD3 positive and CD3 negative) and mast cells (Table 2). In all tumors the mitotic index of the epithelial cells was low. In cat No. 2 multifocal coagulation necrosis was present. The morphology of the epithelial cells included sheets of polygonal cells and cords of cuboidal cells. The variable histologic appearance of the thymomas reported here is in accord with the feline thymoma literature, 1,3–5,7,9,13,16 although in one study 9 the anti-cytokeratin antibody (Z0622, Dako) stained Hassal's corpuscles. After thymectomy in cat No. 5, the cat fully recovered, and the exfoliative dermatitis and alopecia disappeared.
Histopathologic characterization of thymic neoplasms.

The results of this study imply that different morphologic types of thymoma cause a similar histologic entity of exfoliative dermatitis. Besides the cell-poor interface dermatitis described above, we found cases with cell-rich areas. In addition, infundibular lymphocytic interface and infiltrative mural folliculitis and loss of sebaceous glands seem to be typical findings in this paraneoplastic disease. Thus, together with the clinical observation of generalized skin exfoliation, this histologic pattern seems to be typically associated with thymoma. In a recent study 10 the presence of Malassezia yeast in feline skin biopsy samples was found to be suggestive of internal neoplasia. In this study yeasts were detected only in cat Nos. 4 and 5. Thus, Malassezia infection does not seem to be a typical feature of thymoma-associated dermatitis.
As long as the underlying immunologic and molecular mechanisms of exfoliative dermatitis in association with thymoma remain to be elucidated, the precise cause-effect relationship of this entity is not fully proven. However, surgical removal of the thymoma of cat No. 5 resulted in regression of the dermatologic problems, which strongly suggests that this neoplasm is involved in the development of the skin lesions described. The absence of immunoglobins in the skin contradicts the theory of an aberrant antibody production. The presence of CD3+ lymphocytes favors a T-cell–mediated process that might be caused by abnormal antigen presentation of neoplastic thymic epithelial cells. Further studies should address the hypothesis of a potential relationship between the CD3-positive T cells within the thymoma and those in the skin, which would include the clonality determination of these cells.
Footnotes
Acknowledgements
We are grateful to Dr. Michael Goldschmidt, Department of Veterinary Pathology, University of Pennsylvania, Philadelphia, for sharing the tissues of cat No. 5 and Dr. Catherine Nägeli for submission of cat No. 3. We greatly appreciate the review of the thymus slides by Dr. Tom van Winkle, Department of Veterinary Pathology, University of Pennsylvania. We thank Prof. Hans Lutz and his team from the Veterinärmedizinisches Labor for performing the quantitative PCR analysis. We give special thanks to Ursula Forster for technical assistance.
