Abstract
Two cases of feline thymoma with amyloid deposition were encountered between 1982 and 2010. Neoplastic cells were separated by abundant, pale eosinophilic, homogeneous material that was congophilic and birefringent. Ultrastructurally, the neoplastic cells were connected by desmosomes, and the extracellular deposits were composed of nonbranching, hollow-cored fibrils, 8–10 nm in diameter. In the case with sufficient archived tissue for additional sections, the amyloid remained congophilic following potassium permanganate incubation, and the neoplastic cells were immunoreactive for pancytokeratin. The histologic, histochemical, ultrastructural, and immunohistochemical features of both neoplasms are consistent with epithelial-predominant thymoma with the unusual feature of intratumoral amyloid deposition. The affinity of the amyloid for Congo red following potassium permanganate incubation is consistent with non-AA amyloid. The ultrastructural findings were consistent with amyloid production by the neoplastic epithelial cells.
Thymomas are derived from thymic epithelium with variable benign lymphocytic infiltration and thus are classified as lymphocyte-predominant, epithelial-predominant, or mixed. 6 Neoplastic thymic epithelial cells are often elongated or spindle-shaped with poorly defined cytoplasmic margins and are cytokeratin positive. 6 Intratumoral mast cells have been reported in 45% (5/11) 3 to 75% (3/4) 10 of feline thymomas and 20% (3/15) 1 to 85% (17/20) 2 of canine thymomas. Thymoma is uncommon in domestic animals and usually affects adult to aged animals, which often are presented with dyspnea, lethargy, coughing, anorexia, vomiting, regurgitation, or ventral cervical edema.1,6,12 Cats with thymoma frequently have a poorly compressible thorax and muffled cardiac sounds. 12 The purpose of this report is to describe pathologic findings in 2 feline thymomas with intratumoral amyloid deposition.
Case Histories
Case 1
An 11-year-old, spayed female, Himalayan cat was presented to the Lloyd Veterinary Medical Center at Iowa State University in 2010 with mild weight loss in the preceding month and a 2-day history of vomiting and retching. On physical examination, the patient had mild tachycardia and tachypnea with muffled heart and lung sounds and a noncompressible thorax. Radiographically, a soft tissue mass within the cranioventral aspect of the mediastinum displaced the trachea dorsally. With ultrasound, the mass was 7 cm × 5 cm × 7 cm, distinctly marginated, moderately hypoechoic, and heterogeneous, and it extended from the thoracic inlet to the diaphragm with an anechoic, 4.5 cm central cavitary space. Computed tomography further defined the margins of the mass, revealing intimate contact with the pericardium, great vessels, trachea, and esophagus and caudodorsal displacement of the heart. No communication between the mass and the great vessels was detected by injection of intravenous contrast.
Cytologically, ultrasound-guided fine needle aspirates from the mass were moderately cellular with a predominance of small lymphocytes, occasional medium-sized lymphocytes, rare macrophages, and low numbers of mast cells within a variable background of erythrocytes, cellular debris, and proteinic fluid. Cells with dysplastic or anaplastic features were not observed. Fluid from cystic areas within the mass was poorly cellular with rare small lymphocytes, macrophages, and nondegenerate neutrophils. Based on the cytologic features (small lymphocytes and mast cells) and the presence of cystic areas within the mass, 9 the presumptive cytologic diagnosis was lymphocyte-predominant thymoma.
At surgery, the mass was identified within the cranioventral mediastinal area encroaching on the heart base caudally and displacing the lungs dorsally, with the left side more severely affected than the right. The mass was removed via gradual retraction, ligation of tumor-associated vessels, and a combination of sharp and blunt dissection. On gross examination, the multilobular mass was firm and mottled pale tan to brown (Fig. 1 ). Samples for histopathology were fixed in 10% neutral buffered formalin for 24 hours, processed routinely, and embedded in paraffin. Tissue sections were cut at 5 μm and stained with hematoxylin and eosin (HE), Congo red, Gomori trichrome, Giemsa, and the periodic acid–Schiff reaction. An additional serial section and a control section of kidney with glomerular amyloidosis were pretreated with potassium permanganate prior to Congo red staining as described. 11 A sample of formalin-fixed tissue was processed routinely for electron microscopy; sections were contrasted with uranyl acetate and lead citrate. For immunohistochemistry, 3 μm sections were treated with 3% hydrogen peroxide to inhibit endogenous peroxidase and then pretreated with Tris-EDTA (for antibodies to cytokeratin, CD3, CD79a) or citrate buffer (for antibody to vimentin) in a steam bath for antigen retrieval. Immunohistochemistry was performed using an automated cell staining system (i6000, BioGenex Laboratories Inc., San Ramon, CA) with a commercial chromogen (NovaRED, Vector Laboratories Inc, Burlingame, CA) and hematoxylin counterstain. The primary antibodies were mouse monoclonal anti-cytokeratin (Dako North America Inc., Carpentaria, CA), mouse monoclonal anti-vimentin (Dako North America), rabbit polyclonal anti-CD3 (Dako North America), mouse monoclonal anti-CD79a (Dako North America), rabbit polyclonal anti-chromogranin A (Dako North America), or mouse monoclonal anti-neuron specific enolase (BioGenex Laboratories Inc, San Ramon, CA) with a commercial goat anti-mouse/anti-rabbit secondary antibody (MultiLink, BioGenex Laboratories) and horseradish peroxidase–streptavidin conjugate (Zymed, Invitrogen Corp, Carlsbad, CA). Appropriate positive control tissues were used for each antibody, and serial sections of the same tissues with omission of primary antibodies served as negative controls.
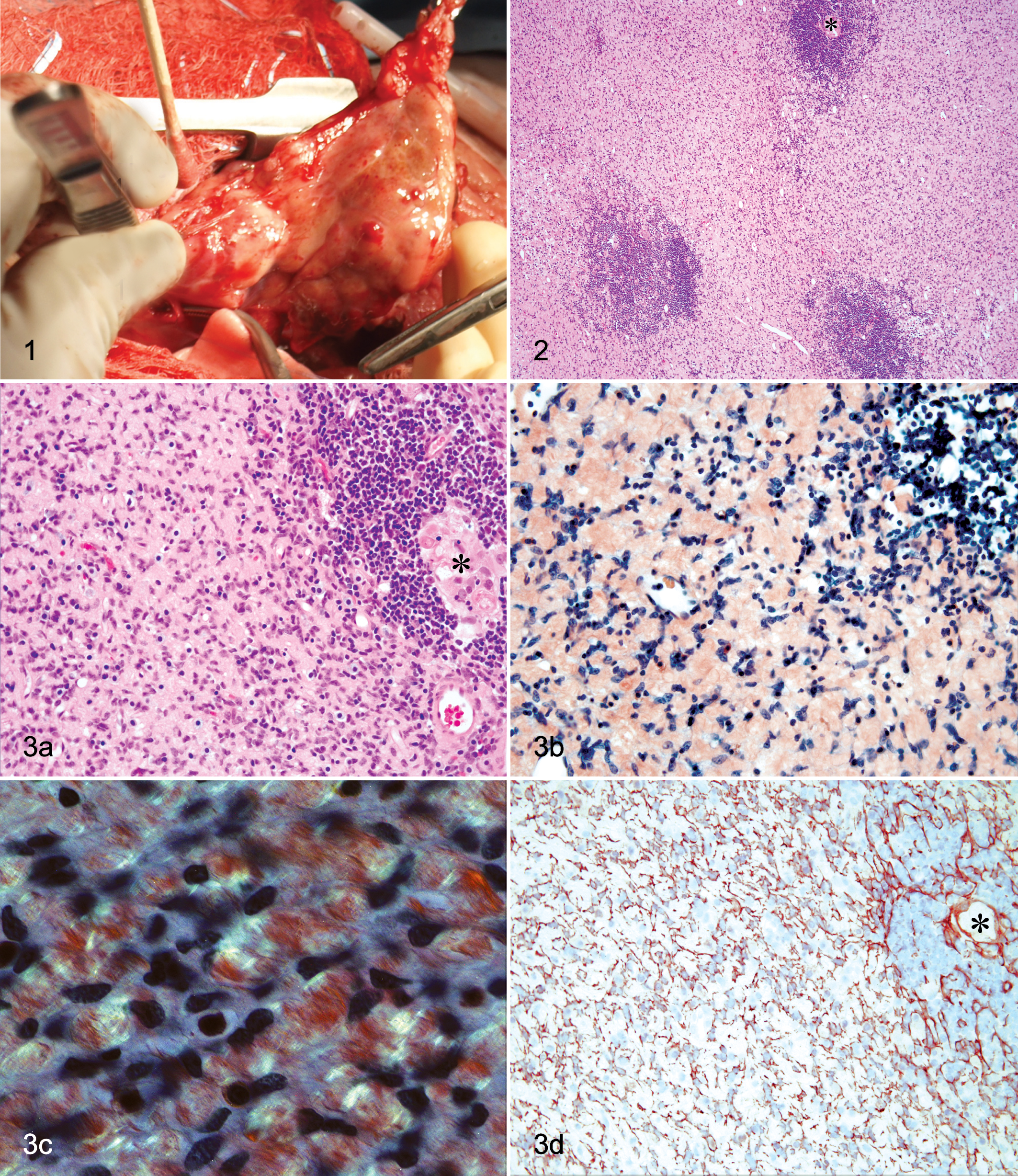
Histologically, native thymic tissue was replaced by a partially encapsulated, mildly infiltrative mass composed of solid sheets of pleomorphic, elongated to spindle-shaped cells with indistinct cell borders; variable amounts of homogeneous to vacuolated eosinophilic cytoplasm; and large, round to ovoid nuclei with finely stippled chromatin and variably prominent nucleoli. Mitotic figures were not observed in 20 high-power (400×) fields. Multifocal necrosis, hemorrhage, mineralization, and cystic spaces containing proteinic fluid were observed in the neoplastic tissue. One cyst was lined by cuboidal epithelium with apical cilia, consistent with a bronchogenic cyst. In all sections, neoplastic cells were separated by abundant, variably dense, pale eosinophilic, homogeneous to fibrillar extracellular matrix (Fig. 2). Infiltrates of small lymphocytes often surrounded structures consistent with Hassall’s corpuscles (Fig. 3a). The extracellular matrix stained red-orange with Congo red (Fig. 3b) and had apple-green birefringence under polarized light (Fig. 3c), consistent with amyloid. These intratumoral amyloid deposits remained congophilic following treatment with potassium permanganate, whereas glomerular amyloid deposits in the control tissue lost their affinity for Congo red. Additionally, the intratumoral deposits were pale gray-blue with Gomori trichrome staining versus the sky blue appearance of the collagenous stroma and appeared light pink with the periodic acid–Schiff reaction. Giemsa staining revealed metachromatic cytoplasmic granules in mast cells, which were scattered throughout the mass. The neoplastic cells were immunoreactive for pancytokeratin (Fig. 3d) and negative for vimentin, CD3, CD79a, chromogranin A, and neuron-specific enolase. The small lymphocytes were immunoreactive for CD3.
Ultrastructural analysis revealed prominent desmosomes between adjacent neoplastic cells (Fig. 4), scattered mast cells, lymphocytes, and numerous extracellular accumulations of nonbranching, hollow-cored fibrils, 8–10 nm in diameter (Fig. 5), consistent with amyloid. 4 Dense areas along the epithelial cell membrane-amyloid interface appeared consistent with zones of amyloid fibrillogenesis. 4

The cat recovered after surgical resection of the mass, with no apparent recurrence noted in radiographs 5 months postoperatively. The patient continued to do well on alternate-day prednisone therapy 7 months after surgery.
Case No. 2
Review of case submissions from 1982 to 2010 identified 1 additional feline thymic mass with similar histologic features. This mass was obtained from a 12-year-old, male domestic cat that was euthanized because of neurological signs and behavioral changes. At necropsy, a 5 cm × 2 cm × 2 cm, elongated and nodular mass with fluid-filled cysts at each pole was in the mediastinum. This mass was histologically similar to the thymoma in cat No. 1, with abundant amyloid deposition separating elongated to spindle-shaped neoplastic cells without mitotic figures. The amyloid was congophilic, birefringent, pale blue with trichrome staining, and pink with the periodic acid–Schiff reaction. Transmission electron microscopy performed at diagnosis in 1982 revealed numerous desmosomes, variably dense accumulations of extracellular fibrils 8–10 nm in diameter (Fig. 6), and scattered mast cells, macrophages, and lymphocytes.
Discussion
Unique to these thymomas is the deposition of abundant intratumoral amyloid. In cat No. 1, the amyloid was potassium-permanganate–resistant and, thus, classified as non-AA amyloid. 11 Amyloid production has been reported in a single case of human thymoma. 7 One case in a review of feline thymic pathology was described as a thymoma with a background matrix of amyloid; 5 however, it is unclear whether that case was primary amyloidosis or, rather, the localized deposition of AA amyloid associated with systemic disease. Amyloid was not reported in any of 28 feline or 67 canine thymomas in multiple reviews.1-3,5,10,12 To our knowledge, this is the first detailed description of primary amyloidosis in a thymoma in a domestic animal.
The production of amyloid in the thymus did not appear to be part of systemic amyloidosis in the cats of this report. In cat No. 1, no diagnostic abnormalities were detected in serum chemistry, hematology, or urinalysis data at diagnosis or throughout the 7-month postoperative period, and survey abdominal radiographs at diagnosis were within normal limits. Although hepatic and renal biopsy was not performed, there was no clinical evidence of amyloid deposition in organs other than the thymic mass. In cat No. 2, amyloid was not observed histologically in any tissue collected at necropsy (including liver, kidney, brain, and lung) except the thymic neoplasm.
Although amyloid deposition is uncommon in thymomas, the potential for its production is supported by the recent identification of amyloid precursor protein and amyloid precursor–like protein 2 in the thymus of mice. 8 These proteins were expressed exclusively by thymic stromal cells. 8 It is unknown whether feline thymic epithelial or stromal cells express amyloid precursor family proteins.
Footnotes
Acknowledgement
We thank Judith Stasko of the National Animal Disease Center (NADC), Ames, IA for her assistance with electron microscopy.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
