Abstract
Free-living amoebae of the genus Acanthamoeba can cause a fatal disease of the brain in humans called granulomatous amoebic encephalitis. We present a case of meningoencephalitis and pneumonitis in a simian immunodeficiency virus (SIV)-infected rhesus macaque caused by Acanthamoeba sp. The animal became ill 176 days after intravenous inoculation with SIVmac251 after a short history of weight loss and a sudden onset of hind limb paresis and abnormal head movements. Histopathologic examination of hematoxylin and eosin-stained tissues revealed multifocal to coalescing necrotizing neutrophilic meningoencephalitis and pneumonitis. Immunofluorescence and polymerase chain reaction were used to identify the genus of amoeba as Acanthamoeba. Immunohistochemistry of immune cell markers was used to characterize the animal's immune response to the opportunistic amoebic infection with features of both innate and adaptive cell-mediated immunity. Although not previously reported, the potential transmission to humans, either through environmental contamination or contact with an infected animal, makes this disease a threat to laboratory animal care staff and pathologists.
Pathogenic, free-living (or amphizoic) amoebae belonging to the genera Acanthamoeba, Naegleria, and Balamuthia cause infections in humans and animals, 12,14 are ubiquitous in soil and water, and are distributed worldwide. 15 Acanthamoeba and Balamuthia (formerly leptomyxid) generally infect humans and animals as opportunistic pathogens. 14 In recent years, the incidence of amoebic infections, including Acanthamoeba species, Naegleria fowleri, and Balamuthia mandrillaris, in humans has increased significantly, particularly in immunocompromised patients with acquired immunodeficiency syndrome (AIDS), cancer, or transplanted organs. 12 These amoebae can cause two types of disease in the central nervous system (CNS). The first is a rapidly progressive infection called primary amoebic meningoencephalitis (PAM) caused by N. fowleri. The second is a chronic, slowly progressive CNS disease called granulomatous amoebic encephalitis (GAE) that can be caused by either Acanthamoeba sp. or B. mandrillaris. 9 Whereas PAM can be seen in young, healthy individuals, GAE generally occurs in immunocompromised hosts. 16 Acanthamoeba species can also cause a vision-threatening infection known as Acanthamoeba keratitis, especially in contact lens wearers.
We present the first documented case of fulminant amoebic meningoencephalitis and pneumonitis in a simian immunodeficiency virus (SIV)–infected rhesus macaque due to Acanthamoeba and characterize the immune components in response to the infection. This animal developed lesions characteristic of those seen in humans with Acanthamoeba, including multifocal necrosuppurative lesions in the cerebrum and lung admixed with macrophages, CD3-positive lymphocytes, and intralesional amoeba cysts and trophozoites. These lesions were associated with marked tissue necrosis.
The pathogenesis of Acanthamoeba infections is still poorly understood. Although it is not possible to definitively know when or where this animal was infected with Acanthamoeba, because we have never seen this condition at New England Primate Research Center (NEPRC) before this case and because animals are housed indoors, we suggest that the animal was exposed to amoebae while living in an outdoor habitat in Puerto Rico. We also suggest that the rhesus harbored Acanthamoeba when transferred to NEPRC and developed disseminated disease subsequent to development of immunosuppression as a result of inoculation with SIV.
Materials and Methods
Clinical history
A 2-year-old, adult male rhesus macaque (Mm 151-99) and 12 other rhesus macaques of similar age were purchased from the Caribbean Primate Research Center (CPRC) by NEPRC. These macaques had all lived outside on an isolated island habitat in Puerto Rico until they were purchased and transported to NEPRC. On arrival, all monkeys were quarantined for 45 days during which time they were screened for simian retroviruses (SRV-D, STLV, SIV) and Herpes simiae (B virus) by serology; Salmonella, Campylobacter, and enteropathogenic Escherichia coli by fecal culture; intestinal parasites by fecal floatation; and tuberculosis by intradermal testing. In addition, they were vaccinated for measles and treated with ivermectin for parasites. They remained in individual cages until assigned to different studies.
Two years later, Mm 151-99 was inoculated intravenously with uncloned, pathogenic SIVmac251 (25 ng p27). It was placed in a BL2 facility and was not exposed to other animals. It was euthanatized 176 days postinoculation at 5 years of age after a short history of significant weight loss and sudden onset of hind limb paresis and abnormal head movements.
Tissue processing and light microscopy
At the time of necropsy, representative tissues from all major organs and the brain, including frontal, parietal, temporal, and occipital cortices, basal nuclei, thalamus, hippocampus, brain stem, and cerebellum, were collected, fixed in 10% neutral buffered formalin, embedded in paraffin blocks, sectioned at 5–7 µm, and stained with hematoxylin and eosin (HE) by routine techniques. Select sections were also stained with periodic acid–Schiff (PAS). Additional sections were used for immunohistochemistry. Adjacent pieces of tissue were fresh frozen in microcentrifuge tubes on dry ice and stored at −80 C for polymerase chain reaction (PCR). Similarly collected and processed tissues were also examined from seven of the 11 additional animals imported at the same time from CPRC that had also been inoculated with SIV and died from AIDS.
Electron microscopy
Formalin-fixed cerebrum and lung specimens were processed for transmission electron microscopy. Formalin-fixed tissues were postfixed in 1% osmium tetroxide (Stevens Metallurgical Corp., New York, NY), dehydrated, and embedded in eponate 12 resin (Ted Pella, Redding, CA). Sections were stained with uranyl acetate and Reynold's lead citrate and examined on a JEOL 1005 electron microscope.
PCR amplification of Acanthamoeba-specific 18S ribosomal deoxyribonucleic acid
Deoxyribonucleic acid (DNA) was isolated from 60 to 100 mg of fresh frozen tissue of two different regions of brain (frontal and occipital cortices) from Mm 151-99 using a Qiagen DNA Tissue Extraction kit as per the manufacturer's recommendations (Qiagen Inc., Valencia, CA). Unfortunately, frozen lung tissue was not available for analysis. In addition, DNA was isolated from stomach of two other macaques from CPRC (Mm 142-99 and Mm 153-99) as additional possible amoeba cases. Genus-specific primers for 18S ribosomal DNA (rDNA) of Acanthamoeba designated Ac.r. (5′-TTT GAA TTC AGA AAG AGC TAT CAA TCT GT) and Ac.f. (5′-TTT GAA TTC GCT CCA ATA GCG TAT ATT AA) 8 were used with 1–2 µg of isolated genomic DNA to amplify a segment 800–1,100 base pairs (bp) long. DNA amplification was performed in a 50-µl reaction tube containing 1.25 U Taq polymerase, 200 µM deoxynucleoside triphosphates, 4 mM MgCl2, and 0.5 µM primers. The amplification reactions were performed at 94 C for 1 minute, 55 C for 1 minute, 72 C for 2 minutes for 30 cycles, and an elongation step of 10 minutes at 72 C. PCR products were separated by electrophoresis on 2% agarose gels stained with ethidium bromide and visualized under ultraviolet illumination. Quality of DNA was checked by PCR amplification with primers against human β-actin (∼350 bp) designated 75 (5′-CAG TGA TCT CCT TCT GCA TCC TGT C) and 76 (5′-GCT ACG TCG CCC TGG ACT TCG AG). 13
Immunofluorescence for Acanthamoeba sp
Single-label immunofluorescence was performed on formalin-fixed, paraffin-embedded sections of lung from Mm 151-99 (because affected brain sections were limited) and two additional sections of stomach from the previously mentioned macaques (Mm 142-99 and Mm 153-99). The immunofluorescence was conducted as previously described 18 using 1 : 400 and 1 : 100 dilutions of several different antisera made from rabbit against A. castellanii, A. culbertsoni, A. healyi, A. polyphaga, A. rhysodes, A. quina, B. mandrillaris, Hartmannella vermiformis, and Entamoeba histolytica.
Immunohistochemistry for cellular markers
To characterize the immune response to the amoebic infection, routine single-label immunohistochemistry was performed on formalin-fixed, paraffin-embedded tissue sections of brain and lung using an avidin–biotin complex method (ABC Standard, Vector Laboratories, Burlingame, CA) with diaminobenzadine (DAB, DAKO Corporation, Carpinteria, CA) as the chromagen. Slides were counterstained with Mayer's hematoxylin. Antibodies used included CD3 (polyclonal, DAKO), CD79a (HM57, DAKO), CD68 (EBM11, DAKO), HLA-DR (CR3/43, DAKO), glial fibrillary associated protein (GFAP) (polyclonal, DAKO), and CD8 (1A5, Novocastra, Burlingame, CA). Information on the antibodies is summarized in Table 1. For each primary antibody used, appropriate isotype-matched irrelevant immunoglobulins (DAKO) were used as negative controls.
Antibodies used for immunohistochemistry.
Results
Gross, histopathologic, and clinical data
At necropsy, macroscopic examination of Mm 151-99 revealed multiple pinpoint foci of hemorrhage in the white matter of the occipital lobe on the left side of the brain and in the basal ganglia and thalamus on the right side. In addition, the lungs were mottled and contained multifocal 0.2- to 1.0-cm, firm white to tan foci.
Histopathologic examination of the cerebrum, basal ganglia, and thalamus revealed occasional randomly scattered 4-mm to 1-cm foci of necrosis with small amounts of hemorrhage, numerous viable and degenerate neutrophils, fewer macrophages and lymphocytes, and rare eosinophils (Fig. 1A)). Near the margins of the lesions were numerous round 15- to 25-µm amoeba trophozoites with abundant pale cytoplasm, a central round nucleus, and a dense nucleolus (karyosome) (Fig. 1B, arrow)). In addition, there were rare 10- to 20-µm double-walled cysts with a wrinkled outer ectocyst and a round to polygonal inner endocyst (Fig. 1B, inset)). In the occipital lobe, the collections of neutrophils, macrophages, lymphocytes, and rare eosinophils were smaller and tended to surround small parenchymal and meningeal vessels and extended into and expanded the meninges.
Histopathology, immunofluorescence, and immunohistochemistry of brain and lung lesions. 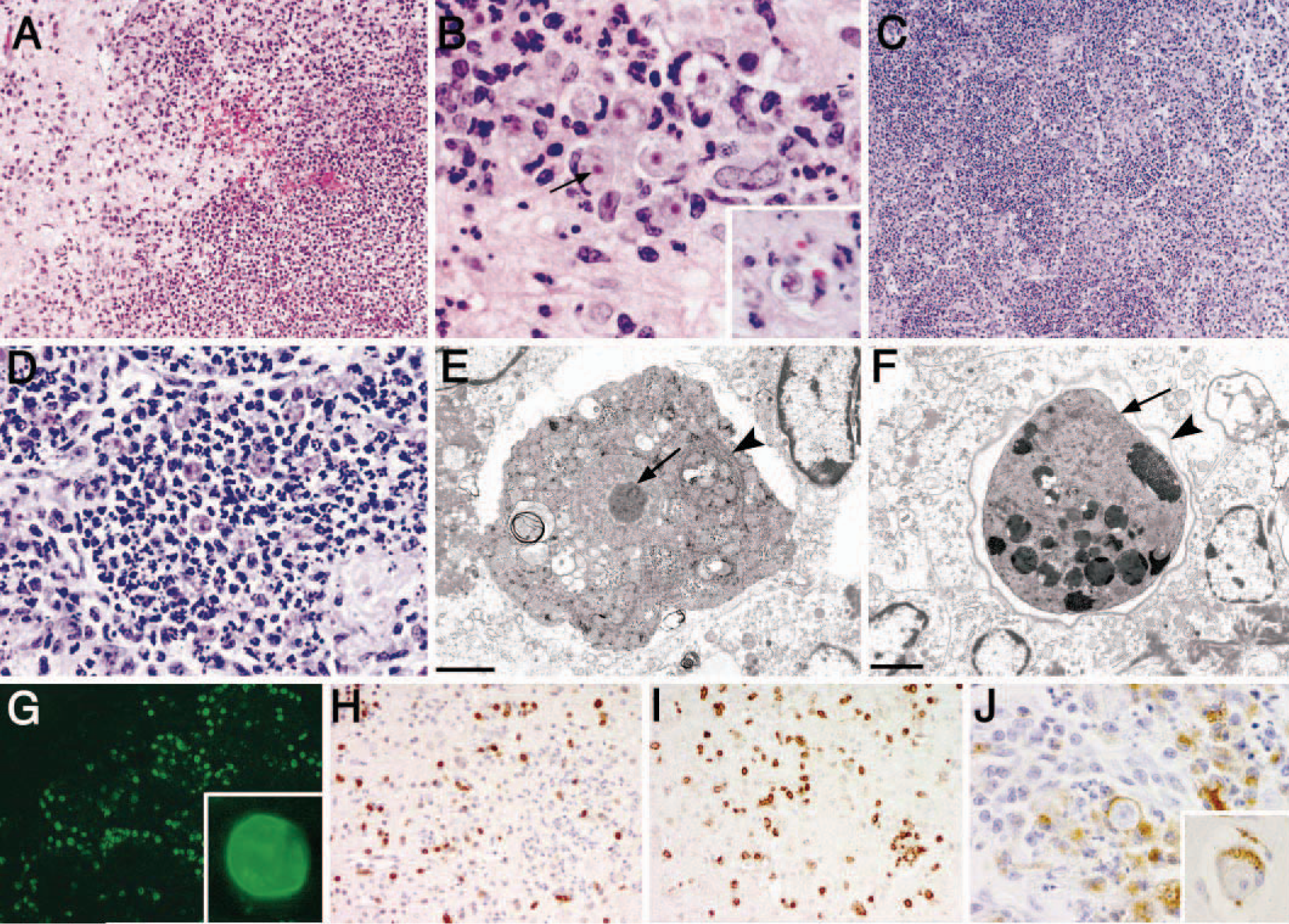
In the lung tissue, there were occasional randomly scattered foci of necrosis and hemorrhage with fibrin, necrotic cellular debris, numerous degenerate and viable neutrophils, fewer macrophages, lymphocytes, and rare multinucleated giant cells and eosinophils (Fig. 1C)). At the margins of the lesions were numerous amoeba trophozoites and occasional cysts as described above (Fig. 1D)). The cytoplasm of trophozoites and the cyst walls stained with PAS stain.
Multifocally, 10–20% of alveoli were partially or completely filled by amphophilic flocculent to granular material consistent with Pneumocystis carinii, an opportunistic infection seen with immunosuppression including AIDS. The presence of P. carinii pneumonia (PCP) in this macaque is an important indication of immunosuppression. There were increased macrophages and occasional binucleated and multinucleated cells in alveoli and terminal bronchioles. There were, however, no giant cells in or around the lesions associated with the Acanthamoeba consistent with a lack or weakened granulomatous response in immunosuppressed patients against the amoebae. SIV in situ hybridization of the lung sections revealed infrequent SIV-infected mononuclear cells in the bronchus-associated lymphoid tissue but not the alveolar histiocytes and giant cells (not shown). Mild abnormalities in other tissues associated with, but not restricted to, SIV included mild follicular hyperplasia and sinus histiocytosis in lymph nodes and mild lymphofollicular aggregates in salivary glands.
Additional data regarding the macaque's disease state include history of weight loss, absolute decline in CD4 count, and plasma viral loads. This animal (Mm 151-99) had lost 1.55 kg or 20% of its body weight during the course of SIV disease, most of which occurred in the last month. Severe weight loss is a frequent sign of advanced SIV disease and simian AIDS. The terminal CD4 count was 847 cells/µl (27% total lymphocyte count) as provided by the investigator (Dr. M. Kaur, Division of Immunology, NEPRC). Although this value is not below 500 cells/µl, usually the upper limit indicative of immunosuppression, the values had been declining during the last 4 weeks to death. In addition, the terminal SIV ribonucleic acid (RNA) plasma load was 1.1 × 107 copy Eq/µl as per Dr. Kaur.
All tissues were examined from the other animals (n = 7) from CPRC that arrived in the same shipment and were used for SIV studies, developed AIDS, and had been necropsied. Several cases had necrotizing lesions of the stomach. On review of all the slides, two cases (Mm 142-99 and Mm 153-99) with necrosuppurative gastritis and amoeba-like organisms were also examined by electron microscopy, PCR, and immunofluorescence. Neither of these cases was positive for Acanthamoeba by PCR or immunofluorescence. Ultrastructurally the amoeba-like organisms were determined to be trichomonads with flagella (Kondova, in press).
Electron microscopy
Formalin-fixed cerebrum and lung specimens were processed for transmission electron microscopy. Both trophozoite and cyst forms were identified. The ultrastructural characteristics of the trophozoite form included a large nucleus and a central nucleolus (karyosome), numerous mitochondria, abundant Golgi apparatus and endoplasmic reticulum, vacuoles, and occasional cytoplasmic pseudopodia (acanthopodia) (Fig. 1E)). Cyst forms ultrastructurally exhibited an undulating or wrinkled outer wall (ectocyst) and a smooth inner wall (endocyst), with variably electron-dense cytoplasm with numerous electron-dense vacuoles (Fig. 1F)).
PCR amplification of 18S rDNA
PCR amplification of genomic DNA isolated from frontal and occipital cortices of Mm 151-99 using the Acanthamoeba genus–specific primers Ac.f. and Ac.r. produced an appropriately sized product (800 bp) (Fig. 2). Unfortunately, frozen lung tissue was not available for PCR analysis. As stated above, two additional cases (Mm 142-99 and Mm 153-99) with necrosuppurative gastritis and amoeba-like organisms were also examined by PCR. Neither of these cases was positive for Acanthamoeba, but both were found instead to contain trichomonads with flagella (Kondova, in press). Stomach from Mm 142-99 was used in the final PCR reaction as a negative tissue control, in addition to water, to guarantee PCR specificity against an unrelated infectious agent. DNA quality and loading was checked by amplification of rhesus β-actin (350-bp product).
Top. PCR-amplified genomic DNA with 800-bp product using Acanthamoeba genus–specific primers in ethidium bromide–stained agarose gel; molecular marker (lane 1), frontal and occipital cortex of Mm 151-99 (lanes 2 and 3, respectively), stomach of Mm 142–99 (lane 4), water control (lane 5). Bottom. DNA amplified with human β-actin primers (350-bp product) to confirm quality of DNA template (lanes as above).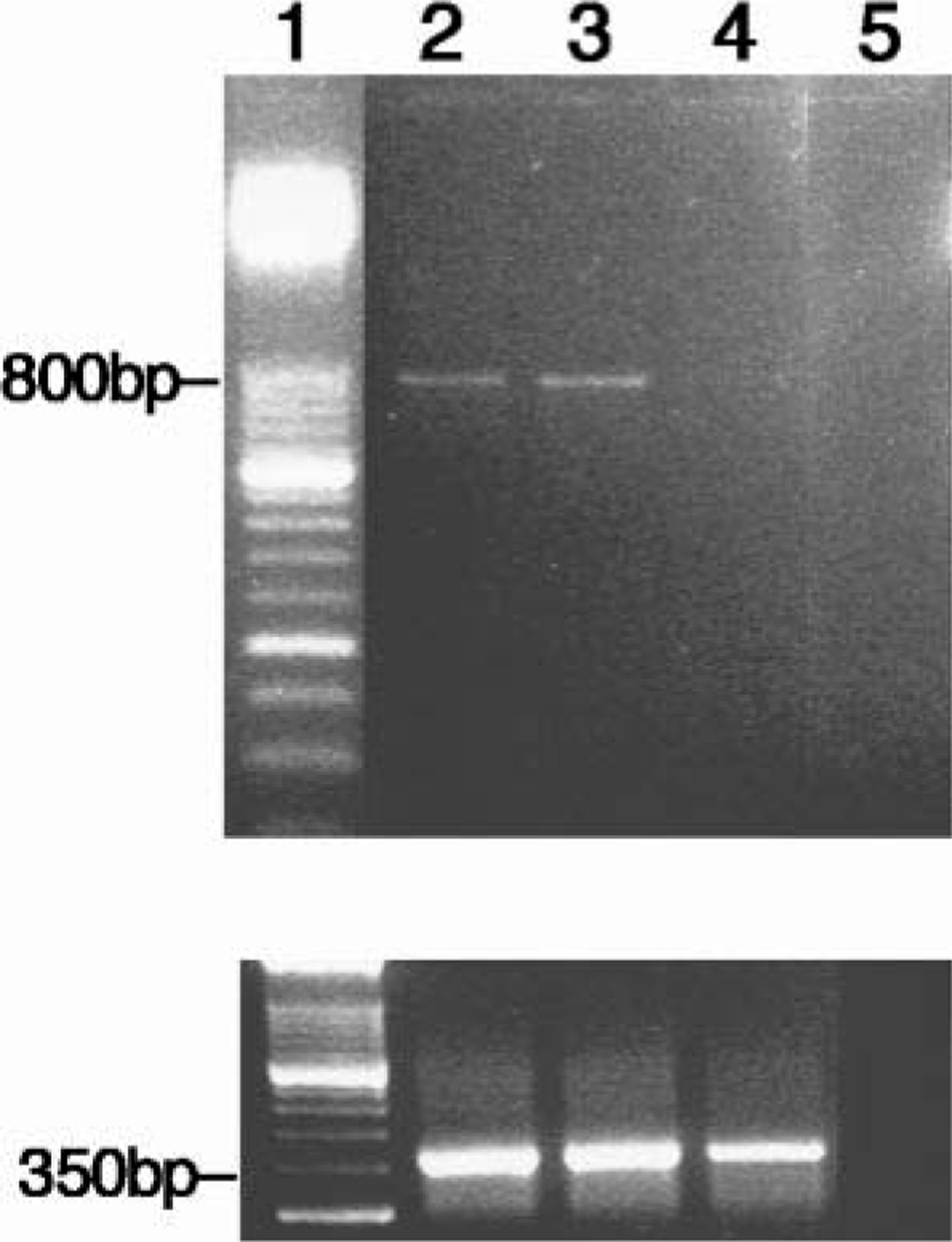
We attempted to clone and sequence the PCR product from brain but were unsuccessful. Based on information in the literature, sequencing Acanthamoeba has proved difficult. 10 There is a high degree of 18S ribosomal RNA (rRNA) sequence diversity within the genus. 7 Various sequencing methods have been used including sequencing mitochondrial DNA and nuclear rRNA, requiring abundant amoebic genetic material. Subgenus classification of Acanthamoeba has been done with a novel riboprinting approach where small-subunit rDNA was amplified and digested for restriction fragment length polymorphism, but this approach is difficult and beyond the immediate scope of this study. 3
Immunofluorescence to identify Acanthamoeba
The amoebae in the lung tissue of the SIV-infected rhesus macaque (Mm 151-99) were identified as Acanthamoeba sp. based on the immunofluorescence assay using several different antisera made against A. castellanii, A. culbertsoni, A. healyi, A. polyphaga, A. rhysodes, A. quina, B. mandrillaris, H. vermiformis, and E. histolytica. The amoebae in the tissue sections did not react with the anti–B. mandrillaris, anti–H. vermiformis, or anti–E. histolytica sera; however, they did react weakly with all the anti–Acanthamoeba sera tested at a 1 : 400 dilution. Tissue amoebae reacted strongly (4+) to a 1 : 100 dilution of anti–A. quina serum (Fig. 1G, inset)) and reacted weakly (2+) with the other anti–Acanthamoeba sera (A. castellanii, A. culbertsoni, A. polyphaga, A. healyi). Based on the antisera available, the amoeba species is most likely A. quina. Stomach sections from Mm 142-99 and Mm 153-99 did not show any fluorescence with any of the tested antisera.
Immunohistochemistry used to identify inflammatory cell response
In this study, we used immunohistochemistry to characterize the immune response to the Acanthamoeba organisms in the brain and the lung. Numerous CD3-immunopositive T lymphocytes infiltrated and surrounded the lesions in both the brain and lung (Fig. 1H)). Most of the CD3-positive T cells were also CD8 positive by immunohistochemical analysis of adjacent tissue sections (Fig. 1I)). There were rare CD79a-immunopositive B lymphocytes associated with the lesions (not shown). There were numerous CD68-immunopositive macrophage-lineage cells in and around the lesions, several of which contained intracellular, phagocytosed trophozoites or cysts (Fig. 1J, inset)). The CD68+ cells are macrophages that had infiltrated into the brain (particularly those in perivascular locations), as well as parenchymal microglia. No multinucleated giant cells were seen in the brain in association with the lesions, suggesting a lack of an adequate granulomatous response to the amoebae. In the brain, there was increased immunoreactivity of GFAP in astrocytes surrounding the lesions (not shown), illustrating regional marked astrogliosis in response to amoebae, cellular infiltrates, and tissue necrosis.
Discussion
This is the first reported case of disseminated Acanthamoeba infection in an SIV-infected rhesus macaque with severe necrotizing meningoencephalitis and pneumonitis. The amoeba was identified by PCR as Acanthamoeba sp. and by immunofluorescence as most likely A. quina. All tissues were examined for evidence of Acanthamoeba infection, but the only active sites of infection in this animal were the brain and lungs. In addition, all tissues were examined from the other seven animals from the CPRC in the same shipment that were also used for SIV studies and necropsied. Two other animals from this shipment (Mm 142-99 and Mm 153-99) developed necrosuppurative gastritis associated with trichomonads (Kondova, in press) that were negative for Acanthamoeba by PCR and immunofluorescence.
Acanthamoeba organisms are ubiquitous in soil and water, occurring in both trophozoite (vegetative) and cyst forms. Infection generally occurs through cyst inhalation or rarely by exposure of skin lesions to contaminated soil or water. 9,12 Once infection has occurred in the lungs or skin, dissemination to the brain is by hematogenous spread, with organisms frequently seen around blood vessels with possible associated vasculitis. 10,12 We believe that infection in Mm 151-99 occurred by inhalation of cyst forms that were in the soil and water of the island habitat in Puerto Rico. There is no other likely time of exposure because this monkey was housed indoors since its arrival in the USA.
Lesions have been reported in humans in other organs including kidney, uterus, prostate, and testis. 1,4,5,17 All tissues in this case were examined, but amoebae were identified only in the brain and lungs. Although Acanthamoeba can develop in immunocompetent hosts, most human cases develop in immunocompromised hosts. 10 Also, immunocompromised hosts have an increased risk of disseminated infection with rapid disease progression. 11,12,16 Once in the brain, Acanthamoeba causes a chronic granulomatous encephalitis or meningoencephalitis termed GAE. In immunocompromised patients, including those with AIDS or cancer, the granulomatous component may be negligible or weak, 12,19 as demonstrated in this case, and there may be no evidence of lymphocytosis in the cerebral spinal fluid. 10 AIDS patients present with other manifestations of Acanthamoeba infection, including chronic sinusitis, otitis, vasculitis, osteomyelitis, endophthalmitis, and disseminated cutaneous nodules or indurated ulcers, the last of which is the most frequent presenting sign of Acanthamoeba infection in AIDS patients. 10
Three other amoebae have been associated with infection of the brain: N. fowleri, B. mandrillaris, and, most recently, Sappinia diploidea. 10 Characteristics of N. fowleri and B. mandrillaris are summarized in Table 2 and compared with those of Acanthamoeba sp.
Comparison between types of amoeba that cause encephalitis.∗.

∗ CNS = central nervous system; GAE = granulomatous amoebic encephalitis; PAM = primary amoebic meningoencephalitis.
A characteristic of Acanthamoeba infection in tissues is the presence of both trophozoites and cysts, as seen in this case. Acanthamoeba trophozoites range from 15 to 35 µm, contain a single nucleus with a large central nucleolus (karyosome), a large contractile vacuole, and characteristic filamentous pseudopodia (acanthopodia). The double-walled cysts range from 15 to 20 µm with a rounded or wrinkled outer wall (ectocyst) and a polygonal to irregular inner wall (endocyst). Pores or ostioles are present where the ectocyst contacts the endocyst. 12
The immune response in Mm 151-99 was first characterized by examination of HE slides, demonstrating that neutrophils were the dominant inflammatory cells present in the lesions frequently clustered around amoebae. Neutrophils are an important component of the innate immune system, functioning both by phagocytosis and releasing lysosomal enzymes in response to organisms. As a consequence of these enzymes, there was marked tissue destruction and abscess formation in areas with numerous neutrophils. Neutrophils are one of the first immune cells to respond to Acanthamoeba organisms. 10
The immune response to the Acanthamoeba was further characterized by identification of immune cell types using immunohistochemistry. In addition to neutrophils, there were numerous CD68-immunopositive macrophages in and around the lesions, several of which contained intracellular, phagocytosed trophozoites or cysts. The presence of macrophages also represents an innate immune response against the amoebae, supporting previous studies in mice demonstrating the importance of innate response against Acanthamoeba infections. 10 Although macrophages were present, however, the absence of granuloma formation or a granulomatous response defined by the presence of epithelioid macrophages and multinucleated giant cells associated with the organisms suggests that this component of the immune response in this immunosuppressed rhesus was weak. In addition, there was evidence of adaptive or cell-mediated immune response against the amoebae with frequent immunopositive CD3 T lymphocytes, most of which were also CD8-immunopositive cytotoxic T cells, associated with the lesions. There were rare CD79a-immunopositive B lymphocytes in and around the lesions examined. Also, the presence of cysts indicates some degree of intact cell-mediated immunity.
In the brain, GFAP-immunopositive activated astrocytes surrounded the lesions and extended into the adjacent brain parenchyma (not shown). Astrocytosis results in the release of the chemokine interleukin-8, which is an important chemoattractant for neutrophils. In addition, activated astrocytes release additional chemokines including monocyte chemotactic protein–1 and macrophage inflammatory protein–1β that initiate and control macrophage infiltration. 2 Astrocytes, however, may also deactivate macrophages that invade the CNS, 6 impairing their ability to effectively phagocytose and eliminate the amoebae.
This case demonstrates that an SIV-infected rhesus developed meningoencephalitis and pneumonitis due to Acanthamoeba infection similar to that seen in AIDS patients. Although the terminal CD4 count was not below 500 cells/µl, usually used to designate severe immunosuppression, the presence of PCP does indicate that the animal was immunocompromised. This suggests that some virulent opportunistic pathogens, such as Acanthamoeba in this case, can cause severe fulminant disease in milder immunocompromised states. Although not completely understood, impaired innate and adaptive immune responses in immunocompromised hosts are important factors in the disseminated disease caused by Acanthamoeba species. Also, the potential transmission to humans, either through environmental contamination or contact with an infected animal, makes this disease a threat to veterinarians, laboratory animal handlers, and pathologists.
Footnotes
Acknowledgements
We thank Dr. A. Kaur for use of animal tissues and for inclusion of CD4 and viral load data for Mm 151-99. We also greatly appreciate the technical assistance of Rama Sriram and Kuei-Chin Lin, the graphics assistance of Kristen Toohey, and the veterinary and pathology services at NEPRC. This study was made possible by the NEPRC Base grant (#RR00168).
