Abstract
This study documents the characteristics of a large series of spontaneously occurring thymomas in a laboratory colony of European hamsters (Cricetus cricetus). Thymomas are rare organotypic neoplasms originating from the thymic epithelial compartment. Because the hamster thymomas largely resembled their human counterparts, the recent World Health Organization (WHO) classification of human thymic epithelial tumors was used. Forty hamsters of both sexes aged 3–29 months were examined macroscopically and histologically. In 22 (55%) of the 40 animals, necropsy revealed enormous whitish masses in the anterior mediastinum, with a diameter ranging from 0.5 to 4.5 cm and a lobulated structure. The anatomy of the thymus region was normal in the remaining 18 hamsters. Histologically, the tumors presented as thymuslike organoid structures with areas of medullary and cortical differentiation and a predominance of lymphoid cells. A network of epithelial cells in the cortical areas, demonstrated immunohistochemically with a cross-reactive antibody against pancytokeratin, supported the diagnosis of thymoma. Cortical lymphocytes showed positive staining with cross-reacting antibodies against CD3 and terminal deoxynucleotidyl transferase, characteristic of immature T cells. On the basis of these findings, the tumors were classified as B1 thymomas, in some cases with AB or B2 components, according to the new WHO classification for human thymic epithelial tumors.
Several possible differential diagnoses must be kept in mind when tumorous masses are found in the anterior mediastinum. The most frequent neoplasms are lymphomas, followed by thymomas, tumors of ectopic thyroid tissue, and neuroendocrine or mesenchymal tumors.
Thymomas are rare malignancies both in human and veterinary medicine. They have been described in a wide range of domestic and laboratory animals and occur more frequently in older animals. In humans, thymomas usually present in the fourth to fifth decade but can occur at all ages without sex predisposition. 28 In dogs and cats, thymomas are also found in older animals. 3 A slight predisposition has been noted in German Shepherd and Retriever Dogs. 1 Thymomas have also been described in laboratory animals, such as rats and mice. 7,20 Two cases of thymoma are also reported in wild European hamsters (Cricetus cricetus). 8 A high thymoma incidence was found in Mastomys natalensis, particularly in females aged more than 2 years. 25,26
Thymomas are neoplasms arising from the specialized thymic epithelial compartments, and the neoplastic cells partly retain the functional characteristics of their normal counterparts, leading to homing of nonneoplastic immature T cells. 18 However, neoplastic epithelial transformation is also associated with abnormalities of the thymic microenvironment, such as architecture, cellular composition, and thymocyte/stroma interactions. The altered thymic microenvironment results in abnormal intratumoral T-cell development and probably contributes to the pathogenesis of paraneoplastic autoimmunity. 16 About one in three of human thymoma patients have symptoms of autoimmune disease at the time of diagnosis, 28 among which myasthenia gravis is the most common. 17 Myasthenia gravis and polymyositis have also been described in dogs with thymomas. 1
The etiology of thymomas is not yet understood. In contrast to lymphomas, there is no evidence of a viral etiology of thymomas in humans or rodents. A single report exists that describes a family with high incidence of thymomas and might point to a genetic predisposition. 4
This study documents a high incidence of spontaneous thymomas in a laboratory colony of European hamsters. Furthermore, morphologic and immunohistochemical characterization of the tumors is performed, and an attempt is made to elucidate potential etiologic factors. Among those, we included a search for oncogenic viruses frequently found in neoplasms of rodents. Because there is no established classification of thymic neoplasms in veterinary medicine and the hamster thymomas bore a striking resemblance to their human counterparts, an attempt to classify these neoplasms according to the new World Health Organization (WHO) classification of human thymic epithelial tumors was made.
Materials and Methods
Animals
Forty European hamsters, 3 to 29 months of age and of both sexes, were examined. Twenty-two of the 40 (55%) hamsters presented clinically with huge masses of variable diameter (Table 1) in the cranial mediastinum, which was also the most prominent macroscopic finding. The anatomy of the thymus region was normal in the remaining 18 hamsters. The animals came from a laboratory colony and were kept individually in macrolon cages (type IV, Becker, Castrop Rauxel, Germany) equipped with a nest box and a running wheel under natural light conditions. Food (Altromin 1314, Lage, Germany) and water were provided ad libitum, and the animals were kept at constant temperature (18 C ± 2 C) and humidity (55 ± 5%).
Summary of all hamsters with thymomas.
The hamsters were sacrificed for brain examination in studies on circadian and circannual rhythms. These studies consisted of monitoring the running wheel activity, monitoring the body weight, and having the reproductive status checked monthly under a short enflurane anesthesia. For additional examination of light and melatonin influence, one group of hamsters was exposed to an artificially long light phase, another group was pinealectomized, and a third group was checked for uric melatonin metabolites collected in metabolic cages.
Morphology
After the hamsters were perfusion-fixed by 7% buffered formaldehyde, a large panel of organs was collected and embedded in paraffin in line with standard laboratory procedures. Sections of 3–5 µm were obtained and stained by hematoxylin/eosin and Giemsa stain.
Immunohistochemistry
Immunohistochemistry was performed with cross-reacting antibodies against CD3 and CD79a (polyclonal A0452 and clone HM57, DAKO, Hamburg, Germany) to identify T and B cells. An antipancytokeratin antibody (clone Lu 5, BMA AG, Augst, Switzerland) was used as a marker for the epithelial elements. Differentiation between mature and immature T cells was performed with an antiterminal deoxynucleotidyl-transferase (TdT) antibody (polyclonal A3524, DAKO). Immunohistochemistry with the cross-reactive antibodies was performed on an automated immunostainer (Ventana Medical systems, Tucson, AZ) according to the manufacturer's protocols, with minor modifications. After deparaffinization and rehydration, the slides were placed in a microwave pressure cooker in 0.01 mol/liter citrate buffer (pH 6.0) containing 0.1% Tween 20 and heated in a microwave oven at maximum power (800 W) for 30 minutes. The sections were immediately cooled in Tris-buffered saline and washed in 3% goat serum for 20 minutes. Incubations with the primary antibodies were performed overnight at room temperature. The rest of the procedure was completed on the Ventana immunostainer with a biotinylated secondary antibody and a horseradish-conjugated streptavidin–biotin conjugated ABC complex.
WHO classification of human thymic epithelial tumors
The classification of human thymic epithelial tumors has its origins in a histiogenetic classification originally proposed by Kirchner and Mueller-Hermelink and others 11,12,14 The new WHO classification basically recognizes the same histologic entities but uses an alphanumerical system, i.e., combinations of letters and numbers. Thymomas with spindle, oval-shaped epithelial cells are designated type A, and those with dendritic or epithelioid epithelial components are designated type B. Tumors combining these features are designated type AB. Nonorganotypic thymic carcinomas are classified as type C thymomas. Type B is further subdivided into B1, B2, and B3 on the basis of increasing epithelial areas and the emergence of nuclear atypia. 15,21
Electron microscopy
For the purpose of etiologic investigation, transmission electron microscopy examination was performed on sternal bone marrow and thymus samples of altogether five selected animals. Thymus and lymph node were embedded in epoxy resin in line with standard laboratory procedures. The sternebrae were fixed in Kuto-fixative 13 for 24 hours, followed by fixation in 1% osmium tetroxide for 24 hours. Sternebra preparation was carried out in liquid nitrogen, and pieces were embedded in epoxy resin. Seventy-nanometer sections of all samples were examined under the transmission electron microscope.
Polymerase chain reaction
Polymerase chain reaction (PCR) was performed on formalin-fixed, paraffin-embedded tissues. Because it is known that papovaviruses induce tumors in hamsters, 22,23 tumor No. 6 (Table 1) was investigated for the presence of DNA sequences related to bovine papillomavirus types 1 and 2 (BPV) and by touchdown PCR for papillomavirus-related DNA sequences. Tumor No. 2 (Table 1) was tested for DNA of hamster polyomavirus (HaPV). For BPV detection, a DNA fragment located in the transformation-associated E5 open reading frame was amplified with the primers E5+ (CAA AGG CAA GAC TTT CTG AAA CAT) and E5− (AGA CCT GTA CAG GAG CAC TCA A). In brief, PCR was performed for 35 cycles with 3 minutes/95 C denaturation, 30 seconds/63 C annealing, and 1 minute/72 C extension. 27 In another assay, we searched for sequences related to the L1 gene of BPV 1, BPV 2, ovine PV 1 and ovine PV 2, European elk PV, deer PV, canine oral papillomavirus, and the papillomavirus of Mastomys coucha using the consensus primers IFNR2 (TTY AAY MGI CCI TAY TGG) and IDNT2 (IST ICC ICK WGT RTT RTC, where the additional letters indicate the following: I = deoxyinosine, K = G or T, M = A or C, R = A or G, S = C or G, Y = C or T, W = A or T) as described recently. 10 Amplification conditions followed a protocol for touchdown PCR, starting with 94 C for 5 minutes, followed by 16 cycles of 94 C for 1 minute, with an annealing temperature that decreased by 1 C each cycle from 50 C to 35 C, and extension at 72 C for 1 minute. The first round of amplification was followed by additional 24 cycles of 1 minute/94 C, 2 minutes/50 C, and 1 minute/72 C. Cloned genomic DNA of BPV 1 (pBPV-1; kindly provided by P. M. Howley, Boston, MA) was used as a positive control. The “touchdown” PCR protocol 10 and HaPV-DNA detection were performed as recently described by Simmons et al. 24 In brief, 0.25 µM of each primer (HaPVf: GCC CAA CCC CAT TTT CAT CC, position within the VP1 sequence: 3277–3296; HaPVr: ACC CTT CAA ATG TGG GAG GC, position: 3718–3737; GenBank accession no. M26281) was used to detect a 460-bp fragment of HaPV-DNA (PCR conditions: 1 minute/94 C, 30 seconds/55 C, 1 minute/72 C). The cloned genomic DNA of HaPV was kindly provided by S. Scherneck (Berlin, Germany) as puc13-plasmid and was used as a positive control. The amplification products were observed in ethidium bromide–stained 2% agarose gels.
Results
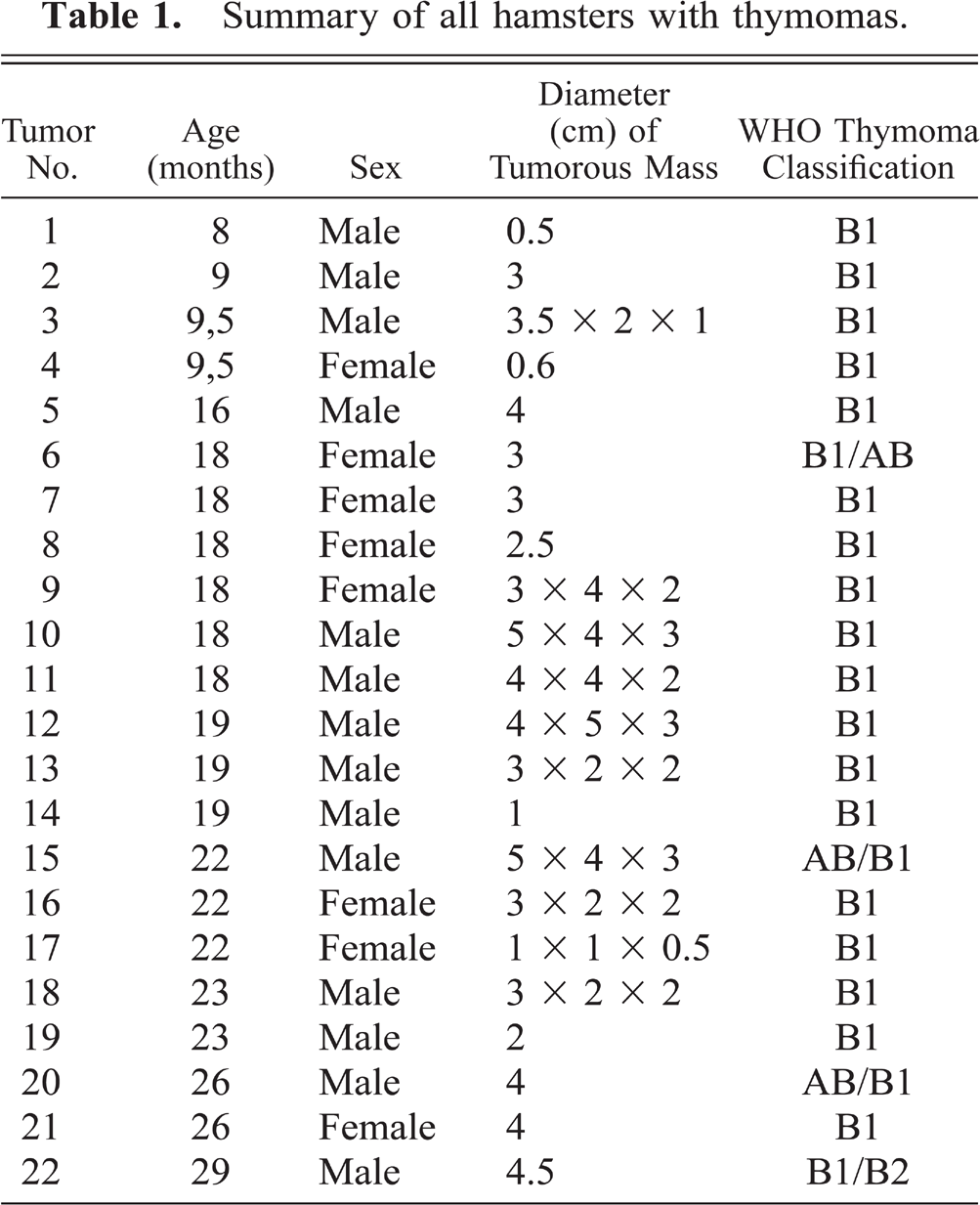
In 22 of the 40 hamsters, a large tumorous mass in the cranial mediastinum was the most prominent macroscopic finding. The tumors were of whitish color and had a lobulated structure, with some of them displaying signs of necrosis. In 18 of the afflicted 22 animals, the diameter of the tumor exceeded 2.5 cm and was combined with compression of the thoracic organs (Fig. 1). The median age of the tumor-bearing hamsters was 18.6 months (range 8–29 months). A total of four animals were aged under 10 months, and a male sex predisposition was seen (14 males versus 8 females).
Tumorous mass in the cranial mediastinum with compression of the adjacent organs.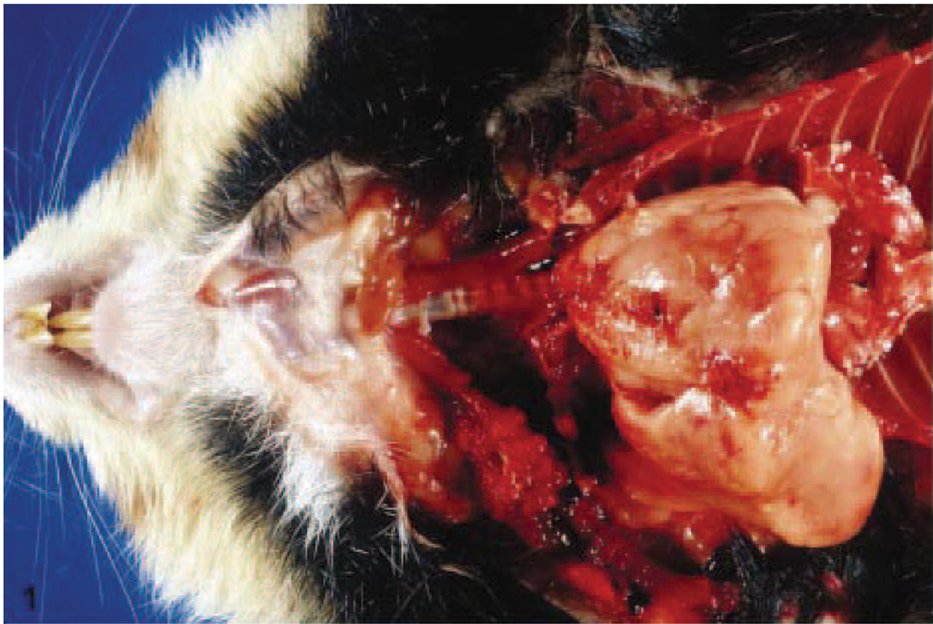
Histologically, all the tumors displayed an overall architecture resembling normal thymus tissue with areas of medullary and cortical differentiation, albeit with less pronounced lobulation than normal thymus. A capsule was also present in all tumors (Fig. 2). Medullary areas appeared as light foci in hematoxylin and eosin and Giemsa stain because of the lower density of lymphocytes and prominent Hassall's corpuscles (Fig. 3).
B1 thymoma: Organoid structure with cortical and medullary differentiation and capsular limitation, HE. Bar = 200 µm.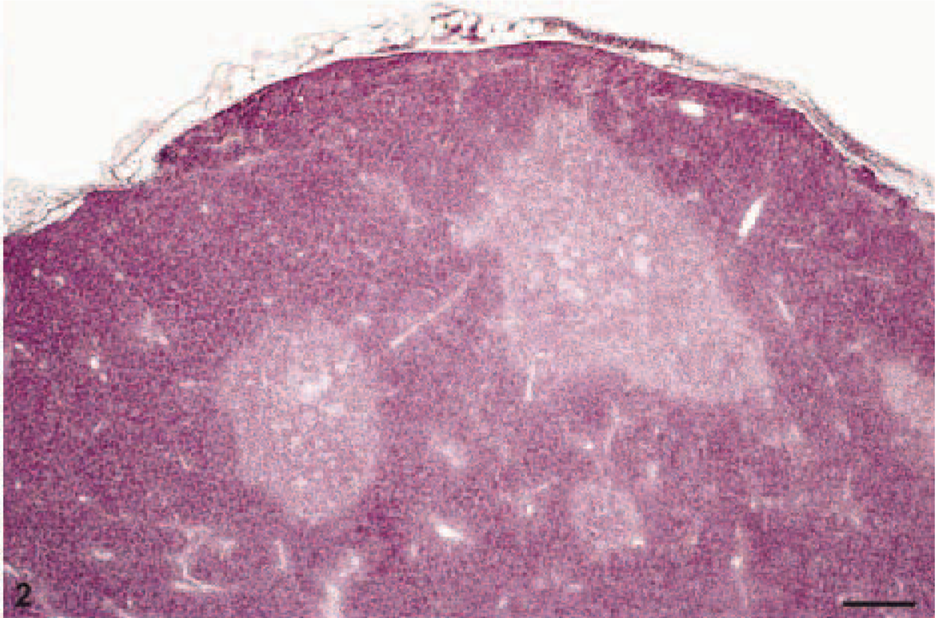
B1 thymoma: Medulla appears as light focus, cortex dominated by lymphatic cells. Giemsa. Bar = 100 µm.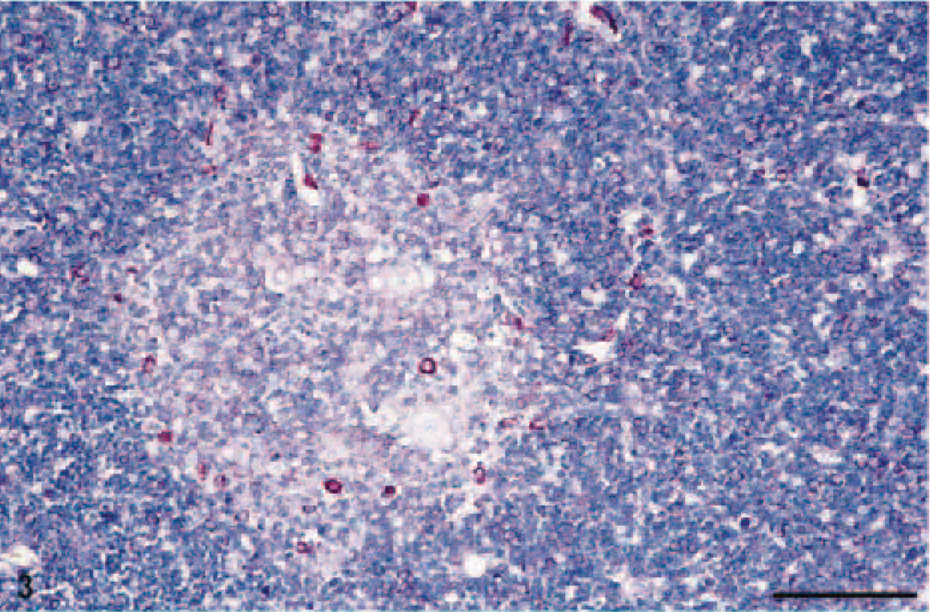
Cytologically, the neoplasms consisted of two distinct cell types: a majority of lymphoid cells without nuclear atypia and a minority of large round-to-polygonal cells with indistinct cytoplasmic borders, large vesicular nuclei, and one or two small nucleoli, representing typical features of epithelial lineage. These epithelial cells formed a delicate network in which the lymphoid cells were embedded. Invasion into adjacent organs was not observed.
Tumor Nos. 6, 15, and 20 (Table 1) also contained large septae, populated by spindle-shaped and occasionally large round cells separating the cortical areas. Tumor No. 22, in addition to lymphocyte-rich areas, contained subcapsular sheets of large, polygonal epithelial cells, mostly lacking a lymphocytic infiltrate.
Immunohistochemistry revealed strong membrane staining for CD3 and nuclear positivity for TdT in the lymphocytes of tumor areas with cortical differentiation (Fig. 4), confirming the presence of an immature T-cell population resembling the normal thymic cortex. Medullary lymphocytes were CD3 positive and TdT negative, and staining for CD79a labeled single interspersed positive B cells. Anti-cytokeratin staining highlighted the epithelial cells forming a network of interdigitating dendritic processes in the cortex and Hassall's corpuscles (Fig. 5).
TdT-positive cortical lymphocytes representing an immature T-cell population. Anti-TdT. Bar = 100 µm.
Cytokeratin-positive network of epithelial cells embedded in lymphatic cells. Antipancytokeratin stain. Bar = 50 µm.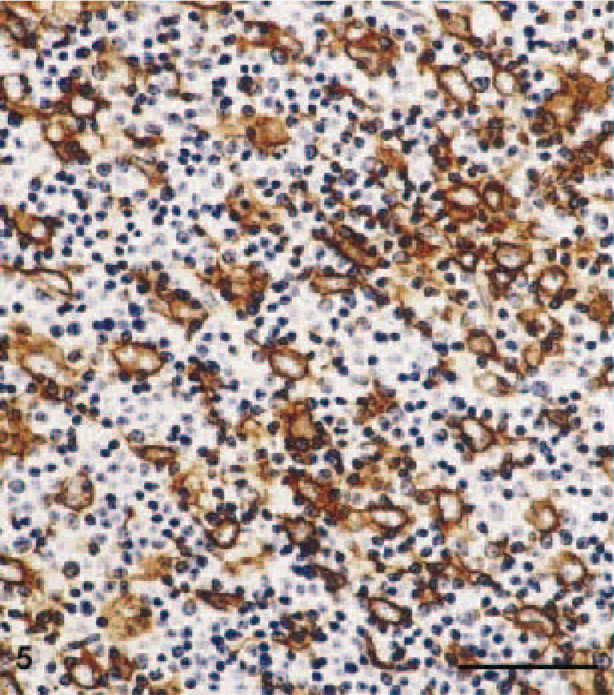
On the basis of these findings, all tumors were classified as B1 thymomas by analogy with the WHO classification for human neoplasms corresponding to the predominantly cortical or organoid thymomas of the histogenetic classification. The epithelial spindle cell areas in tumor Nos. 6, 15, and 20 correspond closely to AB areas in human tumors (Fig. 6), whereas the large-cell, lymphocyte-poor area in tumor No. 22 is best classified as a B2 component (Fig. 7).
Thymoma AB-component with cytokeratin-positive spindle-shaped cells in large septae. Antipancytokeratin stain. Bar = 50 µm.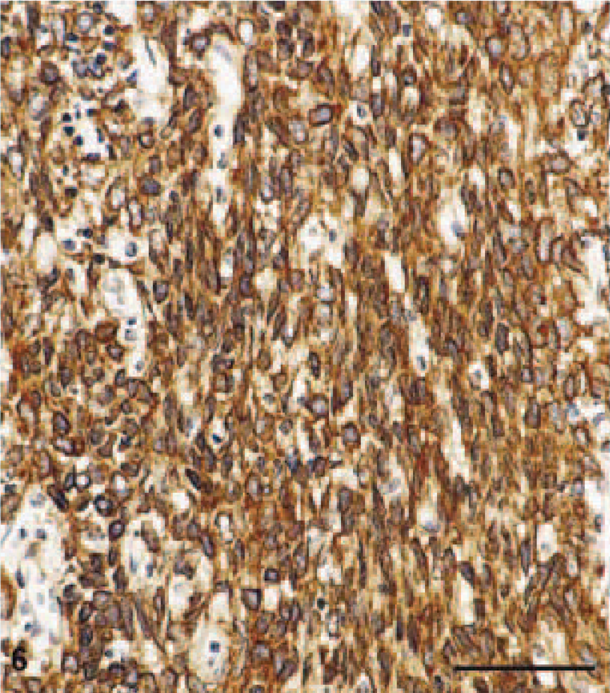
B2 component with subcapsular sheets of large, polygonal cytokeratin-positive epithelial cells with a small lymphocytic infiltrate. Antipancytokeratin stain. Bar = 50 µm.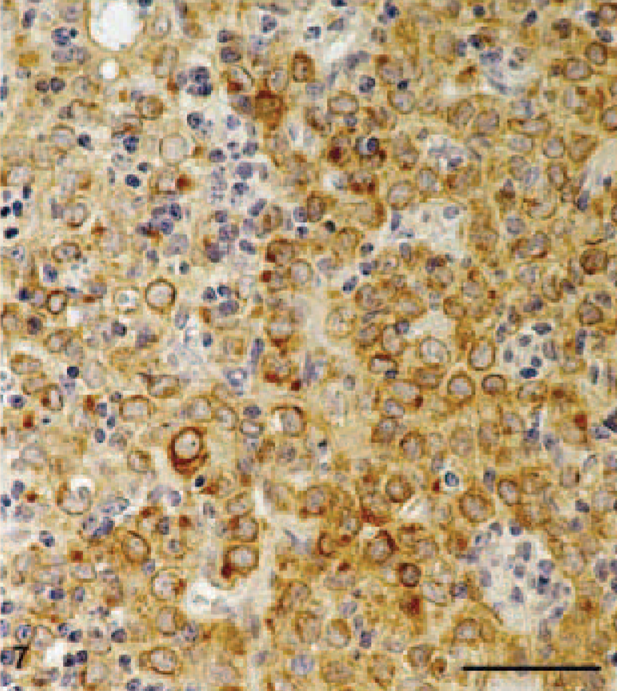
The remaining 18 hamsters were aged 3 to 11 months, with an almost balanced sex distribution (8 females, 10 males). Macroscopically, the thymus region appeared normal. This was also true of thymus histology.
In other organs of all 22 hamsters, variable perivascular lymphocytic infiltrates were a frequent finding, especially in the liver, renal pelvis, stomach, and intestine. Immunohistochemically, these lymphocytes stained positive for CD3 and for CD79a, indicating a mixture of T and B cells.
The bone marrow and thymoma material was examined ultrastructurally for retrovirus or papillomavirus particles. In this rather rigorous examination, no morphologic signs of particles or budding could be seen. Retrovirus reverse transcriptase was not examined because hamster DNA is known to contain genomic endogenous retroviral material. 9 PCR amplification also failed to detect specific products for papovavirus, especially BPV-specific DNA. The 460-bp-long amplicon synthesized with cloned HaPV-DNA as a positive control is shown in Fig. 8.
Agarose gel electrophoresis using the primer set HaPVf and HaPVr. Hamster polyomavirus–specific PCR yielding a 460-bp amplicon (lane 1). No specific amplification products of this size with tumor DNA (lane 2); no template control (lane 3). M = 100-bp molecular weight marker.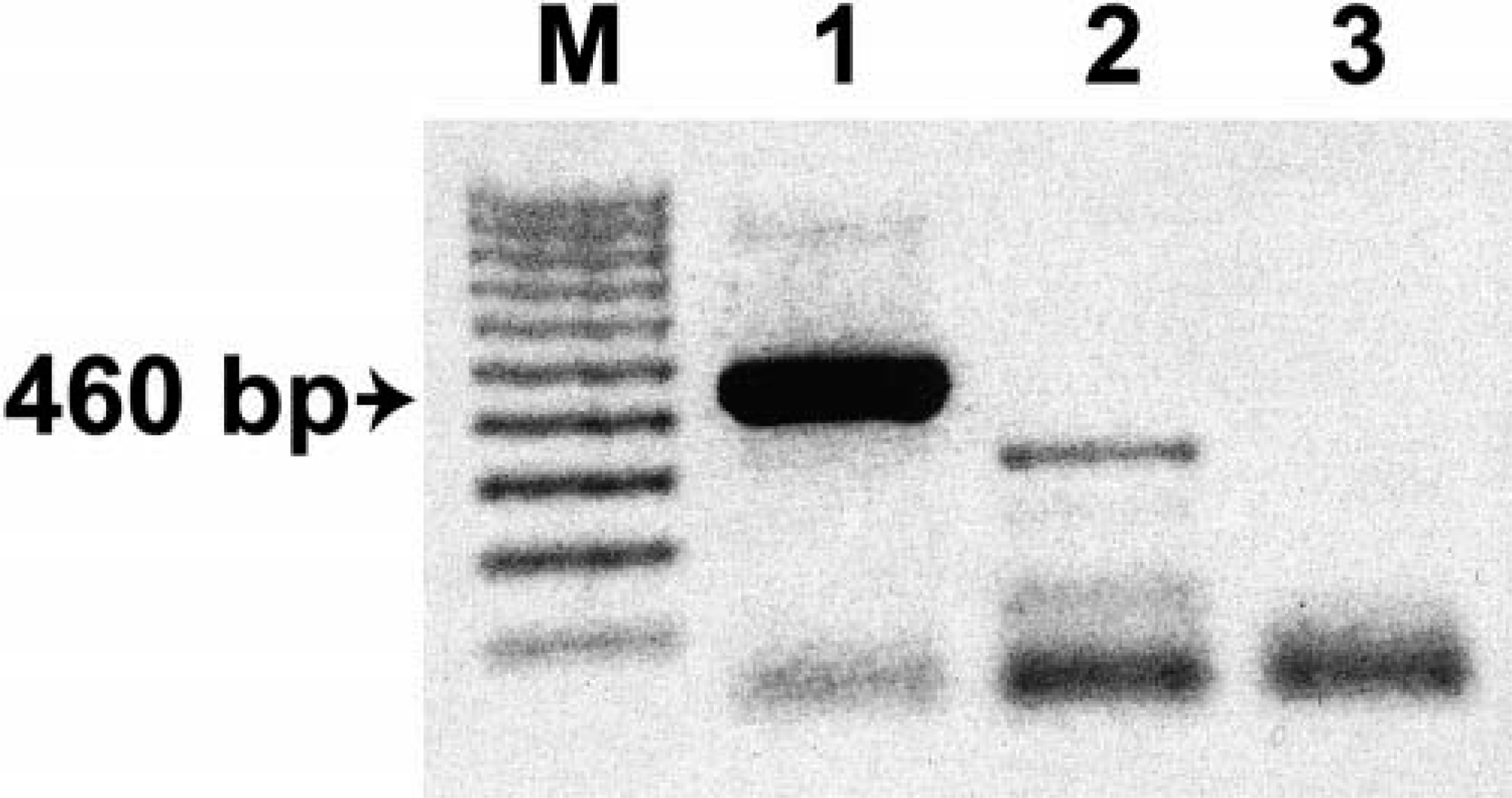
Discussion
Thymic epithelial tumors are rare both in human and veterinary medicine. To our knowledge, this study describes the largest series of thymomas observed in European hamsters. In addition to a description of the morphologic findings, immunohistochemical and etiologic investigations were performed on these tumors and histologic findings compared with their human counterparts. In 1965, Ghadially and Illman described two cases of thymoma in wild European hamsters. 8 A variety of spontaneously occurring tumors and the tumor incidence were evaluated in a large number of laboratory European hamsters (107 males, 178 females). 5 The most frequent neoplasms were lymphoproliferative tumors, followed by adrenal neoplasms (pheochromocytoma). Altogether, the authors identified only two thymomas, corresponding to an incidence of 0.7%. In contrast, the prevalence of thymomas in our study was 22 out of 40 examined hamsters, corresponding to 55%.
Because of the high resemblance of the hamster thymomas to their human counterparts, the current human WHO classification for thymic epithelial tumors was used. This nomenclature is based on histogenetic or functional tumor features. 15,21 Diagnosis of B1 thymoma is mostly arrived at on the basis of morphologic characteristics such as organoid structure and epithelial network. The difference to normal thymus tissue is demonstrated at the macroscopic level by the large extent of lobulated masses in the cranial mediastinum and their expansive growth and at the histologic level by the increased number of cytologically benign, round or spindle-shaped epithelial cells. Immunohistochemistry highlighted the epithelial cells and their network by anti-cytokeratin staining. In addition, anti-CD3 and TdT staining revealed a strong positive signal in the cortical lymphocytes, representing an immature T-cell population. These normal T lymphocytes were strongly augmented probably in response to the increased number of epithelial cells, which provide homing for immature T cells. 2,12 Medullary areas were TdT negative, with an organoid structure similar to that of normal thymus. Staining for CD79a revealed single, interspersed positive B cells. The presence of B cells has also been reported for human organoid thymomas. They were located in the neoplastic epithelial meshwork, mostly in areas of organoid medullary differentiation characterized by epidermoid cells or Hassall's corpuscules. The latter seem to be necessary for specific intrathymic B-cell homing. 6
However, despite the cytologically benign appearance of the epithelial cells, the thymomas examined in our study showed massive local growth with compression, albeit not invasion, of adjacent organs. Thus, lymphatic congestion was seen in the cervical lymph nodes. Metastatic dissemination could not be demonstrated.
Marx and Mueller-Hermelink considered human type A and AB thymoma as clinically benign tumors. Type B thymoma subtypes were found to be low-grade malignant tumors. Invasion into mediastinal fat tissue was observed in 32% of cases and into adjacent organs in 11% of cases; metastasis occurred only in 3% of all cases. 15 The diagnosed AB and B2 components in the hamster thymomas comprised only a small part of the whole tumor mass and neither did they influence tumor classification nor did they have an observable impact on the biologic behavior of the tumors (e.g., in terms of local invasiveness) but rather they represent a morphologic tumor variation.
In other organs of all hamsters with thymoma, variable and sometimes extended perivascular lymphocytic infiltration was found, especially in the liver, renal pelvis, stomach, and intestine. Immunohistochemically, the infiltrates represented a mixed T- and B-cell population. T cells were negative for TdT, and must, therefore, be considered mature T cells. In summary, the infiltrates most probably represent an unspecific inflammatory process unrelated to the presence of a thymoma.
The remaining 18 hamsters, with an average age of 8.4 months (range 3–11 months), presented with a macroscopically normal thymus region and also displayed normal thymus histology. In summary, these animals were considered tumor free.
Thymomas are rare neoplasms compared with other spontaneously occurring tumors in European hamsters. 5 Normally, tumors occur in hamsters aged 2 years and older, the average lifespan of the European hamster being approximately 31 months in males and 34 months in females. Only one animal aged under 1 year presented with a tumor, resulting in an incidence of 0.3%, in the report of Ernst et al. 5 In our study, four animals with tumors were aged under 1 year, constituting a strikingly high incidence in younger animals, too.
Given the high thymoma incidence observed in the present study, the question of etiology naturally arises. Lymphomas and papillomas have been associated with a viral etiology. Because thymomas are epithelial tumors like papillomas, the samples were examined for virus particles and virus DNA. HaPV isolated from skin epithelioma can induce lymphoma or leukemia in newborn and older hamsters. 22,24 Thymoma tissue and bone marrow were therefore examined for virus particles or virus-specific elements by electron microscopy. PCR for bovine and hamster papillomavirus DNA was performed using paraffin-embedded thymoma material. In our samples, neither method revealed virus particles or viral DNA. Retrovirus reverse transcriptase was not examined because hamster DNA contains genomic endogenous retroviral material preventing determination of whether retroviruses are involved in tumor induction or not. 9 The detection of virus particles by an electron microscopy examination depends on the existence of intact virus-specific elements. Electron microscopy will remain negative if the tumor is latently infected and contains only integrated viral nucleic acids without production of viral particles. The E5-BPV–specific PCR detecting two BPV genomic equivalents in about 100,000 diploid cells is considered a highly sensitive assay. In contrast, the touchdown PCR features an expanded specificity but less sensitivity. From these findings, we concluded that these viruses were probably not involved in the etiology of the thymomas in hamsters.
The high incidence of thymomas in hamsters might therefore have a familial background. A human familial lymphoepithelial thymoma associated with a constitutional chromosomal translocation t (14;20) (q24; p13) has been reported. This particular translocation was present in the mother and her two male offspring. 4 In addition, a line of transgenic mice with spontaneous and heritable development of thymomas at a very high penetrance has been found. The available data suggest that thymoma formation in these mice was the consequence of transgene insertional mutagenesis. Fluorescent in situ hybridization localized the transgene integration site at the F2-G region of mouse chromosome 2. Translocation encompassing the analogous region in humans has been implicated in lymphoepithelial thymoma. 19 Spontaneous cases of human type B3 and type C thymomas showed chromosomal imbalances, with a gain in various chromosome segments or loss of entire chromosomes. Type A thymomas did not reveal chromosomal gains or losses. In addition to their possible tumorigenic role, the deletion in type B3 thymoma of chromosome 6, harboring the human leukocyte antigen locus, might play a role in the pathogenesis of the paraneoplastic autoimmunity characteristic of thymoma. 29
The hamster genealogy of the examined laboratory colony revealed one case involving the mating of two littermates with 10 offspring. The male parent developed a thymoma and so did at least seven of his offspring, suggesting a heritable trait.
Stress because of the circadian rhythm experiments could be a possible auxiliary factor for the tumor development and progression.
In summary, detailed analysis of a large series of spontaneous 22 hamster thymomas was performed. Because they were highly similar to human thymomas, the new WHO classification for human epithelial thymic tumors was used. The tumors were diagnosed as B1 type thymomas, and the WHO nomenclature was found to be highly applicable. According to our data, retroviral or papillomaviral pathogenesis appears rather unlikely, whereas a genetic background to thymoma induction seems conceivable. Further investigations should be performed to analyze the genomic aberrations in this hamster colony. These animals may constitute a useful model system for the identification of genes participating in thymoma induction and progression.
