Abstract
Equine cutaneous lymphoma is an uncommon disease that can present with variable clinical signs, immunosuppression, and rapid systemic disease progression. Various subtypes of equine lymphoma have been described and classified according to a veterinary adaptation of the World Health Organization classification system, but little data is available regarding the association between lymphoma subtypes and epidemiological criteria and/or clinical outcome. The objective of the current study was to classify previously diagnosed cases of equine cutaneous lymphoma and correlate subtypes with clinical data to investigate epidemiological trends and prognostic implications. Results from the study confirm T-cell–rich, large B-cell lymphoma (TCRLBCL) as the predominant subtype in equine cutaneous lymphoma. Cutaneous T-cell lymphoma (CTCL) was the second most common phenotype in the current study. To the authors’ knowledge, a description of the morphological and immunohistochemical features of CTCL in horses, an epitheliotropic neoplasm similar to the cutaneous epitheliotropic lymphoma variant (mycosis fungoides) observed in human beings and canids, has not been previously described. Less common lymphoma subtypes identified in the study include diffuse large B-cell lymphoma and anaplastic large T-cell lymphoma. Quarter Horses almost exclusively developed TCRLBCL, while the frequency of CTCL was increased significantly in Thoroughbreds compared with other breeds. TCRLBCLs more frequently presented as multiple masses while CTCLs were more often solitary nodules. When multiple regions of the body were affected, 100% of cases were TCRBCL. Finally, there was an observable trend for longer survival in cases of TCRLBCL without local recurrence, suggesting adequate surgical excision may be an effective treatment modality.
Equine cutaneous lymphoma is a relatively uncommon neoplasm, accounting for 1.7–3% of all equine skin tumors.12,14,16 While generally considered to be nonaggressive, this neoplasm is associated with highly variable clinical signs and may result in immunosuppression and rapid systemic disease progression.1,3,14 Equine lymphoma has been described and subtyped according to a veterinary-adapted World Health Organization (WHO) classification system, and subtypes are defined primarily by their distinct histological features and the immunophenotype of the neoplastic cells (B- or T-cell origin).4,17,18 Although previous studies have shown an increased predilection for certain horse breeds and anatomic locations, relatively little data is available describing the association between individual lymphoma subtypes and epidemiological criteria such as anatomical distribution, tumor size, number, local or systemic recurrence, and geographical region. 12 Furthermore, clinical outcomes associated with different immunophenotypes, treatments, and/or concurrent therapies have not been well characterized.4,14,16
In human beings and in dogs, for which more clinical and prognostic data is available, treatment for lymphoma is directed by a patient’s specific phenotypic subtype and the clinical presentation.2,11,15 As such, additional studies in horses concentrating on specific aspects such as lymphoma subtype, anatomical location, treatment regimen, and/or clinical outcome may be crucial to identify distinguishing features that may lead to better understanding of disease progression and optimal therapeutic intervention. The objective of this retrospective study was to evaluate previously diagnosed cases of equine cutaneous lymphoma, classify them by subtype using the veterinary-adapted WHO classification system, and correlate these findings with available clinical data to identify both epidemiological trends and prognostic features.
A retrospective review was performed on cases of previously diagnosed cutaneous lymphoma from 1999 to 2010 that had been identified by Schaffer et al. 12 (n = 50) and Durham et al. 4 (n = 33) and by additional query of laboratory records at the Colorado State University Veterinary Diagnostic Laboratories (CSU-VDL; Fort Collins, CO; n = 5). Cases were included in the current study if formalin-fixed, paraffin-embedded (FFPE) tissue was available for immunohistochemical staining (IHC) and if histological diagnosis was confirmed as lymphoma by authors. The cases consisted of excisional or incisional biopsy specimens, and necropsy specimens either submitted to the CSU-VDL or other institutions as previously identified. 4
Tissue specimens from each case had been fixed in 10% neutral buffered formalin, routinely processed, sectioned at 3 or 5 μm, collected on charged slides, and stained with hematoxylin and eosin for evaluation by light microscope. Initial diagnosis of lymphoma was made through histological evaluation of tissue specimens by board-certified pathologists at each of the submitting institutions. Tentative diagnoses of T-cell–rich, large B-cell lymphoma (TCRLBCL), cutaneous T-cell lymphoma (CTCL), diffuse large B-cell lymphoma (DLBCL), or anaplastic large T-cell lymphoma (T-ALCL) were made for each case based on histologic features as previously described.4,18 Following histologic evaluation, tissue blocks were then processed for IHC.
For cases submitted to the CSU-VDL, tissues were labeled immunohistochemically for T-cell antigen using monoclonal mouse anti-human cluster of differentiation (CD)3 a (LN10), and for B-cell antigen using monoclonal mouse anti-human, B-cell–specific activator protein PAX-5 b (DAK-Pax5) on an automated staining platform, c following heat-induced epitope retrieval submersion in citrate buffer (pH 6.0). d Immunoreactivity for PAX-5 was highly variable in the current study, ranging from strong to faint immunoreactivity among cells in sample and control tissues. For cases where faint labeling was observed, tissues were also immunohistochemically labeled for B-cell antigen using monoclonal mouse anti-human CD79αcy e (HM57). Immunoreactions were visualized using commercial systems for CD3 antibodies f and for the CD79αcy and PAX-5 antibodies. g Positive immunohistochemical controls consisted of a normal equine lymph node incubated with primary antibodies as described. Primary antibodies were replaced with homologous nonimmune sera for negative controls. For cases submitted to Ontario Veterinary College, University of Wisconsin at Madison, University of Florida, University of Illinois, Ohio State University, and University of Pennsylvania, IHC was performed as previously described. 4 Each case was then classified into the lymphoma subtypes listed above according to the veterinary-adapted WHO classification system as previously described.4,17,18
Histology submission forms, medical records, and diagnostic reports were reviewed for clinical and histological information. The following information was compiled when available: breed, geographical region (Northeastern, Southern, Midwestern, and Western United States; and Ontario, Canada), anatomic location (head/neck, body, limbs, or combination of these), and tumor number (solitary or multiple). For cases from the CSU-VDL, clinical follow-up surveys were faxed to the submitting clinic requesting: last date seen, tumor size (<1 cm, 1–2 cm, 2–3 cm, or >4 cm), number of lesions, anatomic location of lesions, any additional therapy performed in addition to surgical excision, local recurrence or development of lymphoma in another site, and ultimate clinical outcome. Pearson chi-square statistical analyses were performed using commercially available software h to investigate significant differences in distributions of categorical variables.
Overall, 88 cases met inclusion criteria; of these, basic signalment data was available for 90% (n = 79). Quarter Horses (46%, n = 36) and Thoroughbreds (22%, n = 17) were the most represented breeds, followed by Arabians (11%, n = 9), Warmbloods (6%, n = 5), American Paints (5%, n = 4), Morgans (4%, n = 3), ponies (2.5%, n = 2), Appaloosas (2.5%, n = 2), and 1 Missouri Fox Trotter. Geographical data was available for 99% of cases (n = 87), with samples submitted predominately from the western (51%, n = 44) and midwestern (29%, n = 25) United States. Fewer numbers of samples were submitted from the southern (8%, n = 7) and northeastern (7%, n = 6) United States, and Ontario, Canada (6%, n = 5). The majority of cases reviewed in the current study were obtained from the CSU-VDL, which likely accounts for the higher proportion of samples from the West and Midwest.
Anatomic tumor distribution data was available for 58% (n = 51) of cases, with the body alone (43%, n = 22) and the head and neck alone (33%, n = 17) as the most commonly affected sites. Solitary lesions on the limb were less common (6%, n = 3). The body and head were concurrently affected in 8% of cases (n = 4), the body and limbs were both affected in 6% of cases (n = 3), and all anatomical regions were affected in 4% of cases (n = 2). Data regarding lymph node involvement was not available. Approximately 55% of neoplasms (n = 29) with data available arose as multiple tumor masses, occurring slightly more frequently than solitary masses (45%, n = 23).
Data pertaining to tumor size was obtained primarily by clinical follow-up survey, and was only available for 17% of cases (n = 15). Most neoplasms were greater than 4 cm in diameter (53%, n = 8), followed by tumors 2–3 cm in diameter (27%, n = 4). Tumors 1–2 cm in diameter and less than 1 cm in diameter were less common, accounting for 13% (n = 2) and 6% (n = 1), respectively.
TCRLBCL was the most common variant observed in the current study, accounting for 84% (n = 73) of all cases. Histologically, this neoplastic subtype is characterized by an expansile and obliterative neoplasm within the dermis and subcutis that is composed of broad sheets of large round cells with modest amounts of basophilic cytoplasm, admixed with large numbers of mature small lymphocytes (Fig. 1A). Nuclei of the neoplastic cells are round to oval and large (often 2–3 times the diameter of a red blood cell), with clumped chromatin and prominent nucleoli. Mitoses range from 1–3 per single high-powered field (400×). The mature small lymphocytes that accompany these neoplastic cells are strongly positive for CD3 (T-lymphocyte marker; Fig. 1B). Neoplastic lymphocytes are strongly positive for CD79α (B-lymphocyte marker; Fig. 1C), while immunoreactivity for PAX-5 (B-lymphocyte marker) is variable, ranging from strong to faint immunoreactivity among neoplastic cells.
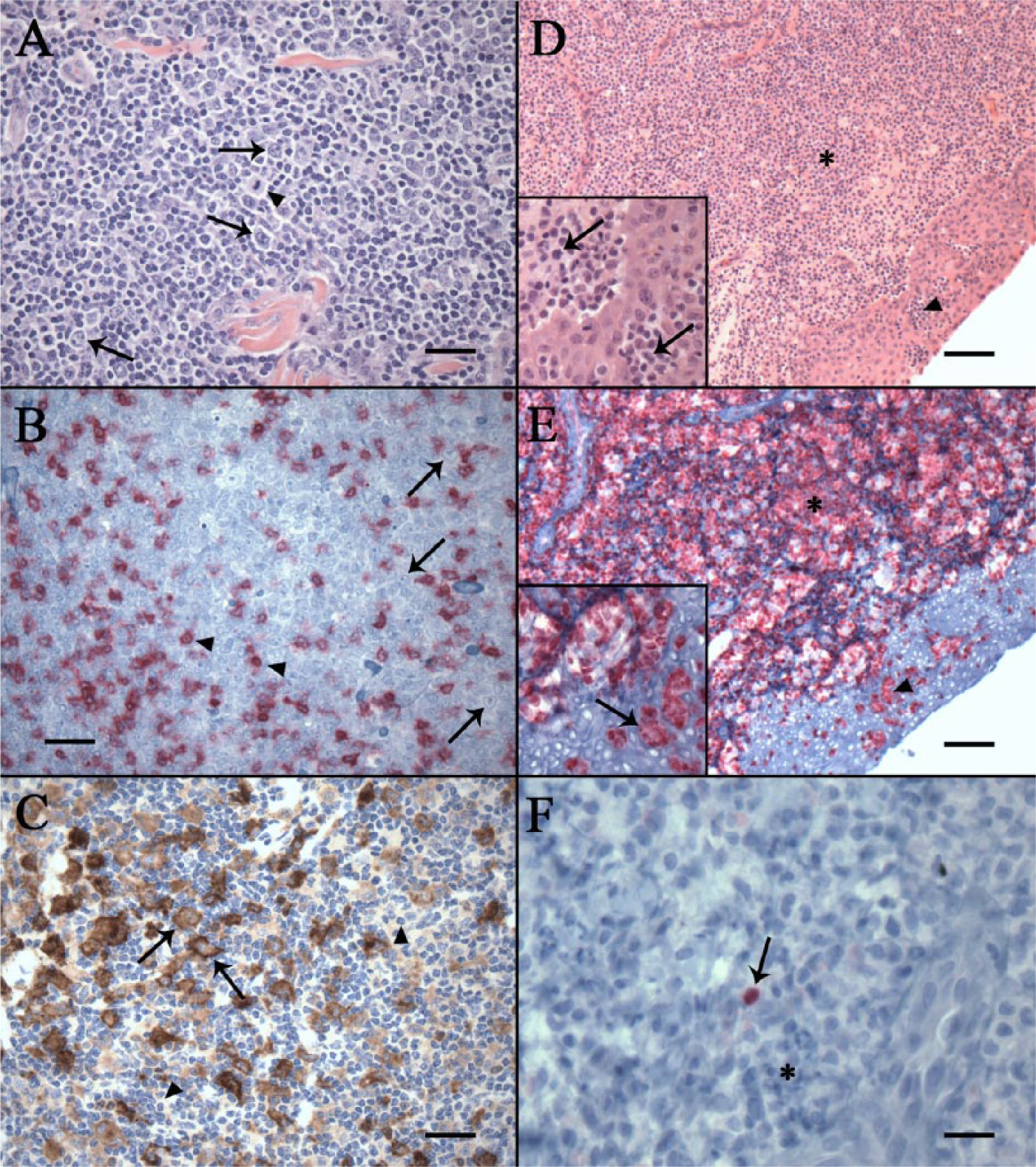
Cutaneous T-cell lymphoma was the second most common subtype observed, accounting for 13% (n = 11) of all cases. Histological features of this neoplastic subtype are characterized by poorly demarcated sheets of neoplastic round cells with scant cytoplasm that multifocally expand and efface the dermis. Within all specimens containing epithelium, these cells exhibit epitheliotropism characterized by multifocal nests and aggregates of neoplastic cells within the epidermis (so called Pautrier’s microabscesses; Fig. 1D). Elsewhere, these cells infiltrate the basal layers of the epidermis where they partially disrupt and efface the dermoepidermal junction. Nuclei are highly pleomorphic among samples, containing 2–5 prominent basophilic nucleoli, varying in shape from round to angular, and ranging in size from 1.5 to 2.5 times the diameter of a red blood cell. Occasionally, cells with hyperchromatic, convoluted nuclei resembling Sézary cells are observed within the epidermis. Mitoses are abundant, varying from 3–5 mitotic figures per single high-powered field (400x), with occasional bizarre mitotic figures observed. Neoplastic cells are strongly immunoreactive for CD3 (Fig. 1E) and are consistently negative for PAX-5 and CD79α (Fig. 1F). Less common lymphoma subtypes identified in the current study consisted of 3 cases of DLBCL (3%) and 1 case of T-ALCL (1%), the latter of which was differentiated from CTCL by the presence of CD3-positive cells with large kidney-shaped nuclei and a paranuclear eosinophilic region.
Following classification, lymphoma subtypes were compared with signalment, and epidemiological and clinical follow-up data. A summary of represented breeds with observed lymphoma phenotypes is presented in Table 1. Of the 36 Quarter Horses in the current study, 94% (n = 34) developed TCRLBCL, while the remaining 2 Quarter Horses had CTCL. While TCRLCBL was also the most common subtype in Thoroughbreds (58%, n = 10), the frequency of CTCL was significantly higher in this breed (33%, n = 5) than in other breeds (chi square, P = 0.019). Arabians were also more likely to develop TCRLBL (78%, n = 7) than CTCL (22%, n = 2); as were American Paints, 75% (n = 3) of which developed TCRLBCL. Warmbloods (n = 5), ponies (n = 2), and Morgans (n = 3) all developed TCRLBCL in this study. Throughout North America, cases were more likely to be diagnosed as TCRLBL (67–89%) than CTCL (11–33%) with no significant variation among different regions.
Summary of represented horse breeds and their respective immunophenotypes.*

TCRLBCL = T-cell–rich, large B-cell lymphoma; CTCL = cutaneous T-cell lymphoma; DLBCL = diffuse large B-cell lymphoma; T-ALCL = anaplastic large T-cell lymphoma. Dash indicates that no cases of the lymphoma immunophenotype were observed in the corresponding breed.
Of the cases for which data was available, associations between immunophenotype and tumor number revealed that TCRLBCLs were more likely to arise as multiple masses (63%, n = 27), while CTCLs were more likely to arise as solitary masses (71%, n = 5). However, this difference was not statistically significant. Both cases of DLBCL for which clinical data was available arose as solitary masses. Lymphomas restricted to the head or neck region alone were more frequently TCRLBCLs (59%, n = 10), compared with all other phenotypes (p = 0.034), and this trend is mirrored for the body alone (87%, n = 20 TCRLBCL and 9%, n = 2 CTCL) and for the limbs alone (100%, n = 3). When multiple regions of the body were affected, 100% of cases (n = 9) were diagnosed as TCRLBCL. There was also a trend for TCRLBCL tumors to be larger in size, with the majority of masses being greater than 4 cm (54%, n = 7/13) or 2–3 cm (31%, n = 4/13) in diameter.
Clinical follow-up surveys (25 cases, 29%) identified 68% of horses (n = 17) that were treated by surgical excision only, compared to 19% of horses (n = 5) that received steroids, 1 horse that received unspecified chemotherapy, 1 horse that received unspecified antibiotics, and 1 horse that received nonsteroidal anti-inflammatory drugs in addition to surgical excision. Twenty cases (23%) had survival data after surgical excision, and consisted of equal numbers of alive (n = 10, 50%) and deceased (n = 10, 50%) horses at the time of this study. Of the horses that were deceased, 33% (n = 7) were reported by clinical follow-up survey to have died from euthanasia related to lymphoma, and 14% (n = 3) died from euthanasia unrelated to lymphoma.
Limited clinical follow-up data was available to assess the correlation between lymphoma subtypes, additional treatments, local recurrence, and/or outcome. For the cases with data available (n = 16), 56% of TCRLBCLs did not recur following excision, while the remaining 7 had local tumor recurrence. In contrast, 100% (n = 3) of the CTCL cases for which data was available had local tumor recurrence. For cases of TCRLBCL with available data (n = 17), 53% of horses (n = 9) were still alive at the time of this study, 35% (n = 6) were reported by clinical follow-up survey to have died from euthanasia related to lymphoma, and 12% (n = 2) were reported to have died from euthanasia unrelated to lymphoma. Interestingly, a higher proportion of horses with TCRLBCL and no local recurrence were still alive at the time of this study (83%, n = 5/6). If local recurrence was reported (n = 7), the proportion of horses still living was reduced to 43% (n = 3), with 43% (n = 3) dying from euthanasia related to lymphoma and 1 horse dying from euthanasia unrelated to lymphoma. No information was available regarding the adequacy of surgical margins. For cases of CTCL with available data (n = 3), 1 horse was still alive at the time of this study, 1 was reported by clinical follow-up survey to have died from euthanasia related to lymphoma, and 1 was reported to have died from euthanasia unrelated to lymphoma. Survival data was obtained solely through information provided by the referring veterinarian in clinical surveys, and information was not available to discern the degree or type of malady leading to a decision to euthanize. Likewise, clinical data regarding additional therapeutics was severely limited for this study, and no significant differences were observed between different lymphoma subtypes, treatment regimen, and recurrence and/or outcome for CTCL or other subtypes.
The present study confirms that TCRLBCL is the most common subtype of cutaneous lymphoma in horses. 4 Histological and diagnostic features of this neoplastic variant were consistent with prior accounts and are characterized by large, neoplastic B cells admixed with large numbers of mature, CD3+ lymphocytes.4,6 Of particular note is the fact that the Quarter Horses in this study almost exclusively developed TCRLBCL. While other horse breeds were observed to exhibit a similar predilection (Arabians, Warmbloods, Morgans, ponies, and American Paints), sample numbers for these breeds were low and may not be representative of the entire horse population. Although Thoroughbreds were also more likely to develop TCRLBCL, the frequency of CTCL is higher in this breed compared with all other breeds in the current study.
As first reported in 1998, 6 TCRLBCLs were demonstrated to be more likely to present as multiple masses than as a single, solitary mass. Interestingly, horses that develop TCRLBCL and experience no recurrence following surgical removal appear more likely to be alive between 3 and 11 years after diagnosis and excision. Only 1 horse under these circumstances died from euthanasia related to lymphoma, suggesting that surgical removal may be an effective method of treatment for TCRLBCL. In contrast, the survival rate appears to drop drastically when there is TCRLBCL recurrence. As no information is available in regard to the adequacy of surgical margins, it is difficult to assess whether incomplete removal resulted in tumor recurrence in these cases.
Although described extensively in human beings and dogs, CTCL is not well documented in horses.5,7,8,10,13 In the current study, we describe histological features of this lymphoma subtype in horses and demonstrate that CTCL in horses is morphologically consistent with features described for the cutaneous lymphoma variant mycosis fungoides in human beings and dogs.7,10 These features include infiltration of the epidermis and the formation of Pautrier’s microabscesses. The occasional presence of Sézary cells within the epidermis may suggest that some of these cases exhibit features consistent with Sézary syndrome, although clinical data is lacking to demonstrate lymphadenopathy and neoplastic cells within the blood. Pagetoid reticulosis is another CTCL variant observed in human beings and dogs and is characterized by a high degree of epitheliotropism in which neoplastic cells are predominately confined to the epidermis. 9 However, in CTCL cases evaluated in the current study, the numbers of neoplastic T cells present within the epidermis were relatively low (limited to nests and small aggregates) and did not exhibit the marked epitheliotropism characteristic of pagetoid reticulosis. Although cases diagnosed as CTCL in this study were exclusively associated with the epidermis, a nonepitheliotropic subtype cannot be ruled out based on a limited sample size. Thoroughbreds developed CTCL more often than other breeds, although TCRLBCL was still most common in this breed. Local recurrence appears to be more common for CTCL than TCRLCBL in the current study, suggesting that CTCL may have a more aggressive clinical course. However, as sample size for this data subset was low, more clinical follow-up data is necessary to confirm this trend. Unlike TCRLBCLs, CTCLs were more likely to arise as solitary masses, suggesting that the number of tumor nodules present at diagnosis may be indicative of lymphoma immunophenotype.
The greatest limitation of the current study was the paucity of clinical follow-up data. As this study was retrospective in nature and spanned over 11 years, obtaining data such as additional treatments, local recurrence, and outcome was exceptionally difficult, especially as many clinics and hospitals do not retain records past 5 years of inactivity. Clinical information in this study was obtained from clinical records and from submitting veterinarian surveys, many of which were incomplete and/or lacking records of such information and thus, making it impossible to obtain a sufficient amount of information to make any definitive correlations between our findings and other clinicopathological data such as complete blood cell count and lymphadenopathy. Furthermore, even when clinical records were available, much of the requested information was still unknown and not documented, underscoring the need for precise, detailed medical records. Future studies should be conducted on a collaborative basis with submitting veterinarians to ensure that complete and precise data can be collected for optimal analysis. As treatment regimens for human and canine lymphomas focus on several factors including clinical presentation and lymphoma subtype, such data is crucial to understanding disease progression and generating effective treatment protocols in horses with recurrent or progressive disease.
In summary, the current study further establishes TCRLBCL as the predominant subtype in equine cutaneous lymphoma, with CTCL as the second most common neoplastic variant. This study describes the morphological and immunohistochemical features of CTCL in horses, an epitheliotropic neoplasm consistent with cutaneous epitheliotropic lymphoma in human beings and canids.7,10 Quarter Horses almost exclusively developed TCRLBCL, while the frequency of CTCL was increased significantly in Thoroughbreds compared with other breeds. TCRLBCLs more frequently presented as multiple masses and were consistently larger in size. CTCLs were more often solitary nodules with no significant size predisposition. When multiple regions of the body were affected, 100% of cases were of the TCRBCL subtype. Finally, there was an observable trend for a higher survival rate in cases of TCRLBCL with no local recurrence following surgery, suggesting that adequate surgical excision may be a highly effective treatment modality for equids with this lymphoma subtype.
Footnotes
a.
LN10, Leica Biosystems Newcastle Ltd, Newcastle Upon Tyne, United Kingdom.
b.
DAK-Pax5, Dako North America Inc., Carpinteria, CA.
c.
Bond-Max, Leica Biosystems Newcastle Ltd, Newcastle Upon Tyne, United Kingdom.
d.
Target retrieval solution (pH 6.0), Dako North America Inc., Carpinteria, CA.
e.
HM57, Dako North America Inc., Carpinteria, CA.
f.
Bond Polymer Refine detection system, Leica Biosystems Newcastle Ltd, Newcastle Upon Tyne, United Kingdom.
g.
Bond Polymer Refine Red detection system, Leica Biosystems Newcastle Ltd, Newcastle Upon Tyne, United Kingdom.
h.
SPSS, IBM Corp., Armonk, NY.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Colorado State University Diagnostic Laboratories with funds provided by Cappy’s Equine Dermatology Research Fund.
