Abstract
Glomerular disease was diagnosed by histopathologic examination in 11 related Bullmastiff dogs, and clinical and laboratory data were collected retrospectively. Four female and seven male dogs between the ages of 2.5 and 11 years were affected. Clinical signs, including lethargy and anorexia, were nonspecific and occurred shortly before death or euthanasia. In five affected dogs serial blood samples were obtained, and dramatically elevated blood urea nitrogen and creatinine levels were demonstrated up to 2.75 years before death. Protein-creatinine ratios were elevated in six of six dogs and were above normal 3.5 years before death in one dog. The kidneys appeared grossly normal to slightly smaller than normal at necropsy. Histologic abnormalities of the kidneys were consistent with chronic glomerulonephropathy with sclerosis. Examination of the pedigrees of related affected dogs yielded evidence supporting an autosomal recessive mode of inheritance.
Inherited glomerulopathies have been described in the Bull Terrier, 12 Cocker Spaniel, 10,16,27 Samoyed, 1,2 Shih Tzu, 13 Soft Coated Wheaten Terrier, 24 and Bernese Mountain Dog. 22,26 A hereditary basis for glomerulopathy is also highly likely in the Doberman Pinscher 4,30 and the Newfoundland dog. 20 The age of onset of clinical signs in familial glomerulopathies ranges from a few weeks to several years of age. Clinical signs include anorexia, lethargy, weight loss, polyuria, polydipsia, and vomiting. Although proteinuria is the hallmark of these glomerulopathies, laboratory findings have varied considerably and can include isosthenuria, aminoaciduria, glucosuria, increased protein-creatinine ratios, nonregenerative anemia, increased blood urea nitrogen (BUN) concentrations, increased creatinine levels, hypoalbuminemia, hyperphosphatemia, and hypercholesterolemia.
In this report we describe and discuss the range of clinical, laboratory, and histologic findings in 11 related Bullmastiffs, along with pedigree relationships and family data supporting an autosomal recessive mode of inheritance.
Materials and Methods
Medical information from four related Bullmastiffs with end-stage renal disease (dog Nos. 1 through 4) was obtained from either patient records from the Veterinary Hospital or Surgical Pathology records at the University of Pennsylvania, School of Veterinary Medicine. Owners, local veterinarians, and breeders provided additional information, clinical observations, complete blood cell counts, serum chemistry profiles, and urinalyses (dog Nos. 5 through 16).
Pedigree information was obtained from the clinically affected (dog Nos. 1–11), suspect (dog Nos. 12–16), and several closely related clinically healthy Bullmastiffs. Dogs older than 6 years of age with normal serum biochemistry values and normal urine protein-creatinine ratios (UPCs) or with no histologic evidence of renal disease were considered normal.
Tissues obtained through case submissions to the Surgical Pathology (dog Nos. 1 and 2) and Necropsy (dog Nos. 3 and 4) Services of the University of Pennsylvania, School of Veterinary Medicine, were fixed in 10% neutral buffered formalin, processed routinely, embedded in paraffin, and sectioned at 3 µm for light microscopy. Blocks of paraffin-embedded renal tissue from three additional cases (dog Nos. 5–7) were obtained from two other diagnostic pathology laboratories. Serial sections of kidney were stained with hematoxylin and eosin, Masson's trichrome for collagen, Congo red for amyloid, periodic acid–Schiff (PAS) for matrix deposition, Jones' methenamine silver for basement membranes, and alcian blue (pH 2.7) for acid mucopolysaccharides. Additional 3-µm sections of renal tissue from dog Nos. 1–3 were evaluated immunohistochemically with rabbit anti-canine IgG, IgA, IgM, and C3 portion of complement (Dako Corp., Carpinteria, CA), using the streptavidin–biotin technique and the chromogen substrate diaminobenzidine (Sigma Chemical Co., St. Louis, MO) as previously described. 7 Tissues were not available for four dogs (Nos. 8–11) diagnosed at other pathology laboratories; therefore, only the information contained in pathology reports was available for review.
Results
Clinical findings
All but one of the affected Bullmastiffs (Nos. 1–10) appeared to be healthy until shortly before death, at which time clinical signs that were reported in each dog included lethargy, weakness, anorexia and weight loss, and polyuria/polydypsia. Occasional episodes of vomiting were noted in dog No. 4, which was also diagnosed with hemangiosarcoma and was subsequently euthanatized. Dog No. 11 was presented to the local veterinarian with sudden onset of severe dyspnea, lethargy, and weakness. During the examination the dog went into cardiac arrest and died before a complete clinical examination could be done. The age of death or euthanasia for all affected dogs ranged from 2.5 to 11 years (mean 5.2 ± 2.5 years; Table 1). Anorexia, weakness, lethargy, weight loss, and polyuria/polydypsia were also reported shortly before death or euthanasia in five dogs suspected of having the same renal disease (dog Nos. 12–16). In this group of two female and three male dogs, the age of death or euthanasia ranged from 1.9 to 6.5 years of age (mean 3.6 ± 2.0 years).
List of Bullmastiffs affected with glomerulonephritis (dog Nos. 1–11) and Bullmastiffs suspected of having the same renal disease (dog Nos. 12–16), including renal panels where available.∗.
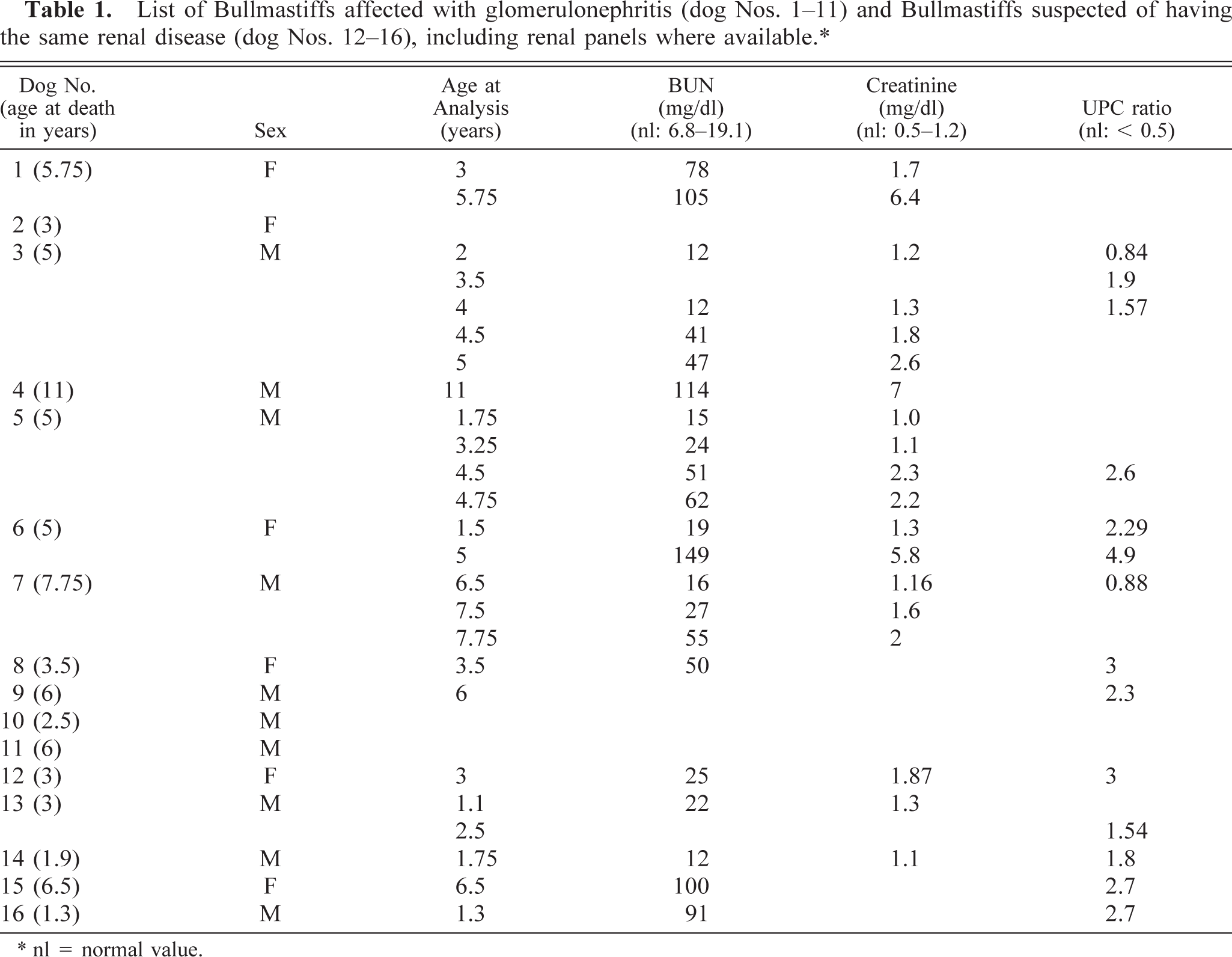
∗ nl = normal value.
Laboratory tests
Some laboratory test data were available for eight of the 11 affected dogs (Nos. 1 and 3–9). Complete blood cell counts were normal, except for lymphocytosis and anemia in one dog with concurrent hemangiosarcoma (dog No. 4). Elevated BUN and creatinine levels were present in all affected dogs for which data were available (Table 1). Examination of the serial serum chemistries for five affected dogs (Nos. 1, 3, 5–7) revealed mild elevations as early as 1.5 years of age and up to 3.5 years before death. Severe proteinuria (4+) occurred in all 11 dogs (Nos. 1–11). UPCs were determined for six affected dogs (Nos. 3, 5–9). All six affected dogs had elevated ratios (Table 1). Serial UPCs were obtained for two of the affected dogs (Nos. 3 and 6) and were elevated 3.5 years before death (UPC = 2.29 at age 1.5 years; normal UPC < 0.5). UPCs were determined in 12 related, clinically healthy dogs (seven female and five male dogs) aged between 6 months and 8 years (mean 2.9 ± 2.5 years). The ratios ranged between 0.03 and 0.26 (mean 0.098 ± 0.074), which is lower than is considered normal (UPC < 0.5) by the Clinical Pathology Laboratory at the Veterinary Hospital of the University of Pennsylvania.
Five related Bullmastiffs (Nos. 12–16) were suspected of having the same renal disease described here, based on similar clinical signs and severe proteinuria (4+). Elevated serum BUN, creatinine, or UPCs were demonstrated in some of these dogs (Table 1). Dog No. 13 was euthanatized after lymphosarcoma was diagnosed, and dog No. 14 was euthanatized because of behavioral problems. In all five suspects no original postmortem results were available. However, glomerulonephritis was noted as the final diagnosis in the records of dog Nos. 13, 15, and 16 provided by the local veterinarians.
Pedigree
A composite pedigree, shown in Fig. 1, contains the 11 Bullmastiffs with glomerulonephropathy (dog Nos. 1–11) and the five dogs suspected to have the same renal disease (dog Nos. 12–16). All dogs confirmed as well as suspected to be affected have male dog No. 17 as a common ancestor to both their sire and their dam. Two of the affected Bullmastiffs (Nos. 4 and 7, both male) were born to clinically normal parents, mitigating against dominant inheritance. Five dogs (Nos. 2, female; 3, male; 5, male; 6, female; and 11, male) were born to matings between one affected and one clinically normal parent (Fig. 1). There were no matings between dogs that were proven to be affected based on histologic findings. However, male dog No. 9 (proven to be affected) was bred to female dog No. 15, which was suspected to be affected based on the information from the local veterinarian and the breeder. This mating produced two offspring, both affected (dog Nos. 8, female, and 10, male). There was no medical information available for the parents of dog No. 1 (female), who was proven to be affected based on the histologic findings. Affected male dogs (proven and suspected) outnumbered affected female dogs by 10 to 6, but this ratio does not differ significantly from a 1 : 1 ratio. The one mating between an affected male dog (No. 9) and an affected female dog (No. 15) produced two offspring, a male and a female dog, both affected. However, it is not known whether there were other littermates. These observations are consistent with a simple autosomal recessive mode of inheritance of glomerulopathy in Bullmastiffs. Fully penetrant X-linked dominant inheritance is excluded by the occurrence of unaffected female dogs in matings between male dog No. 10, which was confirmed to be affected, and an unaffected female dog (Fig. 1). Although not ruled out definitively by the pedigree data in Fig. 1, X-linked recessive inheritance is less likely, with dog No. 1 (female) producing three normal male offspring. However, the sample size was small, a diagnosis was not confirmed in every dog, and information on all littermates of affected dogs was not available to allow an accurate estimation of segregation ratios. Consequently, an autosomal recessive mode of inheritance should be taken as a working hypothesis, with a more complex mode of inheritance also possible.
Composite pedigree of Bullmastiffs affected by glomerulonephropathy. Male dogs are represented by squares and female dogs, circles. The parents of all affected dogs have a common ancestor (dog No. 17), shown at the top of the pedigree. Filled-in symbols represent affected dogs (dog Nos. 1–11), diagonally hatched symbols represent dogs suspected of being affected (dog Nos. 12–16), symbols shaded in gray represent clinically healthy dogs, and empty symbols represent dogs for which no further medical history was available.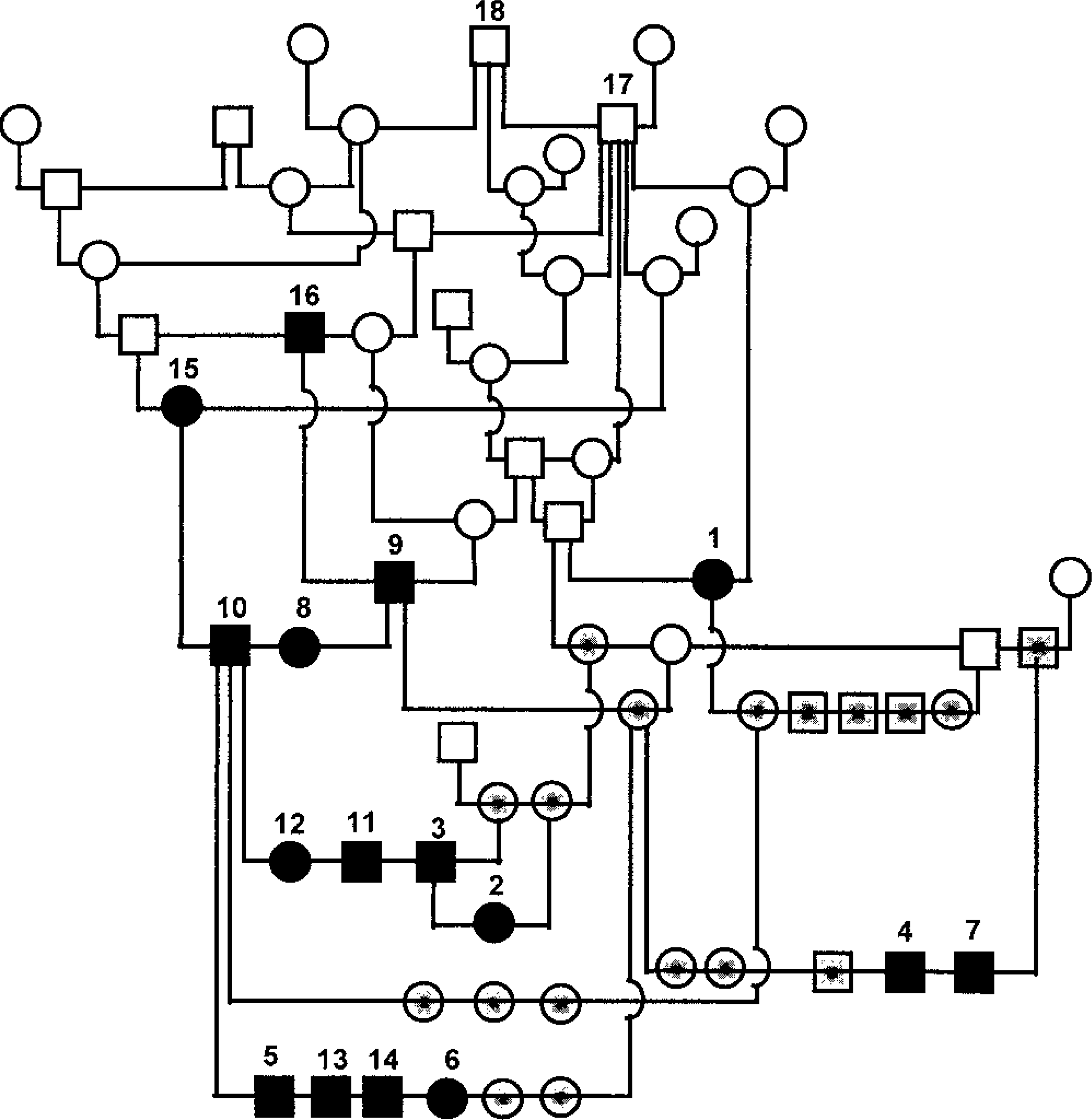
Pathology
Renal biopsy specimens without other pathology data were available for dog Nos. 1 and 2. Postmortem examinations were performed in dog Nos. 3 and 4 at the University of Pennsylvania and on dog No. 11 by the local veterinarian. Gross descriptions of the kidneys from the other eight cases were not available (dog Nos. 1, 2, 5–10). Both kidneys of dog No. 3 were smaller than normal, with three to four linear depressions in the cortical surfaces, which extended toward the medullae. This dog was diagnosed with glomerulonephritis, which was the only significant disease process responsible for the dog's death.
The kidneys from dog No. 4 were grossly normal except for a focal tan area in the corticomedullary region, which corresponded to a regional infarct histologically. Although the significant cause of death in this case was splenic hemangiosarcoma and secondary hemoperitoneum, there was also biochemical evidence of glomerular disease (Table 1).
Dog No. 11 died unexpectedly of heart failure at 3 years of age. On necropsy the local veterinarian found large thrombi in the pulmonary vasculature, with the largest occluding the main pulmonary vein, and the heart appeared mildly enlarged. The kidneys, however, appeared grossly normal.
Glomerular changes predominated and were similar in the seven affected kidneys examined histologically (Table 2). The predominant glomerular change was segmental expansion of the mesangial matrix and an increase in the number of cells in the tufts in the expanded areas (Fig. 2). As such, the glomerular changes were typically diagnosed as either membranoproliferative or membranous glomerulonephritis. Occasional glomeruli had foci of active necrosis, infiltrates of neutrophils or rare plasma cell infiltrates. Although all glomeruli were affected, the expansion of the tuft was typically segmental and often in the hilar region or associated with adhesions to Bowman's capsule. Masson's trichrome and PAS stains revealed that the expanded eosinophilic mesangial material was predominantly collagen (sclerosis) with mildly increased matrix deposition. Less frequently, there was evidence of capillary loop membrane expansion by PAS-positive matrix. In several glomeruli in each case, there were mesangial segments, which were greatly expanded by hyaline material, forming nodular foci within the tufts (Fig. 3). The nodular, hyaline areas were consistent with hyalinosis (PAS positive; red with trichrome and negative with Jone's methenamine silver; not shown). Congo red staining of the kidneys was uniformly negative in all cases, which indicates the absence of amyloid.
Summary of histological findings of renal tissues from Bullmastiffs affected with glomerulonephropathy. Reports were available only for dog Nos. 8–11. Comments included “like Elkhound disease”; (dog Nos. 8 and 9), “chronic glomerular disease” (dog No. 10), and “end-stage kidney disease” (dog No. 11).∗.

∗ N = not present, Y = present, ? = unknown.
Glomerulus; Bullmastiff with glomerulonephropathy, dog No. 5. Segmental expansion of the mesangial matrix and increased cellularity. PAS-methenamine silver. Bar = 20 µm.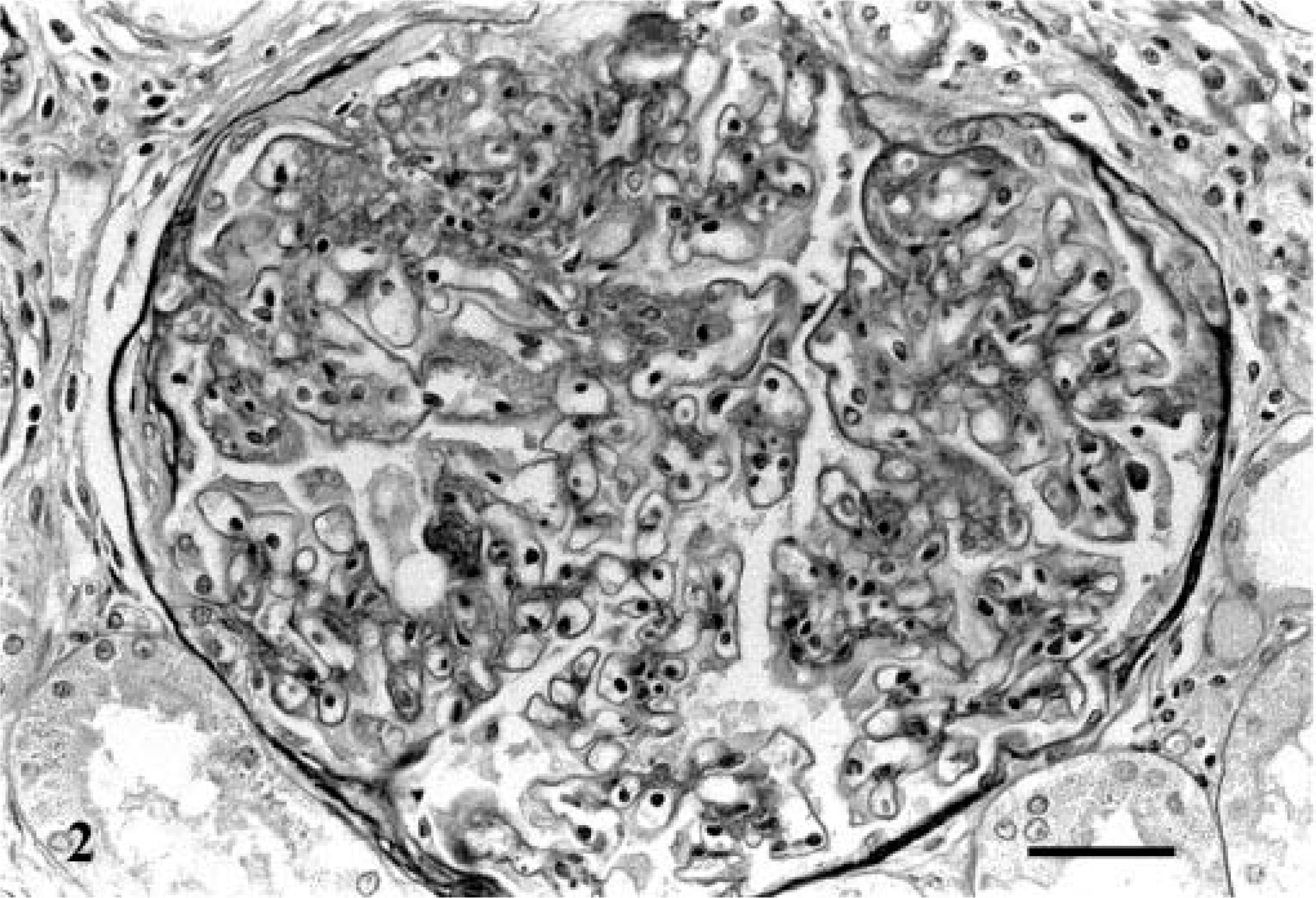
Glomerulus; Bullmastiff with glomerulonephropathy, dog No. 5. Segmental expansion of the mesangial matrix and increased cellularity. PAS-methenamine silver. Bar = 20 µm.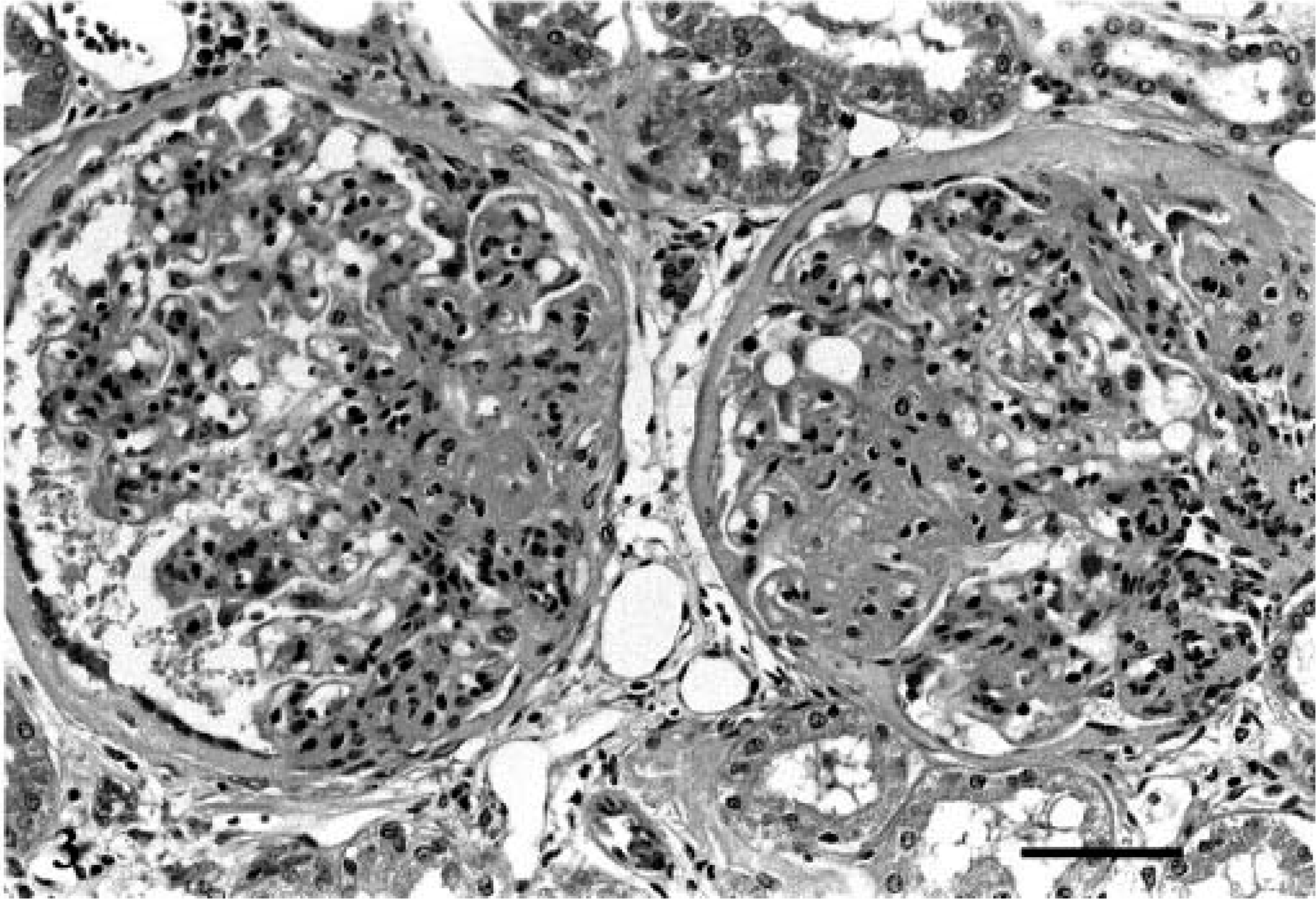
Another striking change associated with the glomeruli in four cases (dog Nos. 2, 3, 5, and 6) was marked dilatation of Bowman's capsule, accentuating the urinary space with only irregular remnants of glomerular tufts remaining (cystic glomerular atrophy). The majority of these irregularly shaped tufts were separated into two distinct lobules that were collapsed and sclerotic with segmental adhesions to Bowman's capsule; however, there were occasional tufts that had remarkably normal components remaining. Also noted in all but five cases were multifocal shrunken glomeruli (approximately 6 per 100) with collapsed and fibrotic, eosinophilic tuft remnants (obsolescent glomeruli).
Thickening of Bowman's capsule by collagen and PAS-positive basement membrane material was present in all seven cases examined by the authors. Periglomerular sclerosis was multifocal and noted histologically or reported in only six cases.
Mild to moderate multifocal interstitial fibrosis was present or reported in nine cases. The fibrosis involved both the medulla and the cortex. It was more diffuse in the medulla, whereas in the cortex the fibrotic areas tended to form radial bands toward the capsular surface. Tubular atrophy was associated with these fibrotic areas in nine cases. There was mild to moderate tubular dilatation present in the seven cases examined histologically, with luminal protein casts evident multifocally. Multifocal tubular epithelial hypertrophy was noted occasionally in three cases. Proliferation of medullary tubular epithelium (metanephric ductlike) was noted in dog Nos. 3 and 5. Lymphoplasmacytic interstitial inflammation was mild to moderate in nine cases and was predominantly associated with the areas of fibrosis and around glomeruli. The findings from all cases are summarized in Table 2.
Immunohistochemical evaluation of the glomeruli of dog Nos. 1–3 revealed minimal segmental deposition of IgM in the capillary loops and mesangium multifocally. Limited deposits of IgG and C3 were detected segmentally along occasional capillary loops in two of the three cases examined. IgA was not detected in any case.
Extrarenal lesions in the suspect and affected Bullmastiffs included splenic hemangiosarcoma (dog No. 4), thromboembolic disease (dog No. 11), and lymphosarcoma (dog No. 13).
Discussion
Familial renal disease of dogs can be separated into three major categories based on histologic patterns: dysplasia, primary glomerular disease, and primary tubular disease. Those renal diseases characterized as dysplastic have features suggesting abnormal maturation of the nephron and its supporting interstitium, with asynchronous glomerular development including retention of fetal glomeruli, primitive tubules with retained metanephric ducts, and retention of primitive mesenchyme. Those diseases characterized as glomerular have clinical evidence of glomerular disease and diffuse involvement of glomeruli with secondary tubular and interstitial changes. Tubular diseases are principally recognized as functional disturbances with subtle to no characterizing histologic features. DiBartola 8 has recently summarized familial renal diseases of the dog in this manner.
The clinical findings in the affected Bullmastiffs described here were nonspecific. Only anorexia, lethargy, weight loss, and occasionally polyuria/polydipsia were noticed shortly before death. This is typical of chronic glomerulonephritis, whereas polyuria, polydipsia, and vomiting are more commonly found in dogs with renal dysplasia. 8 The slow and insidious onset of the disease process was characterized by increased BUN and creatinine, and proteinuria early in the course of disease, well before clinical signs were present. An increased UPC was found up to 3.5 years before the onset of clinical signs in one of the affected dogs. Death occurred between 2.5 and 11 years of age.
Glomerulopathies in the dog are transmitted as an autosomal dominant trait in the Bull Terrier; 12 as autosomal recessive traits in the Bernese Mountain Dog, 26 Cocker Spaniel, 10 Shih Tzu, 13 and Soft Coated Wheaten Terrier; 21,24 or as an X-linked dominant trait in the Samoyed. 15 In the present study in Bullmastiffs, both sexes were affected with about equal frequency, and clinically normal parents produced male and female dogs with glomerular disease. These findings are consistent with an autosomal recessive mode of inheritance. However, additional family studies and breeding studies will be needed to verify this.
All tissues of the affected dogs had histologic changes characteristic of a chronic glomerulopathy. The changes were advanced, with segmental glomerular sclerosis as the typical feature. In three of 11 samples, IgM immunoglobulin deposition was minor, was present only in occasional glomeruli, and is most likely a secondary, nonspecific trapping of the components in glomeruli damaged by the primary process. The morphologic appearance of the glomerular changes and the immunohistochemical findings are not suggestive of an infectious or primary immune-mediated process but more closely resemble the familial diseases of breeds such as the Samoyed, 1,15,29 Doberman Pinscher, 4,25,30 Rottweiler, 6 Bull Terrier, 17 older Soft Coated Wheaten Terrier, 8,21 and Newfoundland dog. 20 This resemblance suggests the possibility of a biochemical or structural defect in the glomerular basement membrane as an underlying etiology, as proven in the Samoyed 29,31 and suspected in the Doberman Pinscher 25 disease. An α-collagen-5 defect has been recognized in the Samoyed glomerulopathy, which is inherited as an X-linked dominant trait. Interestingly, both affected male and female heterozygotes develop proteinuria at 2–3 months of age, but only affected male dogs had a decreased glomerular filtration rate. Focal multilaminar splitting of the glomerular capillary basement membranes was found on ultrastructural examination. Female dogs showed no signs of renal failure by 30 months of age, whereas affected male Samoyeds had usually died by 15 months of age. 14 A dominant mode of inheritance (such as X-linked dominant) is consistent with this concept of an inherited structural defect.
The presence of dilated Bowman's capsules in association with remnants of glomerular tufts, seen in some of the affected dogs, has been termed cystic glomerular atrophy, which is also present in the familial renal diseases of the Doberman Pinscher, standard Poodle, Rottweiler, and Bull Terrier breeds. It is thought to be a secondary change, perhaps related to nephron blockage from fibrosis more distally. 8
The underlying molecular abnormality responsible for the development of the inherited glomerular disease in Bullmastiffs described here is unknown. The materials available for this study were limited to tissues collected terminally in the disease. Clinically, there is evidence that proteinuria develops at a younger age before clinical signs are apparent. The glomerulosclerosis-type changes seen here are commonly caused by immunologic diseases, with associated amyloidosis or deposition of immune complexes in both cats and dogs. 8 However, there was no histologic evidence suggesting immune-mediated renal damage. Interestingly, glomerulosclerosis is quite prevalent (∼ 80%) among captive cheetahs both in South Africa and in the United States of America. 23 However, the form of glomerulosclerosis described in the cheetah more closely resembles diabetic nephropathy of humans. Thus, it was suggested that chronic stress led to hyperglycemia through hyperadrenocorticism, which in turn led to the nephropathy. 3 Alternatively, the changes also resembled those in the rat. Chronic progressive rat nephropathy is characterized by late-onset renal failure and is more common and more severe in male than in female dogs. A familial basis has been proposed because the disease is common in a variety of albino rat strains. However, high-protein diets have also been suggested as a cause of rat nephropathy. 11
The histologic findings described in the Bullmastiffs resemble focal segmental glomerulosclerosis (FSGS) in humans, which is seen in several renal disorders, all of which are characterized by proteinuria and chronic, progressive loss of renal function. Familial forms have been described, 5,9,28 and both autosomal recessive and autosomal dominant modes of inheritance have been suggested. 5 In humans and mice, mutations of cd2ap 19 and actn4 18 have been shown to cause FSGS. Both genes code for structural proteins, and ACTN4 is expressed at high levels in the glomerular podocyte. 18
Elucidation of the mechanisms of the renal disease described here will require a prospective analysis, which could include serial evaluation of biochemical values, renal histology and electron microscopy, and immunohistochemistry in dogs produced from affected parents. Determination of the actual onset of the disease will require serum chemistry and urinalysis at an early age, with continued evaluation at reasonable time intervals.
Footnotes
Acknowledgements
This work was supported in part by National Institutes of Health grant RR02512. We thank the referring veterinarians for submitting biopsy material and the Bullmastiff breeders for supplying pedigree and health information.
