Abstract
Focal segmental glomerulosclerosis (FSGS) recently has been recognized as a common cause of proteinuria in dogs in general, and in Miniature Schnauzer dogs in particular. This study describes the morphologic features present in the kidneys of 8 related proteinuric Miniature Schnauzer dogs. The FSGS, characterized by solidification of portions of the capillary tuft, affected 32% to 49% of examined glomeruli in these dogs. Synechiae, often accompanied by hyalinosis, were present in 13% to 54% of glomeruli and were more prevalent in older dogs. Seven of 8 dogs had arteriolar hyalinosis. Ultrastructurally, all dogs had evidence of a podocytopathy in the absence of electron-dense deposits, glomerular basement membrane splitting, or fibrils. All dogs had multifocal to extensive podocyte foot process effacement. Other podocyte changes included microvillous transformation, the presence of vacuoles or protein resorption droplets, cytoplasmic electron-dense aggregates, and occasional binucleation. Variable amounts of intraglomerular lipid were present in all dogs. All dogs were proteinuric, with measured values for the urine protein-to-creatinine ratio ranging from 1.2 to 6.5. Azotemia was mild to absent and dogs were euthanatized at 5.1 to 14 years of age, in all cases due to nonrenal diseases. The underlying cause of FSGS in these Miniature Schnauzer dogs has yet to be determined, but contributors likely include genetic podocytopathy, lipid abnormalities, and glomerular hypertension.
Focal segmental glomerulosclerosis (FSGS) is a common cause of the nephrotic syndrome in humans and has been recognized as an important cause of proteinuria in the dog. 2,8 –12,15,19,28 In a retrospective study of 501 renal biopsies from dogs with proteinuria, FSGS was the most common non-immune complex glomerulopathy, occurring in 21% of all biopsies. 28 In that study, 3% of the submitted samples were from Miniature Schnauzer dogs and 78% of these Miniature Schnauzer dogs had FSGS, suggesting a predilection for glomerular disease, and FSGS in particular, in this breed.
Histologic findings in FSGS include a pattern of glomerular injury that initially affects a minority of all glomeruli (ie, focal) and involves only a portion of the glomerular capillary tuft (ie, segmental). 12 Via light microscopy, FSGS is characterized by solidification (sclerosis) of a part of the affected capillary tuft with obliteration of capillary lumina by increased mesangial cell numbers and matrix. Adhesions (synechiae) between these sclerotic regions and Bowman’s capsule are common and are accompanied by hyalinosis, the accumulation of eosinophilic glassy material composed of entrapped plasma proteins within the glomerular tuft. In contrast to the focal nature of the histologic changes, the ultrastructural lesion of podocyte foot process effacement (FPE) is present in all glomeruli and affects greater than 50% of the capillary surface area in nonsclerotic areas. 12 This underlying podocytopathy, and eventual podocytopenia, results in the histologic lesion of FSGS. 9
The cause of the podocytopathy may be primary, in which idiopathic or genetic podocyte abnormalities result in podocyte dysfunction and loss, or secondary to podocyte injury due to a variety of insults such as glomerular hypertrophy and hyperfiltration (adaptive FSGS), systemic hypertension, obesity, or other factors that secondarily injure podocytes. 1,3,7,10,27,29 Regardless of its cause, FSGS may progress to diffuse and global glomerulosclerosis, eventually leading to end-stage renal disease.
The aim of this study was to describe the renal histopathologic, ultrastructural, and clinicopathologic abnormalities in 8 related, proteinuric, adult Miniature Schnauzers with FSGS and to evaluate potential causes of the observed lesions.
Materials and Methods
Animals and Clinical Findings
Renal necropsy samples from 8 related proteinuric Miniature Schnauzer dogs were submitted to the Athens Veterinary Diagnostic Laboratory between 2009 and 2014 as part of a study investigating the occurrence of renal disease in this breed, which was initially funded by the American Miniature Schnauzer Club. In this context, complete necropsy tissues were not requested. Medical records for each dog were reviewed and signalment, history, clinical signs, and clinicopathologic data (complete blood count, serum biochemical profile, urinalysis, and urine protein-to-creatinine ratio [UPC]) were compiled. Genetic testing for the protein losing nephropathy–associated variant gene alleles that are found in Soft Coated Wheaten Terrier (SCWT) dogs and Airedale Terrier dogs (PennGen, University of Pennsylvania School of Veterinary Medicine) was performed on buccal swabs from 2 dogs (dog Nos. 7 and 8). 22 Customized Taqman assays, developed to detect these protein losing nephropathy–associated variant alleles (NPHS1 and KIRREL2/Neph3, encoding slit diaphragm proteins nephrin and filtrin, respectively), were used.
Histopathology
Renal tissue was fixed in 10% formalin for at least 48 hours, trimmed, routinely processed for histology, sectioned at 3-µm thickness, and stained with hematoxylin and eosin, periodic acid–Schiff hematoxylin stain (PASH), Jones’ silver stain, PASH and Masson’s trichrome combination stain (PASH-MT), 3 or picrosirius red. Using PASH-stained slides, between 87 and 100 glomeruli were examined per case and the prevalence of segmental (involving less than 50% of the capillary tuft) sclerosis, global sclerosis, synechiae, hyalinosis, and cystic glomerular atrophy was determined. The diameters (in µm) of 20 superficial and midcortical glomeruli were measured in each dog (Photoshop CC 215.5). The largest glomeruli or glomeruli containing the vascular or urinary poles were preferentially measured.
The severity of renal cortical interstitial fibrosis was noted as absent, mild (widely scattered small foci), moderate (streaks extending through the cortex), or severe (diffuse) in PASH-MT–stained slides. Following subjective scoring, percentage area of Type I and III collagen was calculated as an objective measure of interstitial fibrosis using sections of kidney stained with picrosirius red. 21 Digital photomicrographs of 10 consecutive 200× renal cortical fields were obtained under polarized light and the percentage polarized collagen in each image was determined using image analysis software (Adobe Photoshop CS6 Extended, Version 13.0). The histogram function was used to select all colors visible in each image (representing polarized stained collagen), and pixels of positive staining were expressed as a fraction of total image pixels. Vascular lesions and the presence or absence of intraglomerular lipid were also noted.
Transmission Electron Microscopy
Renal tissue was retrieved from formalin-fixed, paraffin-embedded blocks (dog Nos. 1 and 2) or was received fixed in glutaraldehyde (dog No. 3) or 10% buffered formalin (dog Nos. 4–8). For dog Nos. 1 and 2, a portion of tissue was cored from the formalin-fixed, paraffin-embedded block and excess paraffin was removed by gentle heat followed by xylene, hydrated through an ethanol series, and washed several times in deionized water before postfixation. All samples were postfixed in 1% osmium tetroxide, serially dehydrated in graded alcohols, infiltrated with acetone (or propylene oxide) and epoxy plastic, and embedded in plastic. Sections of 1-µm thickness were cut and stained with toluidine blue to locate glomeruli before thin sections were cut at 60 nM using an ultramicrotome (Ultracut S; Reichert Technologies, Depew, NY), placed on copper slot grids coated with Formvar (Electron Microscopy Services, Hatfield, PA), and stained with uranyl acetate and lead citrate. The grids were examined in a transmission electron microscope (JEOL JEM-1210 120Kv), and multiple digital images were acquired (CAB, AMT XR41C Bottom-Mount CCD Camera).
Glomerular cellular components (endothelium, mesangial cells, and epithelial cells), the glomerular basement membrane (GBM), and the mesangial matrix were evaluated for ultrastructural abnormalities. Podocyte abnormalities, including the presence and severity of FPE, microvillous transformation, vacuoles, binucleation, protein resorption vacuoles, and electron-dense aggregates, were scored as not present (–), rare (+), common (++), or extensive (+++).
Statistical Analysis
Pearson correlation coefficients were determined to test for pairwise correlations between prevalence of glomerulosclerosis, percentage interstitial fibrosis, serum creatinine concentration, and glomerular size. An analysis of variance was used to test for differences in glomerular size between individual dogs. Tukey’s test was used to adjust for multiple comparisons. All analyses were performed using SAS (Version 9.3, Cary, NC). A P value < .05 was considered statistically significant.
Results
Pedigree Analysis and Clinical Findings
Seven dogs were within 3 generations of 2 common ancestors (Fig. 1). Although dog No. 8 was related, pedigree data were not available. Three additional dogs in the pedigree were reported to be proteinuric (UPC 3.8 in 1 dog; data not available in 2 dogs); the presence or absence of proteinuria was not determined in the remaining dogs in the pedigree. Dogs were euthanized due to cystic calculi and suspected pyelonephritis (dog No. 1), inflammatory bowel disease (dog Nos. 2, 3, and 5), suspected cardiac disease (dog No. 4), various age-related problems (dog No. 7), and chronic hepatitis (dog No. 8). Dog No. 6 died while under anesthesia during a routine ovariohysterectomy. Conditions diagnosed antemortem included hypertension (dog Nos. 2–5), intermittent urinary tract infections (dog Nos. 1, 2, 5, 7, and 8), gastritis and pancreatitis (dog Nos. 3 and 5), cholecystitis (dog No. 5), the presence of a mummified fetus (case No. 5), and subaortic stenosis (dog No. 8). Systemic blood pressure data were not available for dog No. 2, although reported as hypertensive. Blood pressure data for the remaining 3 dogs included a mean systolic pressure of 182 mm Hg (dog No. 3) and systemic blood pressures of 152/98 mm Hg (dog No. 4) and 169/105 mm Hg (dog No. 5); all 4 dogs were treated with amlodipine and enalapril.
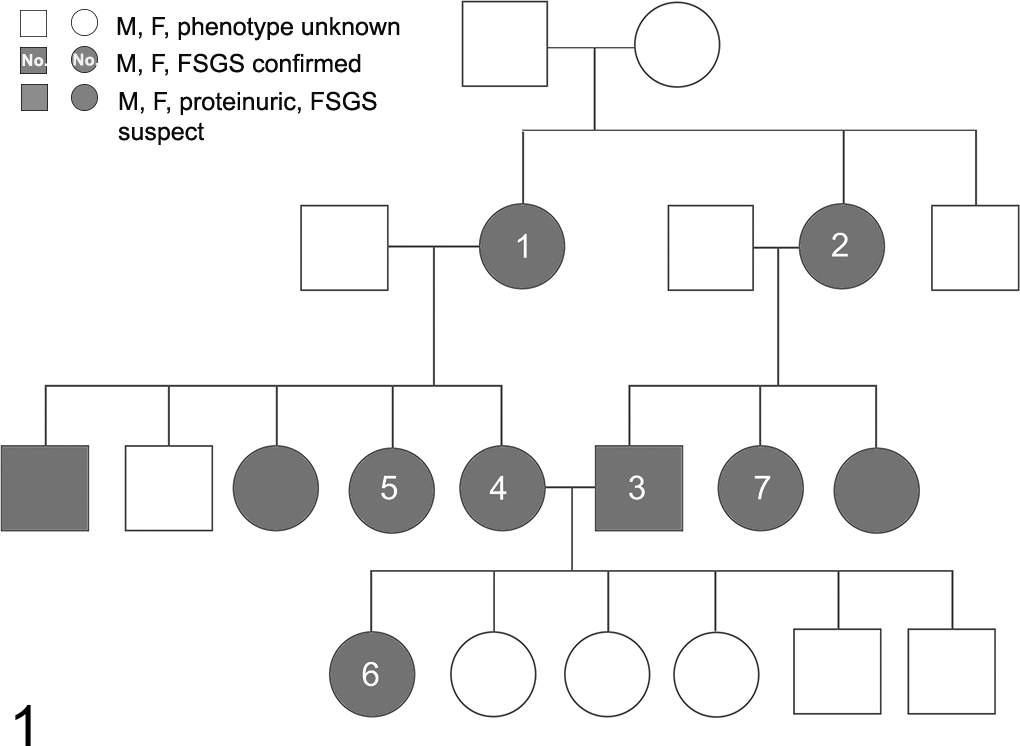
Serial urinalyses revealed persistent proteinuria in the absence of lower urinary tract disease in all dogs, with UPCs within 6 months of death ranging from 1.2 to 6.5 (Table 1). Proteinuria was first detected at 7.9 ± 2.2 (range, 5–11) years of age, with duration of proteinuria relative to death ranging from 2.4 months to 4.1 years. Once identified, proteinuria was persistent. In 7 of the 8 dogs in which UPCs were determined, the UPC was elevated for 2.2 ± 1.5 (range, 0.2–4.1) years prior to death. Overall, the average UPC for individual dogs was 4.5 ± 2.9 (range, 1.2–9.8). One dog (dog No. 8) had a UPC that was borderline proteinuric (0.3) at 6 years of age, followed by UPCs of 1.0 and 2.3 at 7 and 8 years of age, respectively. Serum albumin concentrations were within the normal reference range (2.7–3.9 g/dl) in 5 of the 8 dogs; the remaining 3 dogs (dog Nos. 1, 4, and 8) were mildly hypoalbuminemic (2.3–2.5 g/dl). Dog Nos. 2 and 5 were mildly azotemic within 3 months of death, whereas all other dogs had serum creatinine concentrations within the reference range (Table 1). Other clinicopathologic abnormalities included elevations in serum alkaline phosphatase concentration (dog Nos. 1 and 7), hypercholesterolemia (dog No. 8), and hypertriglyceridemia (dog Nos. 3–5). Serum triglyceride concentrations, measured in 3 dogs at 10 (202 mg/dl, dog No. 3), 7 (504 mg/dl, dog No. 4), and 8 (184 mg/dl, dog No. 5) years of age, were mildly 14 elevated (reference range, 26–108 mg/dl).
Clinical and Histologic Findings in 8 Miniature Schnauzer Dogs With Focal Segmental Glomerulosclerosis
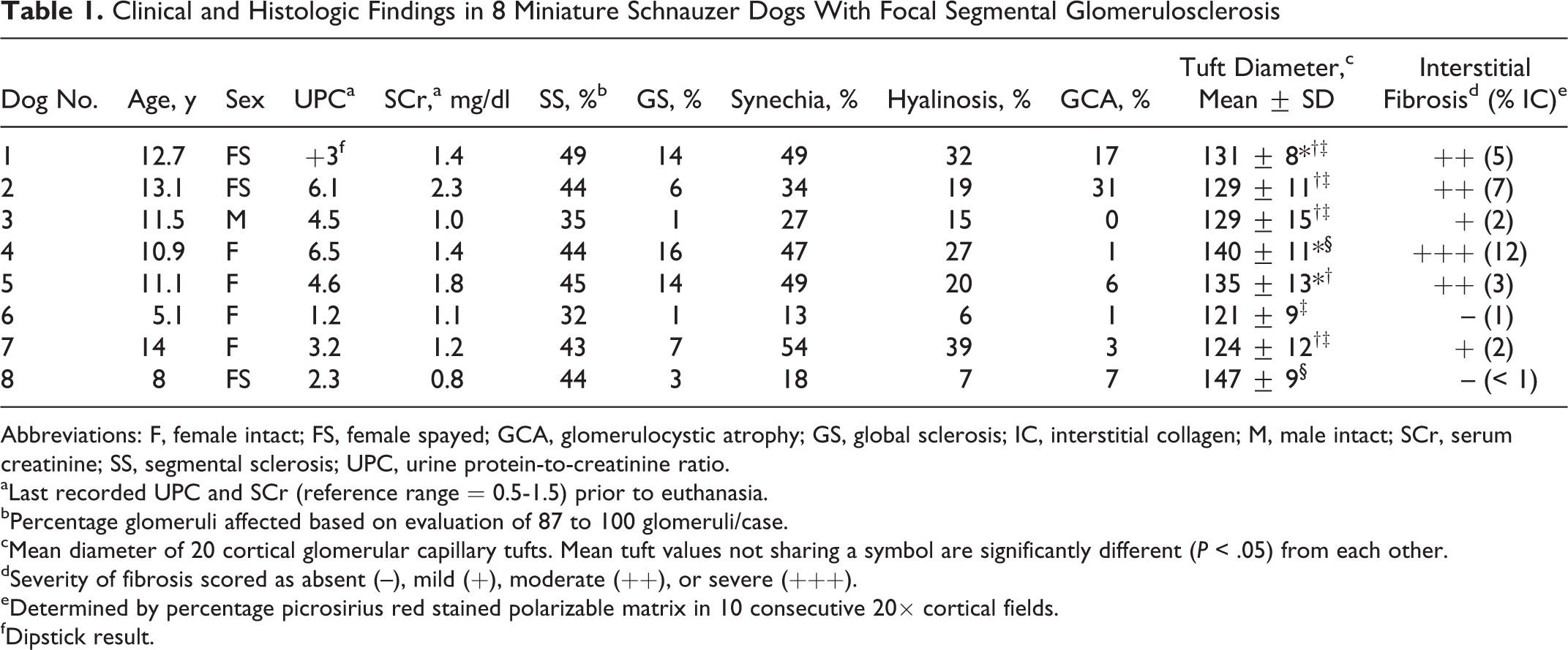
Abbreviations: F, female intact; FS, female spayed; GCA, glomerulocystic atrophy; GS, global sclerosis; IC, interstitial collagen; M, male intact; SCr, serum creatinine; SS, segmental sclerosis; UPC, urine protein-to-creatinine ratio.
aLast recorded UPC and SCr (reference range = 0.5-1.5) prior to euthanasia.
bPercentage glomeruli affected based on evaluation of 87 to 100 glomeruli/case.
cMean diameter of 20 cortical glomerular capillary tufts. Mean tuft values not sharing a symbol are significantly different (P < .05) from each other.
dSeverity of fibrosis scored as absent (–), mild (+), moderate (++), or severe (+++).
eDetermined by percentage picrosirius red stained polarizable matrix in 10 consecutive 20× cortical fields.
fDipstick result.
The 2 tested dogs (dog Nos. 7 and 8) were homozygous negative for the variant NPHS1 and KIRREL2 alleles present in SCWT dogs. 22
Light Microscopy
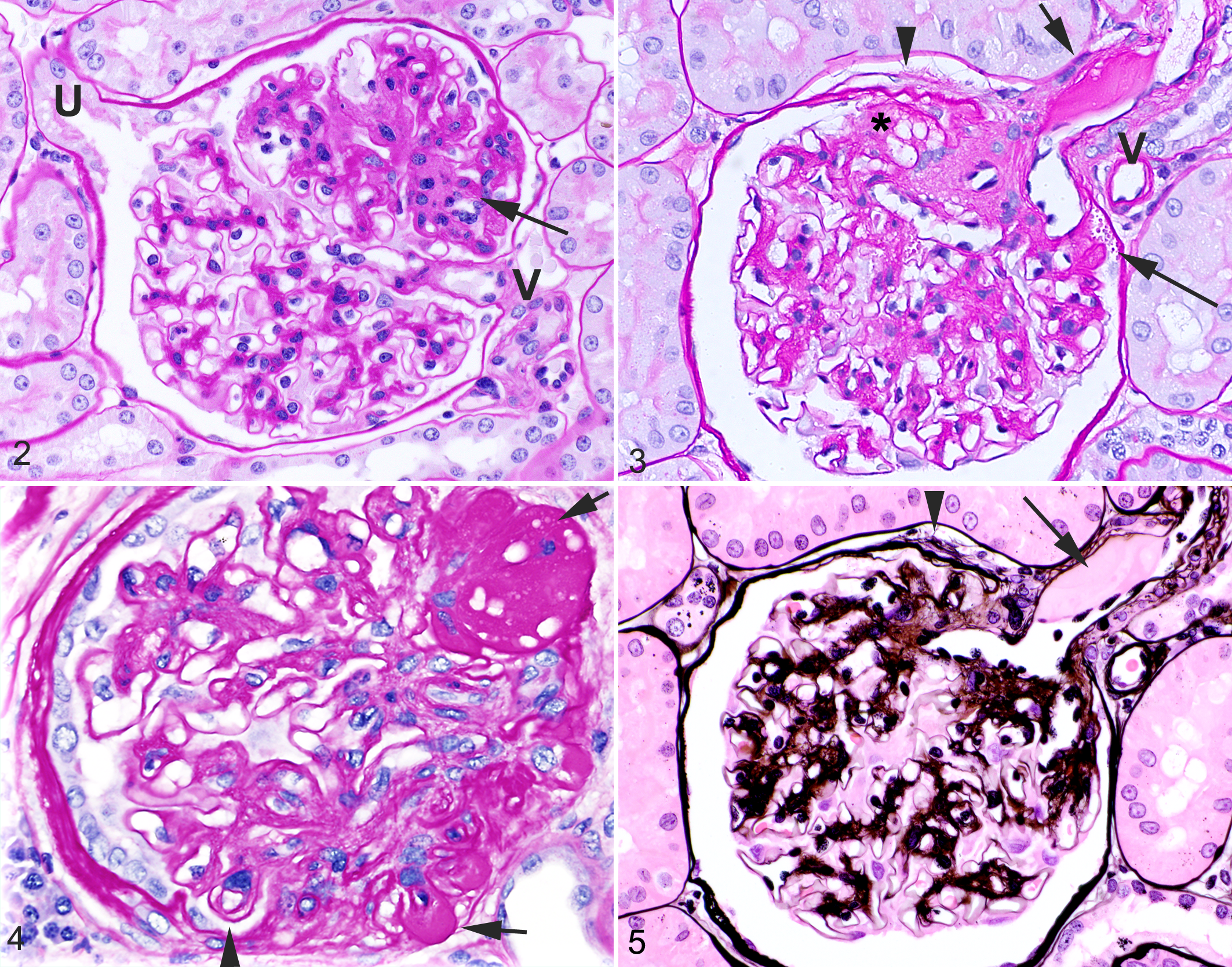
All 8 dogs had light microscopic lesions of focal (affecting 32% to 49% of examined glomeruli) and segmental (affecting up to 50% of the capillary tuft) glomerulosclerosis (Table 1). Global glomerulosclerosis was common, and when global and segmental sclerosis were considered together, sclerosis was present in greater than 50% of glomeruli in 5 of the 8 dogs. Within affected glomerular segments, there was solidification of the capillary tuft with attenuation or obliteration of capillary lumina associated with increased amounts of mesangial matrix and mild to moderate mesangial cell hyperplasia (Fig. 2). This sclerosis was often close to the vascular pole (perihilar), and this distribution was most evident in dogs having smaller segments of sclerosis (Figs. 2, 3). Older dogs had more extensive sclerosis, typically with extensive synechiae and hyalinosis, and the site of the sclerosis could often not be determined (Fig. 4). Synechiae were present in 13% to 54% of glomeruli. Glomerular capillary wall hyalinosis, representing entrapment of plasma proteins within the capillary wall, was observed in 6% to 39% of glomeruli, was best appreciated with PASH (Fig. 4), and was most commonly associated with synechiae. Splitting of the basement membrane of Bowman’s capsule also occurred at sites of capillary tuft adhesion (Figs. 3, 5). Synechiae and foci of hyalinosis were significantly less prevalent in the 2 youngest dogs (dog Nos. 6 and 8; Table 1) compared to all other dogs (P < .001 and P < .003, respectively).
Glomerular diameter is positively correlated with body weight in dogs, with dogs of similar weights to these Miniature Schnauzer dogs (6–8 kg) having mean glomerular diameters of 116 to 138 µm. 13 Six of the 8 dogs in the current study had mean glomerular diameters of 121 to 135 µm and 2 dogs had mean glomerular diameters of 140 and 147 µm (dog Nos. 4 and 8, respectively; Table 1). Mean glomerular diameters in dog Nos. 4 and 8 were significantly (P < .05) greater than mean glomerular diameters in 4 and 6 of the remaining dogs, respectively, suggestive of glomerular hypertrophy in dog Nos. 4 and 8.
Other histologic changes present in glomeruli included duplication of the glomerular basement membrane (double contours or “tram tracks”) indicative of mesangial cell interposition (Fig. 4), parietal epithelial cell hyperplasia and hypertrophy, and intracellular proteinaceous droplets indicative of protein leakage with resorption by parietal or visceral epithelial cells (Fig. 3). In dog Nos. 1 and 2, dilation of Bowman’s capsule with collapse and sclerosis of the glomerular capillary tuft was common (glomerulocystic atrophy; Table 1). Variable amounts of glomerular lipid were observed within some sclerotic areas (Fig. 3) and within hyaline deposits (Fig. 4) in 7 of the 8 dogs; lipid was not observed via light microscopy in dog No. 6.
Interstitial fibrosis was either mild or absent in 4 of the 8 dogs and severe in only 1 dog (dog No. 4). Initial subjective assessment of interstitial fibrosis using PASH-MT staining correlated well with subsequent quantification of interstitial collagen using picrosirius red staining (Table 1). Mean interstitial collagen (%) was 0.86 to 1.20, 1.62 to 1.70, 2.72 to 7.42, and 12.26 in dogs subjectively scored as absent (n = 2), mild (n = 2), moderate (n = 3), or severe (n = 1) interstitial fibrosis, respectively. Tubular changes of atrophy and dilation with mild lymphoplasmacytic inflammation were present in areas of interstitial fibrosis.
The relationship between percentage of sclerotic glomeruli (including those with cystic atrophy), degree of interstitial fibrosis, and serum creatinine concentration was assessed (Table 2). There was a significant positive correlation between prevalence of glomerulosclerosis and serum creatinine (r = 0.74, P = .04). There was also a trend for a positive correlation between glomerulosclerosis and interstitial fibrosis (r = 0.5, P = .17), and between interstitial fibrosis and serum creatinine concentration (r = 0.5, P = .21).
Glomerulosclerosis, Interstitial Fibrosis, and Serum Creatinine Concentration in 8 Miniature Schnauzer Dogs With Focal Segmental Glomerulosclerosis
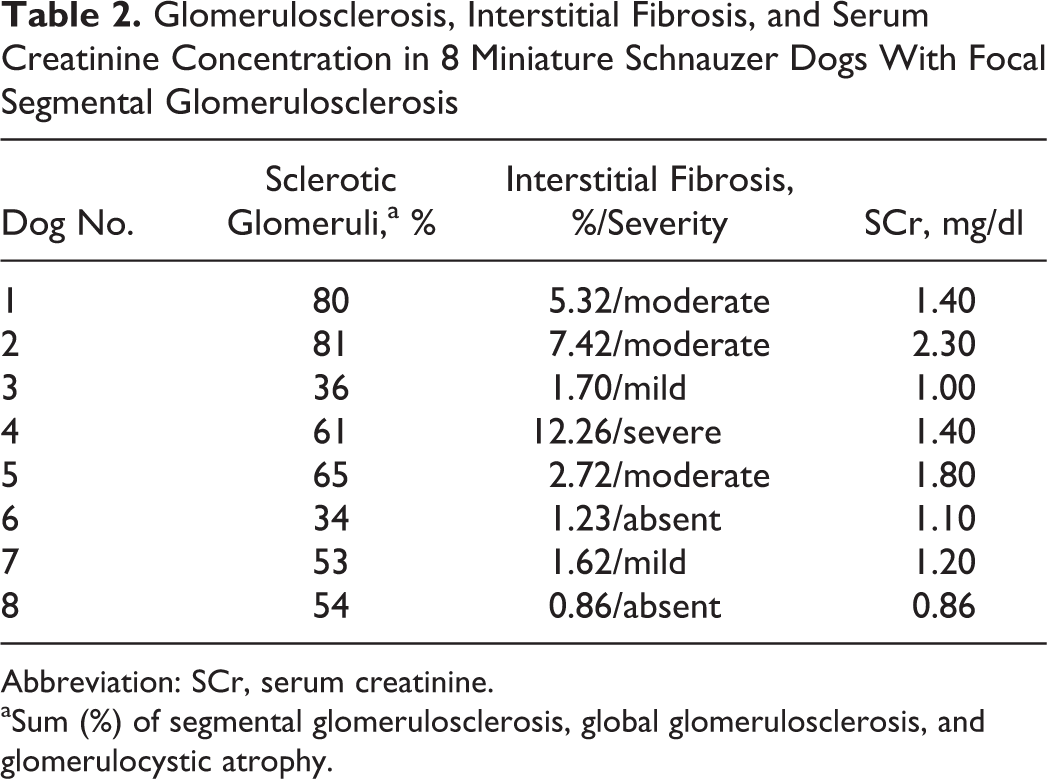
Abbreviation: SCr, serum creatinine.
aSum (%) of segmental glomerulosclerosis, global glomerulosclerosis, and glomerulocystic atrophy.
Seven of 8 dogs (not observed in dog No. 6) had prominent focal accumulations of subendothelial hyaline material within the afferent arterioles (hyaline arteriosclerosis; Figs. 3, 5). Two dogs (dog Nos. 2 and 4) had concentric vascular mural thickening by smooth muscle cells (hyperplastic arteriosclerosis) within small interstitial arteries.
Transmission Electron Microscopy
Glomeruli (2 from dog Nos. 1, 5, 6, and 7; 3 from dog No. 2; and 4 from dog Nos. 3, 4, and 8) were examined ultrastructurally. Whereas segmental sclerosis was not present in all examined glomeruli, all glomeruli had ultrastructural evidence of a severe podocytopathy (Table 3). Foot process effacement was present in all examined glomeruli; intact foot processes were uncommonly observed (Figs. 6, 7). Other nonspecific lesions indicative of podocyte injury included microvillous transformation in 6 dogs (Fig. 6), cytoplasmic pseudocysts in 5 dogs (Fig. 8), intracytoplasmic electron-dense lysosomes (protein reabsorption droplets) in 6 dogs (Fig. 9), and binucleation in 3 dogs (Fig. 10). Variable amounts of lipid were observed via transmission electron microscopy in all 8 dogs, with intraglomerular lipid present as small vacuoles within the subendothelial aspect of the GBM or within the mesangial matrix (Figs. 6, 7) or within foci of hyalinosis (Fig. 11).
Podocyte Ultrastructural Lesions in 8 Miniature Schnauzer Dogs With Focal Segmental Glomerulosclerosisa

aNot present (–), occasional (+), common (++), extensive (+++).
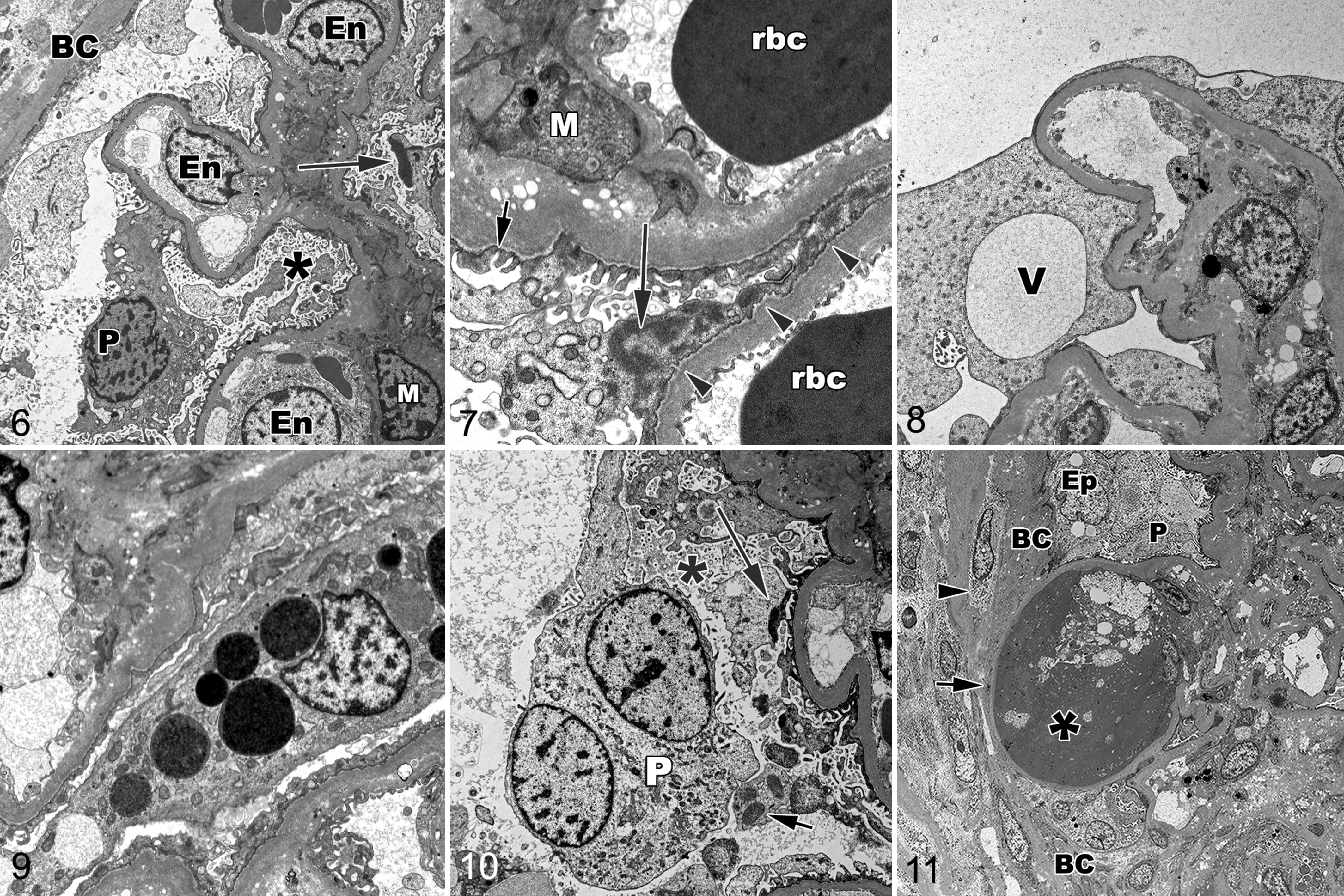
Distinctive, aggregated, electron-dense material was present in the cytoplasm of some podocytes in all 8 dogs (Figs. 6, 7, 10). This material was present within podocyte processes toward the cell body, often in close contact with the cell membrane, and granular in appearance. Similar material was not observed in glomerular parietal epithelial cells, endothelial cells, or mesangial cells.
Other glomerular changes included mesangial interposition (Fig. 7) and multifocal irregular thickening with mild rarefaction of the GBM. No extracellular fibrillary deposits or immune complexes were identified.
Discussion
Miniature Schnauzer dogs have a breed predisposition for developing proteinuric renal disease. 14,28 A finding of FSGS is present in the majority of biopsied proteinuric Miniature Schnauzer dogs and was present in all 8 related, proteinuric dogs of this study. 15,28 Dogs and humans with FSGS exhibit a morphologic pattern of glomerular injury that is a reflection of an underlying podocytopathy with podocyte loss and resultant glomerular scarring. 5,8,10 –12,24,27,29
Podocytes cover the abluminal surface of glomerular capillary loops via interdigitating podocyte foot processes that are bridged by the principle size-selective barrier, the slit diaphragm. 29 Following podocyte injury, there is active reorganization of the actin cytoskeleton and FPE with flattening and widening of podocyte processes, decreased slit diaphragm density, and proteinuria. 29 This universal and reversible podocyte response to injury results in increased contact with the GBM and is thought to be an adaptive response to prevent podocyte loss. 20 However, if injury persists, more severely affected podocytes may become detached from the GBM and are then lost in the urine. As podocytes are terminally differentiated cells with limited or no ability to proliferate following injury, remaining podocytes respond by expansion to cover areas of denuded GBM. Once podocyte density decreases to a critical threshold, further podocyte loss results in exposure of the underlying GBM, capillary adherence to Bowman’s capsule, and focal sclerosis. 11,12 Over time, the sclerosis may progress to involve more glomeruli and a greater proportion of the capillary tuft. 12
A variety of factors have been implicated in the pathogenesis of canine FSGS, including genetic 22 and acquired 4,7 factors. Whereas approximately 80% of FSGS cases in humans are idiopathic, a number of genetic podocytopathies have been recognized in humans and dogs. 1,11,22,27 The protein-losing nephropathy in SCWT dogs is associated with gene mutations encoding the slit diaphragm proteins nephrin and Neph3/filtrin, 22 and some of the renal pathologic findings in these Miniature Schnauzer dogs are similar to those in affected SCWT dogs. 30 Most of the proteinuric SCWT dogs have lesions of FSGS with synechiae and hyalinosis. Whereas SCWT dogs are often hypoalbuminemic, azotemic, and hypercholesterolemic, these clinicopathologic features typically are not present in proteinuric Miniature Schnauzer dogs. In this study, despite long-standing proteinuria, hypoalbuminemia was mild and occurred in only 3 of 8 dogs, only 2 dogs were azotemic, and serum cholesterol was elevated in only 1 dog. Furthermore, 2 out of 2 Miniature Schnauzer dogs in the present study were negative for the specific gene mutations present in proteinuric SCWT dogs.
Although a genetic factor is presumed to have played a role in the development of FSGS in these related Miniature Schnauzer dogs, statements about the potential pattern of inheritance cannot yet be made. In humans, most genetic podocyte defects are characterized by a recessive pattern of inheritance, often involve mutations in slit diaphragm proteins, and present clinically as early-onset nephrotic-range proteinuria. 1,12 In contrast, autosomal dominant forms of FSGS are less common, are more often associated with mutations in podocyte cytosolic or cell membrane proteins, and are usually associated with clinically identifiable disease occurring in late adolescence or adulthood. Identified causes of autosomal dominant FSGS resulting in disease limited to the kidney include mutations in podocyte genes encoding for CD2AP (a slit diaphragm protein linking podocin and nephrin to actin), transient receptor potential cation channel 6 (a cell-membrane–associated calcium transporter protein), α-actinin-4 (a cytosolic actin-binding protein), and inverted formin-2 (a cytosolic actin-regulating protein). 1,12 Light microscopic lesions in all of these podocytopathies are those of FSGS with eventual, secondary, ischemic tubulointerstitial changes. Whereas ultrastructural lesions are also nonspecific indicators of podocyte injury, humans and mice with α-actinin-4 mutations have prominent cytoplasmic electron-dense aggregates within podocytes similar to those observed in these Miniature Schnauzer dogs. 16 –18 Normal α-actinin-4 binds to and cross-links F-actin filaments that are primarily located in podocyte foot processes. 12 The FSGS-producing mutations result in α-actinin-4 conformational changes that expose a normally buried actin-binding site, leading to a gain of function. 31 This increased binding causes increased podocyte rigidity and defects in podocyte adhesion to the GBM, which would be expected to result in increased podocyte loss and FSGS. 12,31,32 The electron-dense aggregates represent accumulations of α-actinin-4 and actin, 17 and although more prominent in humans with mutated α-actinin-4, they may occasionally be observed in other glomerular diseases (J. M. Henderson, personal communication, 26 September 2016). Similarly, review of electron micrographs from dogs with various proteinuric renal diseases by one of the authors (CAB) revealed similar, although less prominent, electron-dense aggregates in 1 of 10 dogs with immune-complex glomerulonephritis (data not shown). Therefore, although all 8 Miniature Schnauzer dogs in this report had prominent cytoplasmic electron-dense aggregates similar to those described in humans with FSGS due to α-actinin-4 mutation, the significance of these deposits remains to be determined.
Histologically, FSGS may be classified morphologically into collapsing, tip, cellular, perihilar, or NOS (not otherwise specified) subtypes. 12 Regardless of the underlying cause, the most common histologic subtype of FSGS in humans and in cases described in dogs is NOS, which lacks features present in the other 4 categories. 9,12 In the dogs in this study, the FSGS was the perihilar variant, characterized by segmental hyalinosis and sclerosis involving the vascular pole in the majority of glomeruli with segmental lesions. In humans, the perihilar subtype is common in adaptive FSGS, hypertensive nephrosclerosis, reflux nephropathy, oligomeganephronia, and in FSGS associated with mutations in α-actinin-4. 12,16 Although urinalyses were not performed in dogs at an early age in this study, at least 1 dog did not have proteinuria (UPC < 0.5) at 5 years of age, suggesting that proteinuria in this breed, like hypertriglyceridemia, develops later in life. 14 Despite persistent proteinuria, only 2 of the 8 dogs were azotemic (serum creatinine > 1.5 mg/dl) and none were uremic. Likewise, humans with α-actinin-4 mutations develop FSGS that manifests as adult-onset proteinuria that, in one report, progressed to kidney failure in only 2 of 12 patients. 16 The late onset of renal dysfunction in some cases of familial FSGS may reflect a decreased capacity of podocytes with structural abnormalities to withstand normal stresses, resulting in podocyte injury and loss.
It has been proposed that idiopathic hypertriglyceridemia, occurring in greater than 75% of Miniature Schnauzer dogs by 10 years of age, plays a role in the podocyte injury observed in this breed. 15 A positive correlation between serum triglyceride concentration and UPC has been reported in Miniature Schnauzer dogs. 14 In that study, all proteinuric Miniature Schnauzer dogs were hypertriglyceridemic, whereas proteinuria was not observed in any Miniature Schnauzer dog with a normal serum triglyceride concentration. Among dogs with hypertriglyceridemia, 40% were not proteinuric, suggesting that additional factors may contribute to podocyte injury by increasing the susceptibility of the podocyte to lipid-associated injury. 5,14 Serum triglyceride concentrations were measured in 3 of the dogs in this study, and all were mildly hypertriglyceridemic. Variable amounts of lipid were present in foci of hyalinosis, in mesangial matrix, and within the GBM in all dogs in this study, as has been previously described in proteinuric Miniature Schnauzer dogs. 15 Whereas extracellular lipid vacuoles are described in areas of hyalinosis as a common nonspecific finding in humans with FSGS, 12 the accumulation of lipid within the mesangium and GBM is not commonly described. Glomerular lipid emboli, a unique finding described in some proteinuric Miniature Schnauzer dogs, 14 were not identified in the dogs of this study. These findings suggest that hypertriglyceridemia and glomerular lipid accumulation may contribute to development of FSGS, although this has yet to be proven.
Glomerular arteriolar hyalinosis was present in 7 of the 8 dogs in this study. Arteriolar hyalinosis has been associated with endothelial damage due to hypertension, diabetes, and calcineurin inhibitor (such as cyclosporine A) toxicity. 26 Only 1 of the dogs of the present study had marked hypertension, 2 dogs were moderately hypertensive, and 1 dog was normotensive. Although blood pressure data were not available for the remaining dogs, 1 additional dog was reported to be hypertensive. In another study, systemic blood pressure was typically reported as normal or only mildly elevated in proteinuric Miniature Schnauzer dogs. 14 Taken together, these data suggest that systemic hypertension is unlikely to be the cause of the lesion in these dogs. However, intraglomerular hypertension may be present in the absence of systemic hypertension. Glomerular hypertension, along with glomerular hypertrophy and hyperfiltration, occurs as a compensatory response to acquired or congenital nephron paucity and may result in podocyte injury and adaptive FSGS. 4,7 Adaptive FSGS is characterized histologically by perihilar glomerulosclerosis and glomerular hypertrophy, and ultrastructurally by focal FPE. 12 Whereas perihilar sclerosis was often present in these dogs, glomerulomegaly was absent in 6 of the 8 dogs in this study. One of the 2 dogs with mild glomerular hypertrophy had severe interstitial fibrosis and severe FSGS. Adaptive FSGS secondary to glomerular hypertension and hypertrophy plays a role in the progression of many forms of chronic kidney disease and was likely present in this dog in addition to the primary FSGS. A second dog with significantly larger glomeruli had minimal interstitial fibrosis and only moderate FSGS. It is interesting that this dog had occasional immature glomeruli and cystic glomerular atrophy, lesions that have been described in juvenile onset renal disease occurring in this breed. 23 Although nephron numbers were not quantified in this dog, congenital nephron paucity with compensatory hypertrophy (oligomeganephronia) is possible.
Ultrastructurally, adaptive FSGS is characterized by FPE that is typically limited to areas of sclerosis. In contrast, the dogs in this study had widespread FPE with only focal foot process preservation. Therefore, although adaptive FSGS may be present in some Miniature Schnauzer dogs, it is likely a secondary lesion and not the primary cause of the FSGS. Whereas the lipid abnormalities in this breed and/or glomerular hypertension may result in endothelial and podocyte injury with resultant FSGS, the presence of an underlying inherited podocytopathy leading to increased susceptibility to these podocyte stressors should also be considered.
Single binucleated podocytes were observed ultrastructurally in 3 of the 8 dogs in this study. In an ultrastructural review of 164 diseased human kidneys, podocyte mitotic figures were present in 1 cell in 1 case, and 6 cases had evidence of 2 or more nuclei in a podocyte. 25 In humans, these binucleated podocytes were hypertrophied, had FPE, and were interpreted to arise following mitosis without cytokinesis. Binucleated podocytes are frequently seen in the urine of human patients with glomerular disease, suggesting that this response to injury may be more likely to result in detachment. 24 Other podocyte ultrastructural lesions present in the dogs of this study included FPE, microvillous transformation, resorption granules, and vacuoles.
In this study, we describe the histologic, electron microscopic, and clinical findings in 8 related Miniature Schnauzer dogs with FSGS. The dogs presented clinically with persistent proteinuria. In most affected dogs, the clinical disease was characterized by adult-onset proteinuria with minimal or no hypoalbuminemia, slow progression of azotemia that did not reach end-stage renal disease, and modest hypertension. All dogs had renal histologic lesions of FSGS with synechiae, hyalinosis, and lipidosis. Whereas most ultrastructural findings were those of nonspecific podocyte injury, unusual electron-dense aggregates were identified in podocytes of all affected dogs. Although these aggregates could represent nonspecific degenerated material or reorganized actin complexes within injured podocytes, their similarity to the aggregates observed in human patients with FSGS due to mutations in α-actinin-4 is interesting and warrants genetic testing for a similar mutation in Miniature Schnauzers. Acquired insults, such as hyperlipidemia and hypertension, may be more damaging to glomeruli that are already fragile due to an underlying genetic podocytopathy. However, with or without an underlying genetic podocyte defect, injury due to lipid abnormalities and glomerular hypertension, along with comorbid conditions such as inflammatory bowel disease or pancreatitis, may be sufficient to cause significant podocyte injury and FSGS in these dogs. In addition to the podocyte lesions, variable amounts of glomerular lipid were identified within endocapillary locations in all dogs, suggesting a relationship between progression of the glomerular injury and lipid abnormalities. Finally, whereas vascular changes in the afferent arterioles suggest that glomerular hypertension may also be present in these dogs, adaptive FSGS does not appear to be an important component of this disease. Serial monitoring of UPC in older Miniature Schnauzers and prompt diagnosis with biopsy may be warranted for Miniature Schnauzers with unexplained and persistent proteinuria, as palliative therapy is available to minimize further podocyte damage and slow the progression of FSGS. 6 Further studies will be needed to fully characterize FSGS in Miniature Schnauzers.
Footnotes
Acknowledgements
We thank Mary Ard from the Electron Microscopy Service at the Athens Veterinary Diagnostic Laboratory for her technical assistance, Dr Paula Henthorn and Michael Raducha at the University of Pennsylvania School of Veterinary Medicine for genetic studies, and The American Miniature Schnauzer Club Health Committee for supporting the submission of postmortem kidney samples in order to better characterize the renal disease occurring in this breed.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support from the Gray Lady Foundation for the research, authorship, and/or publication of this article.
