Abstract
Seventeen cases of canine peripheral nerve sheath tumors (PNSTs), 11 malignant PNSTs (MPNSTs), and six benign PNSTs (BPNSTs) were examined. The prognosis in five of six dogs with BPNSTs was excellent, whereas all dogs with MPNSTs died within 2 years after the last surgical resection. One BPNST formed a recurrent mass with features of a MPNST. Histopathologically, the predominant tumor cell of MPNSTs was either spindle or round in shape with epithelioid characteristics. Other atypical cells had abundant granular cytoplasm or were multinucleated giant cells with periodic acid-Schiff-positive cytoplasmic globules. Furthermore, two MPNSTs contained cartilaginous and osseous metaplasia. On the contrary, most BPNSTs exhibited typical features of schwannoma or neurofibroma, whereas two BPNSTs had atypical morphology. One BPNST consisted of epithelioid cell proliferation with some tumor cells revealing nuclear atypia. Immunohistochemically, the expression of vimentin (100%), S-100 (73%), nerve growth factor receptor (NGFR, 64%), and myoglobin (64%) was commonly found in MPNSTs. The two BPNSTs with atypical histologic appearances were positive for vimentin, S-100, NGFR, and neuron-specific enolase, and one of these had moderate immunore-activity for cytokeratin. Most BPNSTs were positive for glial fibrillary acidic protein, as well as S-100 and NGFR. Although most rhabdomyosarcomas (RMSs) and canine hemangiopericytomas (CHPs) also showed focal immunoreactivity for S-100, most RMSs were intensely positive for myoglobin and negative for NGFR. Most CHPs (80%) exhibited focal α-smooth muscle actin (α-SMA) expression, whereas all PNSTs were negative. These results indicate that immunohistochemistry for NGFR and α-SMA might be useful for differentiating canine PNSTs from RMSs or CHPs, respectively.
Canine peripheral nerve sheath tumors (PNSTs) arise from Schwann cells, perineurial fibroblasts, or both. 17,25 These tumors including schwannomas (neurilemmoma) and neurofibromas were classified as PNSTs by the World Health Organization (WHO) in 1999. Based on the morphologic and biologic behavior, PNSTs are divided into benign PNST (BPNST) and malignant PNST (MPNST) forms with several morphologic features. 17,25,41 In human MPNSTs, variable histologic patterns and heterogenous differentiation have been reported, 10–12,39,42 including epithelioid MPNST 27,29 and MPNST with divergent differentiation such as rhabdomyoblastic (malignant Triton tumor), 5,8,9,24 cartilaginous, osseous, angiomatous, 31 and glandular forms, 6,37,48 or their complex. 47 Similar representation such as epithelioid type, 36 melanotic type, 20,32 cartilaginous and osteogenic, 2 or glandular epithelial differentiation have been found in MPNSTs in dogs or other animals. 25,33,41 Furthermore, atypical cell morphology such as tumor cells with eosinophilic cytoplasmic globules may also occur. 26 Although most PNSTs with divergent differentiation are malignant, BPNSTs do occasionally include glandular components. 4 In addition, several BPNSTs have atypical changes or epithelioid patterns, which may make distinguishing BPNST from MPNST difficult. 23
Immunohistochemically, PNSTs are generally positive for vimentin and S-100, whereas anaplastic or heterogenous MPNSTs tend to be negative for S-100. 12,17,25,39,41,42 Some authors have speculated that the absence of S-100 supports a diagnosis of malignancy. 36 Nerve growth factor receptor (NGFR), expressed in the perineurium of normal peripheral nerves and neoplastic Schwann cells, was frequently demonstrated in human PNSTs. 19,35 To our knowledge, there have been few studies of NGFR immunoreactivity in canine PNSTs. Canine MPNSTs may show variable histologic patterns as is seen with human MPNSTs, although their characteristics are not well established. Therefore, the diagnosis of MPNSTs is often confused with other soft tissue tumors. The present study describes the pathologic features of 11 MPNSTs and six BPNSTs and emphasizes the variety of morphologic features in MPNSTs and some atypical patterns of BPNSTs. In addition, the immunohistochemical characteristics of PNSTs and other soft tissue tumors including one synovial sarcoma (SS), four rhabdomyosarcomas (RMSs), and five canine hemangiopericytomas (CHPs) are described.
Materials and Methods
Case studied
All samples were obtained from biopsy cases between 1999 and 2002 at the Department of Veterinary Pathology, Miyazaki University. Eleven cases of MPNSTs and six BPNSTs were diagnosed by general histopathologic examinations. Clinical data including canine breeds, age and sex, and size and location of tumors are summarized in Table 1. Dog Nos. 7 and 9 are recurrent tumors of dog Nos. 12 and 8, respectively. The average age of dogs with MPNSTs (10.6 years) was higher than those with BPNSTs (8.2 years). The dogs diagnosed with BPNSTs, except for dog No. 12, were alive without evidence of metastasis or recurrence, whereas all 11 dogs with MPNSTs died within 2 years after the last surgical excision. Although little information on the treatments and cause of death could be collected, metastasis or recurrence occurred in most cases.
Histopathology
Tissue samples were fixed in 10% formalin. Paraffin sections of 4 µm thickness were stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), Masson's trichrome, alcian blue (pH 2.5), and Watanabe's silver impregnation. Malignancy was based on the degree of cellular atypia and pleomorphism as well as other histologic features.
Immunohistochemistry
Tissues of 11 MPNSTs and six BPNSTs together with control cases including one SS, four RMSs, and five CHPs were examined. For the primary antibodies, mouse monoclonal antibodies against vimentin (prediluted product, Dako-Japan, Kyoto, Japan), NGFR (1 : 50, Novocastra, Newcastle, UK), neuron-specific enolase (NSE, prediluted product, Dako-Japan), α-smooth muscle actin (α-SMA, prediluted product, Dako-Japan), cytokeratin AE1/AE3 (cytokeratin, prediluted product, Dako-Japan), myelin basic protein (MBP, prediluted product, Zymed, San Francisco, CA), and proliferating cell nuclear antigen (PCNA, prediluted product, Zymed) and rabbit antisera against S-100 (prediluted product, Dako-Japan), glial fibrillary acidic protein (GFAP, prediluted product, Dako-Japan), myoglobin (1 : 400, Dako-Japan), and chromogranin A (prediluted product, Dako-Japan) were used. The immunoreactivity for factor VIII–related antigen (von-Willebrand factor, prediluted product, Dako-Japan) was examined in 11 MPNSTs. The sections for anti-vimentin, NGFR, myoglobin, α-SMA, factor VIII–related antigen, MBP, GFAP, cytokeratin, and chromogranin A were pretreated by hydrated autoclave for 5 minutes at 121 C. No pretreatments were applied for antibodies against S-100, NSE, and PCNA. Each slide was treated with 0.3% hydrogen peroxide in methanol for 30 minutes at room temperature. Immunohistochemistry was performed using an Envision polymer reagent (Dako-Japan). For visualization, 3,3′-diaminobenzidine-4 HCl (Sigma, St. Louis, MO) was used. Tissues were stained with Mayer's hematoxylin for nuclear counterstaining in all sections except for PCNA slides, in which methyl green stain was used.
Immunohistochemical evaluation and grading
When immunoreactivity was multifocal or scattered, the positive index was calculated from 10 randomly selected high-power fields (400×) using image-processing software (Win Roof ver 3.1, Mitani corporation, Fukui, Japan). The immunoreactivity was scored as follows: −, negative; +, 0–10% positive; ++, 10–30% positive; +++, 30–50% positive; and ++++, >50% positive. Labeling indices were estimated by counting the number of PCNA-labeled cells or mitoses per 1,000 tumor cells.
Results
Histologic features of MPNST
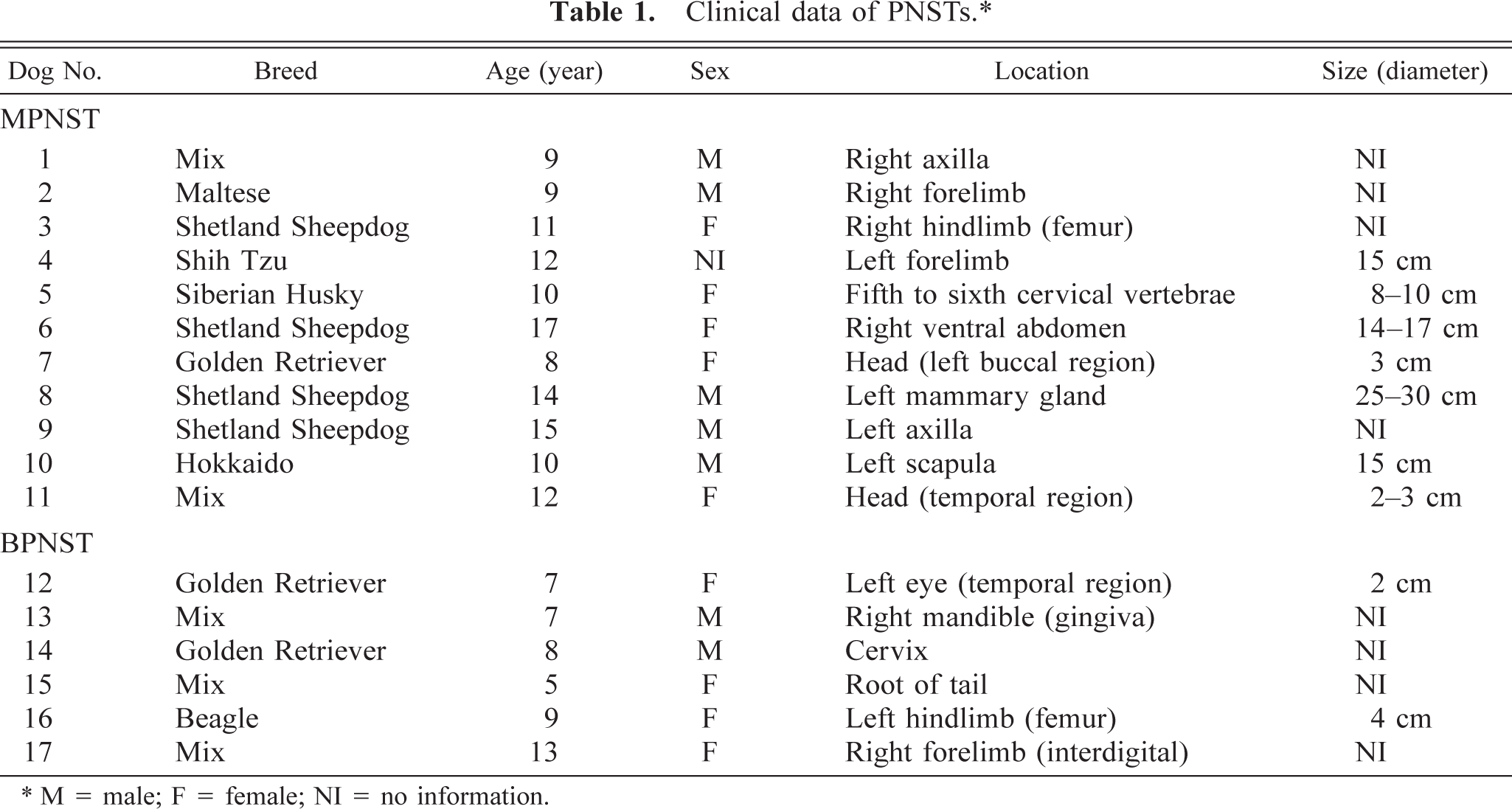
Tumor masses in MPNSTs that were not circumscribed by connective tissue and neoplastic cells often exhibited an aggressive behavior, high cellularity, and various morphologic patterns. Atypical mononuclear or multinucleated cells were consistently observed. More than four mitotic figures per high-power field (400×) were found in most cases. Necrotic foci accompanied by pseudopalisading (Fig. 1) and infiltrates of various numbers of lymphocytes, plasma cells, and macrophages were common. All 11 MPNSTs were divided further into three groups based on their morphologic features.
MPNST; dog No. 1. Necrotic focus is accompanied by nuclear pseudopalisading in MPNST. HE. Bar = 80 µm.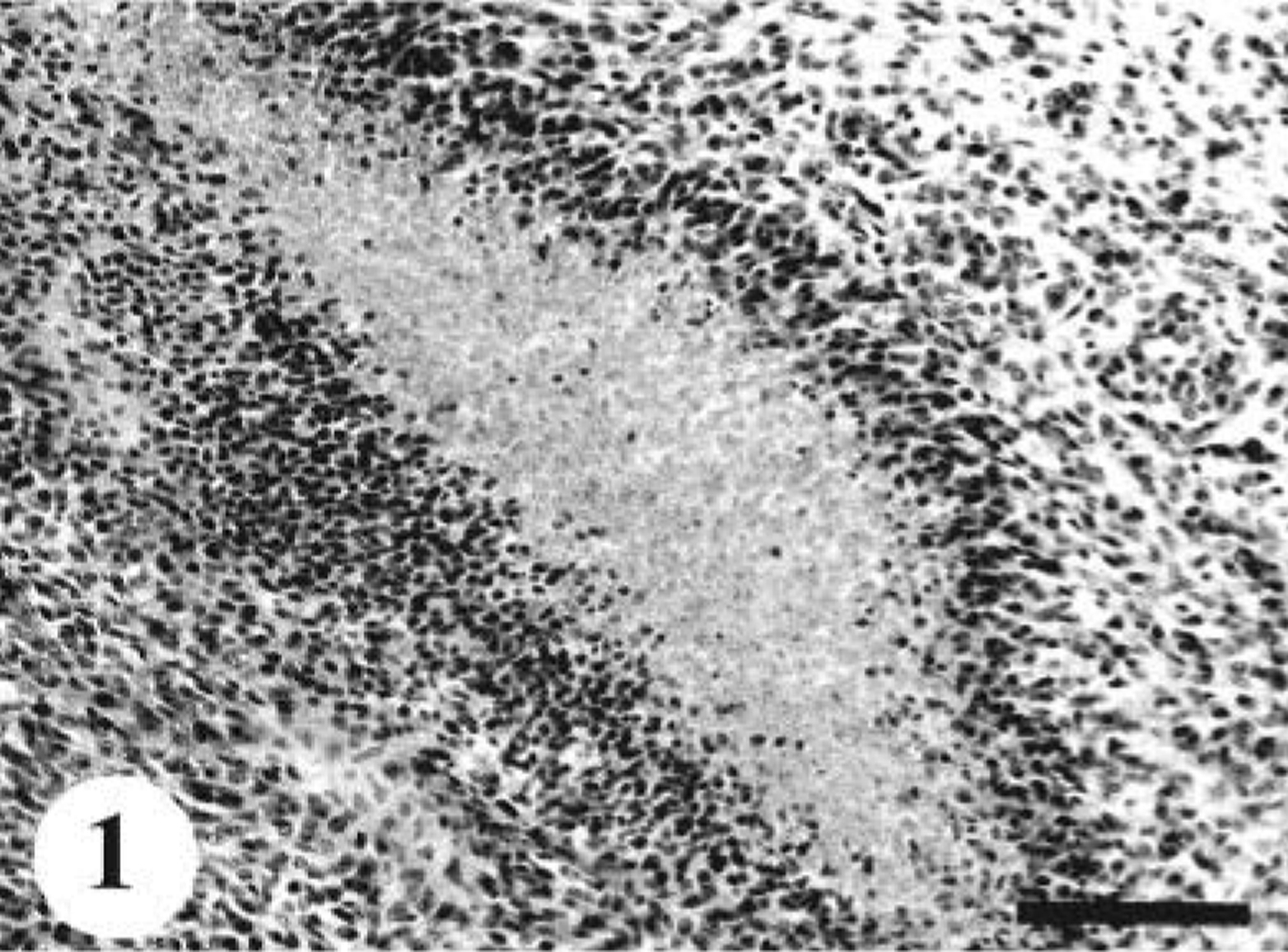
The tumors in dog Nos. 1 and 4 were composed mainly of a homogeneous population of round cells (Fig. 2). These round cells were arranged in sheets or cords with or without a meshwork of reticular fibers. The nuclei were round or oval and vesicular with prominent nucleoli. In dog No. 1, there were neoplastic foci consisting of a proliferation of large cells with pale eosinophilic cytoplasmic granules (Fig. 3). These granules were positive for PAS and alcian blue. In dog No. 4, rosettelike formations of tumor cells were rarely seen.
MPNST; dog No. 1. Round neoplastic cells with scant cytoplasm and round nuclei are arranged in a cordlike pattern. HE. Bar = 40 µm.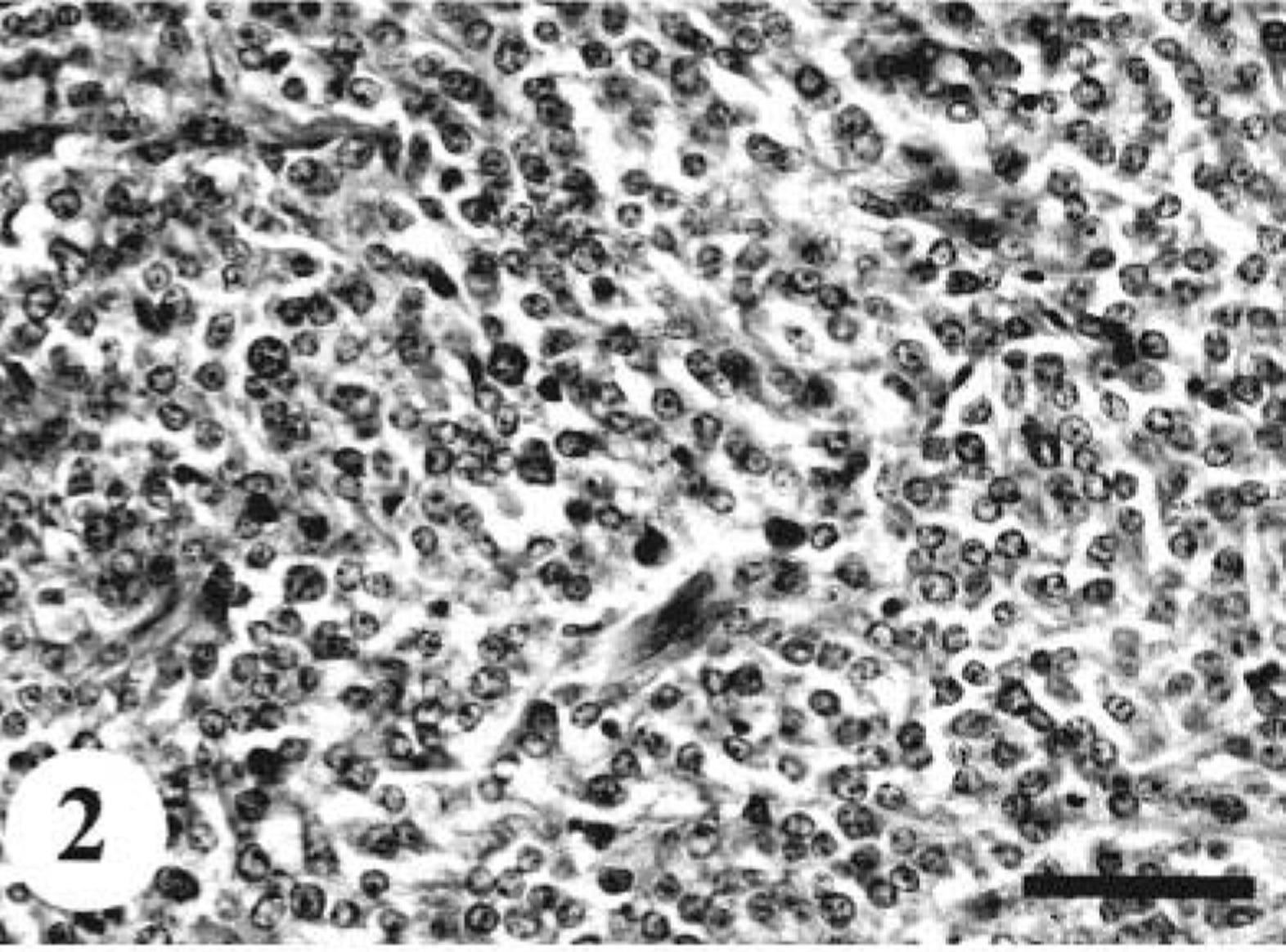
MPNST; dog No. 1. PAS-positive granular cells are focally proliferated in round cell type MPNST. PAS. Bar = 25 µm.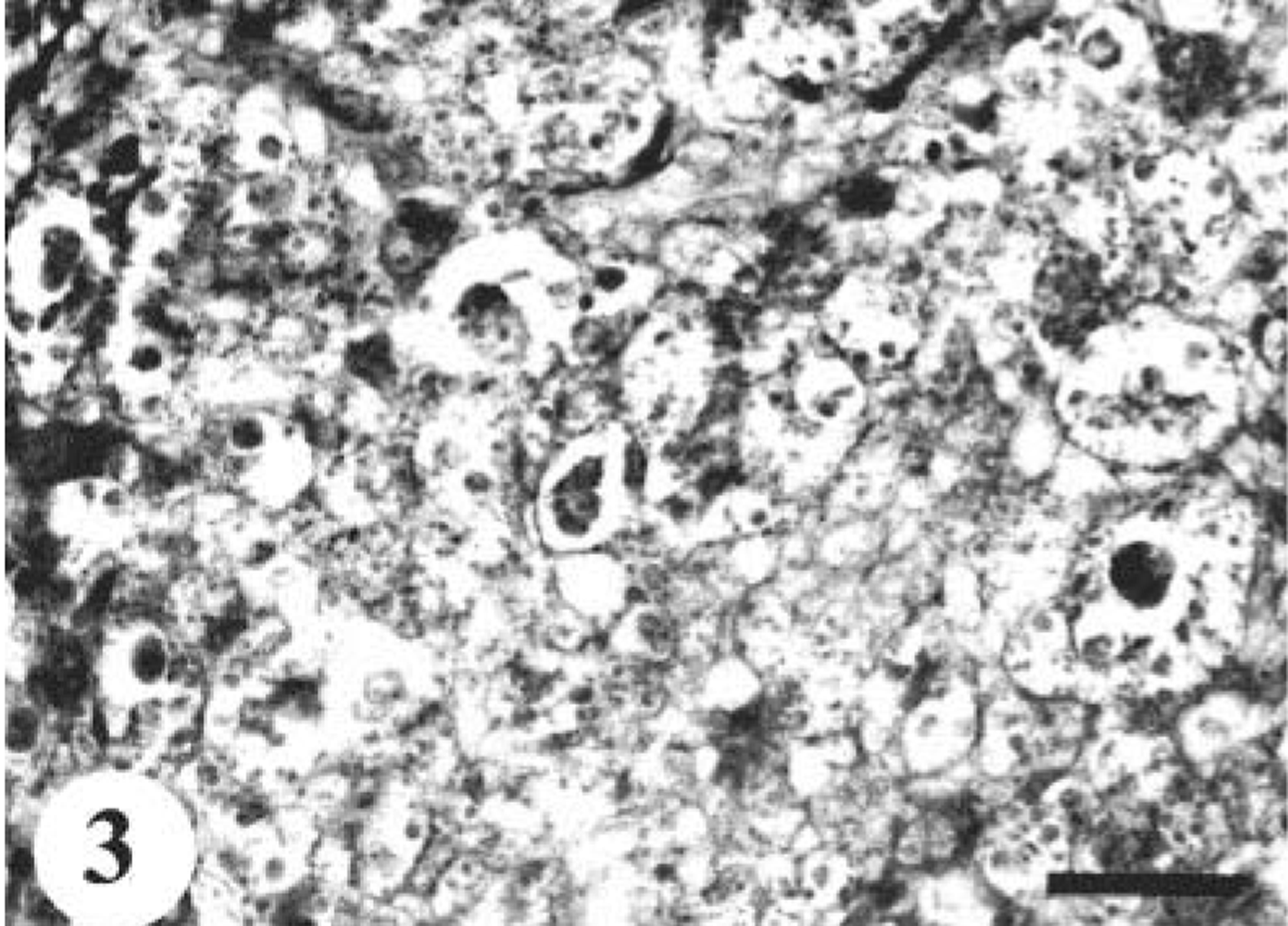
In five cases (dog Nos. 2, 3, 5, 7, and 11), fusiform or spindle cells arranged in densely or loosely sweeping fascicles, interlacing whorls, or storiform patterns were predominant (Fig. 4). Multiple clumps of relatively large, polygonal or spindle cells, designated as onion-bulb–like formations, were commonly observed. A mixture of variable amounts of collagen fibers and cytoplasmic processes poorly stained with Masson's trichrome were interspersed in the area creating a dense fascicular pattern. In dog No. 2, there were multilobular nodules with abundant myxomatous stroma, accompanied by frequent atypical cells. A few clusters of neoplastic cells were embedded in hyalinized stroma. In dog Nos. 3 and 11, epithelioid cells were partly arranged in sheets within delicate collagen. In these areas, quite a few signet ring–like cells with clear cytoplasm were present (Fig. 5). The tumor mass in dog No. 5 exhibited a plexiform pattern. Some neoplastic foci showed hemangioma-like regions (Fig. 6). In the other areas, widespread hyalinization and multifocal cartilaginous or osteoid components were prominent (Fig. 7). Fusiform tumor cells resembling osteoblasts were present within the osteoid tissues. Original structures of the peripheral nerves were almost completely replaced by the proliferation of tumor cells and metaplastic mesenchymal tissues, whereas there were a few remaining degenerative ganglion cells. The tumor in dog No. 7 also contained cartilaginous and osteoid tissues and the same histologic patterns as was seen in the primary tumor (dog No. 12). In dog No. 11, there were multiple neoplastic nodules. Some nodules consisted of epithelioid-like cells, and the others included fusiform or anaplastic spindle cells. Elongated fusiform cells were arranged in interlacing whorls or fascicles occasionally with nuclear palisading, whereas anaplastic plump spindle cells with a number of atypical multinucleated giant cells showed storiform or onion-bulb configurations.
MPNST; dog No. 2. Fusiform or spindle-shaped neoplastic cells are arranged in interlacing fascicles. HE. Bar = 80 µm.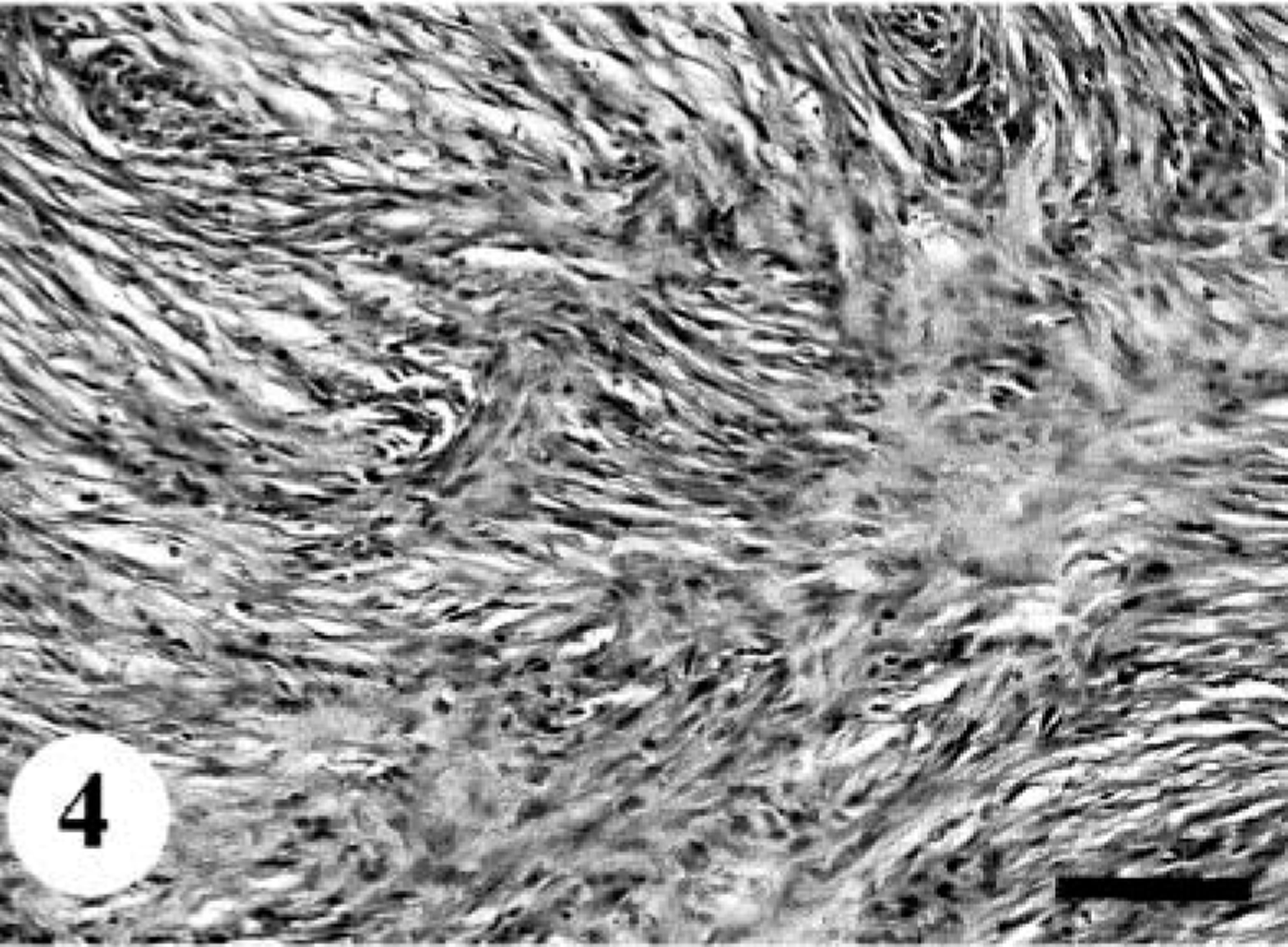
MPNST; dog No. 11. A number of vacuolar cells are interspersed within epithelioid tumor cells. HE. Bar = 40 µm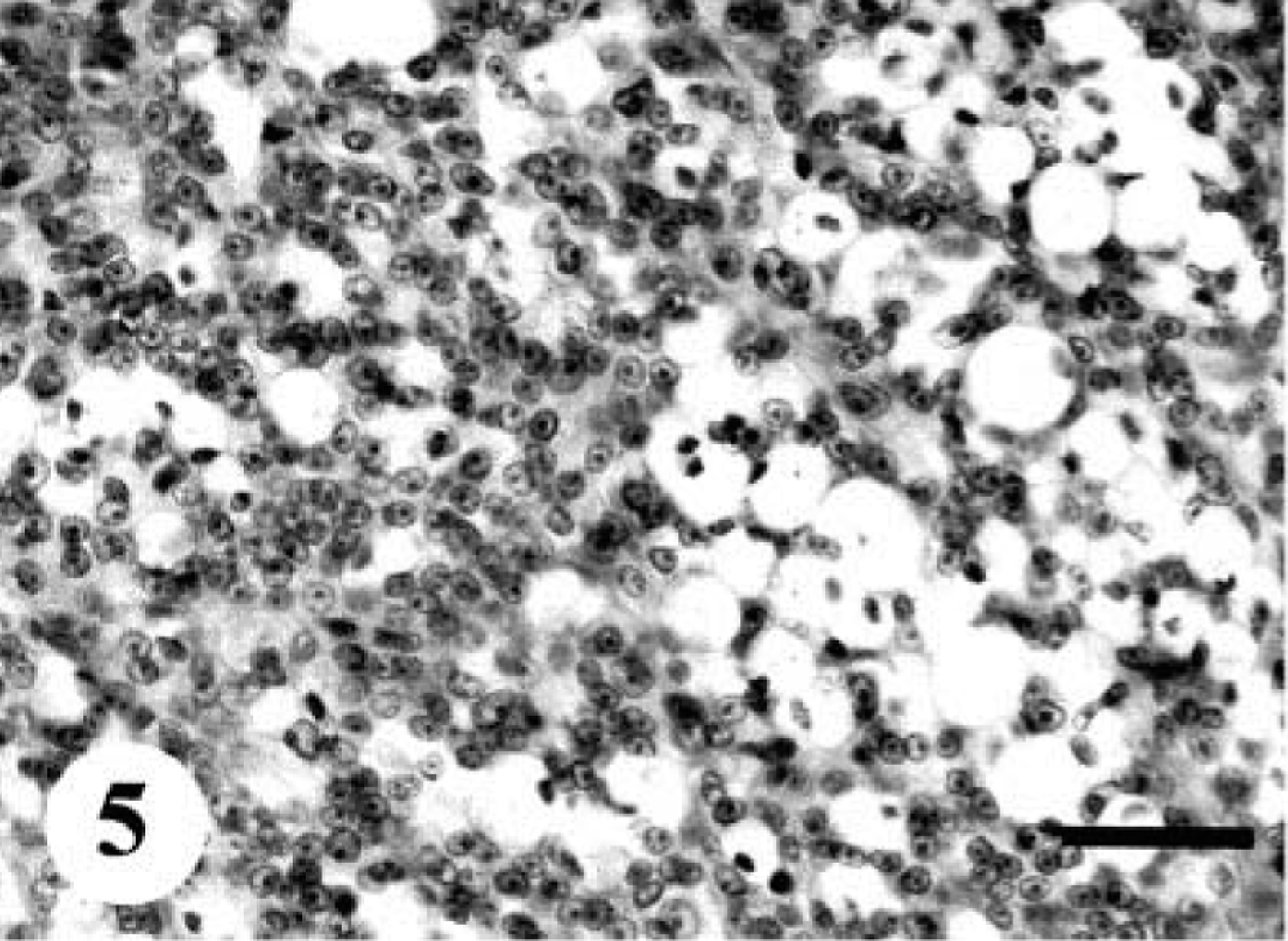
MPNST; dog No. 5. A focus of angiomatous appearances in MPNST (dog No. 5). HE. Bar = 50 µm.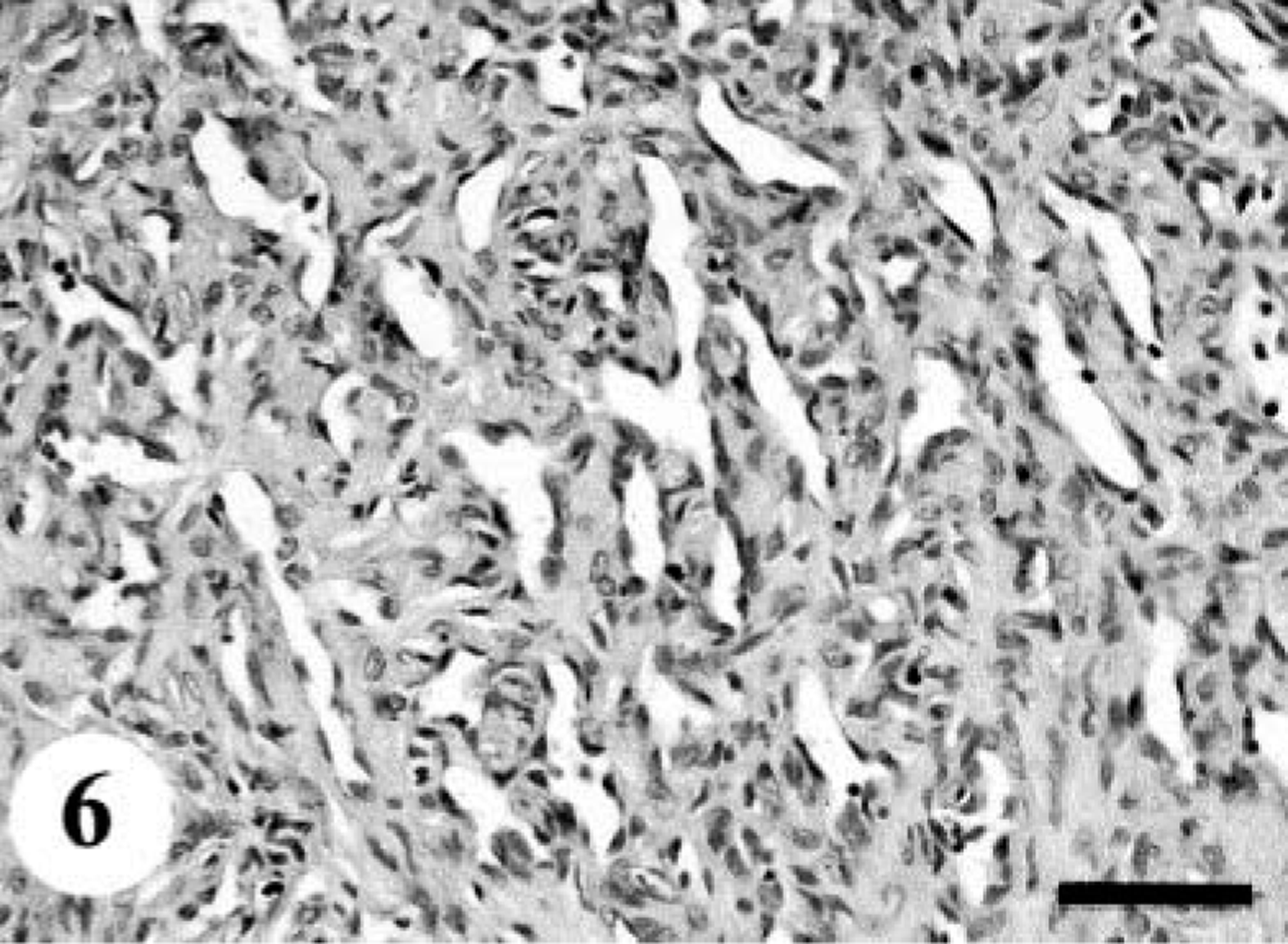
MPNST; dog No. 8. Neoplastic foci consist of widespread cartilaginous tissue accompanied by fusiform cells. HE. Bar = 100 µm.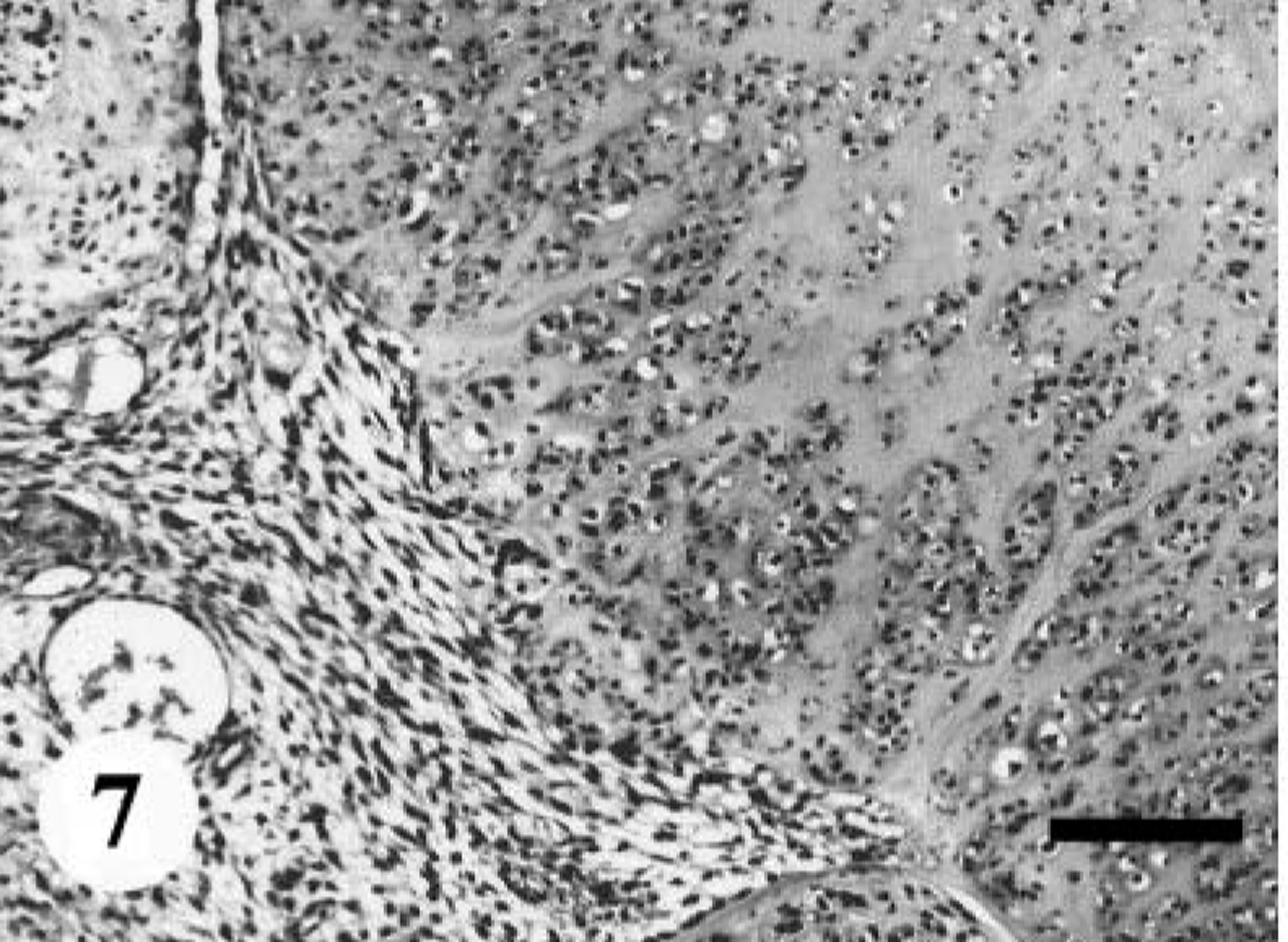
Four cases of MPNSTs (dog Nos. 6, 8, 9, and 10) contained histologic patterns typical of schwannomas (Fig. 8). Some areas were composed of spindle or fusiform cells with a high cellularity designated as Antoni A pattern, and the others comprised a hypocellular abundant mucinous or myxoid matrix with occasional round cells, termed Antoni B pattern. In dog No. 6, intracytoplasmic PAS-positive globules were encountered within only a few atypical multinucleated giant cells and were much larger than those in dog No. 1 (Fig. 9). Compared with the original tumor (dog No. 8), the recurrent tumor cells (dog No. 9) showed more apparent pleomorphism characterized by atypical mononuclear or multinucleated giant cells.
MPNST; dog No. 9. Antoni A area (A) with a high cellularity of spindle cells and Antoni B area (B) with a loose texture of small round cells and abundant myxomatous stroma. HE. Bar = 125 µm.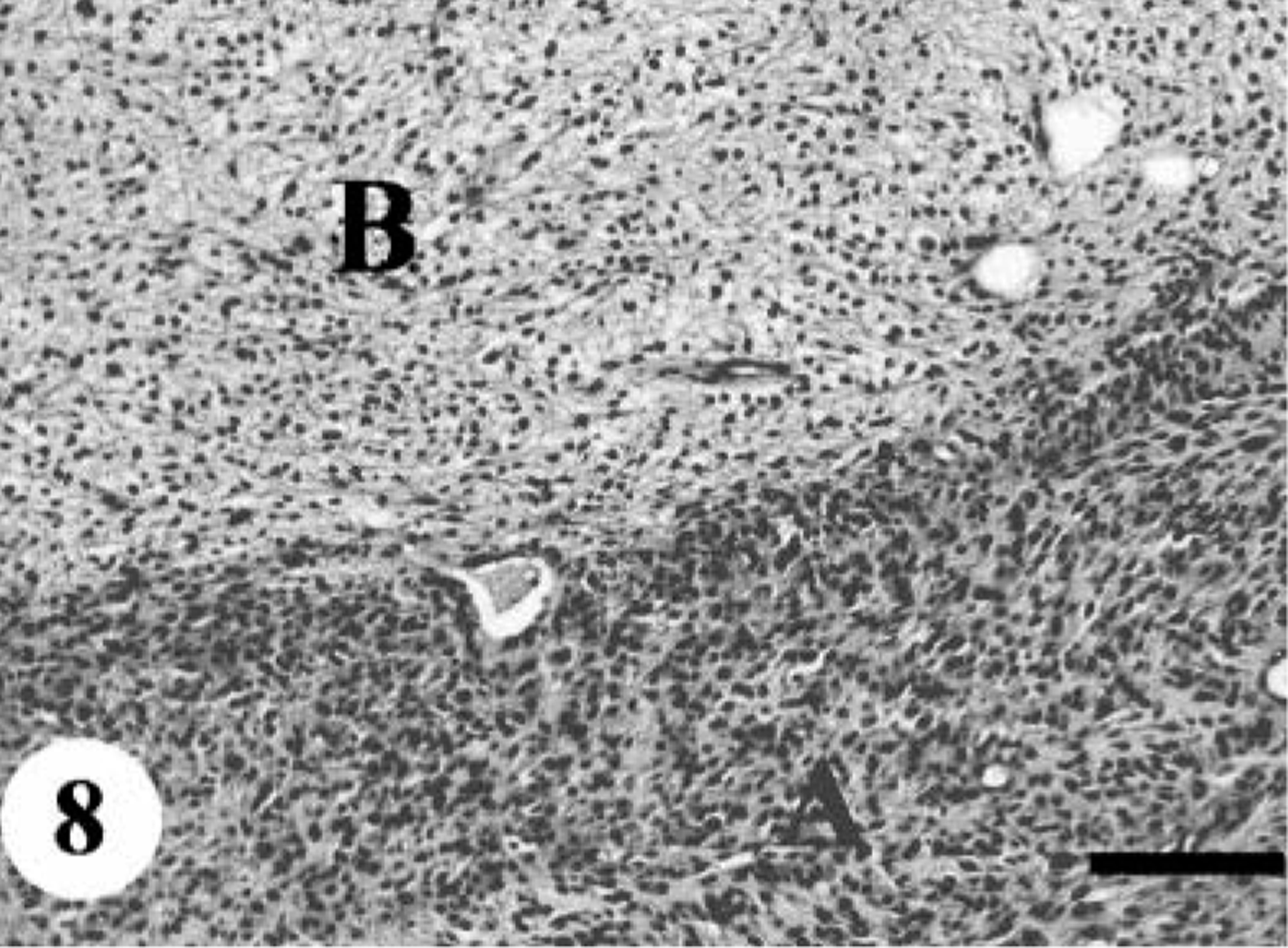
MPNST; dog No. 6. A few multinucleated giant cells with cytoplasmic PAS-positive globules (arrow). HE. Bar = 20 µm.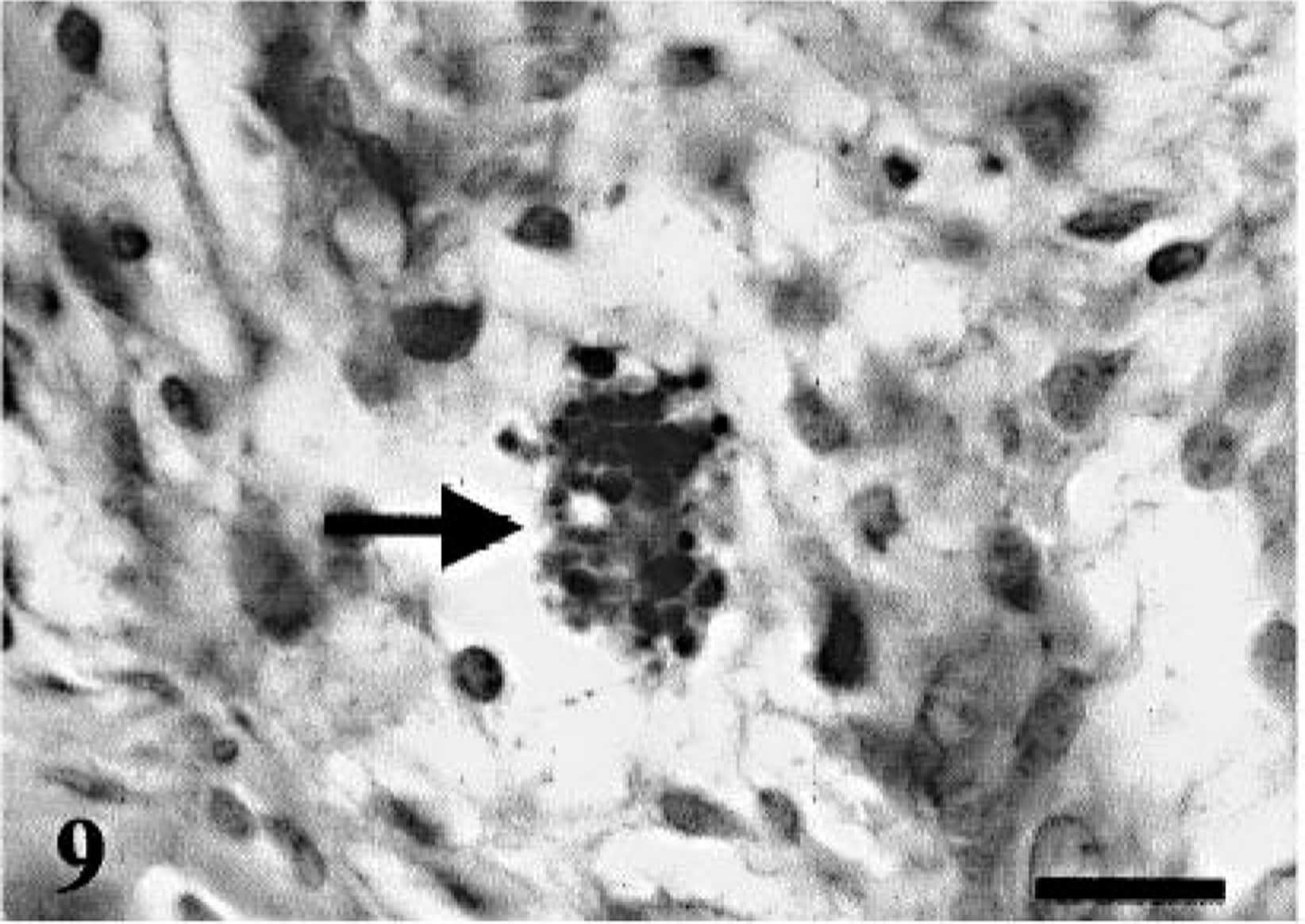
Histologic features of BPNSTs
Two cases of BPNSTs had atypical histologic patterns. The tumors in dog Nos. 12 and 16 consisted of plexiform or multilobular nodules with extensive hyalinization as well as moderate to high cellularities and mitoses. In dog No. 12, small, spindle-shaped neoplastic cells were arranged in interlacing fascicles, whorls, or onionskin-like appearances. Large polygonal cells with bizarre smudgy nuclei termed pseudoinclusions were often observed (Fig. 10). On the other hand, a majority of tumor cells in dog No. 16 resembled epithelioid cells arranged predominantly in tight clusters with abundant hyalinized stroma. Focal regions revealed typical Antoni A and B patterns of schwannoma. A number of large polygonal cells with abundant eosinophilic cytoplasm and hyperchromatic nuclei were also present. Partly, these anaplastic tumor cells infiltrated surrounding collagenous connective tissue, accompanied by moderate infiltration of lymphocytes and plasma cells. The remaining four BPNSTs exhibited typical histologic features of schwannoma or neurofibroma. In these typical PNSTs, neither necrotic foci nor aggressive growth was identified, and mitotic figures were rare.
BPNST; dog No. 16. Anaplastic cells with abundant eosinophilic cytoplasm and large nuclei. HE. Bar = 25 µm.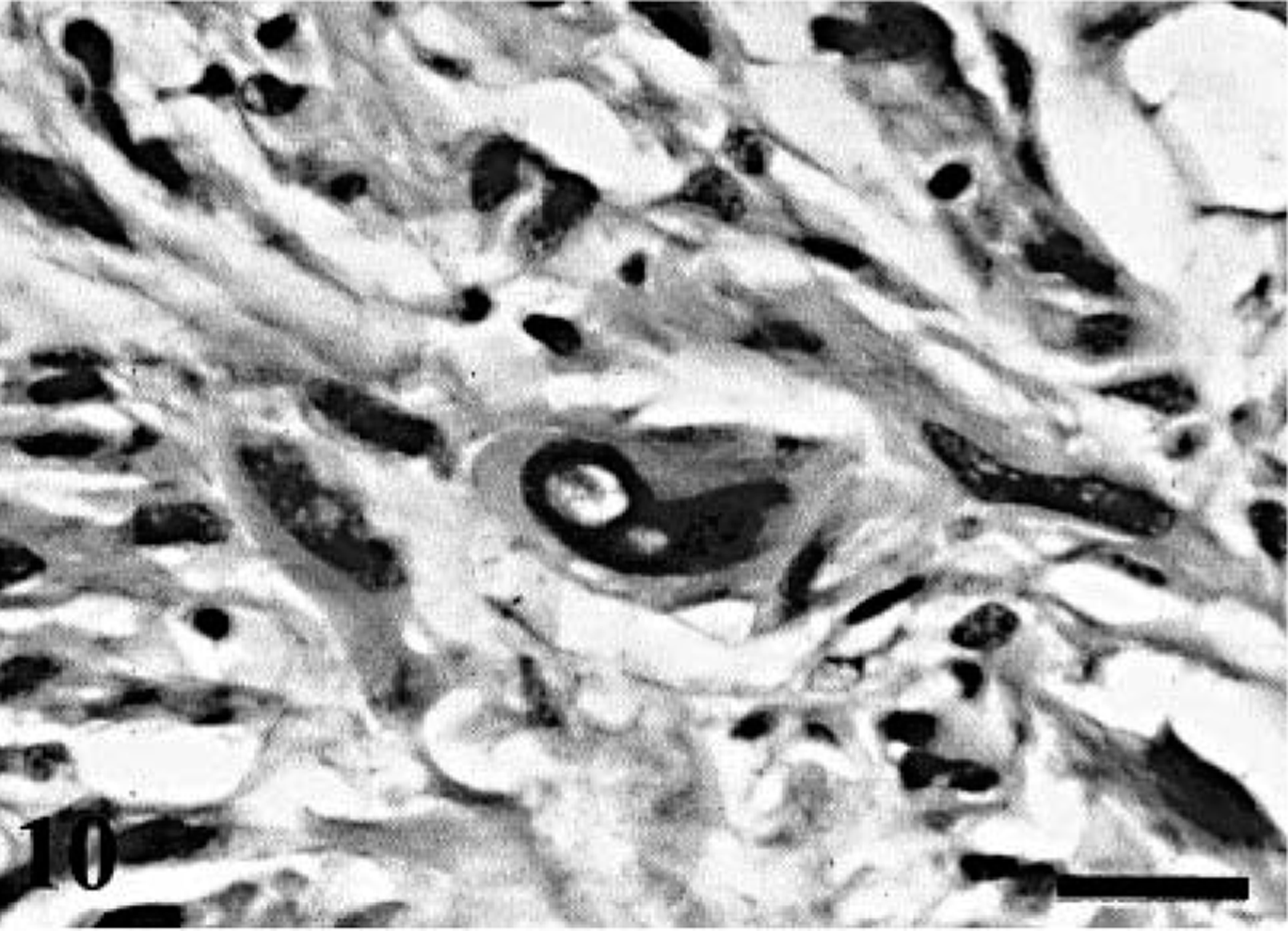
Immunohistochemistry
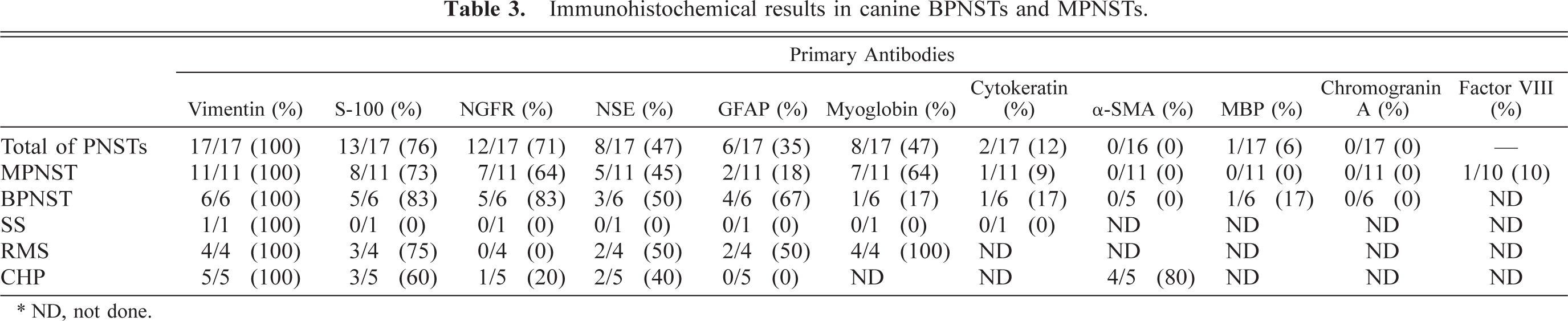
The results of immunohistochemistry are summarized in Tables 2, 3, 4. The immunoreactivity for S-100 was demonstrated in 76% of PNSTs including 73% of MPNSTs and 83% of BPNSTs. Among various cell types in MPNSTs examined, the spindle-shaped neoplastic cells were predominantly positive for S-100 (Fig. 11). On the other hand, BPNSTs including two cases with atypical histologic patterns were intensely positive for S-100 with no relation to cell morphology (Fig. 12). Although 75% of RMSs and 60% of CHPs were also positive for S-100, the expression was consistently focal and less than 20% of tumor cells. Immunoreactivity for NGFR was found in 71% of PNSTs, whereas the other tumors, except for one CHP, were negative. However, the reactivity for NGFR in most MPNSTs and two BPNSTs with atypical histologic patterns was focal or scattered (Figs. 13,14). Interestingly, 64% of MPNSTs were positive for myoglobin without any relation to cell morphology. However, in dog No. 1, round tumor cells tended to show more intense immunoreactivity for myoglobin than granular cells (Fig. 15). Regarding other neuronal markers, immunoreactivity for GFAP was found in one MPNST and 67% of BPNSTs. Focal or scattered expression of NSE and MBP was observed in less than 50% of PNSTs. Both the BPNSTs with atypical histologic patterns were positive for NSE. The morphology of NSE-positive cells mimicked ganglion cells. Immunoreactivity for NSE and GFAP was also detected in several RMSs and CHPs. Cytokeratin expression was found in a MPNST (dog No. 4) and a BPNST (dog No. 16). Cytokeratin positivity was found in a small population of polygonal cells and epithelioid tumor cells (Fig. 16). The positive cells for factor VIII–related antigen were detected only in the angiomatous focus in dog No. 5. None of the PNSTs were positive for chromogranin A or α-SMA, whereas 80% of the CHPs showed focal immunoreactivity for α-SMA.
Immunohistochemical results of each case with MPNSTs, BPNSTs, and other sarcomas examined.

Immunohistochemical results in canine BPNSTs and MPNSTs.
∗ ND, not done.
Results of PCNA labeling and mitotic index of cells within MPNSTs and BPNSTs.
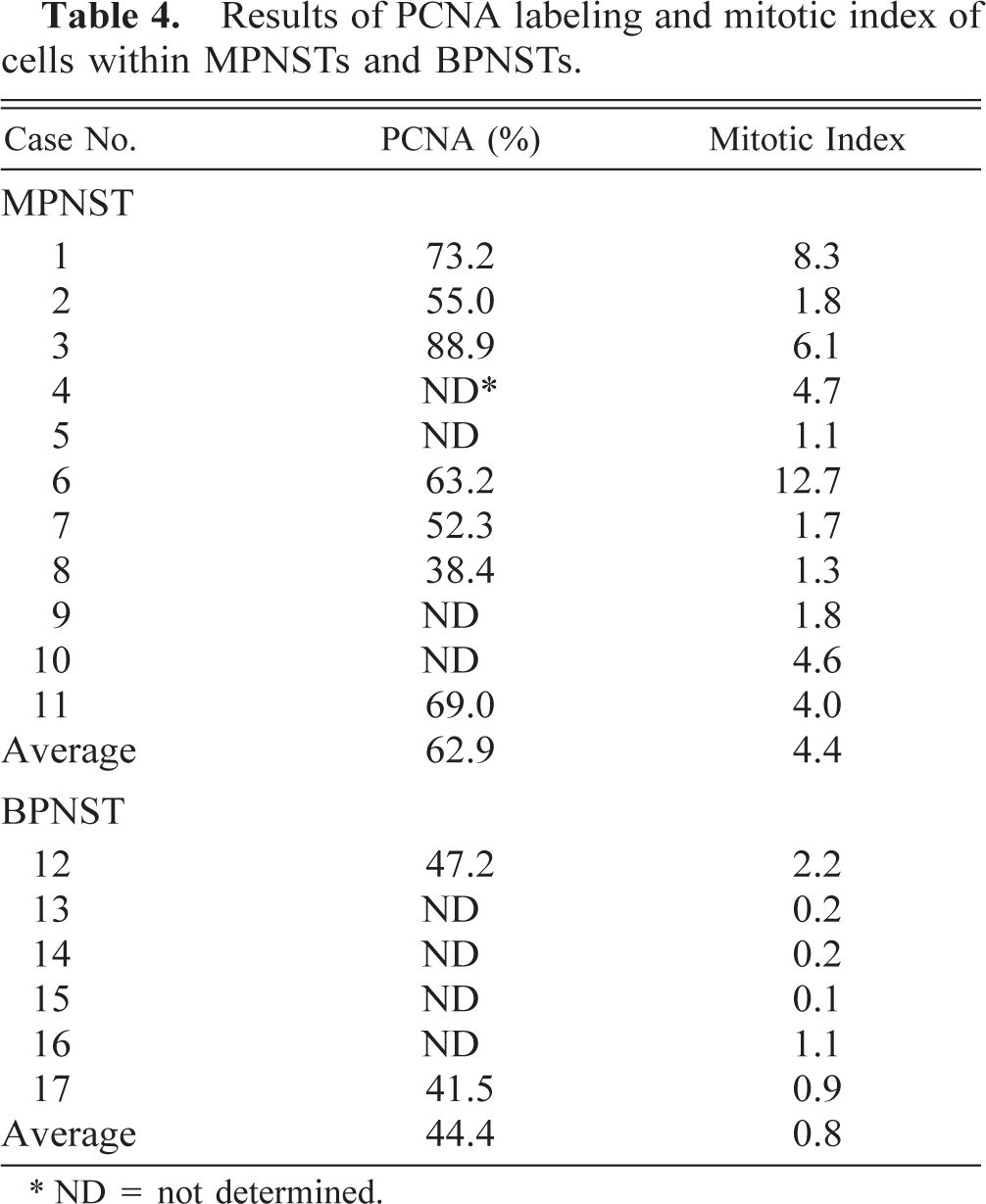
∗ ND = not determined.
MPNST; dog No. 2. Diffuse immunoreactivity for S-100 of the spindle-shaped tumor cells. Immunostaining for S-100. Bar = 40 µm.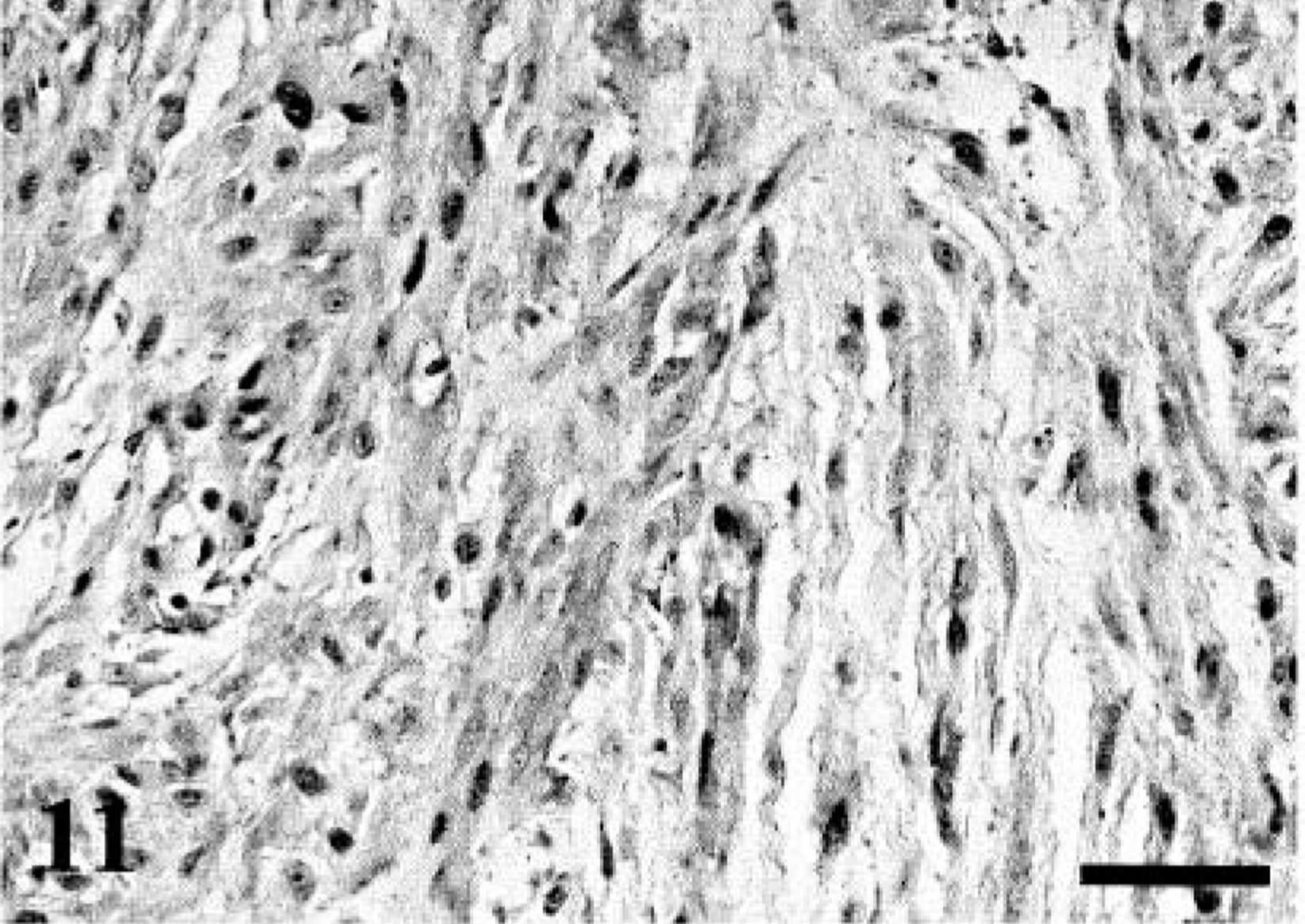
BPNST; dog No. 16. Cytoplasmic and nuclear S-100-immunoreactivity of the epithelioid tumor cells. Immunostaining for S-100. Bar = 40 µm.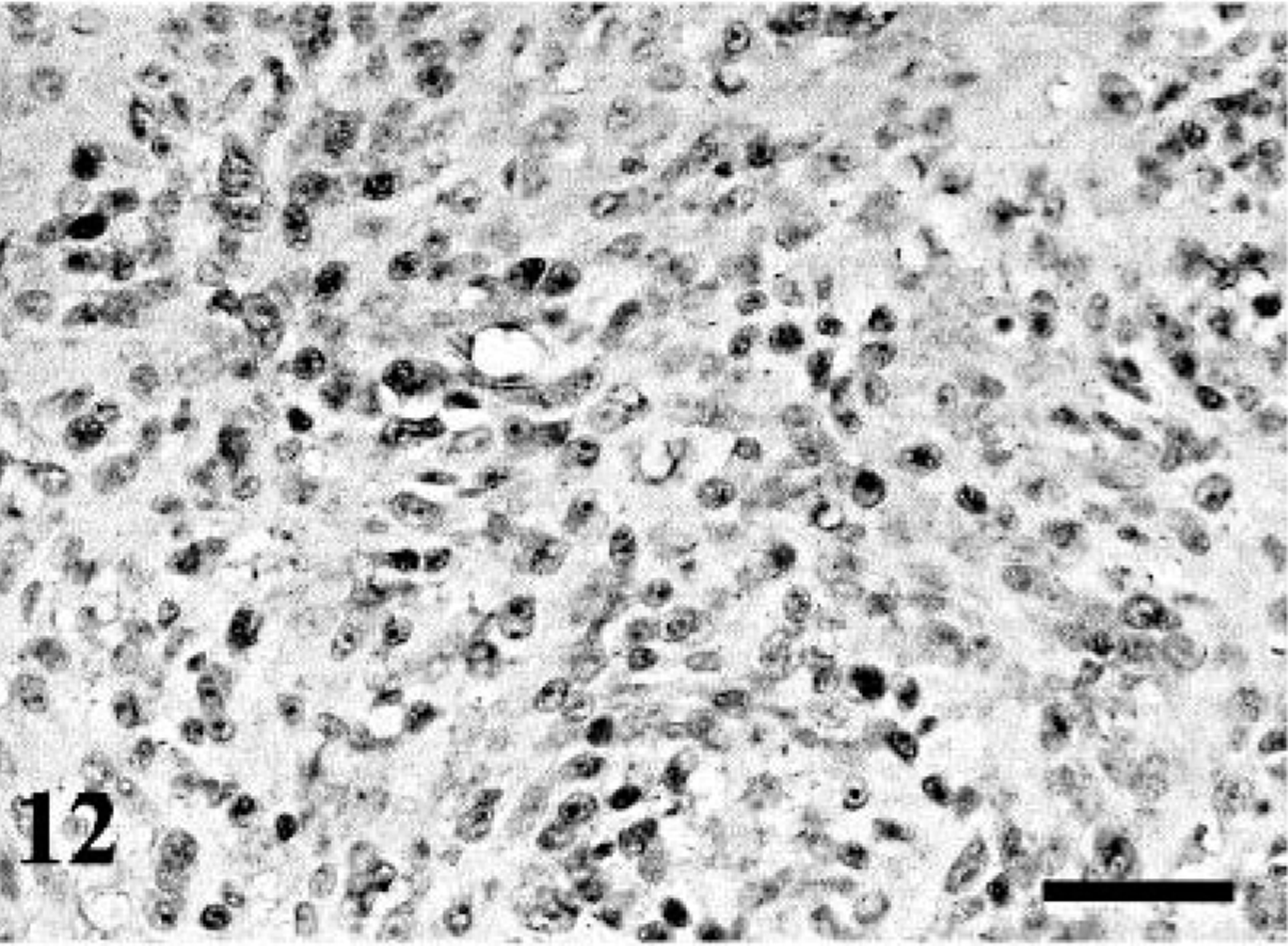
MPNST; dog No. 4. NGFR-positive cells are focally distributed in round cells. Immunostaining for NGFR. Bar = 40 µm.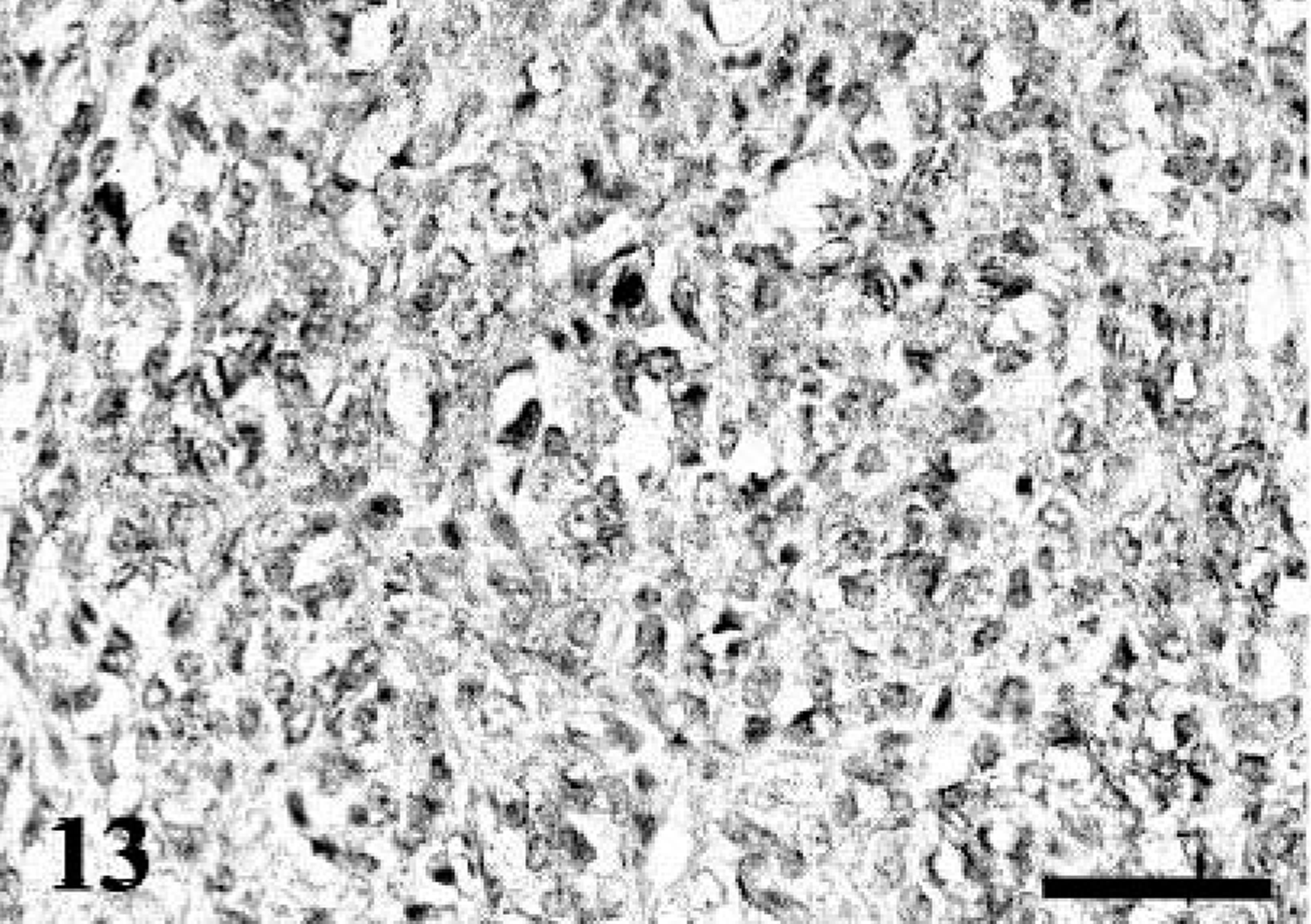
MPNST; dog No. 7. Scattered spindle neoplastic cells show cytoplasmic and membranous immunoreactivity for NGFR. Immunostaining for NGFR. Bar = 40 µm.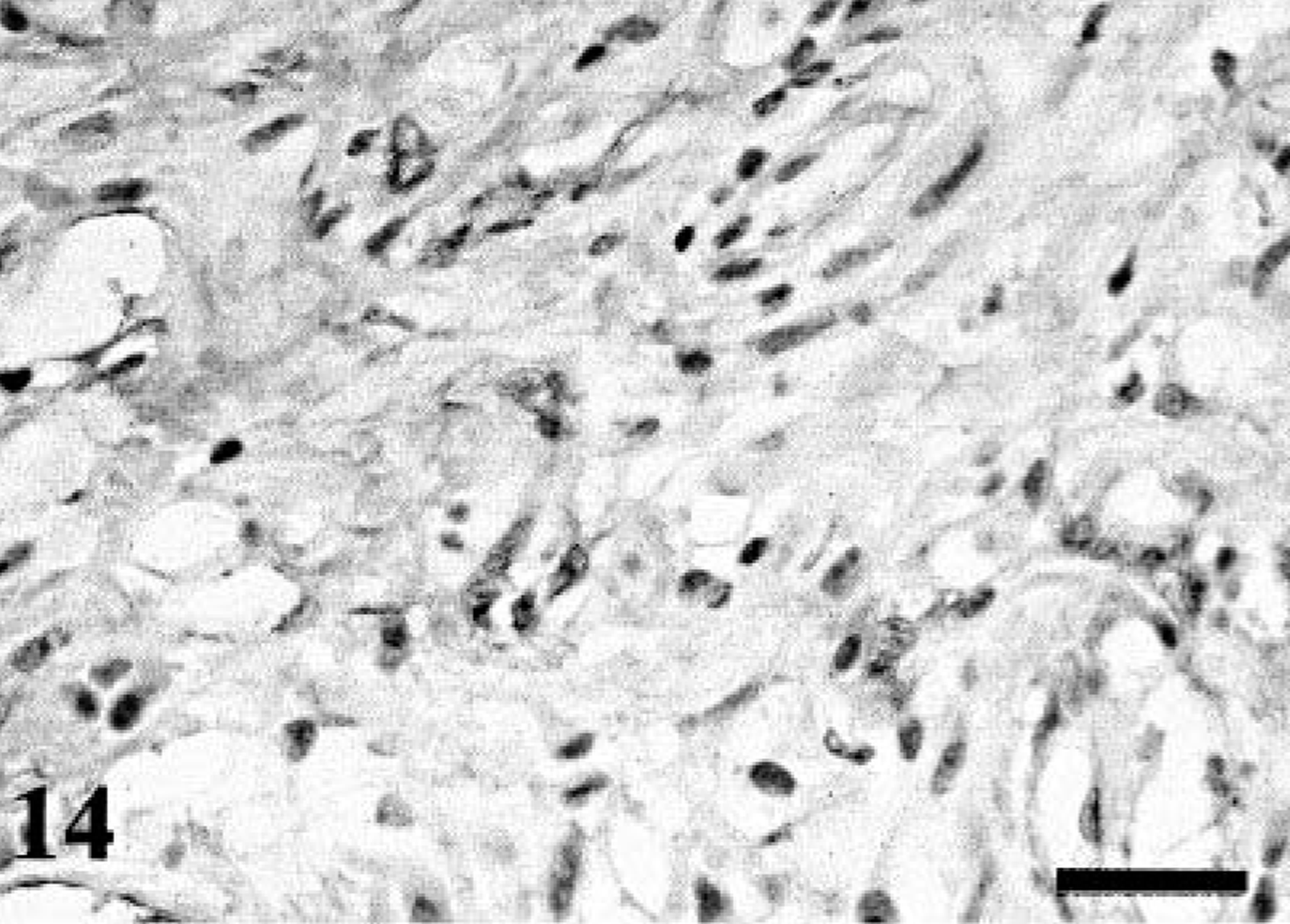
MPNST; dog No. 1. Myoglobin-positive round cells in the right side. Almost granular cells (left side) are feebly positive or negative. Immunostaining for myoglobin. Bar = 50 µm.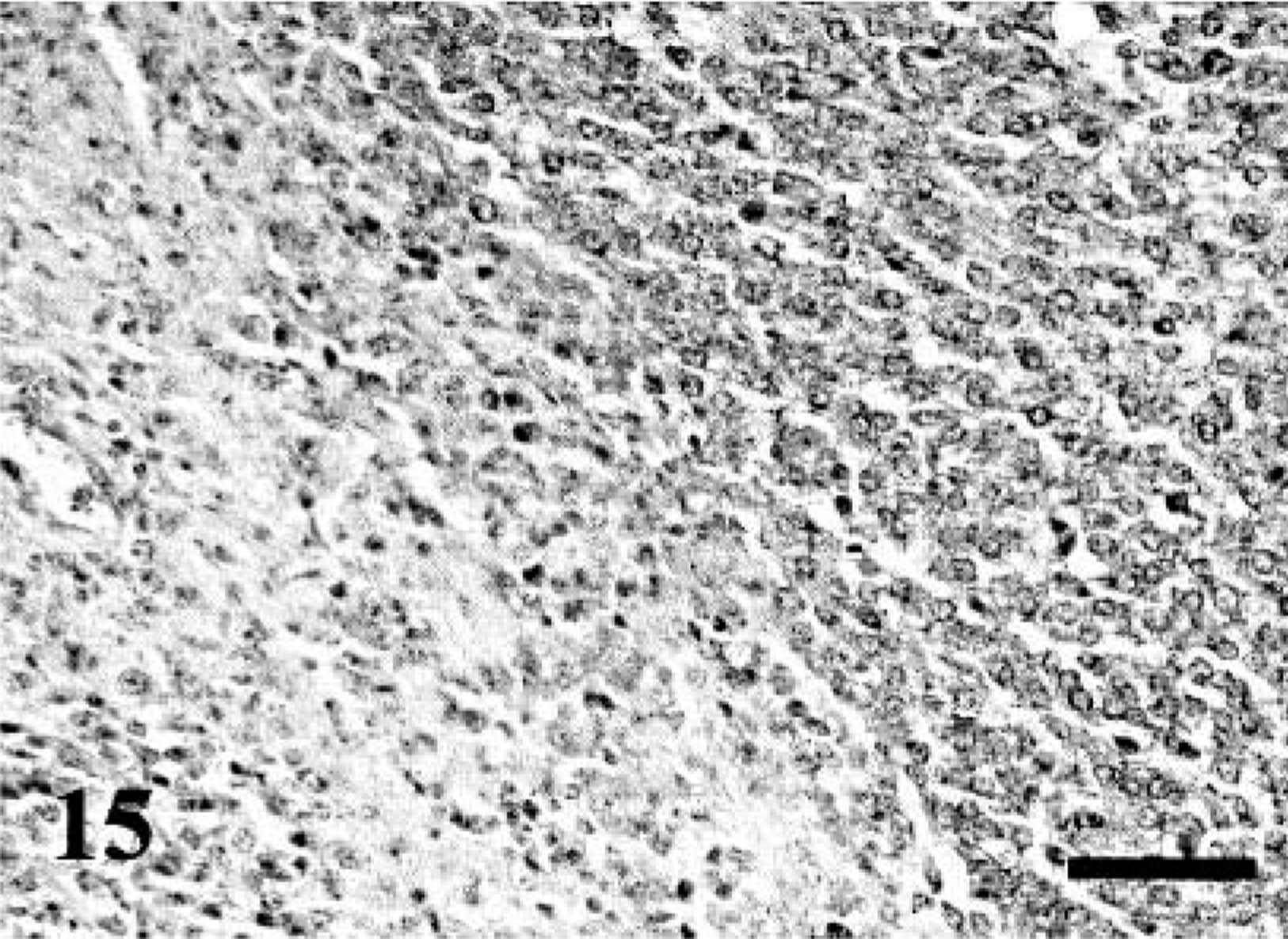
BPNST; dog No. 16. Cytokeratin-positive neoplastic cells are scattered. Immunostaining for cytokeratin. Bar = 40 µm.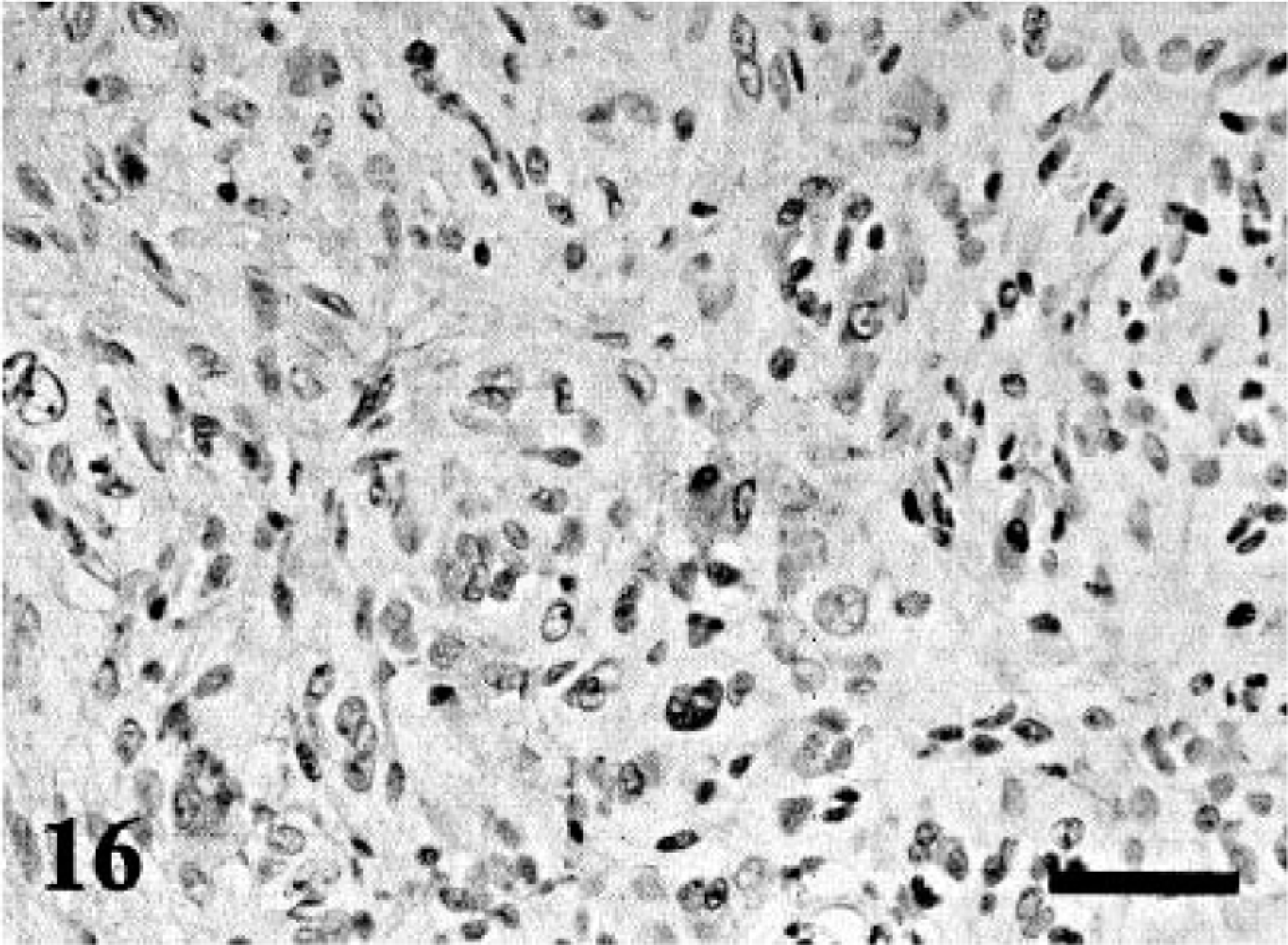
PCNA and mitotic index
The rate of nuclear staining for PCNA and the mitotic index are shown in Table 4. Although PCNA expression in MPNSTs seemed to be higher than in BPNST, there was no statistically significant difference between BPNSTs and MPNSTs (Student's
Discussion
Canine MPNSTs in this study had marked morphologic variations. The round cell type found in two cases (dog Nos. 1 and 4) was morphologically similar to the primitive neuroectodermal tumor described as the small round-cell type in human MPNST 1 or one of the malignant schwannomas, 49 suggesting that the presence of round cells implies a differentiation toward immature neural cells. On the other hand, typical Antoni A and B patterns characteristic for schwannoma were present in four MPNSTs (dog Nos. 6, 8, 9, and 10). Such typical patterns were so identifiable especially in two MPNSTs (dog Nos. 6 and 8) that the classification as malignant schwannomas might be warranted. A majority of the epithelioid types were MPNSTs 27,29,36 though benign epithelioid schwannoma has rarely been reported. 23 In this study, three MPNSTs and a BPNST contained epithelioid-like cells. In addition, a number of vacuolar cells were present together with epithelioid cells in dog No. 11. These vacuolar cells had a morphology similar to previously reported cells containing fat droplets creating a signet-ring appearance. 27 Cellular schwannoma, a subtype of human schwannoma, is frequently accompanied by lipid-laden histiocytes termed histiocytic Schwann cells. 39 However, immunoreactivity for lysozyme, a marker specific for mononuclear phagocytic cells, was not demonstrated (data not shown). A few vacuolar cells in dog No. 11 contained PAS-positive cytoplasmic particles, suggesting that these cells might differentiate into granular cells. The tumor cells with cytoplasmic granules in dog No. 1 appeared similar to granular cells. Similar cells have also been recognized in PNSTs with eosinophilic cytoplasmic globules. 26 Immunohistochemically, these cells are commonly positive for S-100. It is now generally accepted that granular cell tumors are derived from peripheral nerve components. 12,39 Thus, these granular cells likely indicate the divergent differentiation of canine MPNSTs. On the other hand, PAS-positive, large globules in dog No. 6 were morphologically different from the granules in dog Nos. 1 and 11. The granular cells in dog No. 1 were focally distributed, but the globules in dog No. 6 were present only in a few scattered, multinucleated giant cells. Thus, it might also be a unique appearance in the process of divergent differentiation, but an ultrastructural study would be needed to determine this.
Cartilaginous and osseous metaplasia was observed in two MPNSTs. The tumor in dog No. 5 consisted mostly of cartilaginous and osteoid tissues, which could potentially lead to a misdiagnosis as an osteogenic tumor. In such cases, a mixture of fascicles, whorls, and sheets composed of different cell types can be helpful to some extent, but it appears that an evidence of contiguity with peripheral nerves in gross or microscopic examination is more diagnostic. Immunoreactivity for S-100 might have no meaning because most chondrosarcomas and osteosarcomas also show diffuse or focal immunoreactivity for S-100. 12 The reason for diverse differentiation in MPNSTs still remains unclear. Many pathologists have introduced the concept of ectomesenchyme. 10–12,39 Migratory neural crest cells can differentiate not only into melanocytes, Schwann cells, and ganglion cells but also to cells in the leptomeninges and some mesenchymal cells contributing to the formation of muscle, bone, or cartilage in the neck and head regions. This might provide a reasonable explanation for the degree of divergent differentiation observed in MPNSTs.
In this study, two BPNSTs (dog Nos. 6 and 12) revealed atypical histologic features. The tumor in dog No. 12 was quite similar to MPNSTs arising in benign ancient schwannoma 30 or BPNSTs with atypical change including atypical neurofibroma or ancient schwannoma. 12,39 The bizarre nuclei were generally not interpreted as a malignant change but a degenerative change referred to as pseudoinclusions in human BPNSTs. It appears that pseudoinclusions as well as widespread hyalinization are often present in longstanding cases of various types of BPNSTs including ancient schwannoma or atypical neurofibroma and therefore should not be regarded as a sign of malignancy. In addition, one case (dog No. 12) was a recurrent tumor mass diagnosed later as a MPNST (dog No. 7). The major proliferating spindle cells in both tumors had similar immunohistochemical features and PCNA labeling or mitotic index, although the recurrent tumor had cartilaginous and osteoid tissues with frequent atypical cells. Thus, such osseous differentiation and cellular atypia might be more definitive to differentiate MPNST from BPNST. With regard to dog No. 16, a majority of tumor cells had epithelioid characteristics, highly suggestive of epithelioid PNST or epithelioid schwannoma. 23 However, presence of epithelioid cells was considered to be evidence of malignant change. 27,29,36 Moreover, invasion into the surrounding soft tissues and the presence of binuclear or multinucleated atypical cells should also be interpreted as malignant change. Consequently, PNST accompanied by some atypical changes or epithelioid tumor cells should be classified in PNSTs with transformation to MPNST. 30,39 It seems to be difficult to distinguish benign tumors from low-grade malignant tumors in such cases. Therefore, it may be helpful to add additional terminology when describing canine PNSTs, such as use of the terms malignant transformation or intermediate PNSTs.
A correlation between the lack of S-100 immunoreactivity and malignancy has been postulated in MPNSTs. 36 It is true that epithelioid tumor cells seen in two MPNSTs (dog Nos. 3 and 11) were negative or had scattered reactivity for S-100. However, epithelioid BPNST (dog No. 16) and two MPNSTs with heterogenous differentiation (dog Nos. 5 and 7) displayed intense immunoreactivity for S-100, as do a majority of epithelioid MPNSTs in humans. 12,27,29,39 Consequently, the correlation was not as notable in canine MPNSTs, indicating that S-100 reactivity might depend on the origin of predominant tumor cells rather than malignancy as previously reported. 42 That is, MPNSTs are possibly composed of several cell types, Schwann cells, perineurial cells, or perineurial or endoneurial fibroblasts. When predominant neoplastic cells were derived from cells other than Schwann cells, diffuse S-100 reactivity would not be expected. Similar interpretation for S-100 immunoreactivity has been proposed in several previous reports. 13,18,50 For example, typical schwannoma lesions display strong S-100 reactivity but exhibit very limited to no expression of epithelial membrane antigen (EMA), a marker specific for the perineurial cells. 3,13,44 In contrast, typical neurofibromatous lesions frequently demonstrate EMA expression, indicating perineurial differentiation. 13,18,50 However, it is well known that S-100 expression is identified in a wide range of tissues. 12 Even in the present study, 75% RMSs and 60% CHPs were positive for S-100. Thus, diffuse and intense immunoreactivity for S-100 is highly suggestive for canine PNSTs but not definitive. In addition, the results of infrequent MBP expression seen in only one BPNST (dog No. 14) suggest that MBP is not a good immunohistochemical marker for Schwann cell neoplasms. 7
In this study, 64% of MPNSTs showed a positive reaction for myoglobin. Malignant Triton tumor is a relatively rare type of human MPNSTs. 12,39 However, it has been reported that a number of MPNSTs with myoblastic differentiation are categorized as an embryonal type of RMS. 10 In most human MPNSTs, myoblastic cells are apparent in HE-stained tissues, 5,8,9,24 although such myogenic differentiation was indiscernible in our cases by general histologic examinations. In addition, the antibody for myoglobin is accepted as a specific marker for RMSs, but the reactivity tends to be restricted to highly differentiated muscular cells 12 and it also reacts with nonmyogenic tissues. Thus, it might be hard to consider that all myoglobin-positive cells are truly of myoblastic origin. Because myoglobin-positive cells tended to appear frequently in MPNSTs but not in BPNSTs, myoglobin immunostaining might help to distinguish BPNSTs versus MPNSTs. On the other hand, NGFR expression was demonstrated in 71% PNSTs including 64% MPNSTs and 83% BPNSTs. No NGFR reactivity was found in the four RMSs examined. This fact implies that immunohistochemistry for NGFR may help to distinguish MPNSTs from RMSs.
Immunoreactivity for cytokeratin AE1/AE3 was detected in one MPNST and one BPNST. Cytokeratin-positive cells resembling epithelioid cells were scattered throughout or had a focal distribution. PNSTs with glandular differentiation have been reported in both human and animal PNSTs 4,33,37 occasionally with neuroendocrine features, exhibiting immunoreactivity for chromogranin A, somatostatin, or serotonin. 6,48 Moreover, cytokeratin immunoreactivity has also been found on the peripheral regions of a cutaneous plexiform schwannoma in a pig. 43 In PNSTs with glandular differentiation, the characteristic features are visualized by HE-stained tissues and with distinct immunoreactivity for several epithelial markers including cytokeratins, EMA, and carcinoembryonic antigen. No distinct glandular structures within the neoplastic foci with cytokeratin-positive neoplastic cells were found in this study. Thus, these two tumors might not be consistent with glandular PNSTs, but the cytokeratin-positive neoplastic cells might appear as the result of divergent differentiation. The immunoreactivity for cytokeratin in canine MPNSTs might induce some confusion for differential diagnosis from SSs because human SS demonstrate immunoreactivity for S-100, NGFR, 35 and several epithelial markers including some cytokeratins. 14,40 In this study, one case of canine SS was examined, and that exhibited no immunoreactivity for almost all antibodies we used in this study including S-100, NGFR, and cytokeratin. Although MPNSTs and SSs have some morphologic features in common, careful consideration of the anatomic location of the neoplastic mass will be needed to distinguish these tumors.
The most important differential diagnosis of canine PNSTs might be CHPs, which have been categorized as tumors of unknown origin in the current WHO classification. Immunohistochemical investigations of CHPs have been described in several previous reports, 28,34,38 and these unique canine tumors expressed various markers including vimentin, α-SMA, desmin, factor VIII–related antigen, S-100, NSE, and GFAP. As in previous reports, our CHPs also showed immunoreactivity for several neuronal markers such as S-100 (3 of 5 cases), NGFR (1 of 5 cases), and NSE (2 of 5 cases). Our interests focused on the result of positive α-SMA immunoreactivity in 80% of CHPs (4 of 5 cases). On the contrary, no PNSTs exhibited immunoreactivity for α-SMA. Although the immunoreactivity for α-SMA in human MPNSTs has been described, 31,42 the phenomena might be quite unusual. Thus, the utility of α-SMA immunohistochemistry should be considered for the differentiation of PNSTs from CHPs. Because immunoreactivity for several endothelial markers including factor VIII–related antigen and CD34 has been confirmed as characteristic features in human hemangiopericytomas, 46 these markers may also be useful for identification CHPs. In this study, immunoreactivity for factor VIII–related antigen was found in only a limited area of one MPNST (dog No. 5) with angiomatous foci. Some authors suggest that this immunoreactivity might represent vascular differentiation of neoplastic neuroectoderm. 31 Others seem to believe that the lesions are formed by nonneoplastic, simultaneously proliferating cells such as perineurial or endoneurial fibroblasts, 42 perineurial cells, 45 or endoneurial dendritic cells. 16,22 However, such smooth muscular or endothelial differentiation (or both) is very rare in canine PNSTs and common in CHPs. In contrast, neuronal markers including NSE, GFAP, and MBP seemed to be of little utility for the differential diagnosis of PNSTs from other soft tissue tumors.
Several studies of immunoreactivity for GFAP in PNSTs have proven that 30–40% of BPNSTs would be positive.
15,21
In addition, BPNSTs are more often GFAP reactive than MPNSTs.
12,15
Our study also confirmed the previous data, suggesting that GFAP reactivity could help distinguish BPNSTs from MPNSTs. The difference in number of PCNA-labeled cells (
In conclusion, this article described the morphologic and immunohistochemical varieties of canine BPNSTs and MPNSTs. The results indicate that immunohistochemistry for NGFR and α-SMA might be helpful for differentiating canine PNSTs from other sarcomas including RMSs or CHPs. In addition, our results show that immunostains for S-100, GFAP, NSE, myoglobin, and cytokeratin have only limited value in distinguishing between canine MPNSTs with divergent differentiations and other related sarcomas.