Abstract
A 14-year-old male Siberian tiger (Panthera tigris altaica) was admitted with an ulcerating mass on the right thoracic wall. Radiographic and computed tomographic evaluation indicated 2 isolated cutaneous masses without any signs of metastasis. Histology of a Tru-Cut biopsy revealed an anaplastic sarcoma with giant cells. Both tumors were resected with appropriate normal tissue margins. The size of the defect did not allow primary closure of the wound; therefore, a mesh expansion technique was attempted. Three months later, the tiger had to be euthanized due to extensive metastasis to the lungs. Histomorphological features and immunohistochemical results confirmed the diagnosis of malignant peripheral nerve sheath tumor. In contrast to domestic animal experience, the tumor had spread extensively to the lungs without local reccurrence in a short period of time. Correct diagnosis requires various immunohistochemical evaluations of the tumor tissue.
A 14-year-old male Siberian tiger (Panthera tigris altaica) was presented to the Clinic for Zoo Animals, Exotic Pets and Wildlife at the University of Zürich (Zürich, Switzerland) because of an ulcerating mass on the right thoracic wall. After initial immunization for Feline panleukopenia virus, Felid herpesvirus 1, and Feline calicivirus, the tiger received irregular booster vaccinations 1–4 years apart until the age of 9 years. Otherwise, he was in good health, bright and alert, and in good body condition. Body weight was estimated to be 200 kg. For further diagnostic evaluation, anesthesia was induced with 0.05 mg/kg medetomidine hydrochloride a and 2.18 mg/kg ketamine hydrochloride b intramuscularly. The tiger was intubated (internal diameter: 14 mm), c and anesthesia was maintained by administration of isoflurane d in oxygen (end-tidal isoflurane concentration: 1.1-1.3%). Physical examination was unremarkable except for dental tartar, gingivitis, a missing upper left canine tooth, and 2 ulcerating cutaneous masses extending into the subcutis on the right thoracic wall. The masses were well circumscribed, moveable, and spongy in consistency (Fig. 1).
Blood was drawn for hematology and serum biochemistry, all of which were within reference intervals. Right and left lateral radiographs of the thorax revealed a minor generalized bronchointerstitial infiltrate, but no signs of tumor metastases were seen. For the computed tomography (CT) study, the tiger was positioned in left lateral recumbency and scanned with a third-generation CT scanner. e Subsequent to the scout image, 5-mm contiguous transverse images, starting at the level of the ascending aorta to the level of the mid-abdomen, were obtained before and after intravenous bolus injection of a nonionic, iodinated contrast medium f (600 mg iodine/kg body weight). All images were acquired at 130 kVp and 100 mA with a field of view of 450 mm using a soft-tissue algorithm. The CT examination identified 2 cutaneous soft-tissue masses extending into the subcutis of the right side of the body. The larger mass was dorsal to the 12th and 13th rib and measured 6 cm × 19 cm × 15 cm. The larger mass had an irregular surface, and the dorsal, cranial, and caudal margins were ill defined with fusiform subcutaneous extensions approximately 2 cm long. The smaller mass was 15 cm ventral to the larger mass and located at the level of the 13th rib cartilage. The smaller mass measured 2 cm × 5 cm × 6 cm. This smaller mass was oval and well defined. Dorsally, a fusiform subcutaneous extension of the mass was noted. Neither mass involved deeper structures, and contrast enhancement was not noted. The lung parenchyma and the thoracic lymph nodes were unremarkable (Fig. 2).
After the CT examination, the tiger underwent surgery in left lateral recumbency. A constant rate infusion of fentanyl g (500 mg/h) was administered during surgery to provide additional pain relief. An elliptical incision parallel to the skin lines was created including both masses. With continuous dissection of the subcutaneous tissue, the tumors could be easily mobilized and resected. With the help of CT images, 3 cm of grossly normal tissue was excised around the masses in all planes. The size of the defect did not allow primary closure of the wound; therefore, a mesh expansion technique was attempted. Several stab incisions in the cutis allowed the stretching of the cutis to adapt wound edges for primary healing (Fig. 3). Wound closure was achieved in 2 layers, continuous subcutaneous and a continuous intradermal pattern.
Polyglactin 910 h (USB 1) sutures were used. No wound dressing was applied. At the end of surgery, buprenorphine i was administered (0.018 mg/kg, intravenously) for pain relief, and the medetomidine hydrochloride was reversed with atipamezole hydrochloride. j Postoperative treatment included amoxicillin k (10 mg/kg twice a day) and meloxicam l (0.05 mg/kg once a day) orally for 10 days.
Tissue samples were fixed for 24 hr in 4% neutral buffered formalin, routinely processed, and embedded in paraffin. Tissue sections 2- to 3-μm thick were stained with hematoxylin and eosin or for reticulin fibers. The cutaneous, well-demarcated, invasively growing, unencapsulated masses consisted of highly pleomorphic cells arranged in interwoven bundles separated by a fine fibrovascular and collagenous to myxoid stroma. There were no recognizable classical structures such as whorls, palisading, or Antoni A configurations of the neoplastic cells. The cells were partly spindle shaped and partly plump fusiform to epitheloid with an abundant eosinophilic homogeneous cytoplasm. The nuclei were often centrally placed, round to oval, or indented, and the chromatin was coarsely stippled. Most cells contained a large, eosinophilic, prominent nucleolus measuring up to 5 μm in diameter (Figs. 4, 5). In some areas, several multinucleated giant cells were visible. The mitotic rate was low, with 1–2 mitotic figures per 10 highpower fields. Multiple small foci of necrosis were randomly distributed within the larger tumor of the thorax. The surface was extensively ulcerated and diffusely infiltrated by neutrophils, macrophages, plasma cells, and lymphocytes. At the periphery of the neoplasia, no lymphocyte aggregates could be found, as can be seen in feline vaccine-induced sarcomas. With reticulin staining, the basement membranes of the neoplastic cells showed positive reactivity. Additional 3-μm tissue sections for immunohistochemistry were mounted at 37°C on positively charged glass slides, m deparaffinized in xylene, and rinsed in absolute alcohol. The sections were incubated with a panel of antibodies including pan-cytokeratin (Lu 5), neuron-specific enolase (NSE), S-100 protein, glial fibrillary acidic protein (GFAP), nerve growth factor receptor p75, neurofilament 200 (NF 200) and neurofilament 70 (NF 70), vimentin, α-smooth muscle actin, factor VIII-related antigen, melan A, cluster of differentiation (CD)3, CD79a, lysozyme, and MAC 387 (see Table 1 for primary antibodies and antigen retrieval). n Kupffer cells of the liver were used as positive control for the MAC 387 antibody. The tumor cells showed positive reactivity for S-100 protein, NSE (Fig. 6), GFAP (Fig. 7), and p 75 (Fig. 8) but were negative for NF 200, NF 70, MAC 387, and melan A. The histomorphological features and immunohistochemistry results confirmed the diagnosis of malignant peripheral nerve sheath tumor (PNST).
Three months after surgery, the tiger became anorectic and apathic. Recurrent vomiting was observed, and no feces were passed for 3 days. The tiger was anesthetized using the previous protocol of medetomidine and ketamine. The surgical site had healed with the exception of a small superficial scar. No subcutaneous mass was palpable on physical examination. Thoracic radiographs revealed moderate pleural effusion, which made visualization of the heart and diaphragm difficult. A large soft-tissue mass was seen dorsocaudally in the thorax on the laterolateral view. The left cranial lung lobe was only partially ventilated; the main bronchus was pushed dorsally, and air bronchograms were noted. The other lung lobes showed rounded contours and were retracted. The ultrasonographic examination of the thorax confirmed a moderate amount of anechoic free fluid and multiple lobulated masses. Some masses were within several lung lobes, whereas others were in the mediastinum. On the basis of these findings, a tentative diagnosis of metastases to the lung, mediastinum, and possibly the pleura with secondary pleural effusion originating from the subcutaneous tumor on the right thorax was made. Because of the extensive masses, age of the animal, and poor prognosis, the tiger was euthanized with pentobarbital (350 mg/kg intravenously). o
The necropsy revealed multiple gray, firm, lobulated nodules of variable diameter (up to 10 cm) in the lung and mediastinum. There was also a severe hydrothorax with approximately 3 liters of serosanguineous fluid resulting in lung atelectasis. The pulmonary lymph nodes were normal on gross inspection. Tissue samples were taken using the same procedures as described earlier. Histologically, the morphology of the neoplastic cells of the proliferations in the mediastinum and lungs had a similar appearance to the cells of the previously investigated masses. The metastases had less extracellular matrix and were slightly less differentiated. Rarely, the neoplasm showed differentiation to a myxoid pattern with abundant extracellular matrix consisting of mucopolysaccharide with embedded stellate cells. Immunohistochemistry followed the previous protocol and demonstrated the same staining patterns as the previously investigated masses. Based on the similar histomorphological features and the same immunohistochemical results, the diagnosis of malignant PNST was confirmed, and the multiple nodules in the lung and in the mediastinum were interpreted as metastases.
The present case apparently describes the first report of PNST in a tiger. Limited published information is available about PNSTs, despite sporadic reports from a wide range of species including dogs, cows, horse, sheep, goats, humans, rats, snakes, and cats, indicating they are widespread in the animal kingdom. By far, most reports are of tumors in cattle and dogs. 3,6 In dogs, brachial plexus and other spinal nerve root involvement are the most commonly described locations. 2,7 Metastases are rare but may involve the lymph nodes and lung. 1 PNSTs in the cat are uncommon but may present as cutaneous tumors of adult to aged cats. 6,10,13–16 The true incidence of PNST in large felids is unknown, with a single report of a schwannoma in the larynx of a lion. 12
The World Health Organization classification of tumors of the nervous system and mesenchymal tumors of the skin and soft tissues in domestic animals combines malignant and benign forms of neurofibroma and schwannoma under the title PNST. 6 PNSTs arise from Schwann cells, perineurial fibroblasts, or both. 6 PNSTs may arise from any anatomical site in the body, although skin and subcutaneous sites are most common. Tumors occur as slow-growing, nodular, lobulated, and poorly defined tumors, without histologic encapsulation. Clinically, however, the tumors may appear encapsulated, globoid, lobulated, and of variable size and shape, depending on the location. In general, tumors of peripheral nerves are locally invasive and slow to metastasize. 1
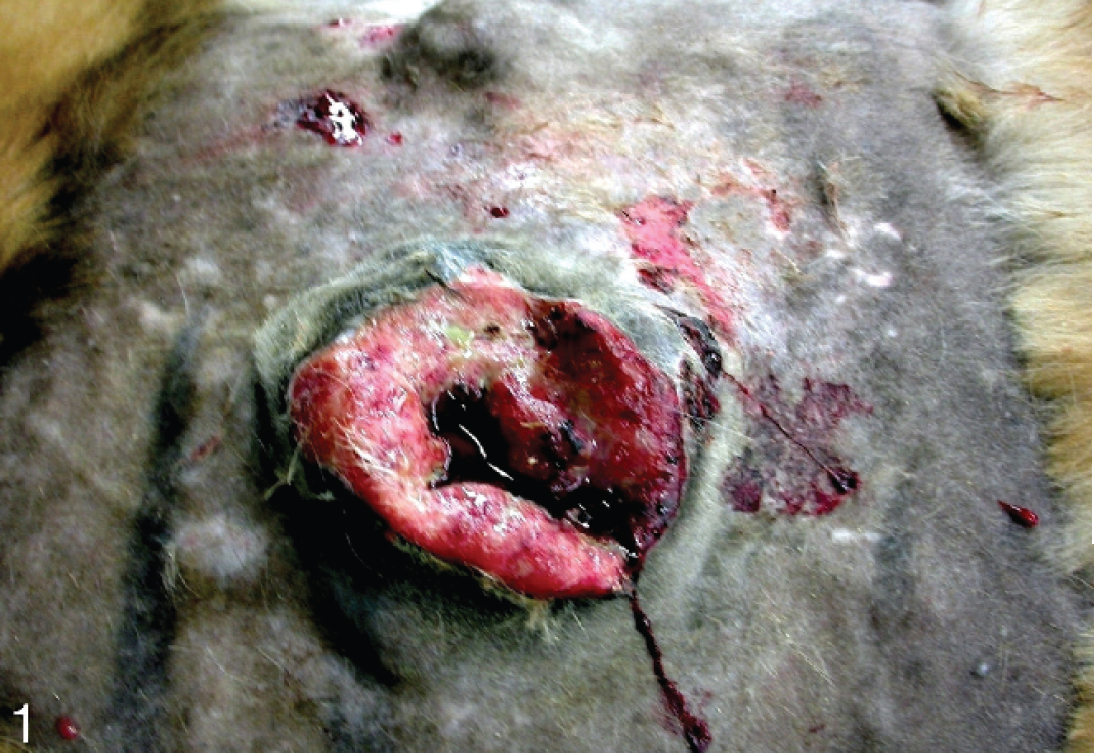
Siberian tiger (Panthera tigris altaica). Macroscopic picture of an ulcerating mass dorsally over the last rib, measuring 20 cm in diameter. The mass was well circumscribed, moveable, and spongy in consistency.

Siberian tiger (Panthera tigris altaica). Transverse computed tomographic image after an intravenous bolus injection of iodinated contrast medium (left: dorsal, right: ventral, top: right). Both tumor masses are located in the subcutis of the right thorax, are hypodense, and have fusiform dorsal extensions (arrows). Deeper layers of the thoracic wall are not involved.
Histopathologic diagnosis is difficult because PNSTs often lack classical morphological patterns and are thus difficult to differentiate from other spindle-cell tumors such as hemangiopericytomas, fibrosarcoma, anaplastic sarcomas with giant cells, melanoma, leiomyoma, or leiomyo-sarcoma. 5,7,10,14 Therefore, diagnosis requires further confirmation with immunohistochemical and/or ultrastructural analyses. 4,5,7,14 PNSTs may stain positively for S-100 protein, GFAP, laminin, NSE, nerve growth factor receptor p75 (CD 271), and vimentin. However, up to 50% of malignant PNSTs do not express S-100 protein. 11,14
In the current case, the histomorphological appearance and the relatively rapid clinical course were initially suggestive of an anaplastic sarcoma with giant cells. However, the positive immunohistochemical staining for neural and mesenchymal markers (S-100, GFAP, NSE, p75, and vimentin) indicated a malignant PNST of the skin with metastases to the mediastinum and lungs. A neurofibroma could be excluded by absence of staining of the neoplastic cells for NF 70 and NF 200. The lack of labeling for MAC 387 and lysozyme made a histiocytic lineage highly unlikely. A mass with similar histological appearance and immunohistochemical results has been described in a domestic cat. 15 Other tumors such as malignant lymphoma, leiomyosarcoma, rhabdomyosarcoma, hemangiosarcoma, malignant melanoma, or an epithelial malignant neoplasm were excluded by a lack of staining for CD3, CD79a, desmin, α-smooth muscle actin, factor VIII related antigen, pancytokeratin, and melan A. Because the definitive diagnosis of malignant PNST requires more extensive laboratory testing, it might be that the occurrence of malignant PNST is more common in felids than currently thought.
Because of the low metastatic rate and poor chemotherapy response, 9 surgical excision and/or radiation therapy are recommended as the preferred treatment for PNST in domestic animals. 1,2,7,9 Radiation therapy is the treatment of choice if surgical excision lacks clean or tumor-free margins. In the present case, CT images were particularly useful to visualize tumor invasion of local tissue to allow surgical excision with 2 3 cm of grossly normal tissue around the mass and 1 additional tissue plane in depth, including the panniculus muscle that was in contact with the tumor. Complete resection of the primary tumor was assumed, and postsurgical histological evaluation indicated complete resection. Therefore, radiation treatment was not considered because of the requirement for several sessions of anesthesia. Further, local occurrence did not occur, supporting the speculation of complete resection of the tumor. Nevertheless, the primary subcutaneous tumor evidently had spread to the thoracic tissues 3 months later, contrary to observations in domestic animals, where metastasis of PNSTs was rarely detected and mostly limited to grade 3 tumors. 8 Although local reccurrence, the most common sequela of PNST, did not occur and lung radiographs did not show any signs of tumor metastasis prior to surgery, it seems that clinically undetectable micrometastases were already present when surgery was attempted and were activated when the primary tumor was removed.
As described in the domestic cat, 5,6,13 cutaneous masses were observed in the tiger in the present study but were initially diagnosed as anaplastic sarcoma with giant cells. Definitive diagnosis of PNST was made following immunohistochemical evaluation of the tumor tissue. Theoretically, early diagnosis would permit a greater chance of treatment success, but definitive diagnosis is possible only with appropriate immunohistochemical staining.
Acknowledgements. The authors thank the Stiftung Forschung für das Pferd, Switzerland, for financial support, and Dr. Lloyd Vaughan for critical review. Hanspeter W. Steinmetz and Maja Rütten contributed equally to this work.

Siberian tiger (Panthera tigris altaica). Wound closure was performed with a mesh expansion technique to allow primary healing after surgical removal of a peripheral nerve sheath tumor of the skin.

Siberian tiger (Panthera tigris altaica). Histological section from a malignant peripheral nerve sheath tumor of the skin showing interwoven streams of spindloid and sheets of epitheloid tumor cells. Hematoxylin and eosin. Bar = 100 μm.

Siberian tiger (Panthera tigris altaica). Histological section from a malignant peripheral nerve sheath tumor of the skin showing spindle-shaped and epitheloid tumor cells. Hematoxylin and eosin. Bar = 20 μm.

Siberian tiger (Panthera tigris altaica). Immunohistochemistry of a malignant peripheral nerve sheath tumor showing neoplastic cells with strong red labeling for neuron-specific enolase. Bar = 50 μm.

Siberian tiger (Panthera tigris altaica). Immunohistochemistry of a malignant peripheral nerve sheath tumor showing neoplastic cells with strong red labeling for glial fibrillary acidic protein. Bar = 50 μm.

Siberian tiger (Panthera tigris altaica). Immunohistochemistry of a malignant peripheral nerve sheath tumor showing neoplastic cells with strong red labeling for nerve growth factor receptor p75. Bar = 50 μm.
Characteristics of primary antibodies and antigen retrieval procedures performed for the definitive diagnosis of a malignant peripheral nerve sheath tumor (primary cutaneous mass and lung metastases) in a Siberian tiger (Panthera tigris altaica). *
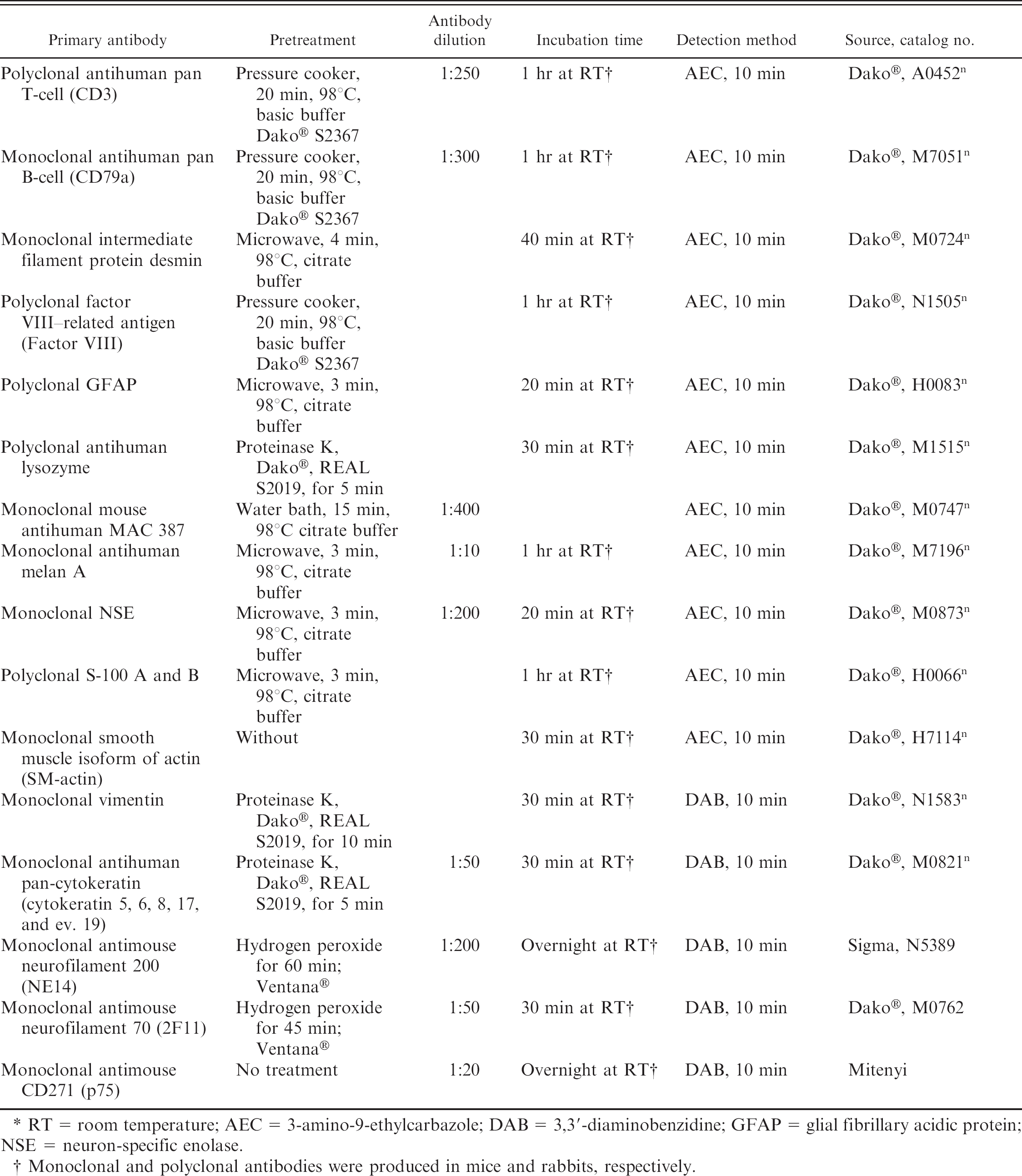
RT = room temperature; AEC = 3-amino-9-ethylcarbazole; DAB = 3,3′-diaminobenzidine; GFAP = glial fibrillary acidic protein; NSE = neuron-specific enolase.
Monoclonal and polyclonal antibodies were produced in mice and rabbits, respectively.
Footnotes
a.
Zalopine®, Orion Pharma Animal Health, Turku, Finland.
b.
Narketan®10, Vétoquinol AG, Belp, Switzerland.
c.
Aire-Cuf®, Bivona Inc., Gary, IN.
d.
Attane™, Provet AG, Lyssach, Switzerland.
e.
Somatom ART, Siemens Schweiz AG, Zürich, Switzerland.
f.
Ultravist® 300, Bayer (Schering) AG, Zürich, Switzerland.
g.
Sintenyl® i.v., Sintetica SA, Mendrisio, Switzerland.
h.
Vicryl, Johnson & Johnson Medical AG, Spreitenbach, Switzerland.
i.
Temgesic, Essex Chemie, Luzern, Switzerland.
j.
Antisedan®, Dr. E. Gräub, Bern, Switzerland.
k.
Amoxicillin 1000 mg, Streuli Pharma AG, Uznach, Switzerland.
l.
Metacam,® Boehringer Ingelheim GmbH, Basel, Switzerland.
m.
Superfrost Color, Menzel, Braunschweig, Germany.
n.
Dako Schweiz AG, Baar, Switzerland.
o.
Vetanarcol,® Veterinaria AG, Zürich, Switzerland.
