Abstract
An 8-month-old Golden Retriever dog was euthanatized because of a large cerebral mass extending from the right frontal lobe to the thalamus that was composed of both mature and immature neuronal cells. The better differentiated cells had abundant eosinophilic cytoplasm with prominent Nissl substance and were generally positive for neurofilament and variably positive for synaptophysin. The generally smaller and less-differentiated cells were infrequently positive for proliferating cell nuclear antigen and were negative for any neuronal and glial markers. No apparent glial differentiation of the immature tumor cells was detected. Based on morphologic and immunohistochemical features, the diagnosis of cerebral ganglioneuroblastoma was made. This neoplasm is very rare in all species, especially in the central nervous system, and has never been reported previously in this site in a dog.
Neuronal neoplasms in the central nervous system (CNS) of domestic animals are extremely rare. 2,3,8 The spectrum of tumors in the CNS showing neuronal differentiation include neurocytomas, neuroblastomas, gangliocytomas, and gangliogliomas. Although ganglion cells are conventionally located outside the CNS, the term may be applied to some CNS tumors containing neoplastic cells with the phenotype of mature neurons. 8 By this definition, ganglioneuroblastomas are characterized by a combination of immature neuroblastic cells with variable numbers of neoplastic cells with advanced neuronal differentiation. To our knowledge, although olfactory and thoracic ganglioneuroblastomas have been reported in dogs 5,7 and adrenal ganglioneuroblastoma was diagnosed in a calf, 6 cerebral ganglioneuroblastoma has not been reported previously in animals. This article describes the histomorphologic phenotype of a cerebral ganglioneuroblastoma in an 8-month-old Golden Retriever dog.
A 7-month-old male Golden Retriever dog was presented to the Veterinary Teaching Hospital of Osaka Prefecture University with a 2-week history of depression, anorexia, circling, hypersensivity on the right side of the face, and left ocular dysfunction. Computed tomography revealed a mass extending from the right cerebrum to the thalamus with scattered focal areas of hyperintensity. Hematologic examination revealed elevated levels of total cholesterol (388 mg/dl) and decreased levels of lactate dehydrogenase (25 U/liter). The neurologic signs gradually worsened over time until 1 month after presentation the dog was euthanatized at the owner's request.
Necropsy revealed a gray mass extending from the right frontal lobe to the thalamus measuring approximately 4.5 × 3.5 × 3 cm (Fig. 1). White foci and edematous areas were observed on the cut surface. No other abnormalities were observed in any organs outside the brain.
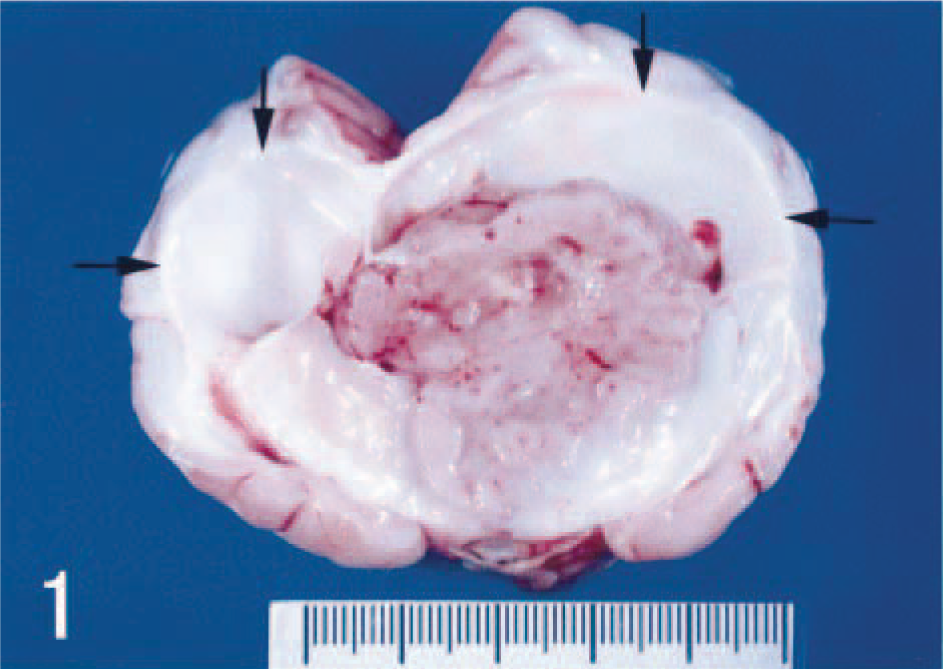
Cerebral ganglioneuroblastoma in the cortex. Cut section shows dilated ventricles (arrows), dorsal median sulcus, and pyriform lobes.
Brain was fixed in 10% neutral buffered formalin and routinely embedded in paraffin. Four-micrometer sections were stained with hematoxylin and eosin and cresyl violet for Nissl substance. Immunohistochemical evaluation was carried out using rabbit polyclonal antibodies against glial fibrillary acidic protein (GFAP, Dako, Copenhagen, Denmark), myelin basic protein (Dako), and mouse monoclonal antibodies against vimentin (Dako), high–molecular weight neurofilament (Serotec, Oxford, UK), synaptophysin (Chemicon International, Temecula, CA), and proliferating cell nuclear antigen (PCNA, Dako) with avidin–biotin–peroxidase kit (Dako). For electron microscopic examination, formalin-fixed samples were cut into 1-mm cubes, postfixed in osmium tetroxide, and embedded in epoxy resin. Ultrathin sections were double stained with uranyl acetate and lead citrate and examined in a transmission electron microscope (H-7500, Hitachi, Tokyo, Japan).
There were extensive areas of necrosis, hemorrhage, edema, and calcification within the large neoplastic mass. Histologically, the mass was composed of both large and compact immature cells (Fig. 2). The large cells had round nuclei and abundant eosinophlic cytoplasm that occasionally possessed short processes. These cells often had Nissl substance, which was clearly observable with special histochemical stains (Fig. 3). Binucleated tumor cells were also seen amongst the large cells. Immunohistochemistry for high–molecular weight neurofilaments revealed positive staining in the cytoplasm and processes of the large cells (Fig. 4), but they were nonimmunoreactive for GFAP.
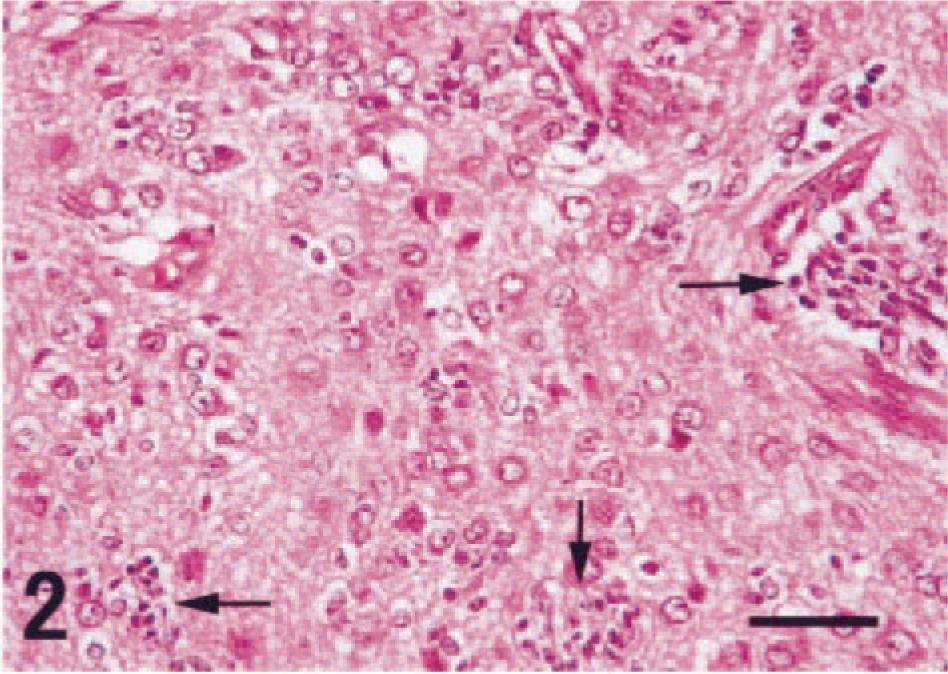
Neoplastic cells with both well-differentiated neuronal cells and immature, less-differentiated compact cells (arrows). HE. Bar = 50 µm.
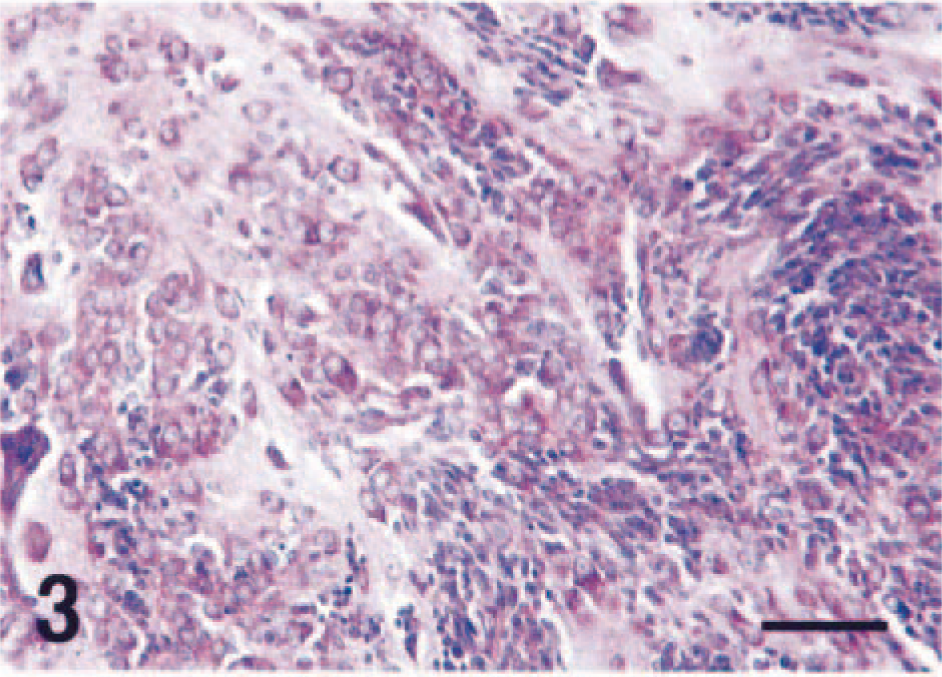
Neoplastic cells exhibiting advanced neuronal differentiation with abundant Nissl substance. Nissl stain. Bar = 60 µm.
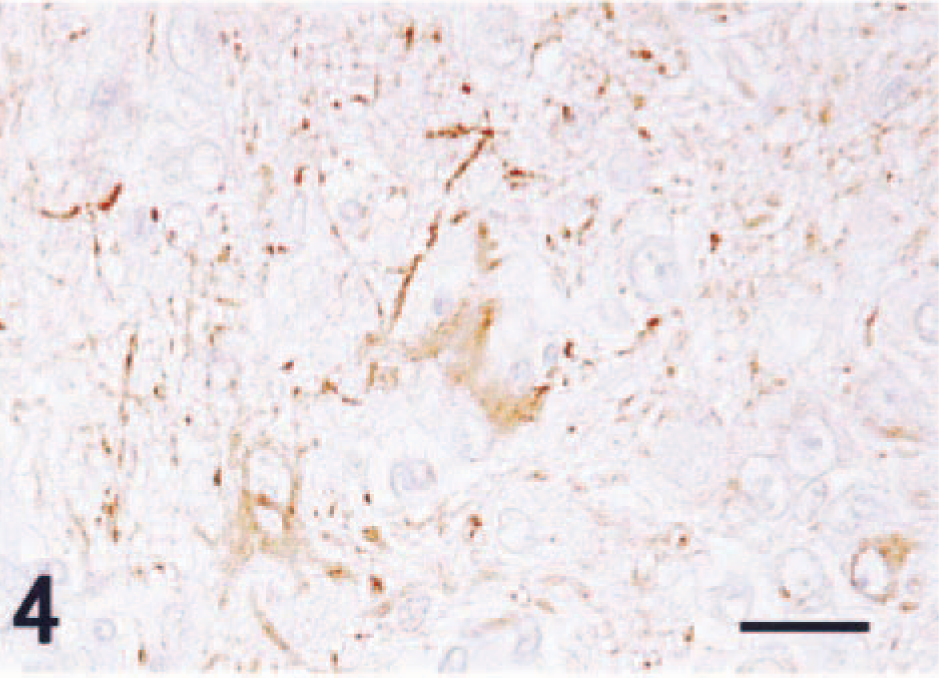
Neuronal cells stained with neurofilament immunohistochemistry. Counterstained with hematoxylin. Bar = 25 µm.
The small immature cells had indistinct cytoplasmic borders, scant cytoplasm, and round to fusiform nuclei and were consistent with neuroblasts. These small cells were often arranged in nests or cords (Fig. 3). Immunohistochemistry for neurofilaments and GFAP failed to demonstrate positive staining in this population. Synaptophysin immunohistochemistry revealed small foci of positive staining consistent with synapselike structures throughout the tumor (Fig. 5). There were relatively few PCNA-positive cells, and these were exclusively observed in the neuroblastic cell component (Fig. 6). GFAP-positive astrocytes were found in the tumor; however, neither cellular nor nuclear atypism was noted, and these were interpreted to be reactive rather than neoplastic. Astrogliosis was prominent in the normal brain tissues around the tumor.
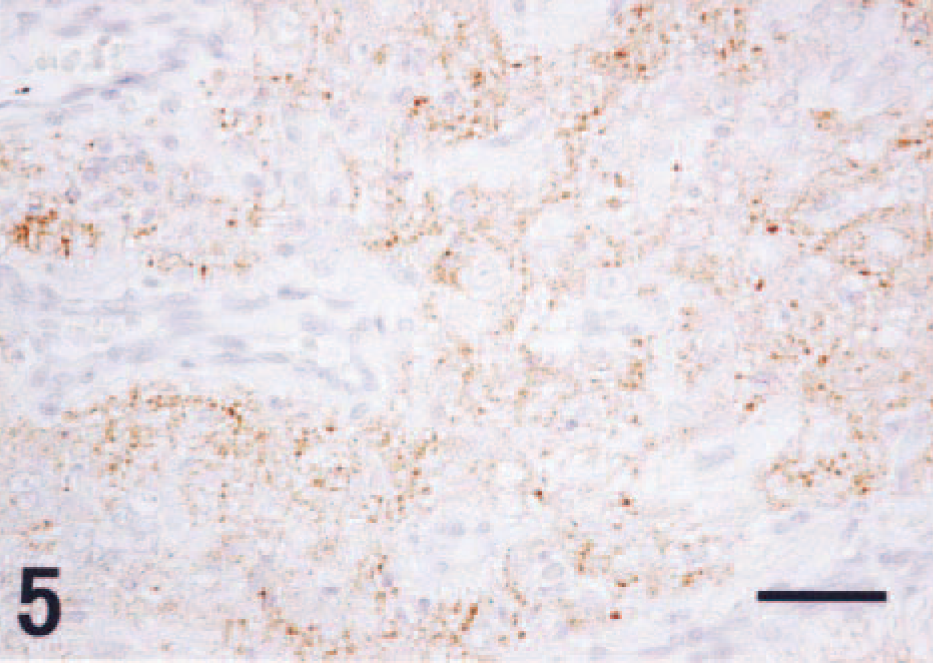
Scattered dotlike immunoreactivity for synaptophysin immunohistochemistry. Counterstained with hematoxylin. Bar = 50 µm.
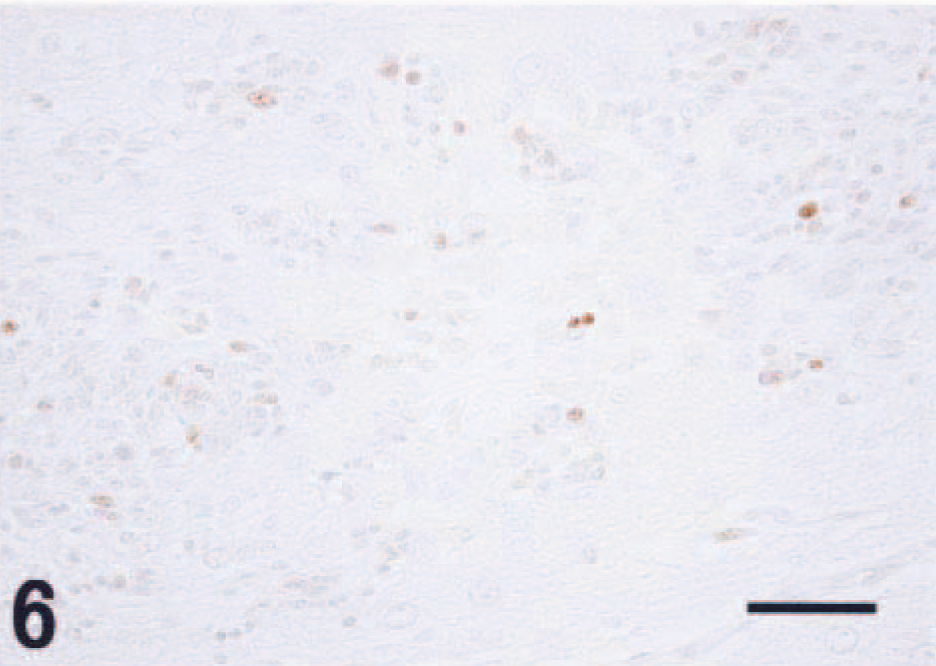
A few PCNA-positive cells in the immature cells. Counterstained with hematoxylin. Bar = 50 µm.
Electron microscopy revealed a well-developed rough endoplasmic reticulum and many mitochondria in the large neuronal tumor cells. The smaller neoplastic cells had a few mitochondria and small dense granules in the cytoplasm. Many synapselike structures containing dense vesicles were observed throughout the tumor.
Embryonic tumors composed of undifferentiated cells with the potential for differentiation into neuronal, glial, muscle, and melanotic cells have been classified within a group referred to as primitive neuroectodermal tumors (PNETs). 1–4,8 PNETs most often occur in the cerebellum as medulloblastomas; however, similar tumors may occasionally be encountered in the cerebral hemispheres in humans. 4 Within this group, differential diagnoses include neuroblastomas, ganglioneuromas, and ganglioneuroblastomas. The presence of both undifferentiated and differentiated neuronal cells is a hallmark histologic criterion for diagnosis of a ganglioneuroblastoma. 5–7
Ganglioneuroblastomas are extremely rare tumors in animals, and a few cases have been reported in dogs 5–7 and a calf. 6 In humans, ganglioneuroblastomas are generally rare tumors and are especially infrequent in the CNS. In humans, the mean age of occurrence is about 13 years, although 10–25% of the tumors develop during the first 5 years of life without a sex predilection. 4 The present case occurred in a juvenile dog, suggesting the hamartomatous nature of this tumor.
