Abstract
Embryonal central nervous system (CNS) tumor is a rare malignant neoplasm that arises from the neural crest. Herein we describe an embryonal tumor in the brain of an adult male domestic goose (Anser anser) that was euthanized after a short history of wandering and lethargy. Grossly, a focal, bright-red, soft plaque was present on the ventral aspect of the brain, extending from the optic chiasm to the midbrain, as well as into the dorsal neuroparenchyma. Histologically, the nodule consisted of a poorly demarcated population of neoplastic cells arranged in streams and bundles, and occasionally palisading around small capillaries (pseudorosettes). Neoplastic cells were elongate and had scant, finely granular, eosinophilic cytoplasm, and elongate nuclei with dense chromatin. Mitoses were uncommon. Scattered foci of intratumoral necrosis and microvascular proliferation were present. Neoplastic cells were immunoreactive for doublecortin and neuronal nuclei. Ultrastructurally, neoplastic cells had elongated-to-polyhedral cytoplasm with short processes, scarce intermediate filaments, and small round mitochondria and rough endoplasmic reticulum. The cell membrane had varied numbers of intercellular anchoring-like junctions, and nuclei were round-to-elongate and had marginal aggregates of heterochromatin. Morphologic, immunohistochemical, and ultrastructural findings in our case are consistent with a poorly differentiated embryonal tumor.
Embryonal central nervous system (CNS) neoplasms encompass a number of tumors including medulloblastoma, medulloepithelioma, neuroblastoma, and ganglioneuroblastoma, among others. 23 Primitive neuroectodermal tumors (PNETs) were first described as part of this group of tumors in humans in 1973, and were characterized as poorly differentiated neoplasms that did not fulfill the morphologic criteria necessary to be classified as a distinct embryonal tumor type. 12 Although controversial, the term PNET was utilized for decades to refer to CNS tumors composed of germinal neuroepithelial cells that can undergo neuronal or glial cell differentiation. 2 The 2016 World Health Organization Classification of Tumours of the Central Nervous System has eliminated PNET from its diagnostic lexicon. As of 2018, the current classification is based on morphologic and molecular features, and classifies these neoplasms into 2 main groups. 23
Medulloblastomas are common and well-characterized infratentorial neoplasms that affect mainly children and arise in the cerebellum or dorsal brainstem. Embryonal neoplasms other than medulloblastomas are classified as embryonal CNS tumors, an umbrella term that includes medulloepitheliomas, neuroblastomas, ganglioneuroblastomas, and other less common or poorly differentiated embryonal tumors. These neoplasms are less well characterized given their rare occurrence and lack of morphologic and molecular features that could allow for a final diagnosis and subsequent classification in a different group. These neoplasms also tend to affect children and, unlike medulloblastomas, typically arise within the telencephalic hemispheres, or rarely brainstem and spinal cord. 23
Embryonal CNS neoplasms are rare in the veterinary medical literature. Medulloblastomas have been described in dogs, cattle, pigs, rats, and non-human primates, with single case reports in a cat, a grizzly bear, and a kowari.1,4,9,11,16,18,19,25–28,31,32 These tumors affect young calves and adult individuals of other species, and occur mainly in the cerebellum. Other CNS embryonal tumors are exceedingly rare and have been described mainly in dogs, with fewer reports in cattle and a colobus monkey.3,5,13,17,20,22,24 Tumors appear to have a predilection for younger animals and occur mainly in supratentorial locations or, rarely, in the spinal cord. Descriptions of avian CNS embryonal tumors are restricted to brief mentions in textbooks and rare case reports of a neuroblastoma in the rostral telencephalon and a medulloblastoma in the cerebellum in psittacines.7,8,30
Herein we describe the pathologic and diagnostic features of a CNS embryonal tumor in the brain of an adult male domestic goose (Anser anser) that was lethargic and wandering aimlessly on a farm. No other geese were present on the farm. Euthanasia was elected because of the suspicion of avian influenza, and the carcass was subjected to autopsy. Gross anatomic changes were restricted to the brain. A focal, bright-red, soft, homogeneous, 5-mm plaque was present on the ventral aspect of the brain, extending from the optic chiasm to the midbrain (Fig. 1). Transverse sections of the brain revealed that the lesion was predominantly intraparenchymal and expanded the left basal nuclei, thalamus, midbrain, and caudal brainstem. No pathologic changes were observed in the pituitary gland. Tissue samples were immersed in 10% buffered formalin, processed routinely for histology, and stained with hematoxylin and eosin. Histologically, a poorly demarcated population of neoplastic cells effaced the left basal nuclei and adjacent telencephalic cortex, thalamus, midbrain, and brainstem (Fig. 2). The neoplasm extended to the adjacent leptomeninges. Neoplastic cells were arranged in closely packed streams and bundles (Fig. 3) and occasionally palisaded around small capillaries (pseudorosettes) (Fig. 4). Neoplastic cells were elongate and had scant-to-moderate finely granular, eosinophilic cytoplasm and variably defined cell borders. Nuclei were elongate and had dense chromatin with indistinct nucleoli. There were 4 mitoses in ten 400× fields. Scattered intratumoral areas of necrosis and microvascular proliferation were also present. The surrounding neuroparenchyma was vacuolated and had areas of astrocytosis. Other examined tissues had no pathologic changes.
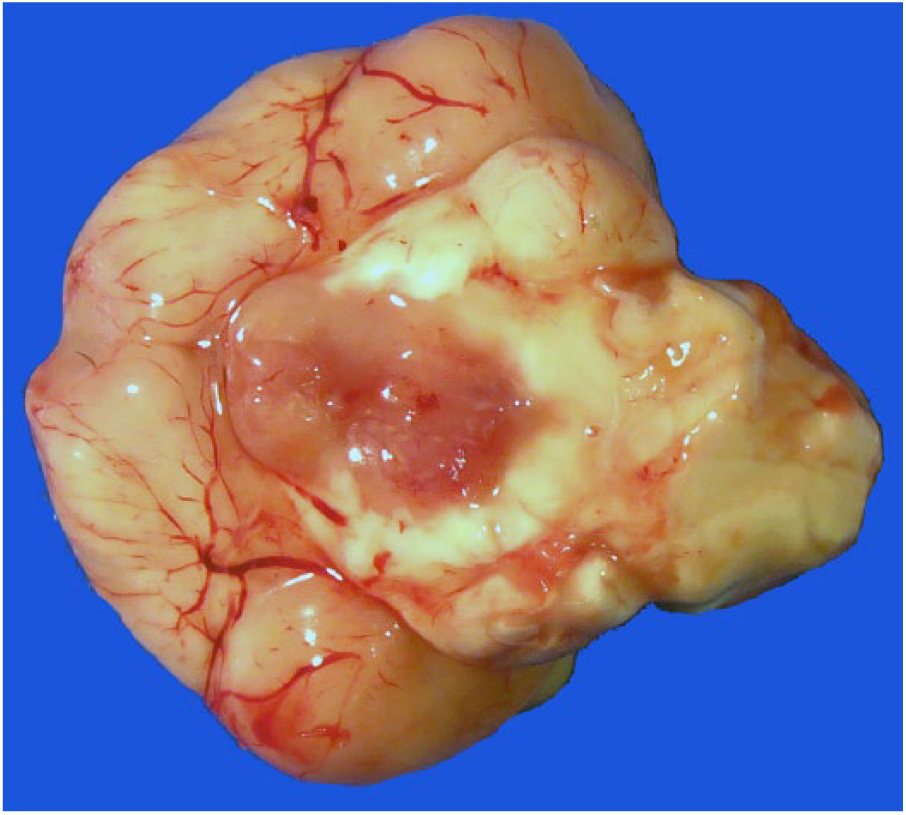
A focal, bright-red, soft, homogeneous, 5-mm neoplasm is present on the ventral aspect of the brain, extending from the optic chiasm to the midbrain.
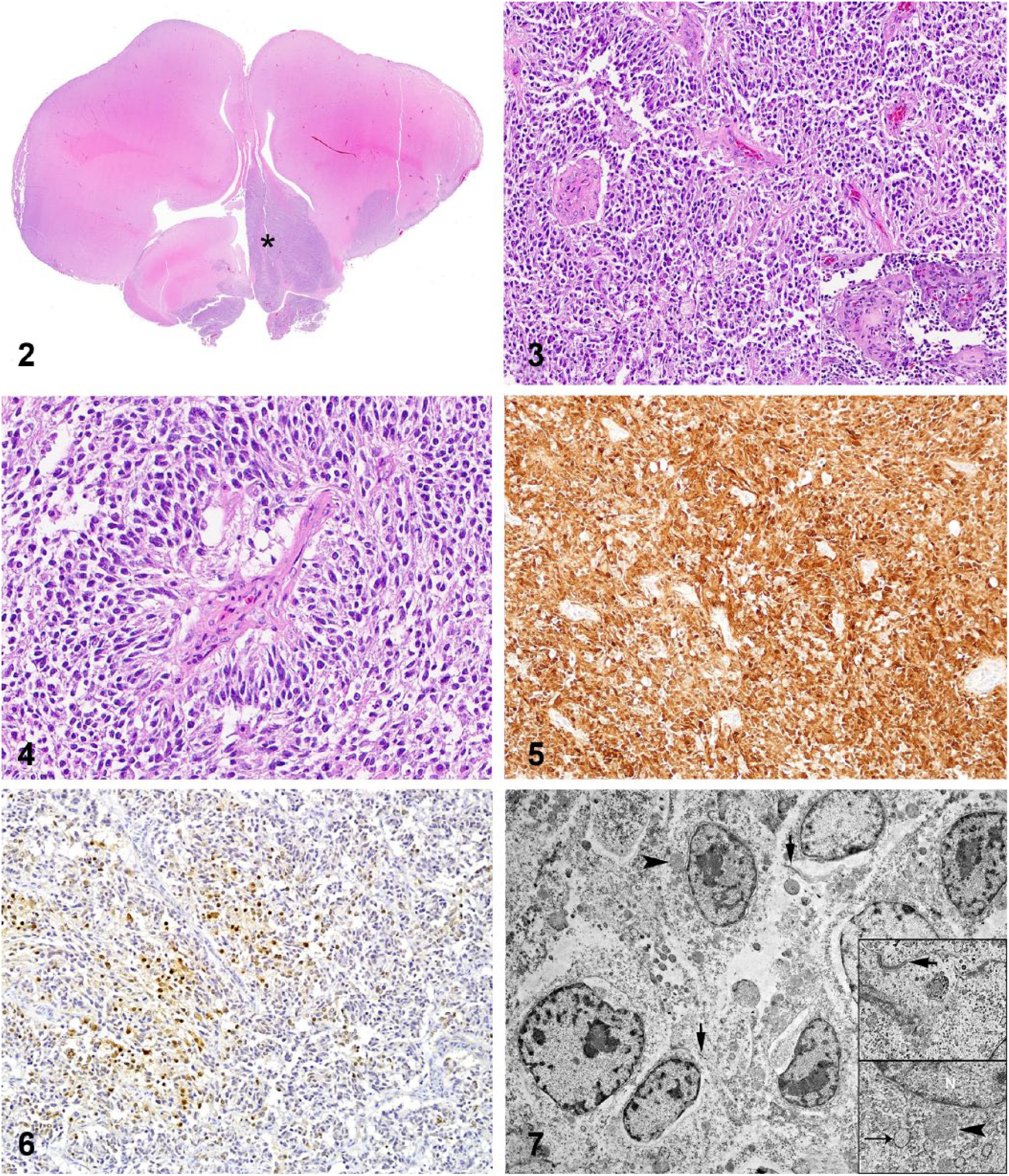
The neoplasm is predominantly intraparenchymal and expands primarily the ventral leptomeninges and the left basal nuclei (asterisk), extending into the adjacent left telencephalic cortex. H&E.
Tissue sections of the neoplasm were subjected to immunohistochemistry (IHC) for doublecortin (DCX; rabbit polyclonal, Abcam, Cambridge, MA; 1:4,000 dilution at 60 min), neuronal nuclei (NeuN; guinea pig polyclonal, Millipore Sigma, Burlington, MA; 1:3,000 dilution at 60 min), glial fibrillary acidic protein (GFAP; mouse monoclonal, Biogen, Cambridge, MA; 1:4,000 dilution at 60 min), oligodendrocyte lineage transcription factor 2 (Olig2; rabbit monoclonal, GeneTex, Irvine, CA; 1:400 dilution at 60 min), neurofilament (NF; mouse monoclonal, Biogen; 1:1,000 dilution at 60 min), synaptophysin (SYP; mouse monoclonal, Biogen; 1:600 dilution at 60 min), vimentin (VIM; mouse monoclonal, BioGenex, Fremont, CA; 1:3,000 at 60 min), and pancytokeratin Lu-5 (PCK; mouse monoclonal, Biocare, Concord, CA; 1:100 dilution at 90 min). Neoplastic cells had diffuse and robust immunoreactivity for DCX (Fig. 5) and patchy immunoreactivity for NeuN (Fig. 6). Scattered reactive astrocytes had immunoreactivity for GFAP. Neoplastic cells were immunonegative for GFAP, Olig2, NF, and SYP. Utilized antibodies had been either previously validated for avian species in our laboratory (Howerth et al. Useful antibodies and techniques for immunohistochemical studies in waterfowl. Proc 31st Ann Meeting Waterbird Society; 2007; Barcelona, Spain) or labeled the adjacent normal nervous tissue (internal control) appropriately.
Sections of the neoplasm were examined by transmission electron microscopy (TEM). Formalin-fixed tissue was post-fixed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer and in 1% osmium tetroxide in 0.1 M sodium cacodylate buffer (Electron Microscopy Sciences, Hatfield, PA). Samples were processed, infiltrated and embedded (Embed 812 resin, Electron Microscopy Sciences). Thin sections (60–70 nm) were contrasted with 5% uranyl acetate and Santos lead citrate (Electron Microscopy Sciences). The neoplasm was composed of a population of cells that had elongated-to-polyhedral cytoplasm with short processes, scarce intermediate filaments, and small round mitochondria and rough endoplasmic reticulum. Cell membranes had varied numbers of intercellular anchoring-like junctions. Nuclei were round-to-elongate and had marginal aggregates of heterochromatin (Fig. 7). Scattered throughout the neoplasm were cells with abundant stellate cytoplasm and robust cell processes, abundant intermediate filaments, and scarce small organelles (mainly mitochondria); nuclei were elongated-to-round with delicate sparse heterochromatin. These cells extended their processes to end on capillaries or arteriolar basement membrane. These features were consistent with astrocytes.
The morphologic, IHC, and ultrastructural findings in our case are consistent with a poorly differentiated CNS embryonal tumor. Streams of neoplastic cells with elongate cytoplasm and elongate dark nuclei with occasional pseudorosettes are hallmarks of CNS embryonal tumors in various animal species.3,8,13,17,20,24,27 Based on the current WHO classification of tumors of the CNS and previous reports in the veterinary medical literature, a medulloblastoma was considered less likely given the supratentorial rather than cerebellar location of the neoplasm.3,5,13,17,20,22–24 The presence of rosettes is variable and not necessarily a feature of these tumors in veterinary species3,5,8,17; rosettes were not present in our case.
No substantial amount of information about the IHC profile of embryonal tumors has been published in veterinary medicine, and documented data have shown a wide range of variability that rarely allows for subclassification of these neoplasms within a specific group.3,5,13,17,20,22,24 DCX and NeuN IHC are useful immunomarkers for diagnostic confirmation of embryonal and neuronal tumors, respectively, but results must be interpreted with caution and in light of tumor morphology and association with additional immunomarkers.6,29 DCX is a microtubule-associated protein involved with neuroblastic migration during brain development, and it is expressed only by a limited number of neurons in the adult brain. 6 Although DXC IHC has been utilized for diagnostic confirmation of embryonal tumors, its expression has also been demonstrated in other CNS tumors of humans and dogs, including gliomas and meningiomas.6,10,15 NeuN has been utilized to confirm neuronal origin in diagnostic neuro-oncopathology, but its expression has also been demonstrated in different gliomas. 29 Although the positive immunolabeling for DCX and NeuN and the lack of GFAP and Olig2 immunoreactivity is supportive of a neuronal origin in our case,6,29 no evidence of neuronal differentiation was observed on histology or TEM. Although most embryonal CNS tumors in humans express Olig2, immunolabeling has been shown to be variable and inconsistent, indicating that this immunomarker should not be relied upon in the diagnosis of these neoplasms. 21 In addition, no immunolabeling of neoplastic cells was detected with more specific neuronal markers, such as NF and SYP, in contrast to previous reports of a cerebral neuroblastoma in a budgerigar and a medulloblastoma in a cockatoo.7,8 The presence of anchoring-like junctions on TEM could be supportive of a neuronal or glial cell origin for this neoplasm. 2 The absence of immunolabeling for glial cell markers undermines a glial cell origin, the absence of cilia and microvilli or secretory granules is not consistent with ependymal differentiation, and the absence of cytokeratin immunolabeling is not consistent with epithelial differentiation.2,14
The morphologic features in our case are consistent with a poorly differentiated CNS embryonal tumour. 23 The scattered GFAP-positive cells throughout the tumor were considered to be reactive astrocytes, as reported previously in dogs.20,25 The location of the neoplasm warranted its differentiation from a pituitary tumor. 7 However, no morphologic, immunohistochemical, or ultrastructural evidence for a pituitary gland neoplasm was present in our case.
Footnotes
Acknowledgements
We thank Nicole Young (Histology Laboratory, Department of Pathology, College of Veterinary Medicine, University of Georgia) for outstanding support with immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
