Abstract
Two distinct nodules developed in a cryptorchid testis of an 8-year-old male West Highland White Terrier. One nodule was a Sertoli cell tumor. The other was a spermatocytic seminoma with focal primitive neuroectodermal differentiation: formation of Homer–Wright rosettes and perivascular pseudorosettes, with immunoreactivity for S-100 protein, neuron-specific enolase, synaptophysin, neurofilament–68 kDa, microtubule-associated protein 2, and vimentin. The dog was alive and healthy 2 years after castration.
The most common testicular tumors in dogs are seminoma, sustentacular (Sertoli) cell tumor, and interstitial (Leydig) cell tumor, 10 which sometimes coexist in one or both testes, especially in cases of cryptorchidism.10,14,21 In humans, classical seminomas are highly malignant and typically occur in young adults. Spermatocytic seminomas, which generally develop in older men, are less common and less malignant than classical seminomas. 17 Classical seminoma is derived from gonocytes, which are undifferentiated testicular stem cells, whereas spermatocytic seminoma is derived from cells capable of maturing at least to the stage of spermatogonia–pachytene spermatocytes.1,19 The neoplastic cells of classical seminoma, like the gonocytes from which they are derived, contain glycogen 16 (periodic acid–Schiff [PAS] positive) and express placental alkaline phosphatase (PLAP) immunohistochemically.9,16 In contrast, spermatocytic seminomas, which are derived from more differentiated cells, do not express PLAP and are PAS negative.3,4
Canine seminomas have also been classified as classical and spermatocytic.6,15 We report a case of spermatocytic seminoma with focal primitive neuroectodermal differentiation and Sertoli cell tumor in the cryptorchid testis of a dog.
Case Presentation
A 7-kg, 8-year-old male West Highland White Terrier, with undescended testes, was presented to a veterinary clinic because of an enlarged left testis (4.0 × 3.5 × 2.0 cm, including the epididymis), which was just deep to the subcutis near the external inguinal ring. The right testis, similarly situated near the contralateral inguinal ring, was atrophied (2.5 × 1.5 × 1.5 cm). Both testes were excised, fixed in 10% neutral buffered formalin, and submitted for routine histologic processing.
Macroscopically, the enlarged left testis contained 2 distinct nodules separated by fibrous tissue. One nodule (2.5 × 2.0 × 2.0 cm) was gray-white; the other (2.0 × 2.0 × 1.7 cm), red-brown. Both nodules were solid and surrounded by nonneoplastic parenchyma. Paraffin sections were stained with hematoxylin and eosin and PAS. For immunohistochemistry, the avidin–biotin complex technique was used with the primary antibodies listed in Table 1. The chromogen was 3,3′-diaminobenzidine; the counterstain was hematoxylin. Mature mouse brain was used as the positive control tissue for all immunohistochemistry except nestin, for which mouse brain at 15 days gestation was used. A classical canine seminoma specimen was used as a positive control for PLAP.
Reagents and Methods for Immunohistochemistry
a Antigen retrieval was by autoclaving at 121°C for 10 minutes, by microwaving at 90°C for 10 minutes in 10 mM citrate buffer (pH, 6.0), or by heat-induced epitope retrieval at 95°C for 40 minutes in preheated Dako Target Retrieval Solution (pH, 9.0).
Histologic and Immunohistochemical Findings
Histologically, the 2 testicular nodules were separated by fibrous tissue. The gray-white nodule was a germ cell tumor composed of 3 cell types arranged in an intratubular to diffuse pattern (Fig. 1). The predominant cell type was of intermediate size with a variable amount of amphophilic cytoplasm and a round-to-oval nucleus, with fine granular chromatin and one nucleolus. The second type of cell was small with a hyperchromatic nucleus and scanty cytoplasm. The third cell type was large and round to polyhedral, with a round, oval, or indented nucleus and ample amphophilic cytoplasm. Many of the large cells had filamentous spireme-like chromatin distribution, like that in spermatocytes, and one to several nucleoli. Scattered bizarre giant cells had 1 to 5 nuclei. There were 2 or 3 mitotic figures per 400× field; atypical mitotic figures were common (Fig. 1 inset). Aggregates of lymphocytes were scattered through the tumor.
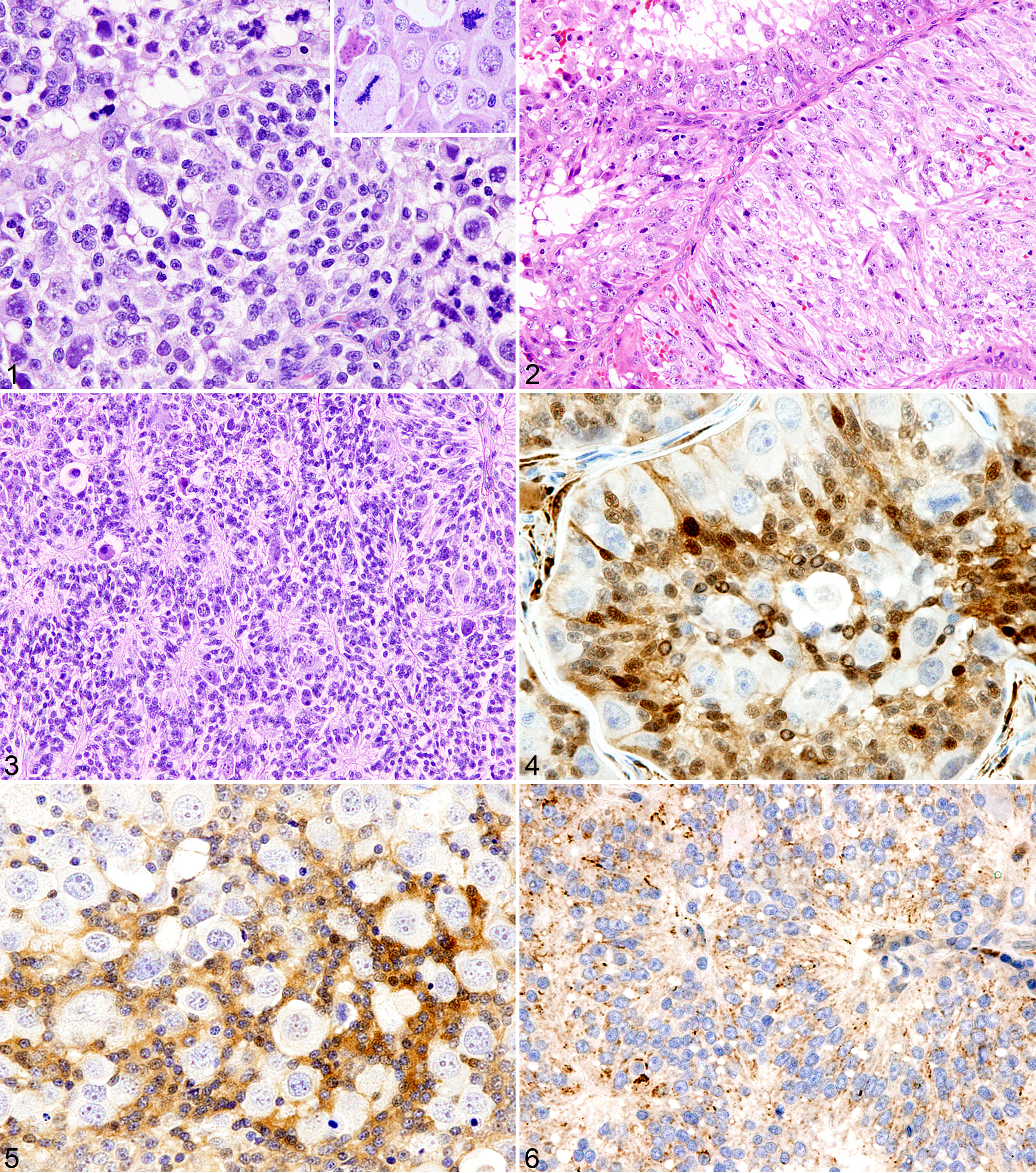
The red-brown testicular nodule was a Sertoli cell tumor (Fig. 2) with intratubular pattern, accompanied by scattered hemorrhagic foci with or without hemosiderin deposition. The elongated neoplastic cells had variable amount of cytoplasm; some contained lipid droplets. The number of mitotic figures varied from 0 to 3 per 400× field. Foci of hemorrhage and lymphocytic infiltration were scattered through the tumor. Neoplastic cells were in vessels in surrounding fibrous tissue.
Histochemical and immunohistochemical results for both tumors are in Table 2. For the germ cell tumor, the large, spermatocyte-like cells were PAS negative and lacked PLAP immunoreactivity. The intermediate-sized cells expressed inhibin α. The small cells that formed nests with Homer–Wright rosettes or pseudorosettes (Fig. 3) were positive for S-100 protein (Fig. 4), neuron-specific enolase (NSE; Fig. 5), synaptophysin (Fig. 6), neurofilament–68 kDa, microtubule-associated protein 2, and vimentin and negative for nestin and glial fibrillary acidic protein. The large spermatocyte-like cells did not express any of these markers (Figs. 4, 5). The cells of the Sertoli cell tumor had strong immunoreactivity for NSE and diffuse moderate cytoplasmic immunoreactivity for vimentin, inhibin α, and synaptophysin but were negative for S-100 protein, neurofilament–68 kDa, microtubule-associated protein 2, nestin, and glial fibrillary acidic protein.
Histochemical and Immunohistochemical Reactivity a
a Reactivity: –, no immunoreactivity; +, mild; ++, moderate; +++, intense.
b Minor portion of neoplastic cells had fine, granular positive reactivity for periodic acid–Schiff.
c Intermediate-sized neoplastic cells population alone had mild immunoreactivity.
In summary, the enlarged cryptorchid testis contained a germ cell tumor, which was classified as spermatocytic seminoma with primitive neuroectodermal tumor (PNET), and a separate Sertoli cell tumor. Nonneoplastic tissue in the enlarged left testis, as well as the cryptorchid right testis, consisted of small seminiferous tubules lined only by Sertoli cells with increased interstitial Leydig cells and fibrous tissue. The dog was alive and healthy without apparent neoplasia 2 years after castration.
Discussion
The distinctive histologic and immunohistochemical features of the 2 testicular tumors and the lack of any continuity between them tend to eliminate a mixed germ cell–sex cord stromal tumor from the differential diagnosis. The immunohistochemical expression of the Sertoli cell tumor in this dog is similar to that reported for NSE in normal and neoplastic Sertoli cells in a canine mixed germ cell–sex cord stromal cell tumor, 18 inhibin α in a canine Sertoli cell tumor,18,21 and synaptophysin in human Sertoli cell tumors. 13
Although seminomas of domestic animals were not subclassified as classical or spermatocytic by the World Health Organization committee, 10 most canine seminomas are similar to human spermatocytic seminomas in histologic appearance and biological behavior. 15 In this dog, the seminoma was negative for PAS and PLAP and had other histologic features of spermatocytic seminoma, such as variable cell size, bizarre giant cells, 1 to 5 nucleoli, high mitotic rate with atypical figures, and filamentous chromatin.6,15,18 Therefore, the tumor was diagnosed as spermatocytic seminoma. Inhibin-α immunoreactivity, observed in this case, has been reported in seminomas, including spermatocytic seminomas.2,21 Interestingly, in a recent study of canine seminomas, spermatocytic seminomas (negative for PLAP and PAS) had lower angiogenesis than classical seminomas (positive for PLAP and PAS), suggesting a basis for their less aggressive biological behavior. 11 Focal lymphocytic infiltration, as observed in this case, is a common feature of human and canine seminomas and may represent an immune response to the tumor and a more favorable prognosis. 7
Part of this germ cell tumor comprised a population of cells that were arranged in Homer–Wright rosettes or pseudorosettes and expressed multiple neuronal markers. This was the basis for the additional diagnosis of peripheral PNET. 8 Peripheral PNETs are reported with testicular germ cell tumors in humans. 20 Furthermore, neuroendocrine tumors often coexist with mature forms of germ cell tumors, such as teratomas, and can be concomitantly present with classical seminomas. 12 However, spermatocytic seminomas are rarely associated with other neoplastic components in humans. 5 In this case, the admixture of neoplastic spermatocytic cells and PNET cells in the same tumor suggests the possibility that both populations were derived from a common progenitor cell population. However, because premeiotic germ cells, which are committed to the spermatogenic lineage, are considered to be the origin of spermatocytic seminomas,1,19 their dedifferentiation to pluripotent neuroectodermal cells may be uncommon.
Footnotes
Acknowledgement
We thank Mrs Shigeko Suzuki for her excellent technical assistance in preparing the histologic specimens.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
