Abstract
Junctional adhesion molecule A (JAM-A) is an immunoglobulin superfamily protein that plays an important role in the assembly and maintenance of tight junctions and the establishment of epithelial cell polarity. The feline JAM-A (fJAM-A) is a functional receptor for feline calicivirus (FCV). Among natural diseases associated with FCV infection, isolates that cause oral vesicular disease are detected in epithelial cells; however, isolates that cause systemic disease are detected in multiple cell types. The distribution of an FCV receptor or receptors in feline tissues is relevant to viral pathogenesis in that it should reflect the wide latitude of clinical sequelae associated with FCV infection. The authors examined the expression of feline JAM-A in the cat by using confocal immunofluorescence localization on normal tissues, with special regard to tissue targets of naturally occurring FCV. As described in the human and the mouse, fJAM-A was widely distributed in feline tissues, where it localized at cell–cell junctions of epithelial and endothelial cells. fJAM-A was highly expressed on feline platelets, with lower levels of expression on feline peripheral blood leukocytes. Additionally, FCV infection of a feline epithelial cell monolayer causes redistribution of fJAM-A to the cytosol of infected cells. It is reasonable to propose that the spectrum of lesions caused by FCV reflects disruption of intercellular junctions that rely on fJAM-A function and tight junctional integrity.
Keywords
Feline caliciviruses (FCVs) are highly contagious pathogens of cats. Despite widespread vaccination, the prevalence of FCV remains high, and disease outbreaks commonly occur in group-housed cats. Of concern are the more rare FCV outbreaks that cause high mortality as a consequence of severe systemic disease. Most common, in acute FCV infection, viral antigen is detected within the cytoplasm of epithelial cells lining the mouth and tongue, and affected cats present with ulcerative glossitis and/or stomatitis. In cats with severe, systemic FCV disease, virus is detected in endothelial cells and multiple epithelial cell types, including those that line the mucosa of the oral cavity, pneumocytes, epidermal cells, hepatocytes, and pancreatic acinar cells. 19,24 Chronic FCV infection is not associated with clinical disease; however, some cats infected with FCV will persistently shed virus in oropharyngeal secretions. 3,26 In addition, FCV has been isolated from feces, 14 indicating that cells in the lower intestinal tracts may support infection.
Feline junctional adhesion molecule A (fJAM-A) is a functional receptor for FCV, and expression of fJAM-A in nonpermissive cell lines—hamster lung (HmLu-1) cells, human kidney epithelial (293-T) cells, and Chinese hamster ovary cells—confers susceptibility to FCV infection. 11,17 In feline cells (Crandell feline kidney cells), FCV infection is blocked by a polyclonal antibody against fJAM-A, indicating that fJAM-A is required. 11,17 Based on these findings, the tissue- and cell-specific distribution of fJAM-A should reflect known viral tropism and pathologic sequelae of FCV infection.
In human and mouse tissues, junctional adhesion molecule A (JAM-A) is concentrated at intercellular tight junctions of epithelial (kidney, lung, intestine) and endothelial cells, and it is found on the surface of leukocytes and platelets. 7,12,28 JAM-A forms dimeric complexes on the surface of cells, and JAM-A homotypic dimer–dimer interactions between adjacent cells are required for maintenance of normal junctional integrity and to mediate transmigration of leukocytes between endothelial and epithelial cell junctions. 6,9,10,12 Antibody cross-linking of JAM-A on the surface of platelets triggers aggregation; however, the role of JAM-A in platelet function is unclear. 7 The distribution of fJAM-A in feline tissues, which has direct relevance to FCV disease pathogenesis, is currently unknown.
Here, we describe the distribution of fJAM-A in normal feline tissues and in feline epithelial cells infected with FCV. We found that fJAM-A was widely distributed in feline tissues, where it localized at cell–cell junctions of epithelial and endothelial cells. In epithelial cells fJAM-A was concentrated in the region of the apical junctional complex. fJAM-A was highly expressed on feline platelets, with lower levels of expression on feline peripheral blood leukocytes. The presence and distribution of fJAM-A in feline tissues and circulating hematopoietic cells in part explains the susceptibility of multiple cell types to FCV infection. In cultured feline epithelial cells, the distribution of fJAM-A was altered during FCV infection.
Materials and Methods
Tissue Preparation and Cell Culture
Tissues from 2 cats submitted and donated for standard, unrestricted necropsy (School of Veterinary Medicine, University of California, Davis) were collected within 2 hours of euthanasia. The cats were < 1 year old. No clinical signs associated with respiratory, oral, or gastrointestinal disease were present in either cat. Sections of tongue, skin, liver, lung, spleen, kidney, pancreas, jejunum, colon, heart, brain, and bone marrow were collected, processed, and analyzed in both cats. Tissues were fixed for up to 12 hours in 10% buffered formalin, incubated in phosphate-buffered saline (PBS) at 4°C for 24 hours, then placed into a 30% sucrose solution until saturated (4–12 hours). Tissues were then embedded in OCT (Ted Pella, Inc, Redding, CA) and flash-frozen on dry ice. Sections, 10 to 12 μm thick, were cut onto Poly-Prep slides (Sigma-Aldrich, Inc, St Louis, MO). Replicate tissue samples were processed for routine hematoxylin and eosin evaluation.
Monolayers of the feline mammary epithelial cell line (FMEC) UCD-04-2, maintained as previously described, 20 were plated in three independent replicate experiments at 1 × 105 cells per well in 8 chambered slides (Nalge Nunc Inc, Naperville, IL) and grown to confluence at 37°C with 5% CO2 for 3 days. Cell monolayers were adsorbed with the UCD-AN123 isolate of FCV at a multiplicity of 10 plaque-forming units per cell for 20 minutes at room temperature, then washed twice and further incubated at 37°C for 6 hours before fixation in 2% paraformaldehyde for 20 minutes at room temperature. FCV isolate UCD-AN123 was originally obtained from an oropharyngeal swab of a shelter-housed female adult cat and grown as previously described. 20
Immunofluorescence and Confocal Microscopy
Fixed slides were washed twice with PBST (ie, PBS containing 0.1% Triton X-100) and blocked with 2% bovine serum albumin (BSA) in PBST for 1 hour or overnight at 4°C. A rabbit antiserum against the purified fJAM-A ectodomain (described previously 17 ) and a monoclonal anti-FCV antibody (Custom Monoclonals International, West Sacramento, CA) were diluted in blocking buffer containing 5% normal donkey serum and incubated for 1 hour at room temperature or 4°C overnight in a humidified chamber. Following three washes in PBST, bound antibodies were detected with Alexa488-conjugated donkey anti-rabbit and Alexa568-conjugated goat anti-mouse immunoglobulin G (Molecular Probes, Invitrogen Corp, Carlsbad, CA). Preimmune serum (in the case of the fJAM-A antibody) and secondary antibody alone were used as negative controls. After three additional washes in PBST, cover slips were mounted with ProLong Gold with DAPI (Molecular Probes, Invitrogen Corp), and images were acquired with an Olympus FV500 series confocal microscope (Olympus, Tokyo, Japan) equipped with argon and krypton lasers and 405 diode. Image stacks were collected on the z-axis, and steps were made at the optimal voxel distance for a plan achromatic 40× objective. Stack depths were from 5 to 8 μm with a pixel resolution of 1024 × 1024. The brightness levels were enhanced using Adobe Photoshop (Adobe, Mountain View, CA).
Flow Cytometry
We used flow cytometry to assay expression of fJAM-A on feline leukocytes and platelets in EDTA-anticoagulated blood. Erythrocytes were first lysed in an ammonium chloride lysis buffer at a 1:25 blood:buffer ratio for 10 minutes at room temperature. Leukocytes were then isolated by centrifugation (300 × g for 5 minutes) and washed in PBS containing 1% BSA and 0.05% sodium azide (ie, PBSA). Cell counts were performed with an electronic impedance counter (Z2 Coulter Counter, Beckman Coulter, Miami, FL). Approximately 1 × 106 washed cells were labeled with fluoroisothiocyanein-conjugated fJAM-A antibody (1:2000) or an isotype control. 17 Platelets in 1% paraformaldehyde-fixed blood diluted in PBSA were immunostained with fluoroisothiocyanein-conjugated anti-fJAM-A (1:2000) and a phycoerythrin-conjugated antibody against human CD61 (1:1000, clone SZ21, Beckman Coulter, Miami, FL [a platelet-specific marker]) or isotype controls. For both leukocytes and platelets, samples were incubated with antibodies for 30 minutes on ice and washed twice (in PBSA, then in PBS); afterward, cells were analyzed by flow cytometry. For data analysis, leukocyte subpopulations (neutrophils, monocytes, and lymphocytes) and platelets were identified and gated separately on the basis of their characteristic location in forward-scatter and side-scatter dot plots (see Results section). A minimum of 10,000 events were counted in regions gated to exclude cell debris. The percentage of gated cells that were positive for fJAM-A and CD61 was determined from histogram plots.
Results
JAM-A Expression in Normal Feline Tissues
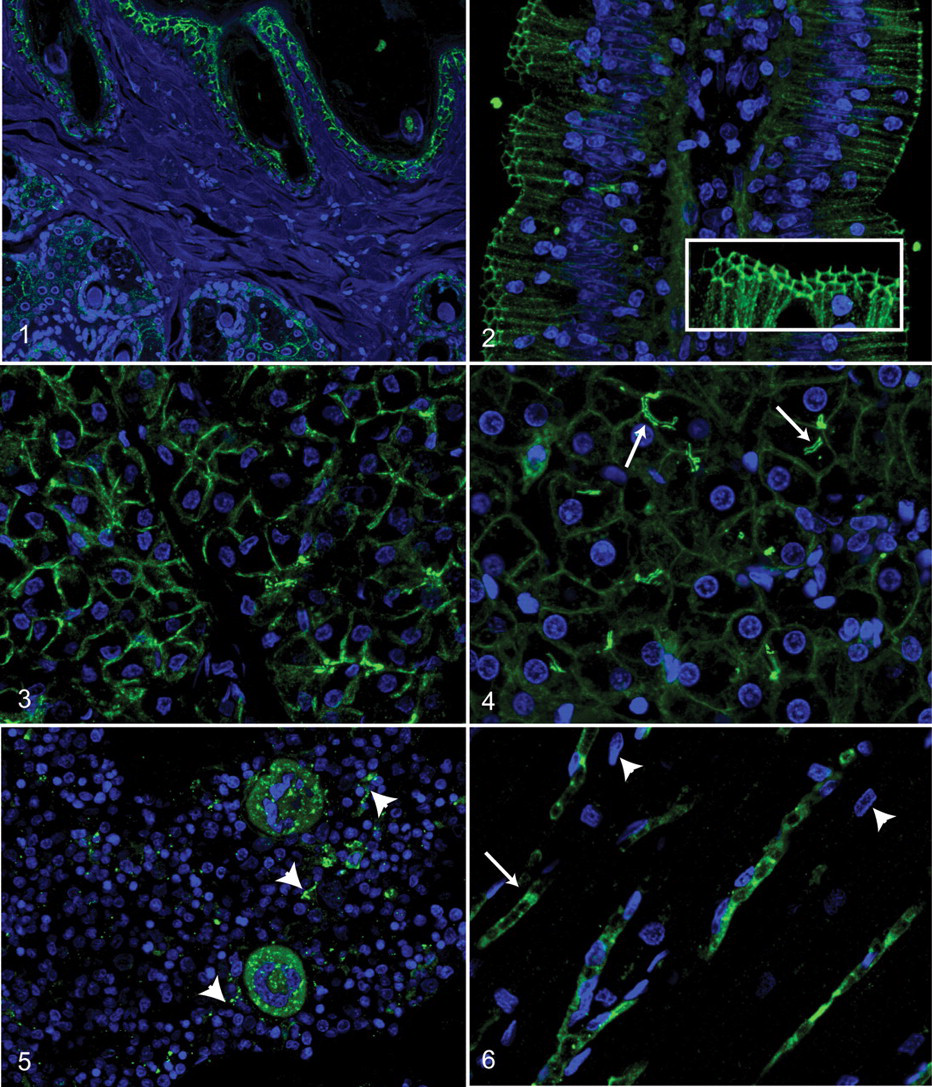
We examined the distribution of fJAM-A in feline tongue, skin, liver, lung, spleen, kidney, pancreas, jejunum, ileum, colon, heart, brain, and bone marrow by antibody-based immunofluorescence microscopy (Figs. 1–6). Tissues from the cats that were analyzed were grossly and histologically normal. Feline JAM-A was consistently found at cell–cell interfaces of epithelial cells. In both mucosal surfaces and epidermis, fJAM-A was present between cells in all epithelial strata, including anucleate superficial keratinocytes of the stratum corneum. In haired skin, fJAM-A was found in follicular and glandular epithelium (Fig. 1). Furthermore, fJAM-A was present along the entire border of the cell–cell junctions of intestinal epithelium (Fig. 2) with the highest expression in the apical region (Fig. 2, inset). In the pancreas, fJAM-A was present at cell–cell junctions of exocrine cells and ductules (Fig. 3). In the liver, in addition to interhepatocellular localization (Fig. 4) there were increased levels of fJAM-A on the cell membrane borders lining the bile canaliculi (Fig. 4, arrows). Within histologic sections of feline bone marrow, fJAM-A was observed diffusely and with focal granular accumulations in megakaryocytes (Fig. 5). Linear diffuse staining in bone marrow (Fig. 5, arrowheads) corresponded to cytoplasmic extensions of megakaryocytes. No other cells of the erythroid or myeloid lineage within bone marrow had detectable levels of fJAM-A by this method. In addition, fJAM-A localized to endothelial cells in all examined tissues, including capillary vascular beds of the myocardium (Fig. 6), gastrointestinal lamina propria (Fig. 2), lung, skin, pancreas, and brain, as well as larger arteries of the heart and intestinal submucosa. In endothelia, fJAM-A was concentrated at intercellular junctions (Fig. 6, arrows) but, in contrast to epithelial cells, was also diffusely distributed within the cytosol.
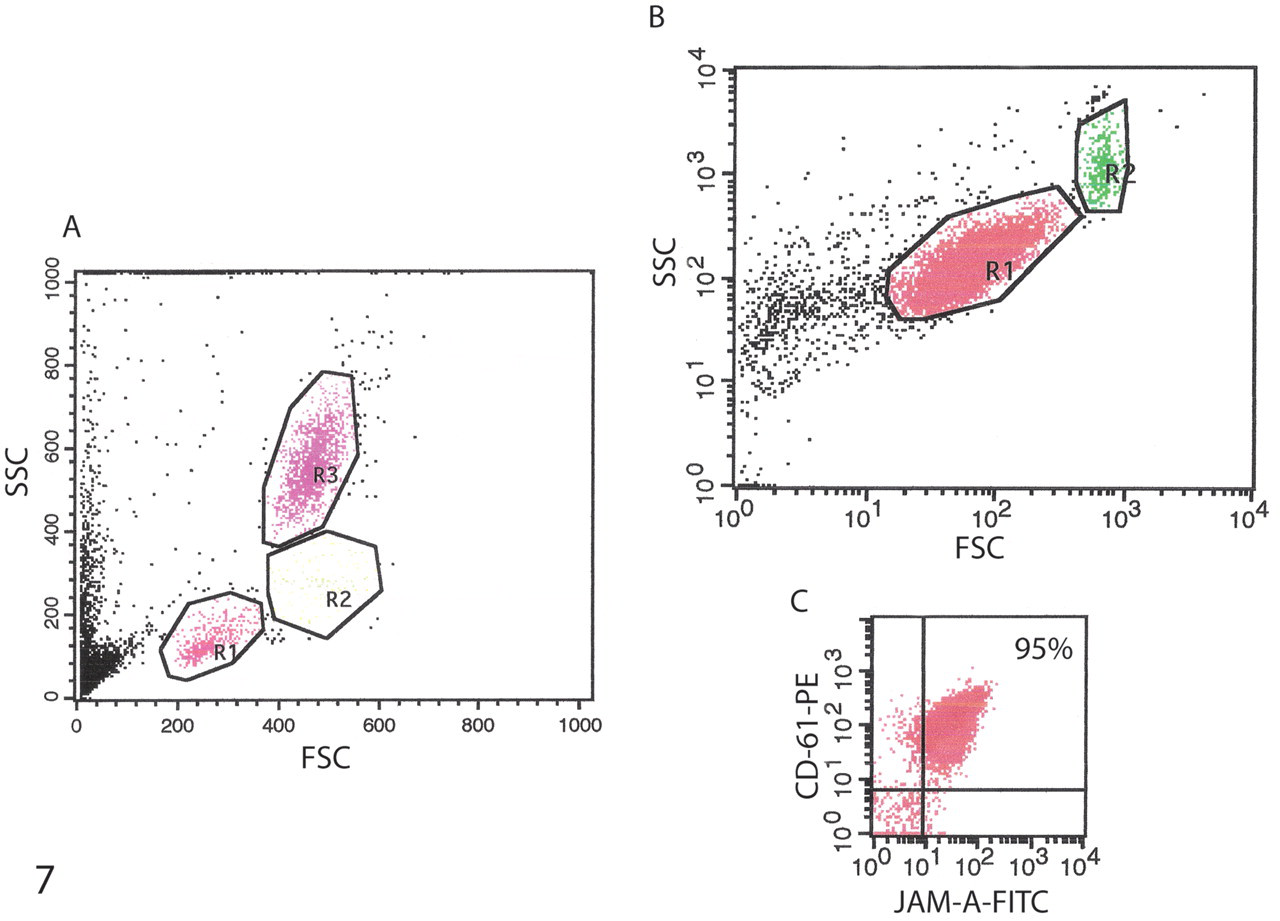
Flow cytometric dot plots of leukocyte subpopulations (A) and platelets (B, C). A, with linear scale acquisition, three leukocyte populations are seen in forward-scatter (FSC) and side-scatter (SSC) dot plots consisting of lymphocytes (R1), monocytes (R2), and neutrophils (R3). Platelets and cell debris compose the ungated events in the lower left. B, with log scale acquisition, the largest population in the FSC and SSC dot plots consists of single platelets (R1), with a smaller subpopulation of aggregated platelets (with higher FSC and SSC; R2). Ungated events in the lower left of the platelet dot plot represent cell debris. Only platelets in the R1 gate were analyzed for fluorescent intensity. C, dot plots of fluorescent intensity of fluorescein isothiocyanate (FITC) fJAM-A and phycoerythrin (PE) CD61 in cells in the R1 region of the log scale dot plot (B). Platelets (95% of cells in the R1 gate) are double positive for fJAM-A and CD61 (upper right).
Distribution of JAM-A on Feline Peripheral Blood Cells by Flow Cytometry
Analysis of leukocytes and platelets in peripheral whole blood by flow cytometry showed that the majority of platelets highly expressed fJAM-A (95% of the CD61+ cells were also fJAM-A+; Fig. 7 ). In contrast, fewer neutrophils (9%), monocytes (27%), or lymphocytes (17%) were positive for fJAM-A.
Effect of FCV infection on JAM-A Expression
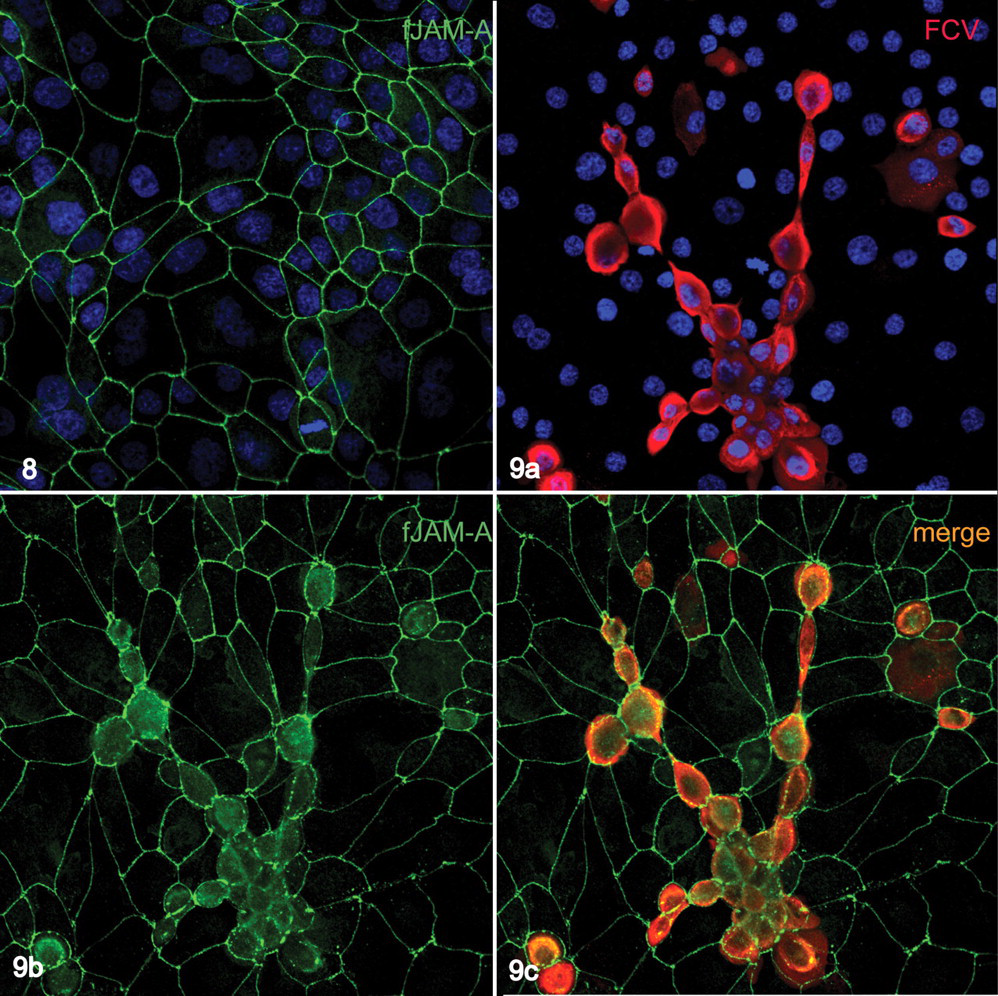
FMECs are a feline primary epithelial cell line originally derived from the mammary glandular epithelium. 20 In uninfected FMEC cells, fJAM was restricted to the cell–cell interface (Fig. 8 ), where it colocalized with the integral tight junction proteins occludin and ZO-1. 20 FMECs were susceptible to FCV infection, and cell monolayers infected with the FCV isolate AN123 at a multiplicity of infection of 10 reproducibly cause plaque formation 15 to 18 hours postinfection. In infected cells visible at earlier time points (6 hours and on), fJAM-A was redistributed throughout the cytosol (Fig. 9 ), although still present at the cell–cell interfaces.
Discussion
FCV belongs to a diverse group of microbes that bind to immunoglobulin superfamily proteins found at intercellular junctions in epithelia and endothelia. 6 The JAM-A molecule is a functional receptor for both FCV and mammalian reoviruses. 1,11 Understanding the distribution and normal function of cell surface molecules that serve as receptors for pathogens provides insight into pathogenesis. In human and murine tissues, JAM-A localizes to the tight junctions between epithelial and vascular endothelial cells and is present on the surface of neutrophils, lymphocytes, monocytes, dendritic cells, platelets, and spermatozoa. 5,28 JAM-A is also a marker for long-term repopulating hematopoietic stem cells in mice. 23 Functionally, JAM-A has been proposed to be involved in intercellular junction assembly and repair, 9,10 leukocyte transmigration, 4,12,18 and angiogenesis, 15 all of which require homophilic or heterophilic interactions. 7,21 In addition, antibody binding of surface-expressed JAM-A on platelets causes platelet aggregation and activation. Last, an expanding number of publications investigate whether expression of JAM-A regulates cancer cell invasion or identifies metastatic potential. 13,16
Binding of virus or virus-specific proteins to adhesion molecules in epithelial or endothelial junctional complexes could disrupt the architecture of these junctions or other functions of JAM-A. Indeed, there is experimental evidence that the fiber protein of adenoviruses, which is secreted in large excess from infected cells, disrupts the epithelial barrier of the respiratory tract by interacting with its junctional receptor Coxsackie–adenovirus receptor. 25 Because fJAM-A is a receptor for FCV, it seems possible that the clinical sequelae of FCV infection might in part be a consequence of disruption of junctional complexes in epithelium or endothelium. Our findings support this hypothesis in that fJAM-A is redistributed from intercellular junctions to the cytoplasm in FCV-infected epithelial cells before their release from the cell monolayer. The significance of the redistribution of fJAM-A in FCV infection will be further investigated. We consider a number of possibilities. Redistribution is potentially important in interactions of inflammatory cells with infected epithelium. Similar redistribution has been described in endothelial cells under inflammatory conditions and is postulated to promote leukocyte movement through the vessel. 27 Redistribution of fJAM-A could also alter FCV paracellular or transcellular viral trafficking. In studies with pathogenic strains of Escherichia coli, for example, infection of epithelial cell monolayers caused redistribution of the tight junction proteins occludin and ZO-1 from an intercellular to a cytoplasmic location and preceded bacterial internalization. 8 A recent study demonstrated that occludin, when redistributed to the cytosol from its tight junctional location, directly activates the extrinsic apoptotic pathway. 2 FCV induces apoptosis, 22 and ongoing experiments include investigating the apoptotic state of infected FMEC cells relative to the distribution of fJAM-A and other proteins of the junctional complex.
We have shown that fJAM-A is broadly expressed at intercellular junctions of epithelial and endothelial cells and is found on the surface of platelets. Although we did not detect fJAM-A in progenitor leukocytes (by immunofluorescence microscopy), we did detect fJAM-A on the surface of circulating leukocytes (by flow cytometry). The distribution of JAM-A in feline tissues parallels its distribution in mouse and human tissues with a few notable exceptions. In murine intestinal epithelial cells, JAM-A is restricted to an apical position that corresponds to the tight junction. 12 In contrast, we found a broader distribution of JAM-A throughout the cell–cell interface of epithelial cells in the intestine and other tissues, with apical concentration in highly polarized cells such as gastrointestinal and pancreatic acinar cells. In uninfected cultured feline epithelial cells, like tissue epithelia, fJAM-A was present along the entire cell–cell interface. In addition to being present at endothelial cell junctions, fJAM-A was found diffusely within the cytosol of endothelial cells, regardless of vessel size.
In cats with severe systemic FCV infection, clinical and pathologic sequelae include edema, ulceration of haired skin and mucosal surfaces, pancreatic and hepatocellular necrosis, and, rarely, hemorrhage. 19 It is reasonable to speculate that this spectrum of lesions reflects disruption of intercellular junctions that rely on fJAM-A function and tight junction integrity. However, given the broad distribution of fJAM-A, interaction with FCV alone fails to explain why the more typical milder infections are restricted to the oral/lingual epithelium. It is likely that additional viral, immunological, and cellular factors determine the outcome of infection with particular FCV isolates. For example, FCV isolates vary in their capacity to infect Chinese hamster ovary cells expressing fJAM-A, 17 which suggests that Chinese hamster ovary cell binding and productive infection by some isolates requires cellular factors other than fJAM-A.
This paper establishes the distribution, in normal cats, of the fJAM-A receptor for FCV. Moreover, in an in vitro epithelial cell culture system, the distribution of fJAM-A is altered during infection. This is a step in establishing whether a primary interaction between FCV and host tissue fJAM-A has an impact on viral pathogenesis.
Footnotes
Acknowledgements
To Drs Niels Pedersen and Kate Hurley for their consistent support and sharing of resources.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
