Abstract
The presence of a heart-base tumor was diagnosed by ultrasound imaging in a 10-year-old, female, domestic shorthaired cat presenting with dyspnea and pleural effusion because of the presence of a modified transudate. Hematology and clinical chemistry were unremarkable. The owner elected euthanasia. At necropsy, a locally extensive, firm, multilobulated nodule surrounded the pulmonary vein. The tumor was composed of lobules of large polygonal cells separated by a fine fibrovascular stroma. Tumor cells infiltrated the myocardium, and neoplastic emboli were present, but no metastases were macroscopically detectable. Tumor cells were immunohistochemically positive for chromogranin A, for synaptophysin and, faintly, for neuron-specific enolase and negative for vimentin, cytokeratin, a smooth muscle actin, glial fibrillary acidic protein, thyreoglobulin, and calcitonin. Based on histologic and immunohistochemical findings, the diagnosis of chemodectoma was made.
Chemodectoma, or extra-adrenal nonchromaffin paraganglioma, is a neoplasm arising from chemoreceptor cells involved in the response to changes in blood pH, oxygen tension, and carbon dioxide content. 4,13,20 Chronic hypoxia is probably involved in the pathogenesis of chemodectoma: a high prevalence of this tumor has been recorded both in humans and in cattle living in mountain areas 1,21 and in brachicephalic dogs, which have an upper respiratory tract anatomic conformation that favors a chronic hypoxic status. 13 Chemodectomas most commonly arise within the aortic or the carotid body 4,13,20 and, less frequently, in the glomus pulmonale, 20 in the glandula suprarenalis, 9 or in ectopic sites. 2,7,8,15,20 Chemodectomas are particularly rare in cats: to date, two carotid body tumors, 5,24 one cauda equina paraganglioma, 8 and eight aortic body tumors 3,6,11,12,19,22–24 have been reported in cats. Aortic body tumors are locally extensive, multilobulated masses, located within the pericardium at the heart base. 4 They are usually encapsulated and organized in small lobules surrounded by a prominent fibrovascular stroma. 4 Neoplastic cells commonly stain for neuron-specific enolase (NSE), chromogranin A (CgA), and synaptophysin (SY). 7,8,15,18
Immunohistochemical characteristics of feline aortic body tumors, however, have not been described. We report the clinicopathologic and immunohistochemical features of a feline aortic body tumor.
A 10-year-old, female, domestic shorthaired cat was presented to the referring veterinarian due to severe inspiratory dyspnea and cyanosis. Clinical and radiographic examination revealed a severe thoracic effusion. Serology for feline immunodeficiency virus, feline leukemia virus, and feline coronavirus was negative. Hematology and clinical chemistry were unremarkable. Cytology of the pleural fluid was characterized by neutrophils, mostly nondegenerate, macrophages, and reactive mesothelial cells (modified transudate). After thoracocenthesis (150 ml of yellowish effusion was removed), radiography and ultrasound imaging of the chest revealed the presence of a 25-mm nodule, located at the heart base and surrounding the large vessels. Because of the poor prognosis, the cat was euthanatized. A full necropsy was performed by the referring veterinarian. Samples of tumor, large vessels, and myocardium were collected, fixed in 10% buffered formalin, routinely processed, and embedded in paraffin. Five-micrometer sections were stained with hematoxylin and eosin. Immunohistochemistry was performed with the avidin–biotin–peroxidase complex technique, 14 using the primary antibodies listed in Table 1.
List of the antibodies used and results of the immunohistochemical staining.∗
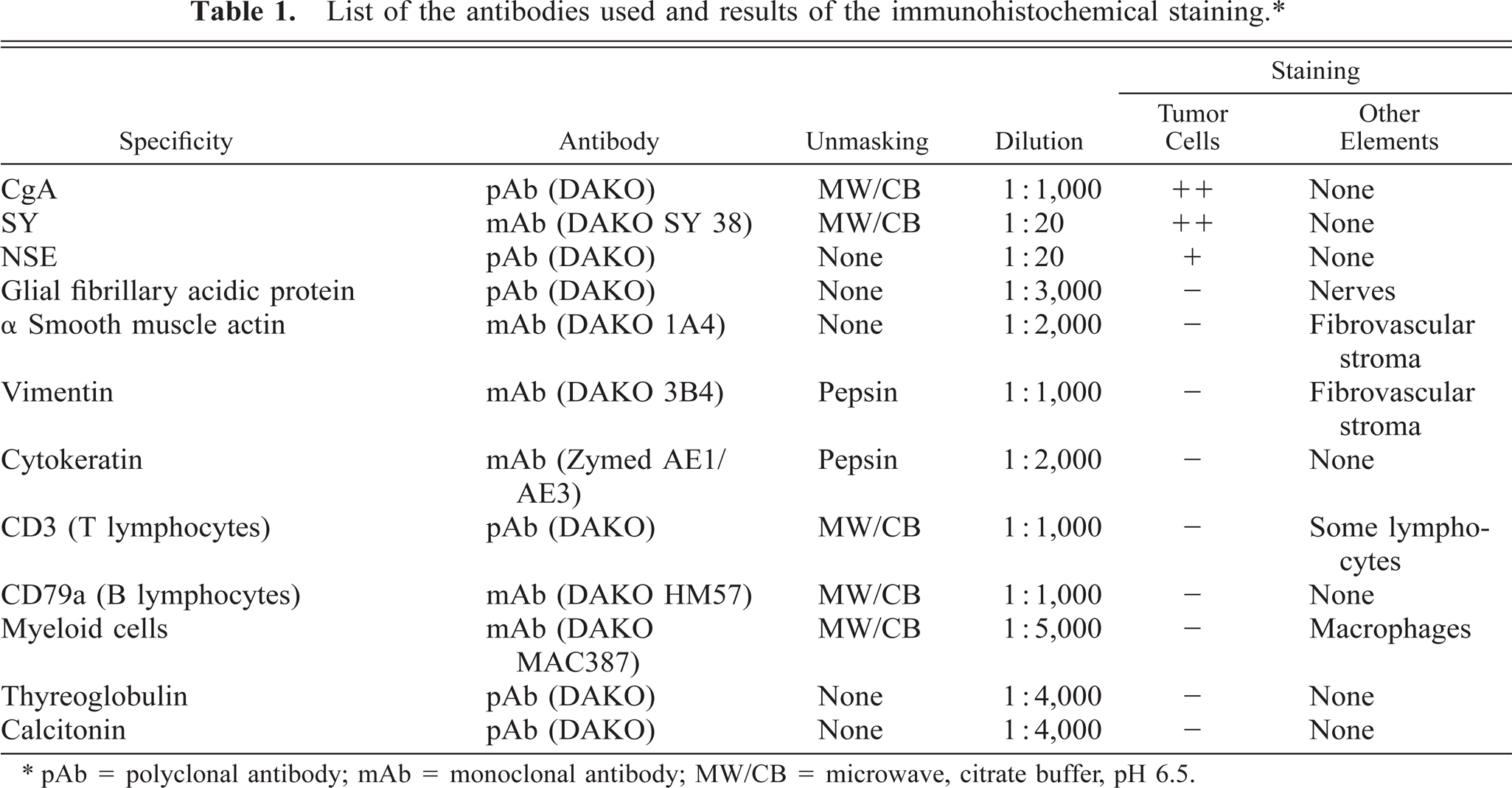
∗ pAb = polyclonal antibody; mAb = monoclonal antibody; MW/CB = microwave, citrate buffer, pH 6.5.
At necropsy, a locally extensive, multilobulated mass, approximately 25 × 35 × 30 mm, was found at the heart base (Fig. 1). The mass was firm, tan to red, and densely adherent to the ventral portion of the aorta, which was dorsally displaced. The pulmonary vein was embedded in the tumor and showed severe stenosis. The pulmonary artery was partially surrounded by the tumor. The vena cava had no connection with the mass. The right atrium and ventriculum were severely dilated. On cut surface, the neoplasm had multiple necrotic foci. No other lesions were detected, except for mild hepatic lipidosis. Histologically, the tumor was only partially encapsulated and focally infiltrated the myocardium of the left atrium. Neoplastic emboli were present within lymphatic vessels. Tumor cells were organized in lobules defined by a thin fibrovascular stroma with numerous small nerves (Fig. 2). Tumor cells were round to polygonal with indistinct cell borders; scant, slightly eosinophilic cytoplasm; and a central, round nucleus, with fine chromatin and one to two prominent nucleoli (Fig. 3). Rare mitoses (zero to one per high-power field), rare multinucleated giant cells, macrophages, and peripheral lymphoid aggregates were visible. Tumor cells stained intensely SY positive and CgA positive and faintly NSE positive (fine granular cytoplasmic pattern). Glial fibrillary acidic protein–positive nerves, vimentin-positive and α smooth muscle actin–positive stromal cells, CD3-positive lymphocytes, and MAC387-positive myeloid cells were also detectable (Table 1). Histological and immunohistochemical findings were consistent with a diagnosis of aortic body tumor. 4,7,10,12,22
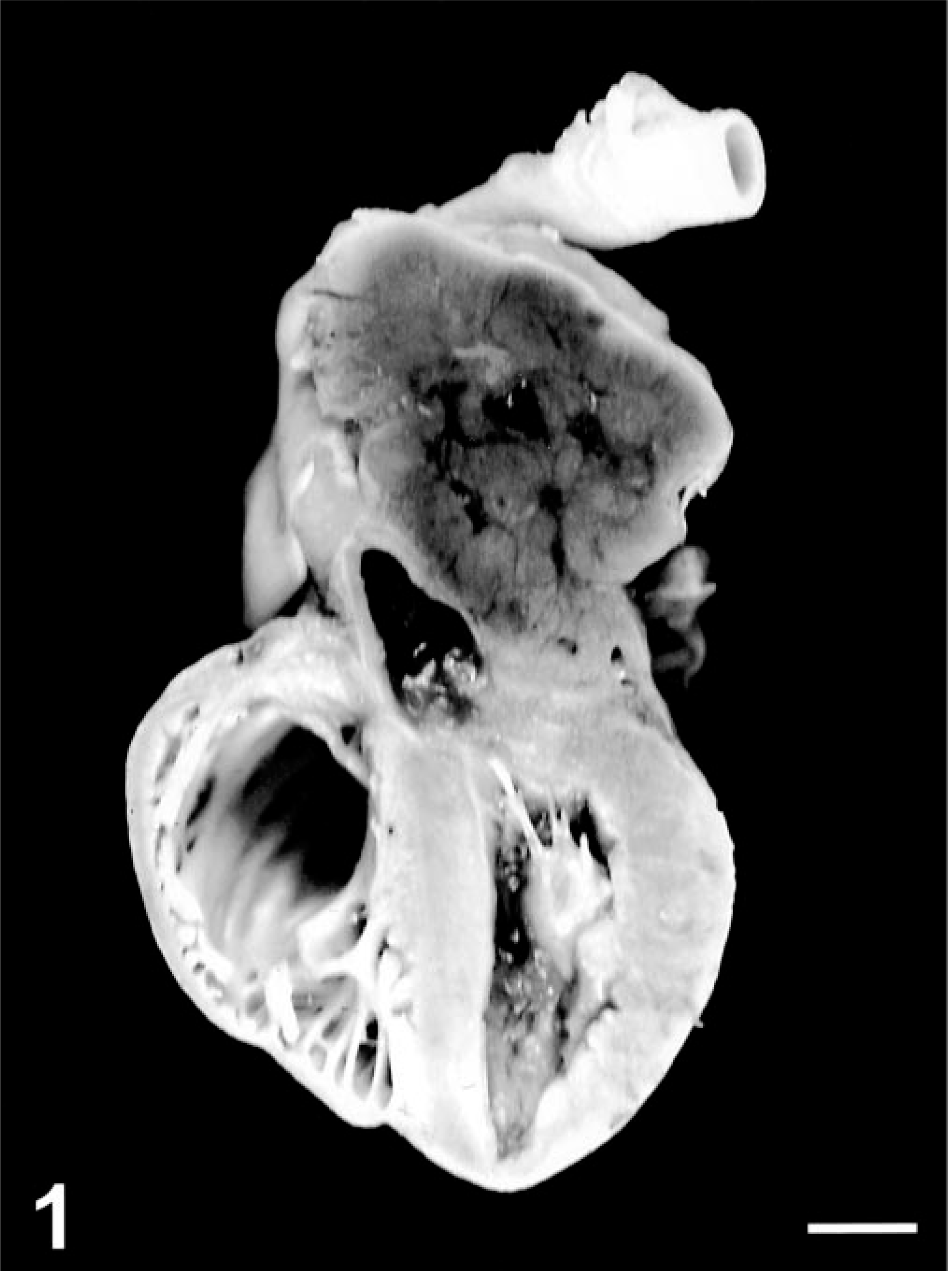
Heart, cut surface; cat. Large, partially encapsulated mass with multiple necrotic foci, which surrounds and constricts the pulmonary vein and pushes the aorta dorsally. Right atrium and ventriculum are severely dilated. Bar = 12.5 mm.
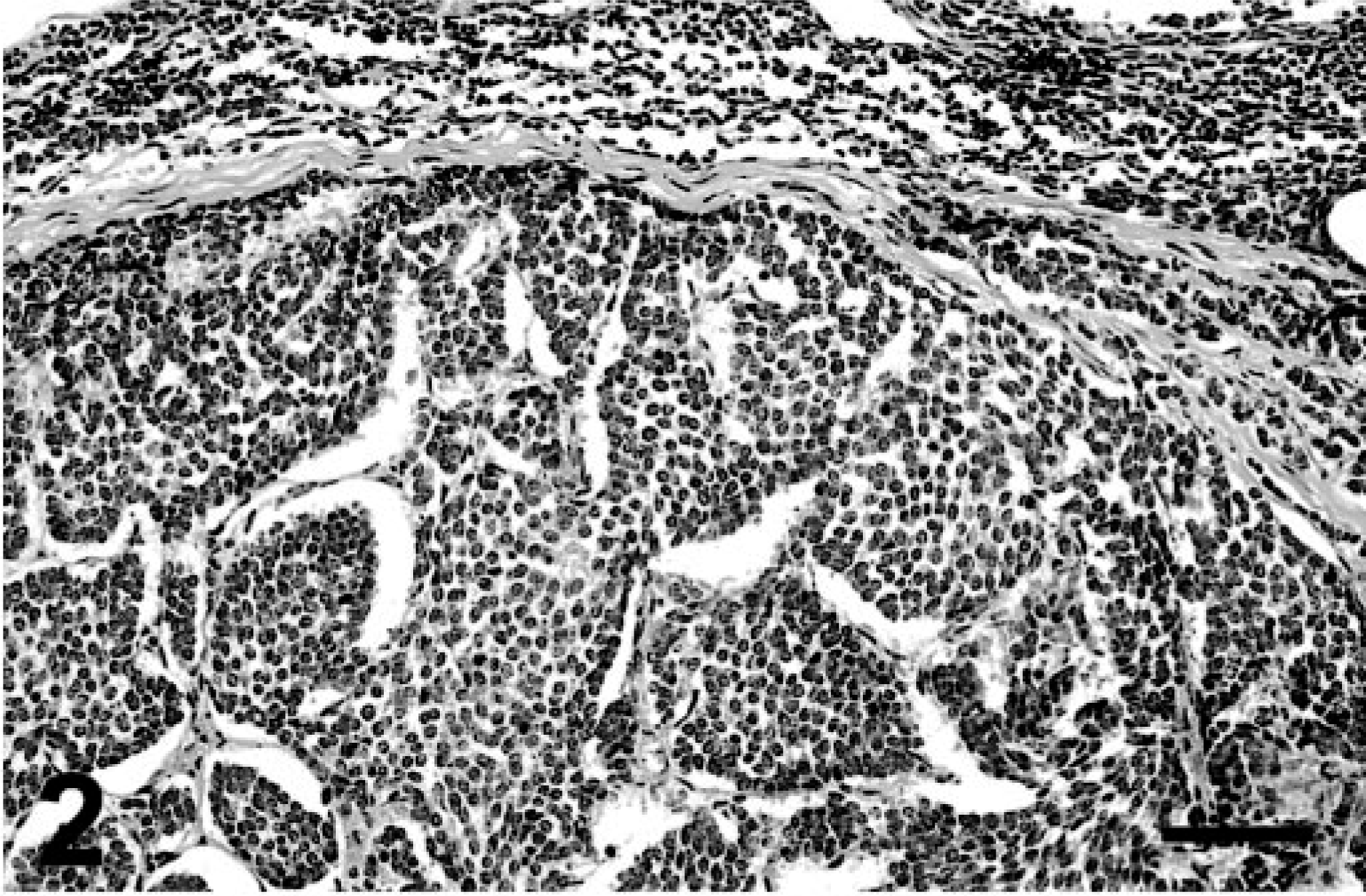
Heart, chemodectoma; cat. Neoplastic cells are arranged in closely packed lobules separated by thin fibrovascular stroma. HE. Bar = 45 µm.
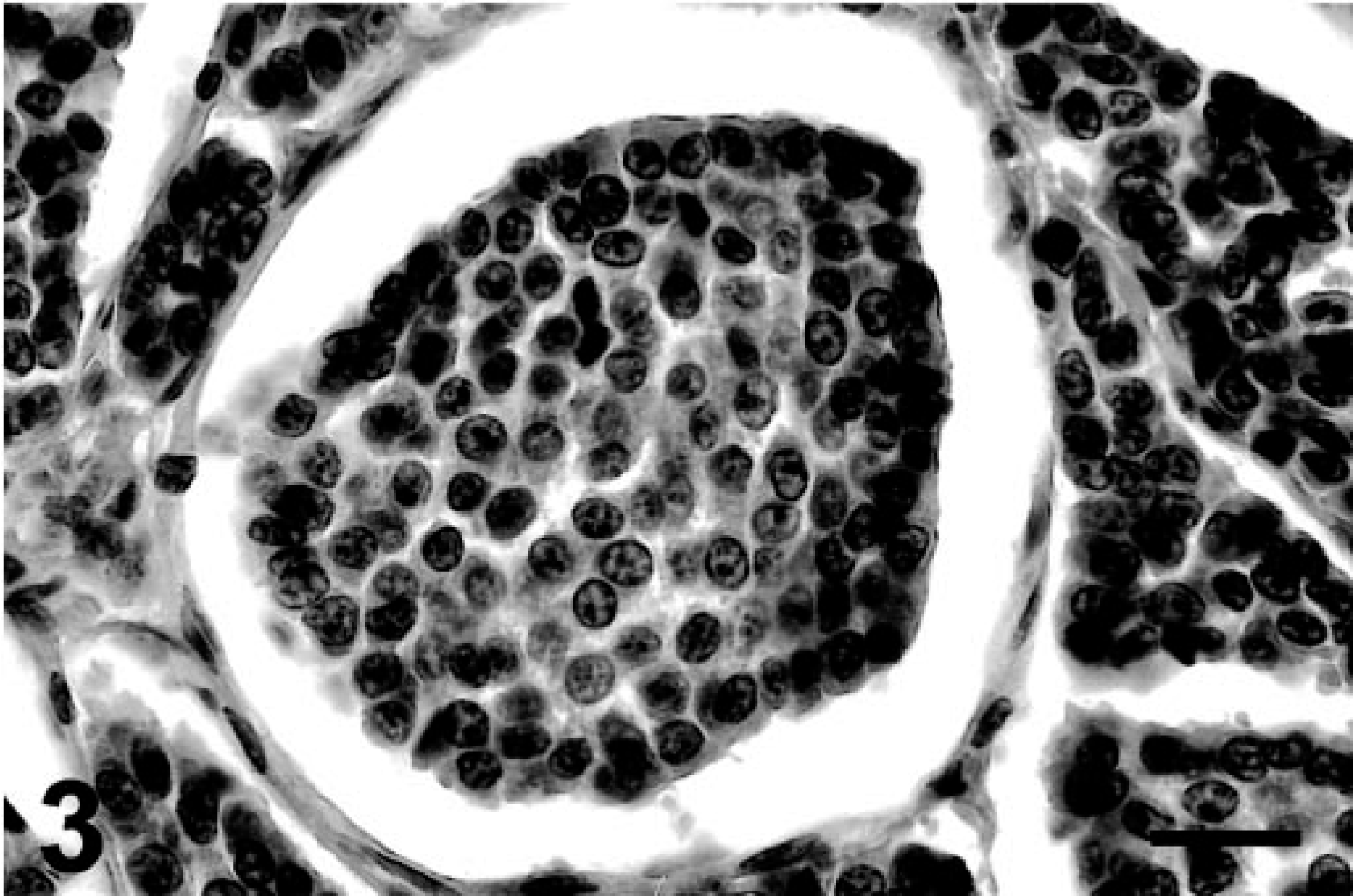
Heart, chemodectoma; cat. High magnification of a neoplastic lobule: neoplastic cells are round to polygonal with indistinct cell borders, scant cytoplasm, and a central, round nucleus with fine chromatin and one or two prominent nucleoli. HE. Bar = 220 µm.
This case shared common features with previously reported feline aortic body tumors. 3,6,11,12,19,22–24 Specifically, there were no lesions associated with a chronic hypoxic status, no other tumors or metastases were found, and no clinical signs due to abnormal chemoreceptor function was observed. However, the severe stenosis of the pulmonary vein was an unusual finding. The in vivo diagnosis of heart-base tumor should be based on excisional biopsies or on fine-needle aspirates. 6,19,22 These approaches, however, have practical limitations because of the need for general anesthesia and because of the fragility of exfoliated cells, which may require sequential sampling for identification. 6,19 Our report confirms the value of immunohistochemistry in confirming the diagnosis of feline aortic body tumor. Specifically, neoplastic cells expressed both SY and CgA. SY is considered the most important marker for neuroendocrine tumors: SY-positive cells have been found in equine, bovine, and feline extraadrenal paragangliomas, 8,15,18 but their presence has not previously been described in feline aortic body tumors. The presence of CgA-positive granules is a typical feature of neuroendocrine tumors. 10 Nevertheless, it has been described only in bovine and canine chemodectomas. 1,16 We found only weak positivity for NSE, which is strongly positive in tumor cells in chemodectomas of different species, including the feline. 7,8,15,18 This was probably due to the fact that chemoreceptor cells rapidly undergo autolysis and lose some of their NSE immunoreactivity. 4 Somatostatin, insulin, glucagon, gastrin-colecistokynin have also been immunohistochemically detected in canine paragangliomas, 9,16 but it is our opinion that their identification is not necessary if tumor cells express NSE, SY, and CgA. The other antibodies used in this study are useful in differentiating aortic body tumors from other lesions characterized by a similar histological pattern, such as ectopic thyroid tumors, 4,19,22 which would also have stained for cytokeratin, thyreoglobulin, and calcitonin.
Footnotes
Acknowledgements
We are grateful to Drs Paola Roccabianca, Luca Pirovini, and Raffaella Capobianco, and to Mr Maurizio Barzani.
