Abstract
To study the ectopic chondrogenesis in canine mammary mixed tumors, the expression of bone morphogenetic protein-6 (BMP-6) and specific BMP receptors (BMPRs), BMPR-IA, BMPR-IB, and BMPR-II, was examined using immunohistochemical and immunoblot analysis in 39 canine mammary gland tumors. Immunohistochemically, BMP-6 and all three types of BMPRs were coexpressed in the myoepithelial cells and chondrocytes in six of eight benign mixed tumors. In complex adenomas, myoepithelial cells showed an expression pattern of BMP-6, BMPR-IA, and BMPR-II similar to those in benign mixed tumors, whereas immunoreactivity for BMPR-IB was very mild. The myoepithelial cells proliferating within the basement membrane showed more intense immunoreactivity for BMP-6 and all BMPRs as compared with those proliferating in the interstitial areas. Western blotting analysis revealed immunopositive bands at 40–45 kDa for BMP-6 in the samples from simple and complex adenomas and benign mixed tumors. The BMPR-IB-specific bands at 45 kDa were most detected in benign mixed tumors. Because among BMPRs, BMPR-IB is thought to be the major receptor for BMP-6 for primary chondrogenesis, these findings suggest that the expression of BMP and its receptors on the myoepithelial cells might play a role in the ectopic cartilage formation in canine mammary gland tumors, especially in benign mixed tumors.
Mammary gland tumors are the most common tumors in female dogs. 22 These tumors have various histologic features that are characterized by complex proliferation of abundant myoepithelial cells, mesenchymal components such as cartilage, bone, and bone marrow, or both. The tumors accompanied by the proliferation of myoepithelial cells are classified as complex tumors, and the tumors containing mesenchymal tissues, such as cartilage and bone, together with the proliferation of myoepithelial cells are called mixed tumors. The morphologic features of these canine mammary tumors are in conformity with those of human salivary and mammary pleomorphic adenomas at the point of ectopic mesenchymal tissue formation. 7
Recent studies of human salivary gland pleomorphic adenomas indicate that bone morphogenetic proteins (BMPs) participate in their morphologic pleomorphism. 10,17,18 BMPs are multifunctional proteins originally isolated from bone and are identified by their ability to induce ectopic cartilage and bone formation. 1,16 BMPs regulate various aspects of organogenesis during development 13,23 and osteogenesis. 24 These proteins also have been detected in various mesenchymal and epithelial tumors, and they participate in ectopic ossification or epithelial-mesenchymal interaction. 9,10,16–18,29,34,35 It has been found that type IV collagen is important for the actions of BMPs because some types of BMPs, such as BMP-2 and BMP-7, can intimately bind to type IV collagen. 8,25 BMP-2 coexisting with type IV collagen induces psammoma bodies in human ovarian cancer. 15
BMP-6 was originally isolated from murine embryonic complementary deoxyribonucleic acid library. 20 BMP-6 messenger ribonucleic acid (mRNA) and its protein were expressed in the suprabasal layer of skin, hypertrophic chondrocytes at endochondral ossification sites, and the central nervous system in vivo. 13,20,32 BMP-6 induced differentiations of osteoblastic cell lines and keratinocytes in vitro 5,6,11 and was detected in human neoplastic epithelial cells including breast, salivary, rectal, and thyroid carcinomas. 2,9 It has been suggested that BMP-6 inhibits cell division, promotes cell differentiation, induces ectopic bone formation, or regulates epithelial-mesenchymal interaction (or all). 2,5,17,32
BMPs transduce their effects through binding to two different types of serine-threonine kinase receptors, similar to other members of the transforming growth factor (TGF)-β superfamily. Both type I and II receptors are required for signaling. BMPs including BMP-6 can bind to three type I receptors, BMP type IA receptor (BMPR-IA), BMPR-IB, and Activin type I receptor (ActR-I), and three type II receptors, BMPR-II, ActR-II, and ActR-IIB. 33 BMPR-IA and BMPR-IB are structurally similar to each other but show distinct spatial and temporal expression patterns during bone formation and organogenesis. 4,12,37 BMPR-IA is strongly expressed in prehypertrophic chondrocytes, osteoblasts, and periosteum in skeletal structures, although the expression of BMPR-IB is not detectable or detected at a low level in these sites. On the other hand, BMPR-IB is expressed in precartilaginous mesenchymal condensations and perichondrium at higher levels than BMPR-IA. The expression of BMPR-IB is predominantly detected in epithelial cells in various organs of the embryo, although both mesenchymal and epithelial cells express BMPR-IA in the same sites. It is considered that BMPR-II, like other type II receptors for the TGF-β superfamily, acts as the primary ligand-binding receptor and produces phosphorylation of type I receptors, which is essential for downstream signal transduction. 19,27 BMPR-II has been detected in osteogenic cells and lung epithelial cell lines in vitro and in the developing central nervous systems, adult rat brains, and ossification sites in vivo. 27,28,33 BMP type I and II receptors also are detected in some tumors including human osteosarcomas and prostate carcinomas. 14,21
A recent study of canine mammary gland tumors indicates that BMP-6 is expressed in mammary gland tumor cells, especially myoepithelial cells and ectopic cartilage of benign mixed tumors. 30 This study is performed to evaluate whether BMP-6 functions as an essential factor for ectopic cartilage and bone formation using immunohistochemical methods for staining BMP-6 and its specific receptors, including BMPR-IA, -IB, and -II.
Materials and Methods
Tissue preparation
Thirty-nine mammary tumors from 32 dogs were studied. Fresh tissue samples were stored at −80 C. For diagnosis and immunohistochemical study, specimens were fixed in methanol-Carnoy's solution. Paraffin sections, 4 µm thick, were made and stained with hematoxylin and eosin for histopathologic diagnoses based on the World Health Organization classification. 22 Briefly, 39 mammary tumors were classified into simple adenomas (n = 5), complex adenomas (n = 11), benign mixed tumors (n = 8), simple adenocarcinomas (n = 10), complex adenomas (n = 3), and carcinoma or sarcoma in mixed tumor (n = 2).
Immunohistochemistry
Goat antisera against human BMP-6 (1 : 20, Santa Cruz, Santa Cruz, CA), BMPR-IA (1 : 10, Santa Cruz), BMPR-IB (1 : 20, Santa Cruz), and BMPR-II (1 : 10, Santa Cruz) were used as primary antibodies. Immunostaining was performed using a system kit of avidin–biotin–peroxidase complex (ABC, PK4000, Vector Laboratories, Burlingame, CA). The sections were treated with methanol containing 0.5% hydrogen peroxide for 10 minutes at room temperature to block endogenous peroxidase. Slides were incubated with phosphate-buffered saline (PBS) (pH 7.4) containing 3% bovine serum albumin for 60 minutes at 37 C to avoid nonspecific binding. Then, the sections were incubated with appropriate concentrations of primary antibodies for 45 minutes at 37 C, followed by incubation with biotinylated rabbit serum against goat immunoglobulin G (Dako-Japan, Kyoto, Japan) and ABC reagent for 30 minutes at 37 C. The specimens were washed with PBS three times for 10 minutes after each step. For visualization, 3,3′-diaminobenzidine tetrachloride (DAB, Sigma, St. Louis, MO) was used. The slides were counterstained with hematoxylin. According to previous reports, 3,31 mammary cells were classified into four cell types: 1) glandular epithelial cells, 2) resting and proliferating myoepithelial cells within the basement membrane, 3) spindle- and star-shaped myoepithelial cells proliferating in the interstitial areas, and 4) chondrocytes of ectopic cartilage. The immunohistochemical results were evaluated for each of the four cell types using semiquantitative analysis as follows: −, 0% positive cells; +, less than 10% positive cells; ++, 10–50% positive cells; and +++, more than 50% positive cells.
Western blotting
Frozen samples were washed and divided into two pieces. These were homogenized with sample buffer for BMP (0.5 M Tris-HCl, pH 6.8, 10% glycerol, and 2% sodium dodecyl sulfate [SDS]) or with sample buffer for BMPRs (0.01 M Tris-HCl, pH 8.0, 1% Triton X-100, 0.1% SDS, 1% sodium deoxycholate, 1 mM iodoacetamide, 0.2 mg/ml aprotinin, 1 mM phenylmethylsulfonyl fluoride, 0.14 M NaCl, and 0.025% NaN3) at 4 C, as in previous reports. 35,36 After centrifugation at 1,000 rpm for 5 minutes, insoluble debris was removed, and another sample buffer (0.13 M Tris-HCl, pH 6.8, 4% SDS, 10% 2-mercaptoethanol, and 14% glycerol) was added. Samples were boiled for 5 minutes, loaded on 10–20% SDS-polyacrylamide gels (ATTO, Tokyo, Japan), and transferred to polyvinylidene difluoride membranes (clearblot P, ATTO). After the transfer, membranes were incubated with 3% bovine serum albumin for 60 minutes at 37 C. Then, these were incubated with appropriate antibodies for 45 minutes at 37 C, followed by incubation with secondary antibody and ABC reagent for 30 minutes at 37 C. The membranes were washed with PBS-0.1% Tween three times for 10 minutes after each step. After washing with PBS for 10 minutes, immunoreactive bands were visualized using DAB.
Results
Immunohistochemistry
The immunoreactivity for BMP and its receptors varied in foci within tumors examined. The results of typical foci in each mammary tumor and intact region are summarized in Tables 1–3.
Immunoreactivity for BMP-6 and BMPRs in 24 benign mammary gland tumors.∗
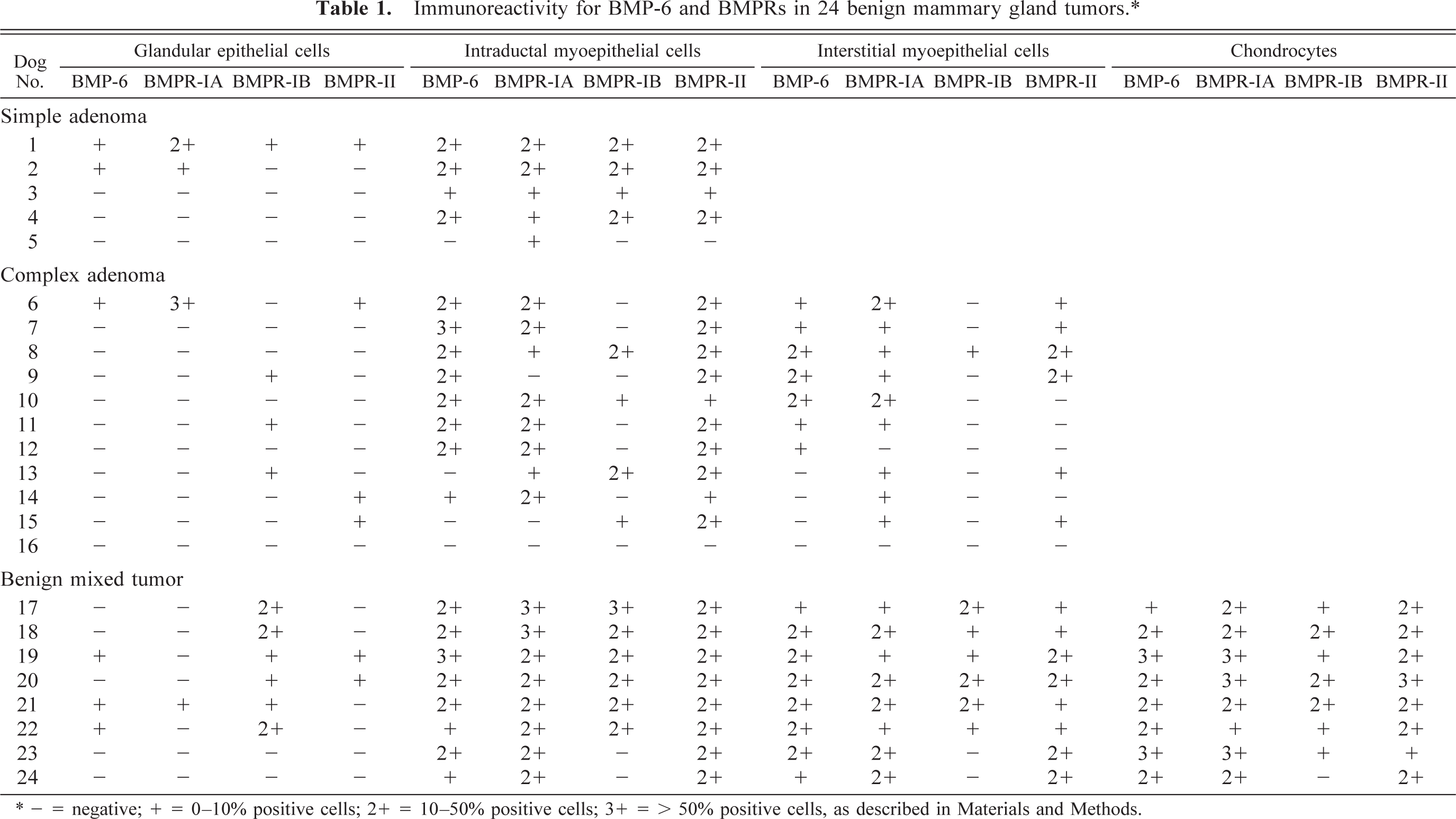
∗ - = negative; + = 0–10% positive cells; 2+ = 10–50% positive cells; 3+ = > 50% positive cells, as described in Materials and Methods.
Immunoreactivity for BMP-6 and BMPRs in normal mammary tissue adjacent to 18 benign mammary tumors.∗

∗ - = not detected; +a = detected in myoepithelial cells; +b = detected in both glandular and myoepithelial cells.
Simple adenomas (n = 5). The tumors were composed of well-differentiated glandular epithelial cells with resting myoepithelial cells. Proliferation of myoepithelial cells in either the ducts or interstitial areas was not evident. Immunoreactivity for BMP-6 and all three BMPRs was found in the glandular epithelial cells of one dog and in the resting myoepithelial cells of four dogs (Table 1). The immunoreactivity for BMPRs was found on the cell membrane. Although the number and distribution pattern of positive cells for BMP-6 and BMPRs varied in each of the five dogs, resting myoepithelial cells were most intensely positive for BMP-6 and BMPRs. The immunoreactivity for BMPR-IA and BMPR-II tended to coexist in these myoepithelial cells. BMPR-IB immunoreactivity was not always found in BMPR-IA–and BMPR-II–positive areas, but frequently localized in the BMP-6–positive glands.
Complex adenomas (n = 11). The tumors consisted of mixed proliferation of glandular and myoepithelial cells. The foci of myoepithelial proliferation sometimes extended to the interstitial area. The myoepithelial cells proliferating within the glands or tubules could be distinguished as resting or proliferating myoepithelial cells. The morphology of resting myoepithelial cells was very similar to that of normal cells. The proliferative myoepithelial cells formed distinct foci within the glands or tubules. The myoepithelial cells proliferating in the interstitial areas could be divided into spindle- and star-shaped cells. Spindle-shaped myoepithelial cells proliferated in the interstitial areas forming solitary foci. These cells had distinct eosinophilic to clear cytoplasm. On the other hand, the interstitial foci consisting of proliferating star-shaped cells had abundant mucinous stroma, occasionally accompanied by hyaline or chondroid changes. Immunoreactivity for BMP-6 was most intense in proliferating myoepithelial cells within the tubules (Fig. 1A). Star-shaped myoepithelial cells with hyaline or chondroid stroma also showed intense immunoreactivity for BMP-6. Spindle- and star-shaped myoepithelial cells without these chondroid changes exhibited very mild immunoreactivity for BMP-6, despite moderate BMP-6 reactivity of myoepithelial cells within the adjacent tubules. Regarding BMP type I receptors, myoepithelial cells within the tubules showed intense immunoreactivity for BMPR-IA (eight dogs) and BMPR-IB (three dogs, Table 1). Figure 1B–D shows a focus containing BMPR-IA–and BMPR-II–positive (Fig. 1B, D) and BMPR-IB–negative (Fig. 1C) myoepithelial cells within the tubules of dog No. 8. Interstitial myoepithelial cells tended to exhibit very mild immunoreactivity for all three BMPRs, as for BMP-6, although myoepithelial cells in the adjacent tubules showed moderate reactivity for all BMPRs. Proliferating myoepithelial cells in the interstitial areas showed no immunoreactivity for BMPR-IB except for one dog (Table 1).
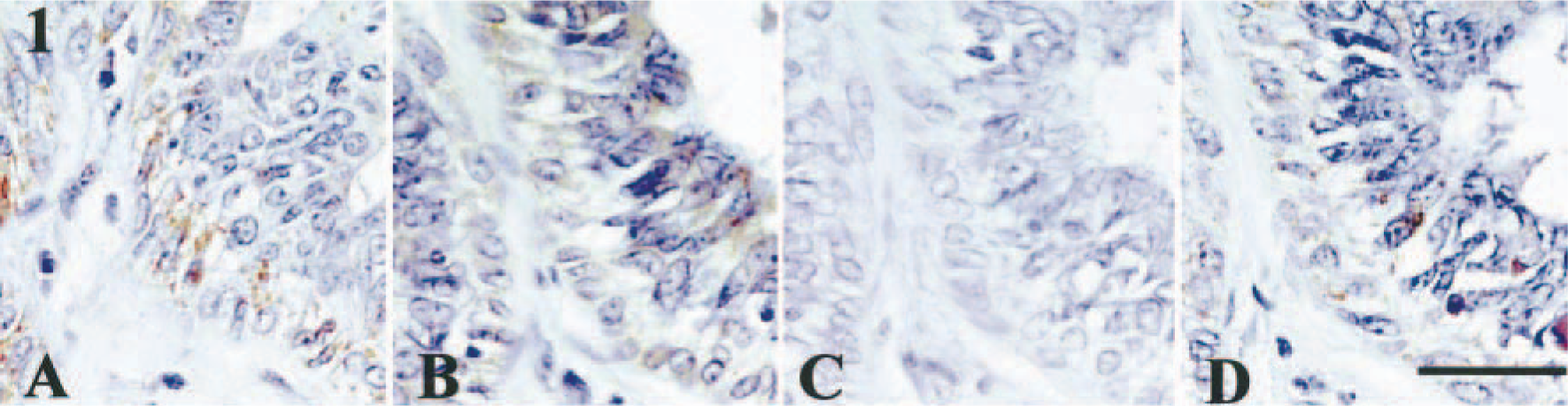
Mammary gland, complex adenoma; dog No. 8. Expression of BMP-6 and BMPRs in proliferative myoepithelial cells. The absence of immunoreactivity for BMPR-IB was due to variable expression in different areas of the tumor. Immunostaining for BMP-6 (A, +), BMPR-IA (B, +), BMPR-IB (C, −), or BMPR-II (D, +). See Table 1 for explanation of + and −. Bar = 30 µm.
Benign mixed tumors (n = 8). These tumors had ectopic cartilage formation, together with abundant proliferation of both myoepithelial and glandular epithelial cells. Two of eight dogs also had additional endochondral ossification. In six dogs, proliferating myoepithelial cells within the tubules were commonly immunoreactive for BMP-6 and all the BMPRs, whereas the intensity of staining varied (Table 1). Spindle- and star-shaped myoepithelial cells in the interstitial area also were positive for BMP-6 and all three BMPRs in these six dogs, although the intensity was weaker than that of intraductal myoepithelial cells. Star-shaped myoepithelial cells with chondroid stroma exhibited immunoreactivity for both BMP-6 (Fig. 2A) and BMPRs. Figure 2A–D represents a focus containing BMP-6–, BMPR-IA–, and BMPR-II–positive and BMPR-IB–negative star-shaped myoepithelial cells in dog No. 19. Myoepithelial cells adjacent to the ectopic cartilage also were moderately positive for BMP-6 and all BMPRs. Tubular epithelial cells in these areas were positive for BMPR-IA or BMPR-IB (or both). The cells, considered as immature chondrocytes within the rough irregular cartilage, exhibited very mild immunoreactivity for BMP-6 and BMPRs (Fig. 3A, D). In some foci, immunoreactivity for BMPRs, such as BMPR-IA and BMPR-IB, was absent (Fig. 3B, C). Mature chondrocytes within the complete cartilage were frequently immunoreactive for BMP-6 and for all BMPRs (Fig. 3E, H). Small tubular epithelial cells adjacent to immature cartilage were positive for BMP-6.
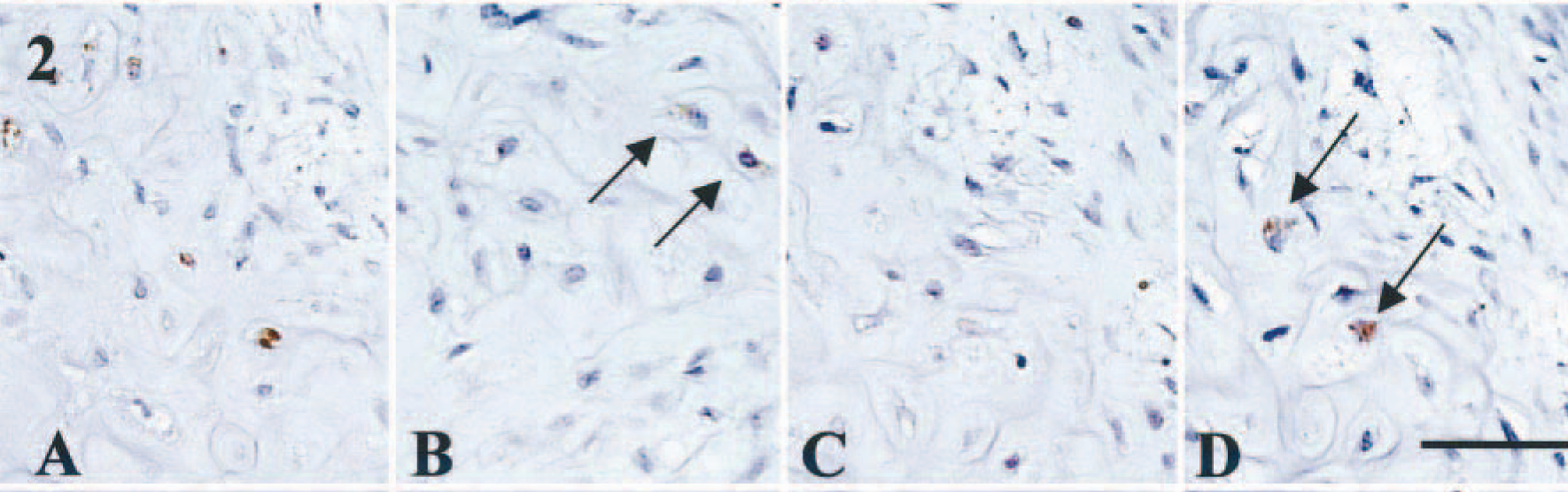
Mammary gland, benign mixed tumor; dog No. 19. Expression of BMP-6 and BMPRs in myoepithelial cells with chondroid change. Note the coexpression of BMP-6 and BMPRs in myoepithelial cells with chondroid stroma. Immunostainings for BMP-6 (A, +), BMPR-IA (B, +, arrows), BMPR-IB (C, −), or BMPR-II (D, +, arrows). See Table 1 for explanation of + and −. Bar = 40 µm.
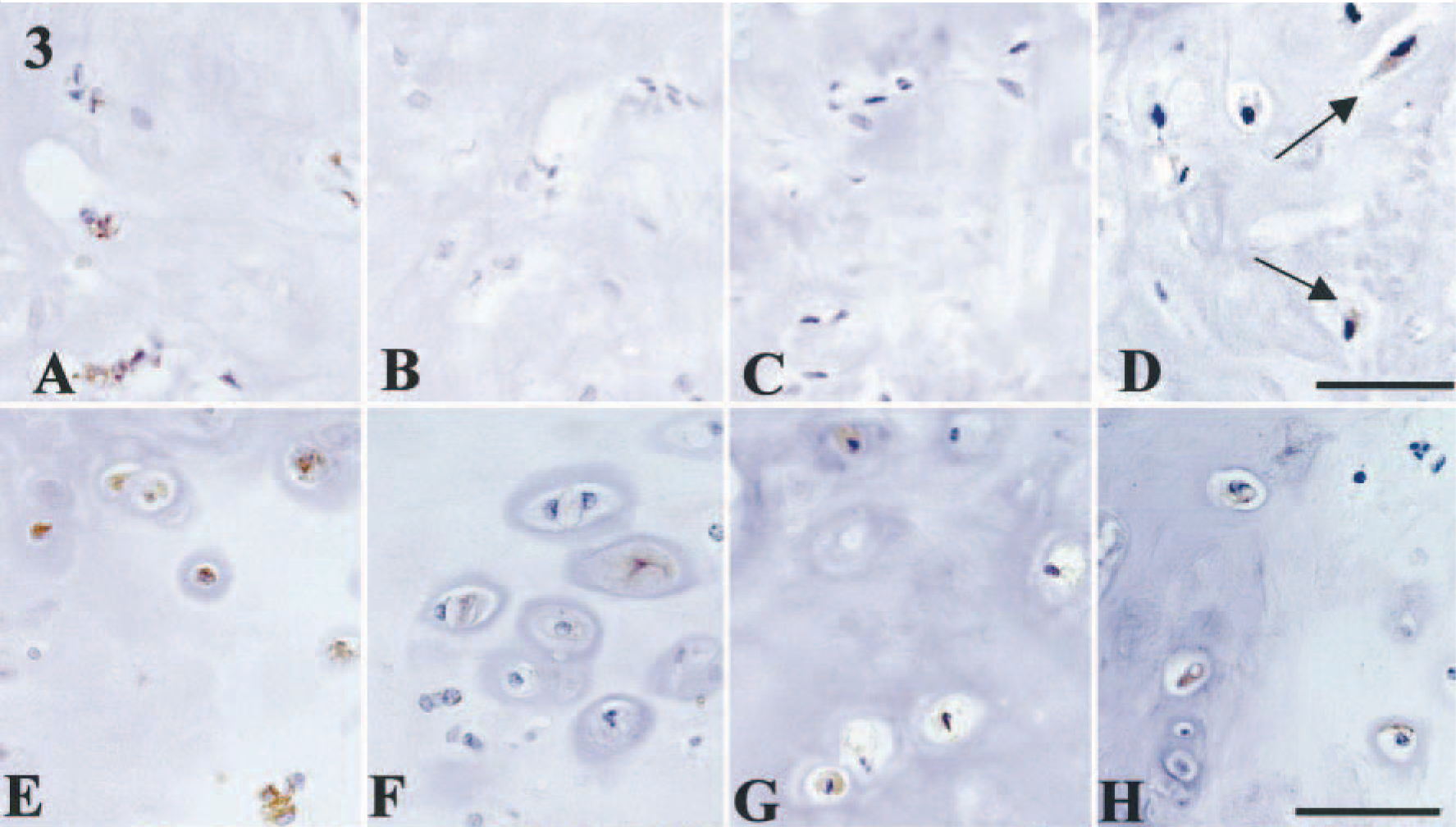
Mammary gland, benign mixed tumor; dog No. 22. Expression of BMP-6 and BMPRs in immature chondrocytes within incomplete cartilage (
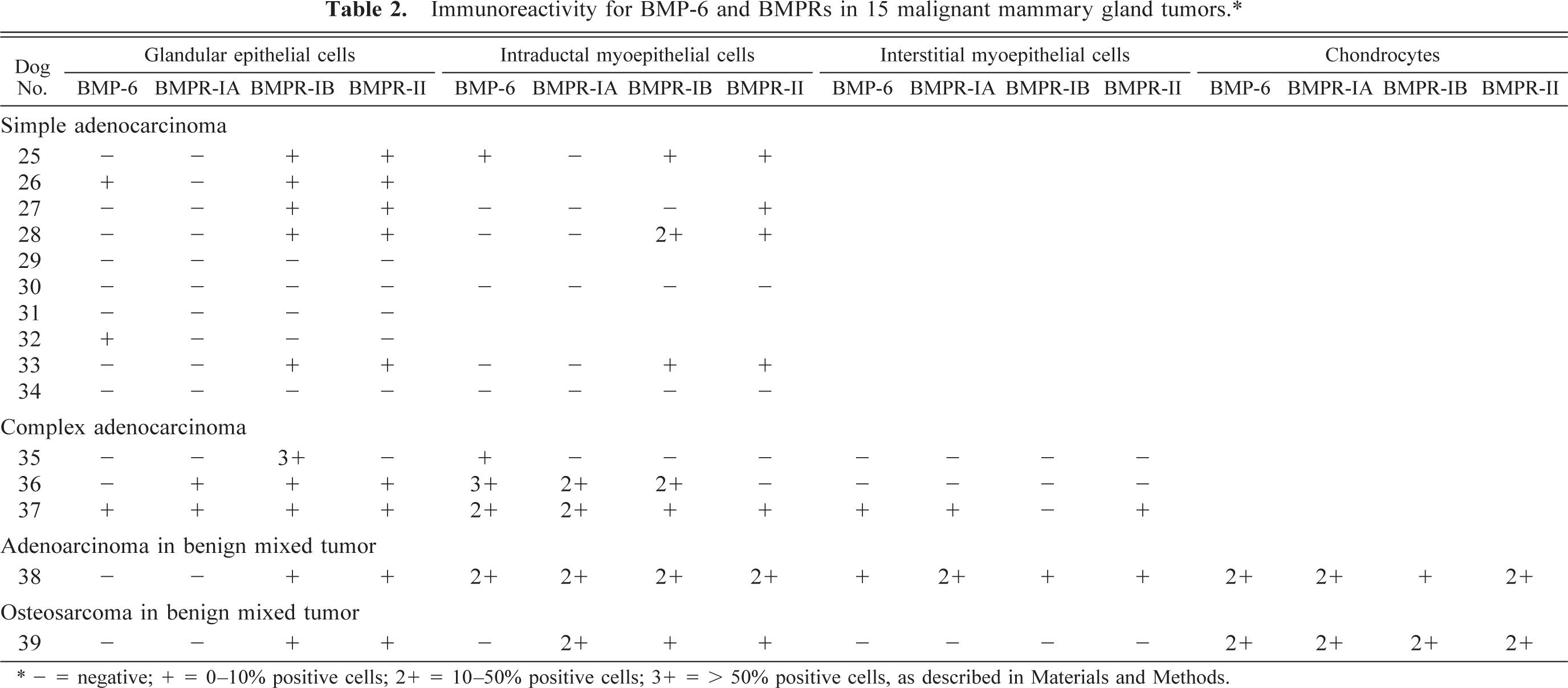
Simple adenocarcinomas (n = 10). These tumors were composed of a pure proliferation of poorly differentiated tubular and glandular epithelial cells. Four dogs had no myoepithelial component. The immunohistochemical results are summarized in Table 2. In only one dog, resting myoepithelial cells were weakly positive for BMP-6. Immunoreactivity for BMPR-IA was not detectable in either glandular epithelial or myoepithelial cells. In the foci with squamous cell metaplasia, coexpression of BMPR-IB and BMPR-II was observed in three dogs (Fig. 4A, B).
Immunoreactivity for BMP-6 and BMPRs in 15 malignant mammary gland tumors.∗
∗ - = negative; + = 0–10% positive cells; 2+ = 10–50% positive cells; 3+ = > 50% positive cells, as described in Materials and Methods.
Mammary gland, simple adenocarcinoma; dog No. 33. Expression of BMPRs in tubular epithelial cells with squamous metaplasia. Squamous tubular epithelial cells are positive for BMPR-IB and BMPR-II. Immunostaining for BMPR-IB (A) or BMPR-II (B). See Table 1 for explanation of + and −. Bar = 30 µm.
Complex adenocarcinomas (n = 3). The morphologic features of these tumors were in conformity with those of complex adenomas, whereas epithelial or myoepithelial cells (or both) exhibited cellular atypia and invasive activity. The myoepithelial cells within the tubules were positive to varying degrees for BMP-6 in three dogs. Weak immunoreactivity for BMPR-IA was found in the glandular epithelial cells and myoepithelial cells within the tubules in two dogs. Glandular epithelial cells were positive for BMPR-IB in three dogs in various intensities. Myoepithelial cells within the tubules, especially proliferative cells, showed moderate to intense BMPR-IB expression in two dogs. The myoepithelial cells proliferating in the interstitial areas were negative for BMPR-IB. Mild to moderate BMPR-II immunoreactivity was found in the glandular epithelial cells in two dogs and in interstitial myoepithelial cells of one dog. BMP-6 and all three BMPRs were colocalized in glandular epithelial cells and myoepithelial cells within the tubule in one dog.
Adenocarcinoma in benign mixed tumor (n = 1). Briefly, the tumor had similar morphologic features to a benign mixed tumor, whereas the epithelial component had malignant features. The glandular epithelial and myoepithelial cells showed marked proliferation. Necrotic foci were evident around the mature cartilage. Intraductal and interstitial myoepithelial cells coexpressed BMP-6 and all three BMPRs. BMPR-IB and BMPR-II were also detected in glandular epithelial cells with squamous metaplasia.
Osteosarcoma in benign mixed tumor (n = 1). This tumor was composed of an irregular proliferation of spindle-shaped osteoblastic cells with distinct osteoid and chondroid stroma, together with the characteristic features of mixed tumors. Hypertrophic chondrocytes were moderately positive for BMP-6 and BMPR-IA and were weakly positive for BMPR-II. Immunoreactivity for BMPR-IB was found in immature chondrocytes. Resting myoepithelial cells adjacent to the neoplastic foci showed weak immunoreactivity for all BMPRs but not for BMP-6.
Normal mammary gland tissues (n = 18). Immunoreactivity for BMP-6 and BMPRs in intact mammary tissue around the benign tumors was examined. Immunohistochemical findings are summarized in Table 3. Among normal mammary glands adjacent to simple adenomas (n = 5), complex adenomas (n = 6), and benign mixed tumors (n = 7), immunoreactivity for BMP-6 and BMPRs was most frequently detected in those of benign mixed tumors (five of seven dogs). In normal mammary glands adjacent to simple adenomas (n = 5), only one dog had coexpression of BMP-6 and BMPR-IB in resting myoepithelial cells. In two of five dogs, there were no immunoreactive cells for BMP-6 and BMPRs (Table 3). Resting myoepithelial cells of intact glands around complex adenomas (n = 6) were weakly immunoreactive for BMP-6 and BMPR-IA in only one of six dogs. Resting myoepithelial or glandular epithelial cells (or both) in normal glands within benign mixed tumors showed coexpression of BMP-6 and all three BMPRs in two dogs.
Western blotting
Specific immunoreactive bands for BMP-6 at 40–45 kDa were detected in samples from benign mixed tumors, complex adenomas, and simple adenomas (Fig. 5). In the sample from adenocarcinoma in a benign mixed tumor, no immunoreactive bands were detectable. In the homogenates from two benign mixed tumors, there were specific immunoreactive bands for BMPR-IB at 45 kDa, but the reactivity for BMPR-IA and BMPR-II was weak or not detectable (Fig. 6). In one sample from a simple adenoma, the immunoblot pattern was similar to that in a benign mixed tumor. In complex adenomas, there was very weak immunoreactive band for BMPR-IB at 45 kDa.
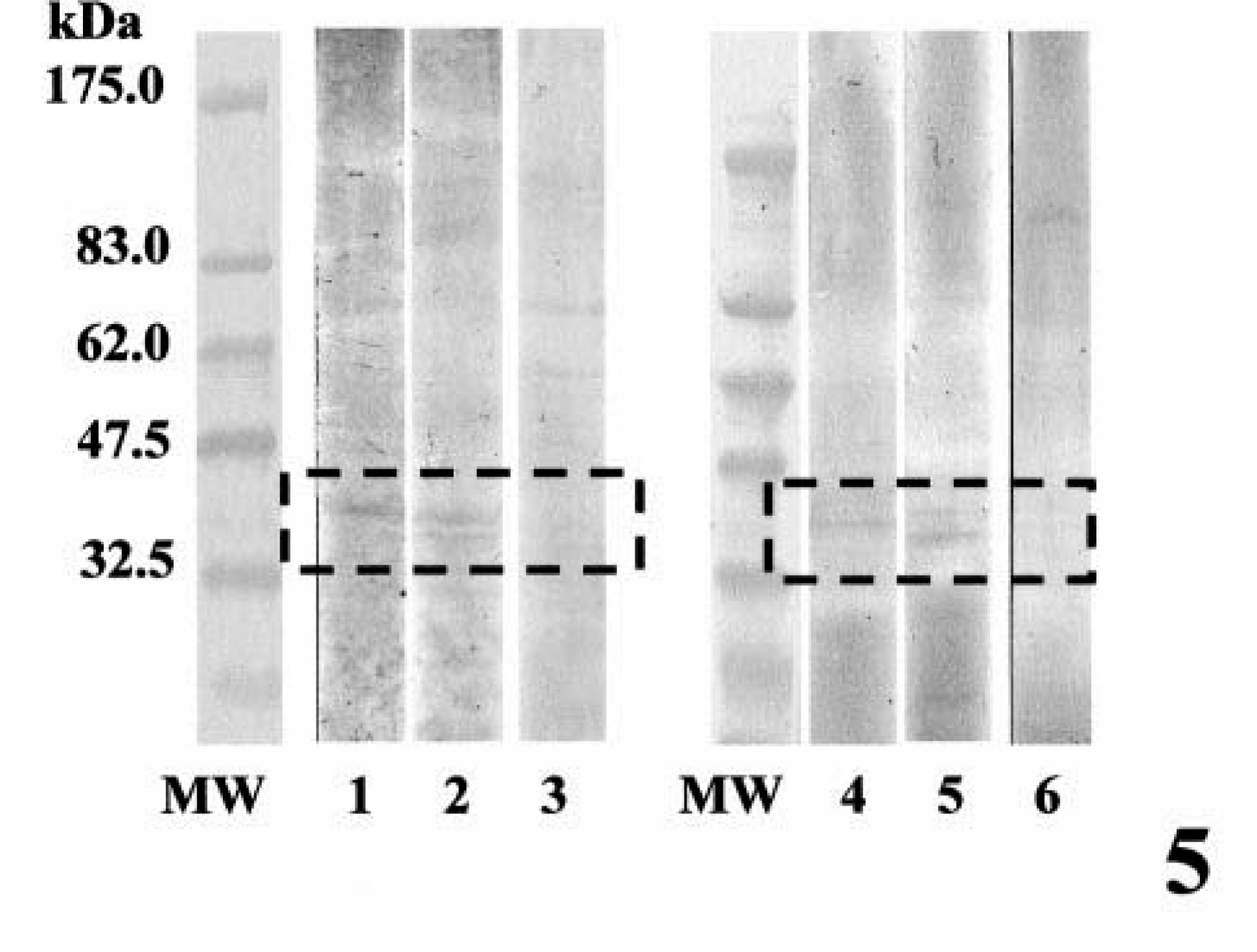
Western blotting analysis for BMP-6. MW, molecular weight markers; lanes 1 and 2, benign mixed tumor; lane 3, adenocarcinoma in benign mixed tumor; lane 4, complex adenoma; lanes 5 and 6, simple adenoma. Note the immunopositive bands at 40–45 kDa (lanes 1, 2, 4, and 5) for BMP-6.
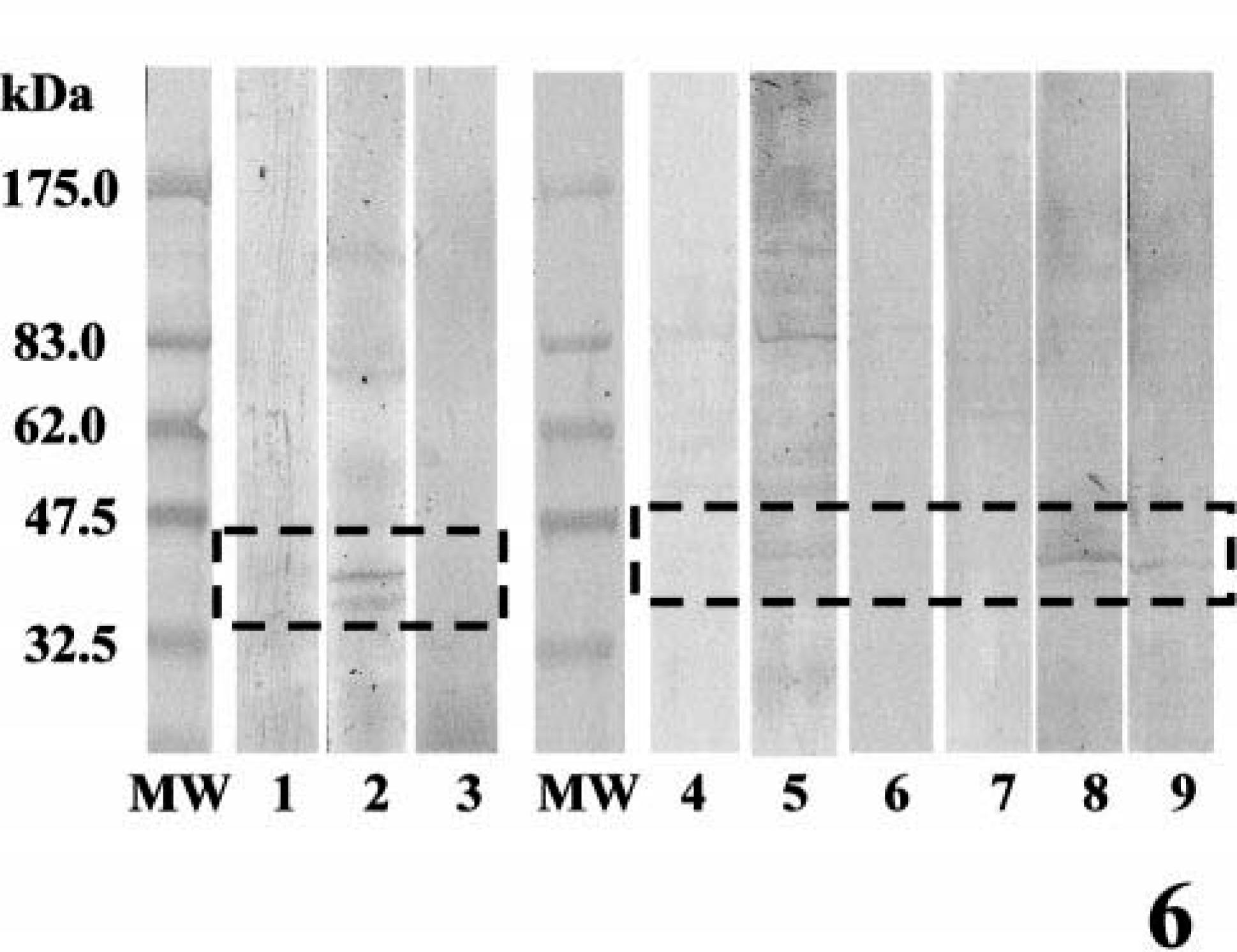
Western blotting analysis for BMPRs. MW, molecular weight markers; lanes 1–3, benign mixed tumor; lanes 4–6, complex adenoma; lanes 7–9, simple adenoma. Lanes 1, 4, and 7, BMPR-IA; lanes 2, 5, 8, BMPR-IB; lanes 3, 6, 9, BMPR-II. Note the immunoreactive bands (lanes 2, 5, and 8) for BMPR-IB at 45 kDa. Specific bands for BMPR-IA and BMPR-II were weak or undetectable.
Discussion
In this study, coexpression of BMP-6 and BMPRs has been confirmed predominantly in the myoepithelial cells in several types of canine mammary gland tumors. The findings, together with our previous report, 30 emphasized that myoepithelial cells expressing BMP-6 and BMPRs might play an important role for ectopic cartilage formation in canine mammary gland tumors. Expression of BMPR-IB, but not BMPR-IA, has been found in the early cartilage primordium of chick limb and in the perichondrium of developing cartilage of mice embryo. 12,37 During later chondrogenesis, BMPR-IA and BMPR-IB are coexpressed in chondrocytes and osteoblasts. These results suggest that BMPR-IB is involved in the differentiation of immature mesenchymal cells into primordial chondrocytes and that BMPR-IA regulates later chondrogenesis. This study revealed the absence of BMPR-IB expression in spindle- and star-shaped myoepithelial cells of most complex adenomas (7 of 11 dogs). In contrast, most benign mixed tumors (six of eight dogs) had moderate immunoreactivity for BMPR-IB. The data may suggest a role for BMPR-IB in metaplastic change of myoepithelial cells into chondrocyte lineage. A limited number of complex adenomas (4 of 11 dogs) with BMPR-IB expression in myoepithelial cells might indicate incipient formation of cartilage and bone by myoepithelial metaplasia in these tumors. Proliferative myoepithelial cells within the basement membrane and interstitial myoepithelial cells of complex adenomas and benign mixed tumors were immunoreactive for BMPR-IA. Because BMPR-IA tended to be coexpressed with BMP-IB in both tumors, corroborative function of these two BMPRs are suggested for chondroid metaplasia in canine mammary tumors. BMPR-II also was detected in various types of myoepithelial cells of all mammary tumors examined. BMPR-II was not always coexpressed with BMPR-IB. Several previous studies showed that other type II receptors for BMPs, such as ActR-II and ActR-IIB, could bind to BMPs. 6,33 It is known that ActR-II–deficient mice have mandible development defects, whereas the abnormality is not found in activin-deficient mice. 33 In canine mammary gland tumors, the type II receptors other than BMPR-II might also play some role for the stimulation of type I receptor and cartilage formation.
Immunoreactivity for BMP-6 in the interstitial myoepithelial cells tended to be mild compared with those of myoepithelial cells within the basement membrane. Some types of BMPs, such as BMP-2 and BMP-7, are known to induce osteogenesis and psammoma body formation in the presence of type IV collagen that contributes to trapping secreted BMPs. 9,15 When BMP-secreting cells are not covered with extracellular matrix, the secreted BMPs are easily hydrolyzed with extracellular protease or dispersed. The connection between myoepithelial cells and basement membrane consisting of type IV collagen may keep levels of BMP-6 high in canine mammary tumors. This phenomenon might also be related to the intense immunoreactivity for BMPRs on the myoepithelial cells within basement membrane.
Interestingly, metaplastic squamous cells showed intense immunoreactivity for both BMPR-IB and BMPR-II. Several previous studies showed that squamous cell carcinomas expressed BMP-2, BMP-5, or BMP-6 (or all). 9,29 BMPR-IA and BMPR-IB were detected in the epidermis of mouse embryos. 5,33 These findings may indicate the possibility that squamous metaplasia in canine mammary adenocarcinomas is also mediated by BMP signaling through BMPR-IB and BMPR-II. BMP-6 and BMPRs expression also was found in normal mammary gland tissues around the mammary neoplasms. A previous study of mouse mammary gland development has elucidated that mesenchymal and epithelial cells expressed BMP-2 and BMP-4 mRNA temporally and spatially and that these molecules participated in the extension of ducts of mammary gland, which was dependent on epithelial-mesenchymal interaction. 23 Extension of mammary ducts also is stimulated by estrogen. It has been reported that BMP-6 synthesis is stimulated by estrogen in vitro. 26 Considering these reports and our findings, BMP-6 may act on duct development in both normal and neoplastic canine mammary glands.
In conclusion, this study suggests the participation of BMP-6 signaling through BMPR on primary ectopic cartilage formation in canine mammary tumors. In addition, BMP signaling may mediate mammary development and metaplastic changes including squamous cell metaplasia of glandular epithelium.
