Abstract
This study addressed histologic and immunopathologic changes in ocular tissues and investigated the distribution of major histocompatibility class II (MHC class II)-positive cells in Atlantic salmon (Salmo salar) suffering from severe postvaccination disease. Twenty-nine fish with generalized inflammation, probably a result of vaccination, were investigated. One individual that had escaped vaccination was included in the study. Material was investigated by cultivation methods for fungi and bacteria. Histology using conventional staining procedures and immunohistochemistry with antisera against MHC class II β chain were performed. No growth was observed from the cultivation investigations. Histology revealed occlusion of the lumen in the larger choroid vessels and in the choriocapillaris, inflammatory infiltrations and loss of structure in the choroid rete, and, in some cases, aggregations of multinucleated giant cells (MGC) and Splendore-Hoeppli material. Immunohistochemistry demonstrated massive MHC class II+ cellular infiltrations in the uveal tract. Such infiltrations were also seen in the ventral ciliary cleft, a condition that is associated with glaucoma. Immunoreactive cells included dendritelike cells, epithelioid cells, and MGCs. The endothelia of smaller vessels were frequently MHC class II+, and immunoreactive infiltrations were seen in the optic nerve in several individuals. No pathologic changes were detected in the unvaccinated individual. In conclusion, generalized inflammatory reactions in fish may lead to severe ocular inflammation, occlusion of uveal vessels, and perivascular changes with MHC class II+ upregulation in cells in the uveal tract and optic nerve.
Keywords
A major challenge for fish pathologists has been the classification of cellular host responses in pathologic changes. Reliable histologic description and identification of immune cells have to be supported by useful cell markers, and for the Atlantic salmon (Salmo salar), very few such markers have been created. 23,32,33 The increasing information on genes encoding various putative immune molecules including cytokines in several fish species, as exemplified in the zebra fish (Danio rerio), 41 has not yet been matched by accompanying protein markers. Consequently, piscine pathology relies on the detection of various infective agents and histopathology, whereas immunopathologic studies are nearly absent.
The major histocompatibility class II (MHC class II) molecule is essential for the initiation of adaptive immune responses. It consists of two glycosylated heterodimers of one α and one β chain, which can bind and present antigen to CD4+ T cells. This may in turn induce a clonal selection and proliferation of B lymphocytes, leading to specific humoral immune responses. As a general rule, professional antigen-presenting cells, such as macrophages and dendritic cells, are constitutively MHC class II+ with an ability to up- or downregulate their expression of this molecule. Expression may also be induced in populations of nonprofessional antigen-presenting cells given appropriate stimulation. Bacterial lipopolysaccharides (LPS) are potent inducers, acting by binding to special receptors and thereby triggering the release of cytokines. In particular, tumor necrosis factor-α and interferon-γ (IFN-γ) activate MHC class II expression. 12
All jawed vertebrates investigated so far harbor the MHC genes. 9 In the Atlantic salmon, Hordvik et al. 17 cloned and sequenced the MHC class II β chain. Expression experiments showed that transcriptional regulatory mechanisms appeared similar to those in mammals. 22,24 The production of antisera recognizing the β chain allowed us to investigate several tissues using immunohistochemic methods, and we suggested a similar distribution of MHC class II+ cells as that seen in mammals. 23
Fish possess camera-type eyes, similar to their mammalian counterparts. However, some important differences exist. In fish, the choroid is nourished by the ophthalmic artery, which receives its blood from the pseudobranch. When entering the eye, the ophthalmic artery and vein form a countercurrent system in a horseshoelike structure curving round the head of the optic nerve. This structure is a part of the choroid and is termed as the choroid rete (Fig. 1). The pseudobranch and choroid rete are involved in elevating the oxygen pressure in the choriocapillaris adjacent to the retina. 5,15,43 The choroid is divided into an outer suprachoroid and the medial lamina vasculosa, containing the larger chororidea vessels, which branch into the third layer, viz. the inner lamina chroriocapillaris. The choriocapillaris nourishes the retina, which is avascular in most fish species including the Atlantic salmon. 5,15 The ciliary body is rudimentary and lacks ciliary processes, and the aqueous-producing region includes parts of the iridic epithelium in addition to the ciliary body. The lens is spherical, and focusing is regulated by the retractor lentis muscle. The drainage site of the aqueous humor is believed to be the ventral ciliary cleft (Fig. 1). 4,15
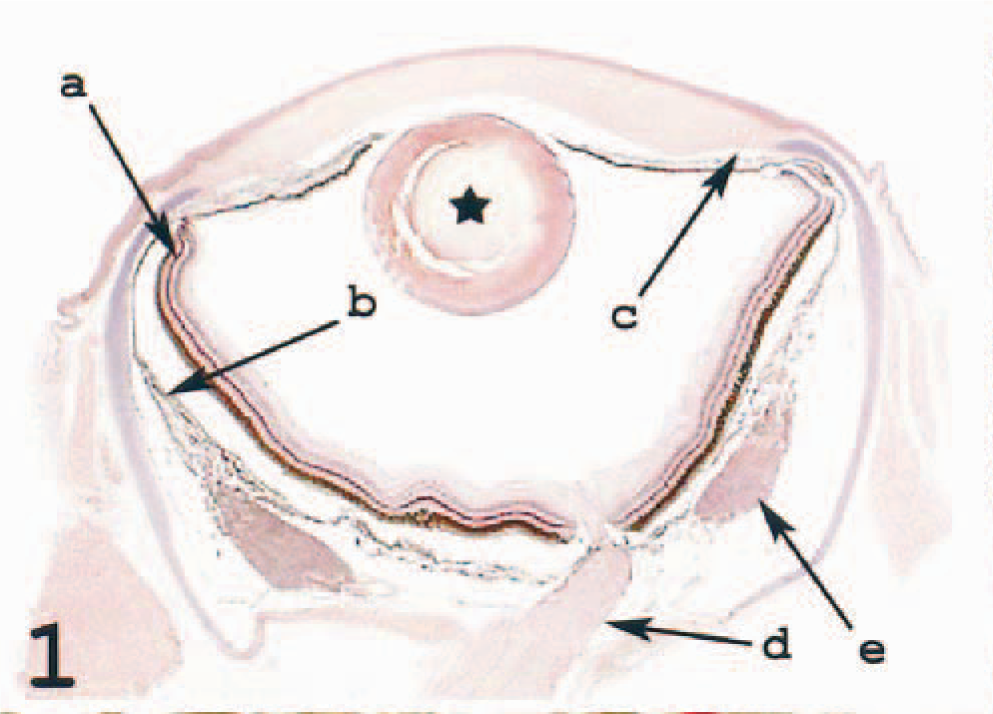
Eye; Atlantic salmon. Section shows the retina (a), the choroid (b), the ventral ciliary cleft (c), the optic nerve (d), and the choroid rete (e). HE.
Although exophthalmus and ocular hemorrhage are general and nonspecific clinical signs in a number of fish diseases, limited attention has been paid to eye pathology. 42 Recent attention to cataract has changed this. Bjerkås et al. 3 showed an astonishingly high incidence of this condition in wild out-migrant Atlantic salmon smolts caught at sea, suggesting a possible explanation for low return rates to rivers. Various pathologic ocular conditions of both wild and farmed fish leading to reduced feed intake, reduced ability to orientate, and increased exposure to predators may be highly underdiagnosed. Consequently, the eye deserves the attention of fish pathologists.
This study addressed ocular changes related to MHC class II–expressing cells in a population of farmed Atlantic salmon with severe postvaccination granulomatous inflammation in the abdominal cavity. 35 We have reported the distribution of MHC class II+ cells in the ocular tissues of nondiseased fish and shown that vaccination induces expression of this molecule. 21 Diseased fish have not been investigated for MHC class II expression, and because much of our focus has been on the eye, this was the organ of choice to start describing immunopathologic changes.
Materials and Methods
Fish
Atlantic salmon of the AquaGen strain (Kyrksæter⊘ra, Norway) were hatched in early spring 2001. The fish suffered an infectious pancreatic necrosis virus (IPNV) outbreak in fall 2001 but recovered. Intraperitoneal vaccination with Alphaject 6100 (Alpharma, Oslo, Norway) containing mineral oil adjuvant and antigens against Aeromonas salmonicida subspecies salmonicida, Moritella viscousus, Vibrio anguillarum serotypes 01 and 02, V. salmonicida, and formalin-inactivated IPNV serotype Sp was performed in May 2002 when the fish had reached a size of between 30 and 65 g. At vaccination, the water temperature varied between 11.4 C and 15.5 C. Fourteen days after vaccination, the smolts were transferred from freshwater tanks to sea cages. In August, routine autopsy of the fish performed by the local veterinarian revealed extensive intraperitoneal reactions with severe adhesions and indurated changes as previously described by Poppe and Breck. 35 The fish did not perform well and stopped feeding. Sixty eyes from 30 fish were sampled in November (first sampling, n = 10) and December (second sampling, n = 10) in 2002 and in February in 2003 (third sampling, n = 10). The eyes were fixed whole in 4% ice-cold nonbuffered formaldehyde. In the last sampling, one unvaccinated individual from the same school was included together with nine affected fish as a negative control. At autopsy, this individual revealed no signs of injection-site changes associated with vaccination. 29 Another fish in this sampling showed severe unilateral exophthalmus. All eyes were dissected after fixation, with vertical cuts through the anterior and posterior poles, and were processed for paraffin embedding following standard procedures. 2
Cultivation and staining for bacteria and fungi
Paraffin-embedded sections from the eyes and peritoneal adhesions from all samplings were Gram stained to identify bacteria, Ziel-Neelsen-stained for mycobacteria, and stained with both periodic acid–Schiff reaction and Gomori methenamine silver. 2,13
Five affected fish from the third sampling were investigated for the presence of pathogens using cultivation. For each method, samples from head kidney and adhesions from the abdominal cavity were harvested and cultivated. Inoculates on blood agar (Bacto heart infusion agar, Difco Laboratories, Detroit, MI) supplemented with 5% bovine blood and with a final NaCl concentration of 0.5% were incubated for 2 weeks at 15 C. Investigation for marine mycobacteria followed standard procedures, 31 with the exception that incubation was performed for 8 weeks at 10 C, 22 C, and 30 C. Mycological investigation was performed by incubation on Sabouraud agar (Bacto Sabouraud agar modified, Difco Laboratories), following the manufacturer's instructions, for 2 weeks at 15 C.
Histology and immunohistochemistry
Sections of eyes, 4 µm, were mounted on glass slides and stained with hematoxylin and eosin (HE), following standard procedures. 2
For immunologic histology, sections of 4 µm were mounted on slides coated with 3-aminopropyl triethoxy silane (Sigma Chemical Company, St. Louis, MO), dried overnight at 50 C, and stored at room temperature. A polyclonal antiserum recognizing a fraction of the β chain was used to detect MHC class II molecules. 23 Negative controls used rabbit preimmune serum.
Tissue sections were dewaxed in xylene and rehydrated in graded ethanol baths. To unmask the MHC class II epitopes, the sections were subsequently autoclaved in a solution of 0.01 M citrate buffer, pH 6.0, kept at 120 C for 15 minutes, cooled to room temperature, and then transferred to phosphate-buffered saline (PBS). 6
To prevent nonspecific binding, the sections were incubated for 20 minutes in normal caprine serum diluted 1 : 50 in 5% bovine serum albumin (BSA) in Tris buffer solution, 0.05 M Tris-HCl, pH 7.6, diluted 1 : 10 in 0.9% NaCl (TBS) before incubation overnight at 4 C with pre- or postimmune isolated sera, diluted 1 : 1,000 in 1% BSA in TBS. The next morning, the sections were washed in PBS and incubated at room temperature for 30 minutes with biotinylated goat anti-rabbit IgG (Vector Laboratories, Burlingame, CA) diluted 1 : 200 in 1% BSA in TBS. The sections were subsequently washed in PBS.
To inhibit endogenous peroxidase activity, the sections were treated with 3% H2O2 in methanol for 10 minutes and then washed in 0.01 M PBS, pH 7.3. The inhibition step was followed by incubation for 30–35 minutes in avidin–biotin complex–horseradish peroxidase (PK-4000, Vector Laboratories) according to the manufacturer's instructions. After washing in PBS, bound antibody was detected using 3-amino-9-ethyl-carbazole (Sigma) for 17 minutes at room temperature.
The sections were washed in distilled water and either left unstained or counterstained with hematoxylin, 45 seconds, washed in distilled water, and mounted with polyvinyl alcohol mounting medium, pH 8.2 (Ullevål Apotek, Oslo, Norway), for light microscopic analysis. Sections treated with preimmune rabbit antiserum served as negative controls in the experiments.
Results
Cultivation and staining for bacteria and fungi
None of the staining techniques used on eyes and peritoneal adhesions revealed any presence of fungi or bacteria, and no growth was observed on cultivation.
Histology and immunohistochemistry
The eyes of the unvaccinated fish displayed findings as described in the control material in a previous study. 21 In brief, no immunoreactive cells were present in the cornea proper. In the limbus region, dendritelike MHC class II+ cells were seen. In the anterior chamber, a very limited number of macrophage-like cells reacted with the antiserum. Scattered immunoreactive cells were present in the uveal tract, including the choroid rete. A very limited number of immunoreactive cells were seen in the retina with the exception of the photoreceptor layer.
In vaccinated fish with severe intraperitoneal changes, marked histo- and immunopathologic reactions were identified in the eyes. The changes were uniform in all eyes sampled at different times unless otherwise mentioned. One animal from the third sampling showed unilateral exophthalmus with an enlarged globe. Histologic examination of this eye revealed severe retinal and uveal degeneration, probably a consequence of ischemia. Apart from this, other classical signs of glaucoma were not observed. 26 Four animals, one from the first sampling, one from the second, and two from the third, displayed multinucleated giant cells (MGCs) in the choroid rete adjacent to Splendore-Hoeppli material with asteroid configuration. Apart from these observations, the data from all diseased fish are presented together.
No pathologic changes were observed in the cornea. Immunoreactivity for MHC class II in the cornea proper was absent, but positive cells were seen in the area of the limbus.
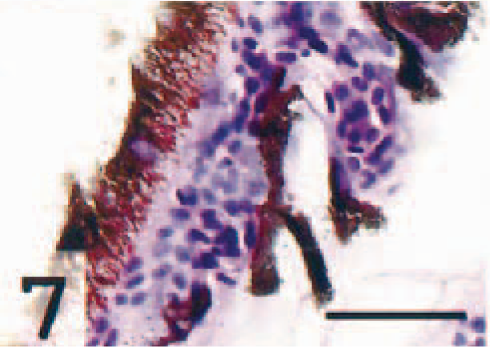
Populations of epithelioid cells were frequently seen in the region of the ventral ciliary cleft (Fig. 2). Immunohistochemistry revealed that MHC class II molecules were frequently expressed at this site, particularly in large cells embedded in the trabecular meshwork. However, immunonegative cells were also detected (not illustrated).
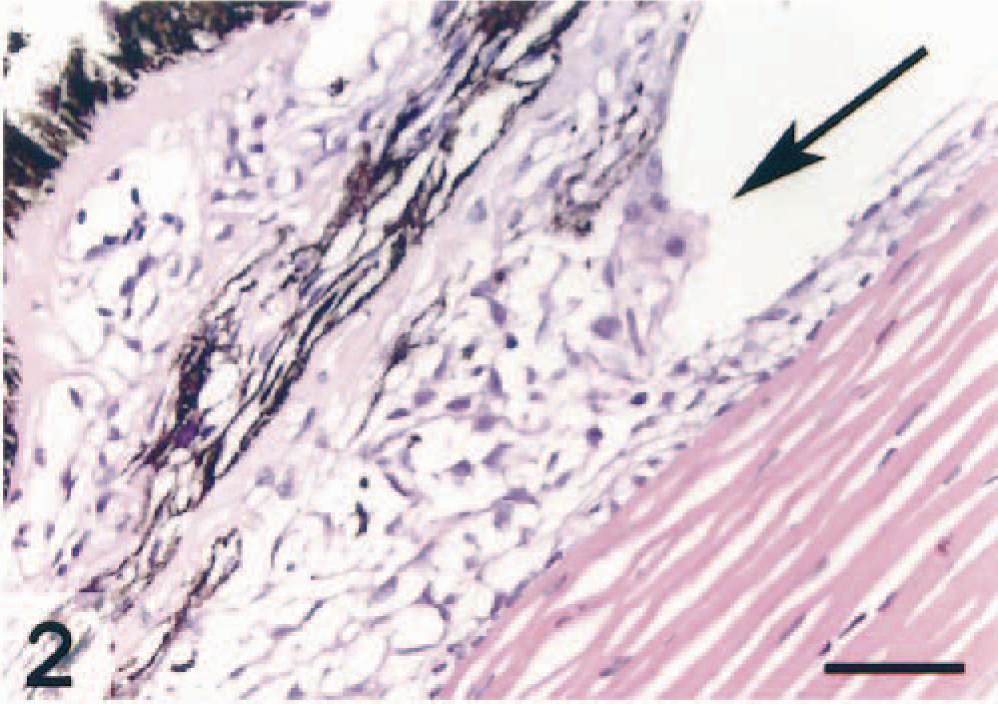
Ventral ciliary cleft; Atlantic salmon. Occlusions by epithelioid-like cells are seen at the entrance of the cleft (arrow). HE. Bar = 50 µm.
Cells within the iris of affected fish were dendritelike and appeared to be loosely arranged. Immunoreactive cells were seen in abundant numbers in the iris and in the rudimentary ciliary body (Fig. 3). In iridial vessels, the endothelium was frequently immunoreactive and surrounded by MHC class II+ dendritelike cells.
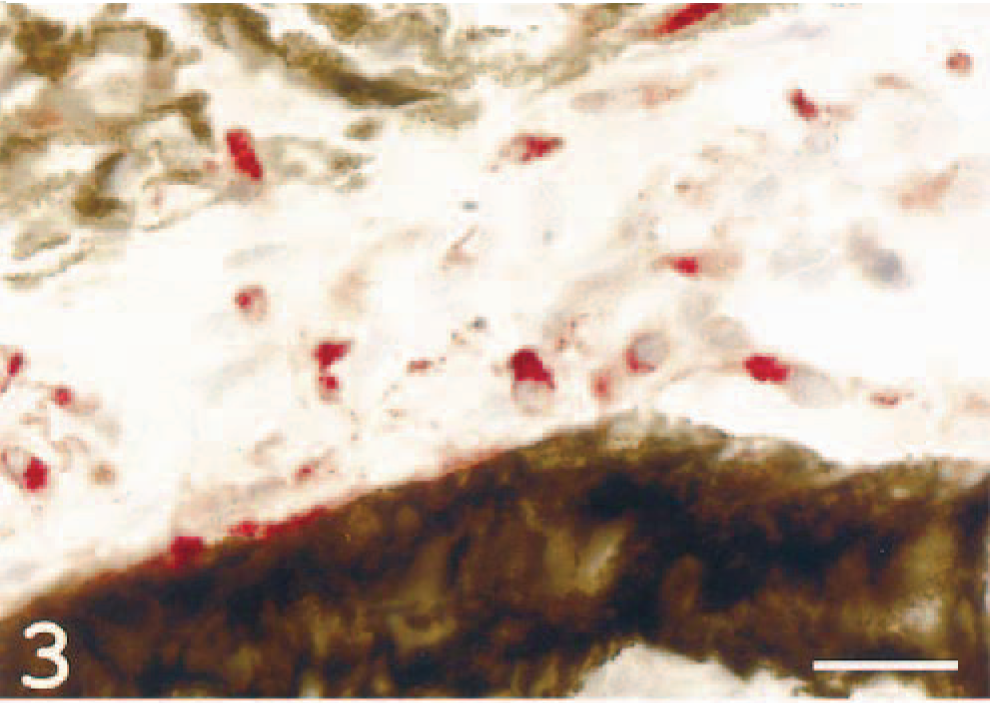
Iris; Atlantic salmon. MHC class II immunoreactive, dendrite-like cells are seen (red signal). MHC class II immunostain, hematoxylin counterstain. Bar = 20 µm.
In the larger choroid vessels, variably sized and variably dense perivascular infiltrates consisting of epithelioid cells were seen (Fig. 4). A large proportion of endothelial cells and the perivascular epithelioid cells were MHC class II+ (Fig. 5). These findings are consistent with inflammatory vasculopathy.
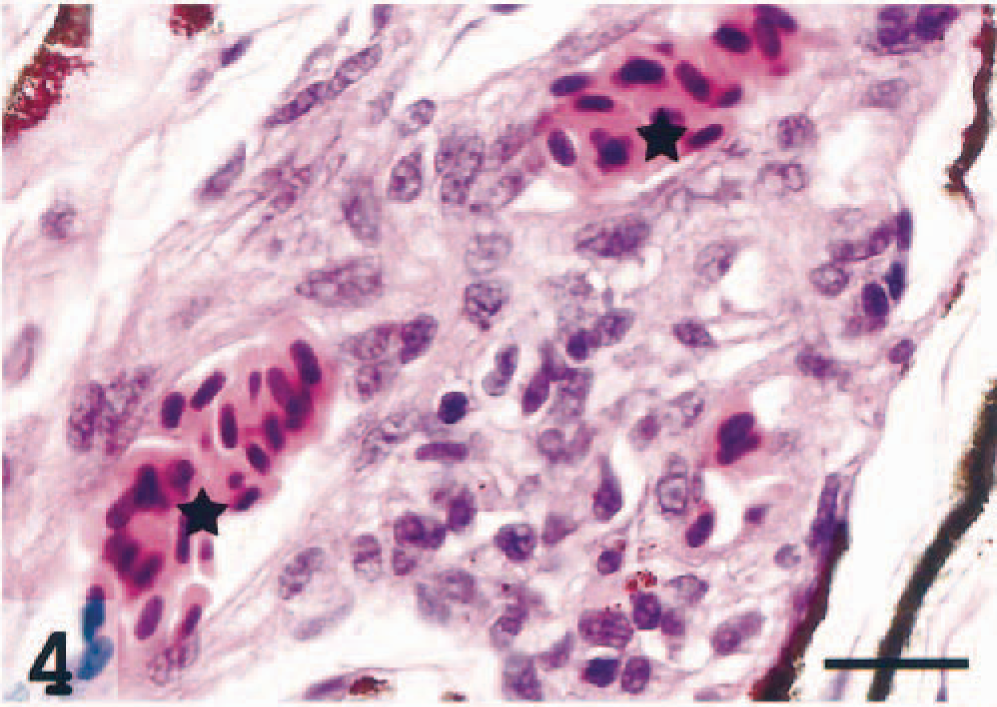
Choroidea; Atlantic salmon. Massive infiltrations of epithelioid-like cells between and around two uveal vessels (asterisks). HE. Bar = 20 µm.
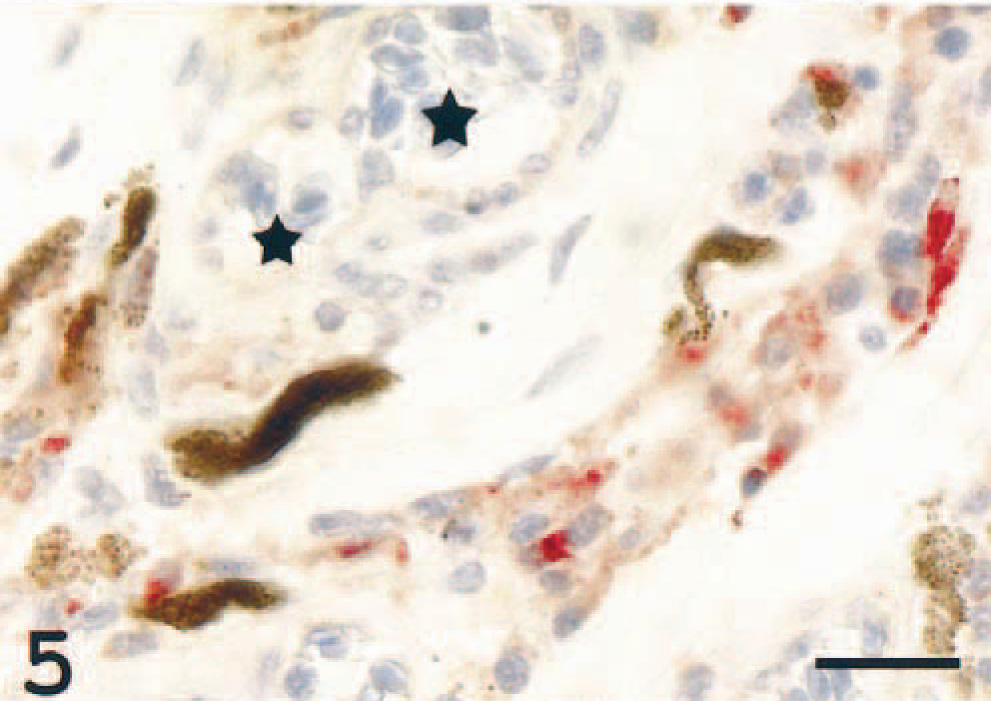
Choroidea; Atlantic salmon. Several cells are immunoreactive in the tissue surrounding two choroid vessels (asterisks). MHC class II immunostain, hematoxylin counterstain. Bar = 20 µm.
In the choriocapillaris, moderate to severe infiltrations, proliferations, or both, sometimes occluding the vascular lumen, were seen in all individuals. The cells in these areas were mostly MHC class II+, both in occluded and in partially occluded vessels, suggesting an inflammatory choriocapillaropathy (Figs. 6, 7). Focal retinal detachment was seen occasionally (not illustrated).
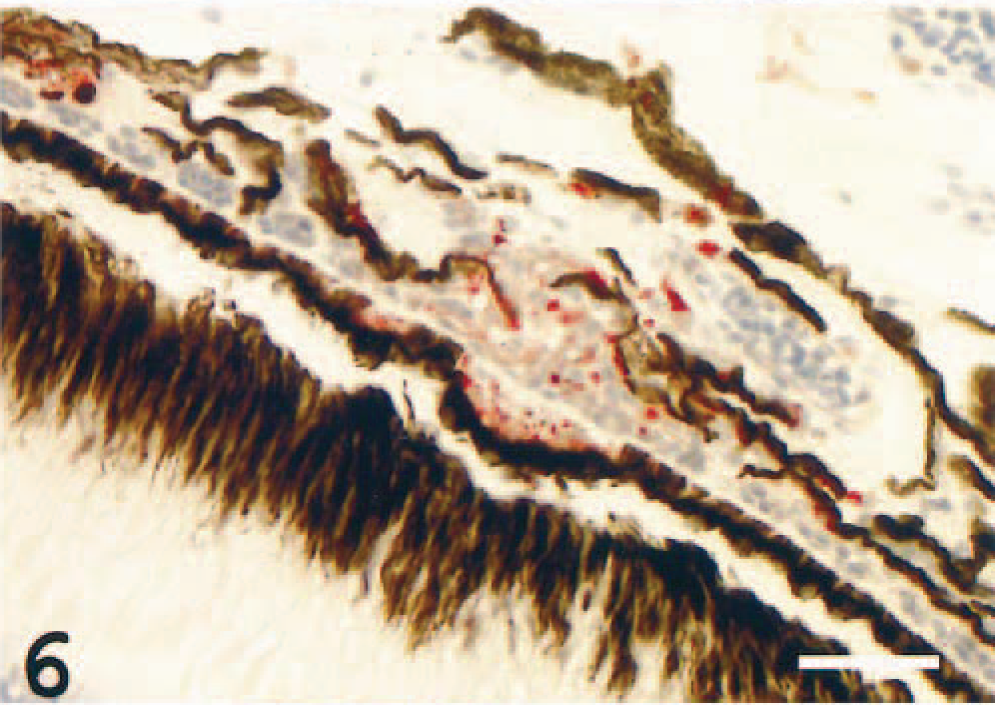
Choriocapillaris; Atlantic salmon. Occlusions by positively stained epithelioid-like cells are seen in the capillary structures. MHC class II immunostain, hematoxylin counterstain. Bar = 50 µm.
Choriocapillaris; Atlantic salmon. Occlusions by epithelioid-like cells in and around the capillary lumen. HE. Bar = 40 µm.
All degrees of occlusion of the larger choroid vessels were seen within both eyes of each individual (Fig. 8). The occlusions contained abundant MHC class II+ cells (Fig. 9). In vessels devoid of occlusions, the endothelium was frequently immunoreactive (Fig. 10), but immunonegative vessels were also identified.
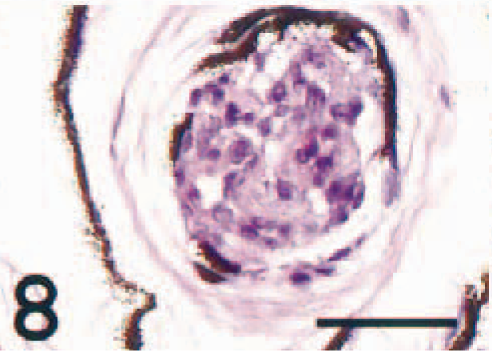
Choroidea, larger vessel; Atlantic salmon. The vascular lumen is totally occluded by epithelioid-like cells. HE. Bar = 40 µm.
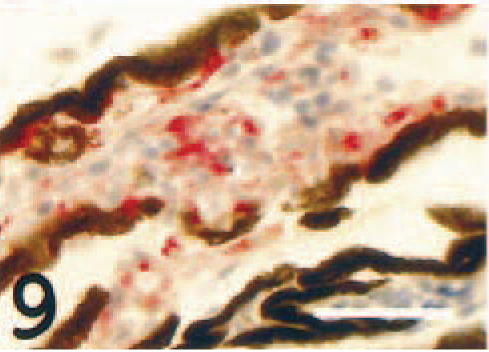
Choroidea, occluded larger vessel; Atlantic salmon. Strong reaction present in several cells in the occlusion. MHC class II immunostain, hematoxylin counterstain. Bar = 40 µm.
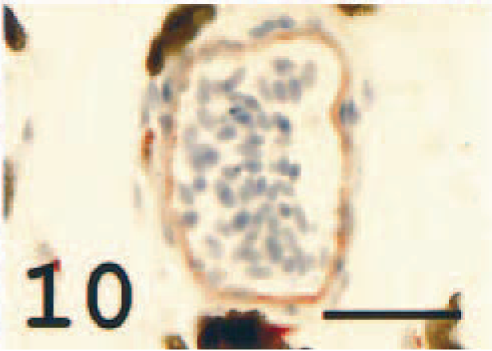
Choroidea, larger vessel; Atlantic salmon. Strong endothelial reaction. MHC class II immunostain, hematoxylin counterstain. Bar = 40 µm.
Varying amounts of loosely arranged dendritelike and epithelioid cells were seen in the extravascular choroid tissue and choroid rete. The dendritelike cells frequently displayed varying contents of melanin granules, probably melanosomes (Fig. 11). Such melanosome-containing cells also tested positive for the MHC class II antiserum (Fig. 12). Taken together, the findings indicate a severe choroiditis in diseased fish.
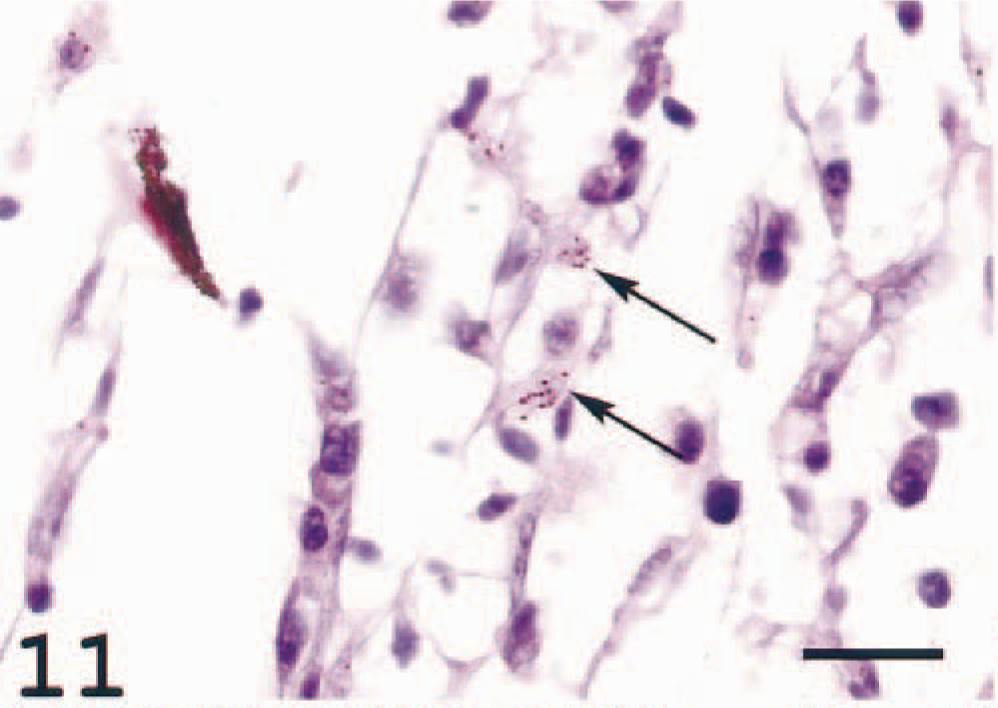
Choroid rete; Atlantic salmon. Loosely arranged dendrite-like cells located in the periphery of the choroid rete close to the sclera. Arrows indicate cells with cytoplasm containing melanin granules, probably melanosomes released from melanin-producing cells elsewhere. HE. Bar = 20 µm.
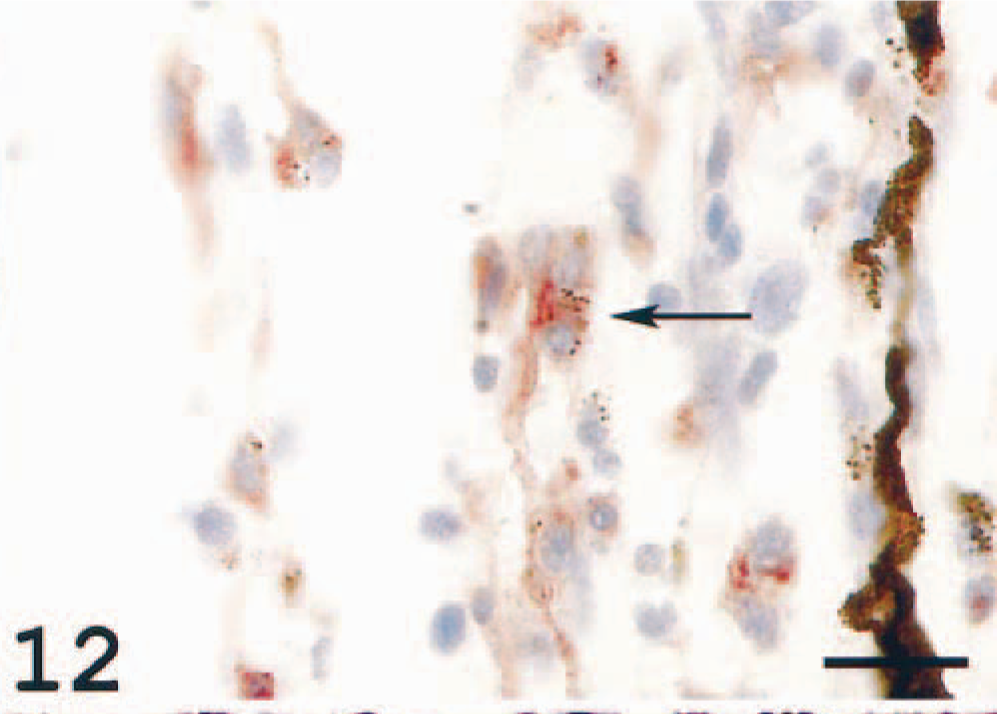
Choroid rete; Atlantic salmon. Cells in the same localization as those seen in Fig. 11 and containing melanin granules show immunoreaction. MHC class II immunostain, hematoxylin counterstain. Bar = 20 µm.
In the choroid rete, varying degrees of degeneration of the countercurrent system due to inflammatory reactions were seen (Fig. 13). In four individuals, giant cells were present in the choroid rete of both eyes. The nuclei in the MGCs were arranged in a circular, peripheral arrangement typical for Langhans' giant cells. 1 There were also deposits of homogenous, deeply eosinophilic to basophilic material, resembling the Splendore-Hoeppli phenomenon and surrounded by macrophage-like cells (Figs. 13, 14). The MGCs showed weak or no reaction with the MHC class II antiserum, and the Splendore-Hoeppli material was surrounded by MHC class II+ cells (Fig. 15). Large numbers of immunoreactive cells were seen elsewhere in the choroid rete. Frequently, MHC class II+ cells were detected not only adjacent to the large vessels at the base of the choroid rete but also in the recurrent capillary system, leading to occlusion and destruction of these fine structures.
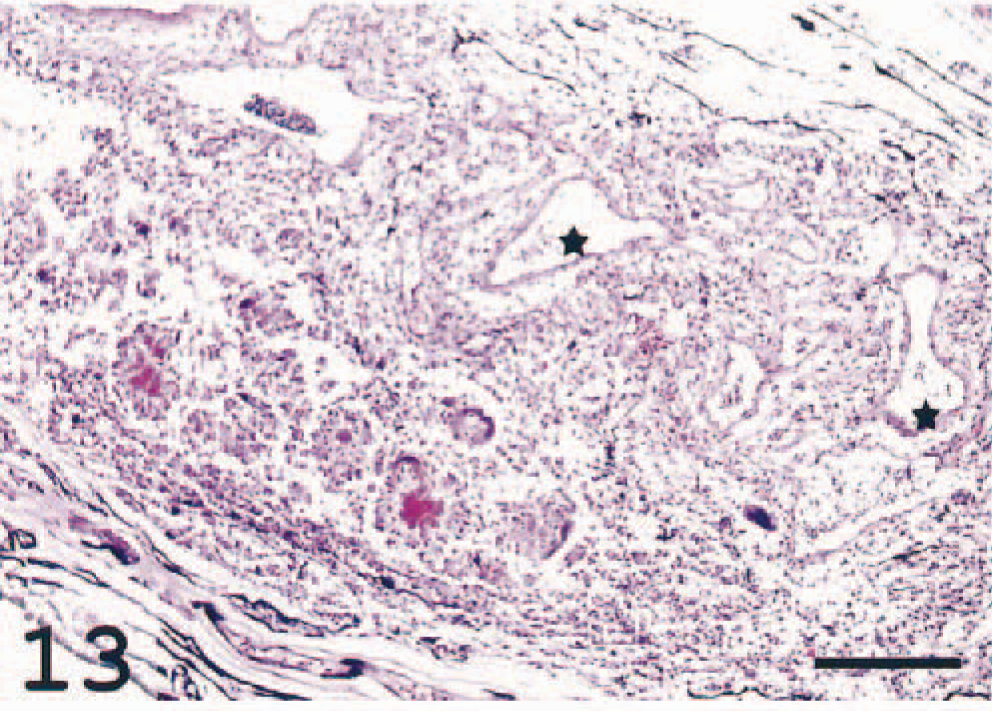
Choroid rete; Atlantic salmon. The structure of the vascular capillary rete is destroyed and replaced with inflammatory cells. Asterisks indicate larger vessels. HE. Bar = 200 µm.
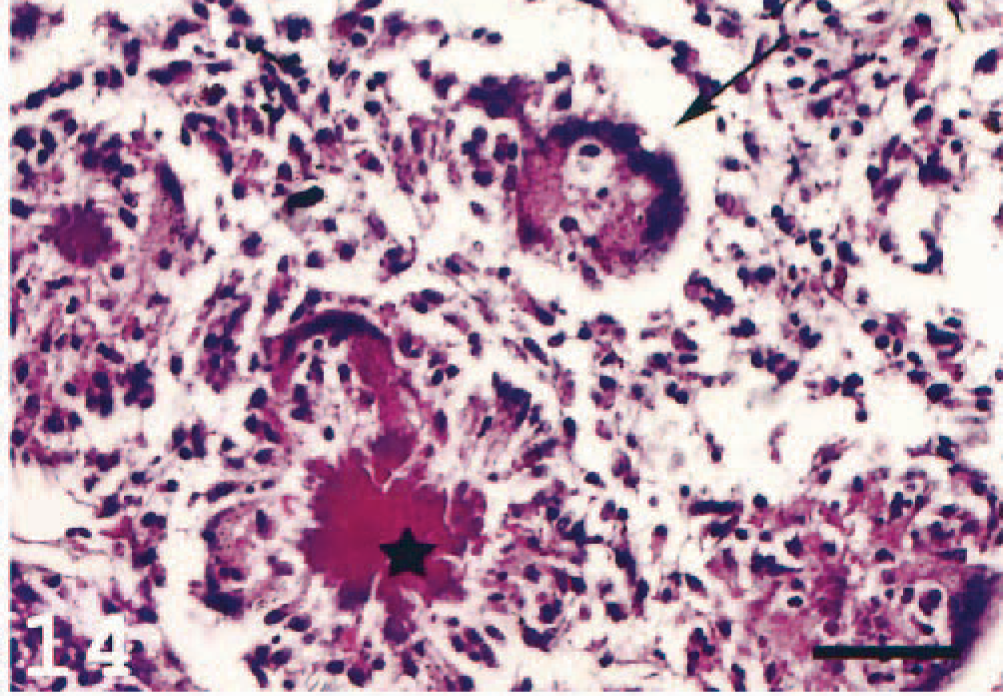
Choroid rete; Atlantic salmon. Arrow indicates a MGC containing debris. Asterisk indicates Splendore-Hoeppli–like material with characteristic radiating clubs. HE. Bar = 50 µm.
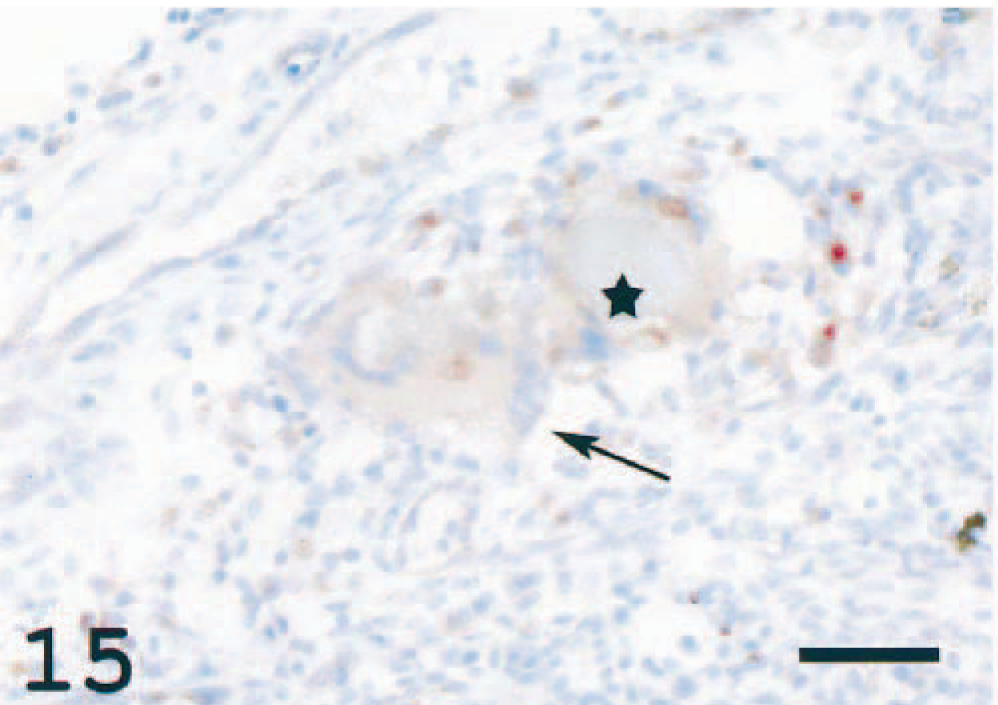
Choroid rete; Atlantic salmon. Arrow indicates a weakly staining MGC. Asterisk is placed in Splendore-Hoeppli material surrounded by immunoreactive cells. Reaction is also seen in scattered cells elsewhere. MHC class II immunostain, hematoxylin counterstain. Bar = 50 µm.
No signs of cellular degeneration or infiltration were observed within the retina. Antiserum against MHC class II revealed scattered immunoreactive cells in all layers with the exception of the photoreceptor layer (not illustrated).
In the optic nerve, accumulations of cells were seen in a majority of the diseased fish. These accumulations, resembling focal inflammatory processes, were MHC class II reactive (Fig. 16), and the finding was consistent with neuritis.
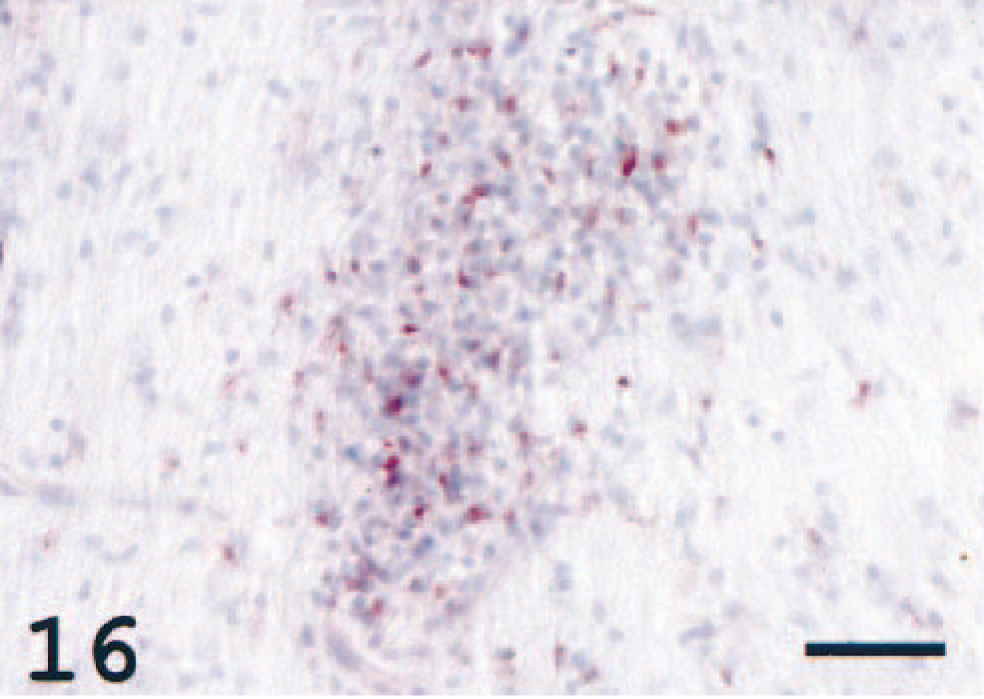
Optic nerve; Atlantic salmon. Clusters of immunoreactive cells are seen in the nerve. MHC class II immunostain, hematoxylin counterstain. Bar = 50 µm.
In the periglobular tissue, perivascular MHC class II+ cellular infiltrations were frequently observed in adipose and muscular tissues (not illustrated).
Discussion
The mammalian uveal tract is particularly susceptible to inflammatory reaction as identified by MHC class II induction. 10 With regard to fish, the current opinion is that the uveal response to various antigens is often remarkably limited, and if reaction occurs, necrosis or granuloma formation is most common. 42 In our material, changes were prominent in the uveal tract with peri- and intravascular infiltrations and MGCs in the choroid rete. Immunohistochemistry revealed MHC class II reactivity in the infiltrates, suggesting similar inflammatory mechanisms as in mammals. No pathologic cellular infiltrations were observed in the retina. Immunoreactive cells were seen in the optic nerve.
The macrophage and its derivates, epithelioid cell and MGCs, are considered a histologic hallmark of granuloma. 37 These cells may be either MHC class II positive or negative, depending on their age, stimulation, and localization in the inflammatory changes. 7,18,38 Epithelioid cells are highly differentiated, tightly interdigitated macrophages, whereas MGCs are formed by macrophage fusion. They are present in inflammatory responses against persistent microbial infections or nonphagocytozable foreign objects. 8 Because no microorganisms were detected in our material and because one nonvaccinated individual showed no signs of such reaction, we believe that the severe inflammation observed was a consequence of adverse reactions of unknown etiology to vaccination.
In the mammalian cornea, the MHC class II expression is absent in Langerhans cells, probably because of tight downregulation of expression, 14 and similar mechanisms may be present in fish. 21 In the present material, no immunopathologic changes were seen in the cornea.
The ventral ciliary cleft, the probable site of aqueous humor drainage, harbored epithelioid cells located adjacent to and within the trabecular meshwork. Macrophages that transverse the anterior eye structures leaving the posterior chamber and follow the aqueous humor flow to the anterior chamber and from there escaping the eye via the iridocorneal drainage apparatus 39 are the probable precursors. Such cells may well block drainage, consequently leading to glaucoma as seen in mammals. 26,30 Because the development of glaucoma is associated with uveitis, this may be an explanation for the unilateral exophthalmus clinically observed in one individual. Macrophage-like cells have previously been identified in the anterior chamber of the Atlantic salmon eye. 21 Unfortunately, no studies have addressed glaucoma or investigated the normal intraocular pressure in fish. Given the importance of exophthalmus as a clinical sign in a number of different fish diseases, surprisingly little effort has been made to clarify the pathogenesis of this condition.
Studies in mammals have shown that there is a fine meshwork of dendritic cells and resident tissue macrophages in the uvea, and following induction of uveitis, infiltration of monocytes and lymphocytes occurs in the iris vasculature in addition to an increased recruitment and migration of dendritic cells, all MHC class II+. 28 In our material, it was impossible to distinguish between dendritic cells and cells of macrophage origin. Both can be MHC class II+, but in fish, markers to differentiate these cells are missing. The presence of dendritic cells in fish is still a controversy. There was a high density of immunoreactive cells in the iris and the ciliary process of affected fish, and we believe this response is of similar character to that described in mammals. 28
The nature of the vessel reactions was highly diverse in various regions within a single eye of vaccinated fish. Choroid vessels with both immunoreactive and immunonegative endothelial cells were seen in addition to perivascular and luminar cellular infiltrations. Some of these cells, resembling epithelioid cells, were MHC class II+, whereas others were negative. In a previous study, we showed that vaccination could rapidly induce endothelial expression, which disappeared within some days thereafter. 21 In mammals, such expression can either be constitutive or induced by IFN-γ. However, because endothelial cells are nonprofessional antigen-presenting cells, the reason for and role of MHC class II molecules here are disputed, but together with other factors, expression may play a role in controlling the extravasation of immune cells into inflamed tissue. 20,34
In the choroid rete, inflammatory reactions with abundant epithelioid cells and MGCs were present in vaccinated fish. Material resembling the Splendore-Hoeppli phenomenon 11,19 was also visible. These findings indicate intense localized antigen-antibody reactions. In mammals, the Splendore-Hoeppli material has been described as being eosinophilic to deeply eosinophilic and usually radiating in an asteroid fashion. Its pathogenesis is poorly understood. The presently observed phenomenon was morphologically similar but basophilic. Such material has as yet not been reported in fish. We suggest that our findings are analogous to the mammalian Splendore-Hoeppli phenomenon. Taken together, pathologic vascular changes were prominent, and as a result of the frequent occlusions seen, ischemia of the ocular tissues would be a plausible consequence of these reactions. It is likely that cellular debris from the melanin-containing cells in the choroid pigment layer and the retinal pigment cell epithelium was released and subsequently trapped in MHC class II+ cells in the choroid rete.
In addition to the oxygen pressure regulatory function of the choroid rete, the recurrent blood system 43 is ideal for antigen surveillance and trapping in afferent and efferent blood. The most prominent pathologic changes in the eye were detected in this structure, and we propose that in addition to a function related to retinal oxygen pressure control, this structure is vital for the immune function of the ocular tissues.
Macrophages colonize the retina in early developmental stages of teleost embryos and differentiate to microglial cells. 16 No obvious reaction was observed in the retina of the affected fish. This demonstrated that despite a massive MHC class II induction in the uveal tract, the retina may remain relatively unaffected, indicating an ocular immune privilege as seen in mammals. 40 However, previous investigations have shown that there is a retinal reaction to various cytokines and LPS in mammals, and that MHC class II expression may be induced in the Atlantic salmon after vaccination. 24 Because the retinal function is of vital importance to the animal, actions are taken to limit inflammatory responses as effectively as possible. 39 This is the probable explanation for the lack of MHC class II retinal induction in the present material.
In mice, it has been observed that leukocytes may infiltrate the optic nerve head in connection with LPS-induced uveitis. 27 Inflammatory cell aggregations as detected in the present study may consist of several cell populations including activated microglial cells, T cells, macrophages and their derivates, and MGCs, 25,26 all of which may be MHC class II+.
The uveal inflammatory reaction in the present material was severe compared with previous results from vaccinated and nonvaccinated fish studied in our laboratory. 21 Because MGCs and epithelioid cells were presently identified, the reaction can be characterized as a granulomatous inflammation.
Recurrent uveitis may lead to cataract and glaucoma, 26 and cataract is increasingly documented in fish populations. 3 The etiology is largely not clarified. The diagnosis of glaucoma has still not been applied in fish, but exophthalmus, frequently seen in diseased individuals, may well be a result of this condition. This study has shown that uveitis with extensive expression of MHC class II molecules may develop in Atlantic salmon, and it is likely that diseases that evolve from this condition also exist in fish.
In addition to this study, morphologic approaches to the distribution of MHC class II+ cells in fish have been published three times. 21,23,36 Increased knowledge of ocular immunology and immunopathology is warranted to determine the pathogenesis of fish eye diseases.
Footnotes
Acknowledgements
We thank Mrs. Laila Aune, Department of Basal Sciences and Aquatic Medicine, for excellent laboratory work. Bacteria and fungi were cultivated by Professor Henning S⊘rum, The Norwegian School of Veterinary Science, Oslo, and Dr. Berit Dj⊘nne, The National Veterinary Institute, Oslo. This study was supported financially by grants from Stiftelsen Thomas Fearnley, Heddy og Nils Astrup, Oslo.
