Abstract
Intranasally inoculated neurotropic influenza viruses in mice infect not only the respiratory tract but also the central nervous system (CNS), mainly the brain stem. 26 , 34 Previous studies suggested that the route of invasion of virus into the CNS was via the peripheral nervous system, especially the vagus nerve. To evaluate the transvagal transmission of the virus, we intranasally inoculated unilaterally vagectomized mice with a virulent influenza virus (strain 24a5b) and examined the distribution of the viral protein and genome by immunohistochemistry and in situ hybridization over time. An asymmetric distribution of viral antigens was observed between vagal (nodose) ganglia: viral antigen was detected in the vagal ganglion of the vagectomized side 2 days later than in the vagal ganglion of the intact side. The virus was apparently transported from the respiratory mucosa to the CNS directly and decussately via the vagus nerve and centrifugally to the vagal ganglion of the vagectomized side. The results of this study, thus, demonstrate that neurotropic influenza virus travels to the CNS mainly via the vagus nerve.
Keywords
Influenza is an acute disease of humans and animals caused by infection with an influenza A virus. Human influenza is commonly a rhinitis followed by a tracheobronchitis or an interstitial pneumonia; 5,31 however, encephalitis and encephalopathies associated with or etiologically related to influenza virus infection can also occur. 13 These encephalitis and encephalopathies include acute necrotizing encephalopathy of childhood (influenza-associated encephalopathy of childhood), 21,39 hemorrhagic shock and encephalopathy, 18 Reye's syndrome, 6,32 encephalitis lethargica (von Economo's encephalitis), 7,29,42 and postencephalitic Parkinson's disease. 4,9 Mice infected with influenza virus develop either pneumonia or progressive upper respiratory disease resembling that in adult humans, but neural infections can occur depending on the neuropathogenicity of the viral strain, mouse strain, and route of infection. 10,11,19,22,30,40 Intranasally inoculated virulent strains of influenza viruses caused neural infection, and viral antigens preferentially appeared in the brain stem, olfactory bulbs, and thoracic cord of mice. 26,34 In these reports, transneural spread, especially transvagal infection, from the respiratory tract to the central nervous system (CNS) was postulated. If the virus invades the CNS via the vagus nerve, vagectomy may protect the CNS from transneural infection. In this study, we inoculated unilaterally vagectomized mice with a virulent influenza virus by the intranasal route and examined the effects of vagectomy on the distribution of viral antigens, genomes, and histologic changes in the CNS and peripheral ganglia over time.
Materials and Methods
Virus strain
Avian influenza virus strain 24a5b has high pathogenicity in chicks (100% mortality for 3-day-old chicks by air-sac infection) and neuropathogenicity in mice on intranasal inoculation. 15,35,36 This strain does not cause any pathologic changes due to hematogenous viral spread in mice. 34 This virus was obtained from the strain A/Whistling swan/Shimane/499/83 (H5N3) by 24 serial passages in the air sacs of chicks and five passages in the brains of chicks.
Mice and vagectomy
Thirty-five, 8-week-old female BALB/cA Jcl mice (CLEA Japan Inc., Tokyo, Japan) were used. Twenty mice received unilateral (right) cervical vagectomies 5 days before viral inoculation. The mice were anesthetized by intraperitoneal injection of 0.3 ml of pentobarbital sodium per animal, an incision was made in the medial neck region, and approximately 3 mm of the right vagus nerve was removed under a dissection microscope. The incision was closed after it was confirmed microscopically that the resected tissue consisted of nerve bundles. Fifteen intact mice served as control animals.
Infection of mice with a virus strain
Five days after the surgery, all vagectomized and nonvagectomized control mice were inoculated in both nostrils with 5 µl of the virus (106 50% egg infectious doses/ml). The mice were observed every day and killed on days 2–6 postinoculation (pi) (four vagectomized and three control mice for each day).
Histology and immunohistochemistry
The liver, spleen, kidneys, heart, lungs, adrenal glands, pancreas, duodenum, trachea, thymus, brain (including olfactory bulbs), cranium (including nasal mucosa, pterygopalatine, and trigeminal ganglia), vertebrae (including spinal cord, dorsal root, and sympathetic trunk ganglia), and superior cervical and vagal (nodose) ganglia were collected and fixed in 10% phosphate-buffered formalin (pH 7.2). The cranium and vertebrae were decalcified in an 8% formic acid solution after formalin fixation. The organs were sectioned at 4 µm and stained with hematoxylin and eosin for light microscopy. For detection of avian influenza virus antigens in the tissues, all sections were stained by the streptavidin–biotin–immunoperoxidase complex method (Histofine SAB-PO kit, Nichirei Corp., Tokyo, Japan) using rabbit antistrain 499 (A/Whistling swan/Shimane/499/83 [H5N3]) hyperimmune serum at a 1 : 2,000 dilution as the primary antibody and counterstained with Mayer's hematoxylin.
In situ hybridization
To detect viral genomes and confirm viral infection, representative tissues (lungs, brain, spinal cord, and superior cervical and vagal ganglia) were subjected to in situ hybridization. Paraffin-embedded sections were dewaxed in xylene and rehydrated in graded ethanol solutions. After treatment with proteinase K (100 µg/ml) for 30 minutes at 37 C, the sections were fixed again with 4% paraformaldehyde for 10 minutes. Slides were transferred to 0.1 M triethanolamine and incubated in the same solution containing 0.25% acetic anhydride for 10 minutes at room temperature. Sections were washed with phosphate buffer (PB), rehydrated through graded ethanol solutions, and dried at room temperature. The sections were covered with 80 µl of hybridization mixture consisting of digoxigenin (DIG)-labeled riboprobe, 50% vol/vol formamide, 10 mM Tris-HCl (pH 7.6), 0.2 mg/ml transfer ribonucleic acid (Boehringer Mannheim, Indianapolis, IN), 1× Denhardt's solution, 100 mg/ml dextransulphate, 600 mM NaCl, 0.25% wt/vol sodium dodecyl sulfate, and 1 mM ethylenediaminetetraacetic acid (pH 8.0). The DIG-labeled riboprobes used were for the detection of nucleoprotein of A/Hong Kong/483/97 (HK483) (H5N1). 26 Homology between the riboprobes and nucleoprotein of 24a5b was 88.3%. The slides were then covered with coverslips and incubated at 42 C for 16 hours in a humid chamber. After hybridization, the coverslips were carefully removed in 5× sodium citrate–sodium chloride (SCC) at 42 C. The slides were washed once at 42 C with 2× SCC and 50% formamide for 30 minutes, twice with 0.2× SCC for 20 minutes at 42 C, and finally, once in 0.2× SCC at room temperature. For detection of hybridization, sections were incubated with anti-DIG antibodies conjugated with alkaline phosphatase (Boehringer Mannheim) diluted 1 in 500 in 1× blocking reagent. After three washes in PB, substrate consisting of nitroblue tetrazolium and 5-bromocresyl-3-indolylpholphate was layered over the sections. Tissues were counterstained with methyl green.
Results
Clinical signs and macroscopic findings
Mice showed a slight decrease in body weight and ruffled hair after the vagectomy, but they had recovered by the day of viral inoculation. The mice infected with the virus showed anorexia, emaciation, and ruffled hair from day 3 pi and distention of the abdomen on day 6 pi, but prominent neurologic signs did not appear during the experimental period. At necropsy, hemorrhagic foci in the lungs and retention of gas in the stomach and intestines were observed on days 4 and 6 pi, respectively.
Histologic and immunohistochemical findings
Histologic lesions were observed in the respiratory tract, peripheral ganglia, brain stem, and thoracic spinal cord. In the respiratory tract, mild rhinitis, tracheobronchitis, and bronchointerstitial pneumonia were observed on day 2 pi and later became more prominent. Lesions included desquamation of the mucosal epithelial cells; fibrinous exudate; infiltration of neutrophils, macrophages, and lymphocytes in the tracheobronchial walls and pulmonary interstitium; and enlargement of the alveolar epithelial cells. Immunohistochemical examination revealed viral antigens in the nasal, tracheobronchial, and bronchiolar mucosae and in the alveolar epithelium from day 2 pi (Fig. 1). Positive staining appeared mainly in the nucleus of infected cells, but their cytoplasm sometimes stained positively. There were no significant differences in the extent of the respiratory lesions and in the distribution of viral antigens between vagectomized and control mice. Except for splenic follicular hyperplasia in both groups, histologic and immunohistochemical abnormalities were not found in other visceral organs.
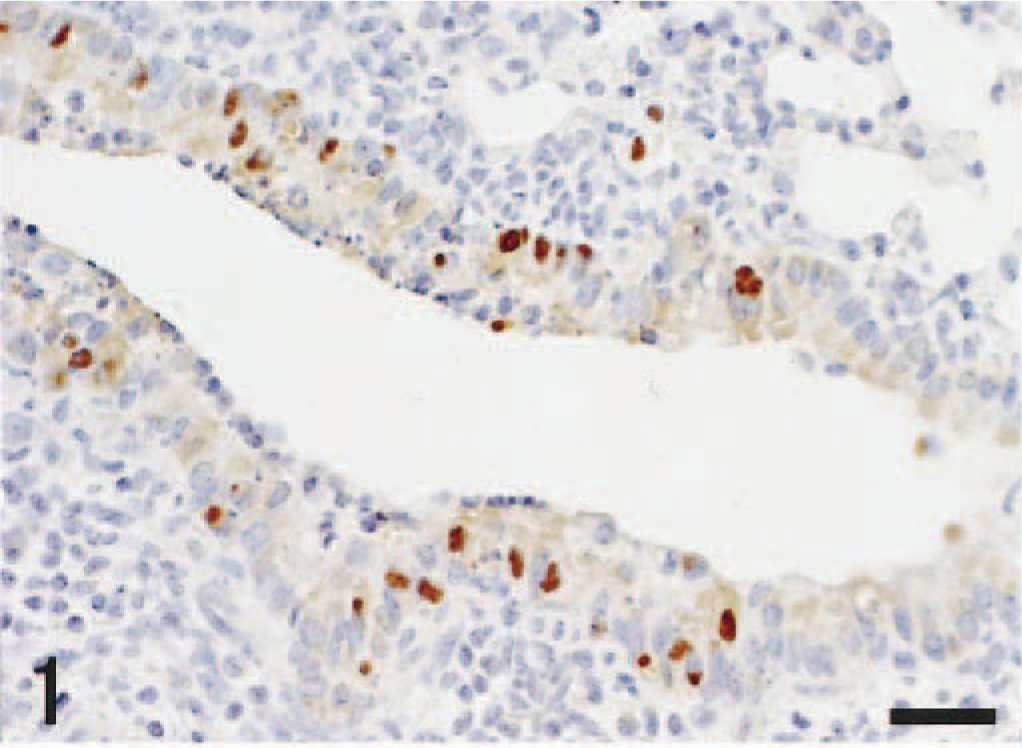
Lung; mouse, unilaterally vagectomized and infected with influenza A virus strain 24a5b, day 3 pi. Most of viral antigens are located in the nuclei of the bronchiolar epithelial cells. Immunohistochemistry by the streptavidin–biotin–immunoperoxidase complex method. Mayer's hematoxylin counterstain. Bar = 25 µm.
In the CNS, histologic changes in both groups were confined to the brain stem and thoracic spinal cord. In the brain stem, histologic lesions were observed from day 5 pi; lesions appeared mainly in the nucleus of the solitary tract (NTS) and nucleus ambiguus (NA) (Fig. 2). They comprised nuclear pyknosis of oligodendrocytes and increased numbers of microglial cells. More advanced lesions included perivascular cuffing of mononuclear cells, necrosis of nerve cells, and neuronophagia. Histologic lesions were always associated with the presence of viral antigens in the nucleus and sometimes in the cytoplasm of nerve and glial cells (Fig. 3). A slight asymmetry of lesion severity and size was observed in each mouse. Spinal lesions were restricted to the gray matter of the thoracic cord in vagectomized mice on day 6 pi. The peripheral ganglia such as the pterygopalatine, vagal (nodose), trigeminal, superior cervical ganglia, and sympathetic trunk ganglia of thoracic area showed minimal histologic changes and contained viral antigens in the nucleus and cytoplasm of gangliocytes. The localization of viral antigens in the nasal mucosa, lungs, and neural tissues of nonvagectomized and vagectomized mice is summarized in Tables 1,2. In nonvagectomized mice, the viral antigen distribution was almost symmetric in each ganglion. In vagectomized mice, the viral antigens appeared in the vagal ganglion of the nonvagectomized side (left) from day 3 pi (Fig. 4), whereas the antigens never appeared until day 5 pi on the vagectomized side (Fig. 5). This type of asymmetry was not apparent in other peripheral ganglia. Numbers of antigen-positive cells were small to moderate in the pterygopalatine, vagal, and trigeminal ganglia and large in the superior cervical and the sympathetic trunk ganglia.
Distribution of influenza virus antigens with days in nonvagectomized mice.∗

∗ PPG = pterygopalatine ganglion; VG = vagal (nodose) ganglion; V = trigeminal ganglion; SCG = superior cervical ganglion; STG = sympathetic trunk ganglion; DRG = dorsal root ganglion of spinal cord.
† Numbers under the columns Nasal Mucosa to Brain Stem represent heads of mice immunohistochemically positive for the viral antigen.
‡ Left/right side
Distribution of influenza virus antigens with days in vagectomized mice.∗

∗ PPG = pterygopalatine ganglion; VG = vagal (nodose) ganglion; V = trigeminal ganglion; SCG = superior cervical ganglion; STG = sympathetic trunk ganglion; DRG = dorsal root ganglion of spinal cord.
† Numbers under the columns Nasal Mucosa to Brain Stem represent heads of mice immunohistochemically positive for the viral antigen.
‡ Left nonvagectomized/right vagectomized side
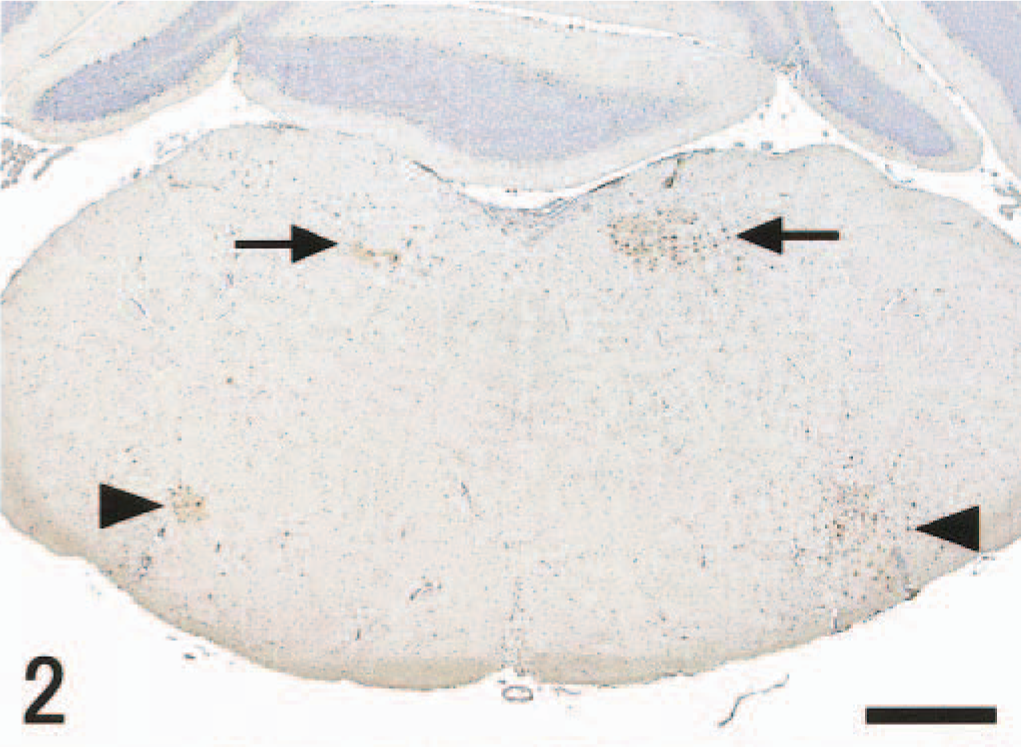
Brain stem; mouse, unilaterally vagectomized and infected with influenza A virus strain 24a5b, day 5 pi. Viral antigens are detected bilaterally in and around the NTS (arrows) and NA (arrow heads). Immunohistochemistry by the streptavidin–biotin–immunoperoxidase complex method. Mayer's hematoxylin counterstain. Bar = 500 µm.
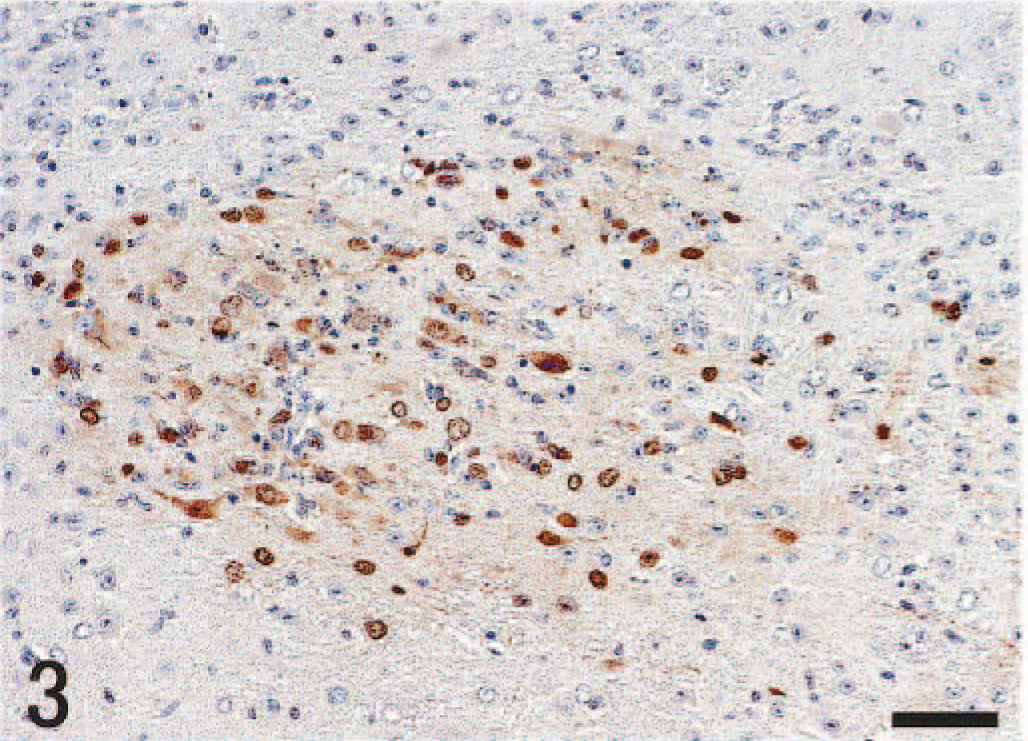
NTS, brain stem; mouse, unilaterally vagectomized and infected with influenza A virus strain 24a5b, day 5 pi. Higher magnification of the region indicated by the right arrow in Fig. 2. The nuclei of neurons and glial cells and, sometimes, the cytoplasm are positive for the viral antigen. Immunohistochemistry by the streptavidin–biotin–immunoperoxidase complex method. Mayer's hematoxylin counterstain. Bar = 50 µm.
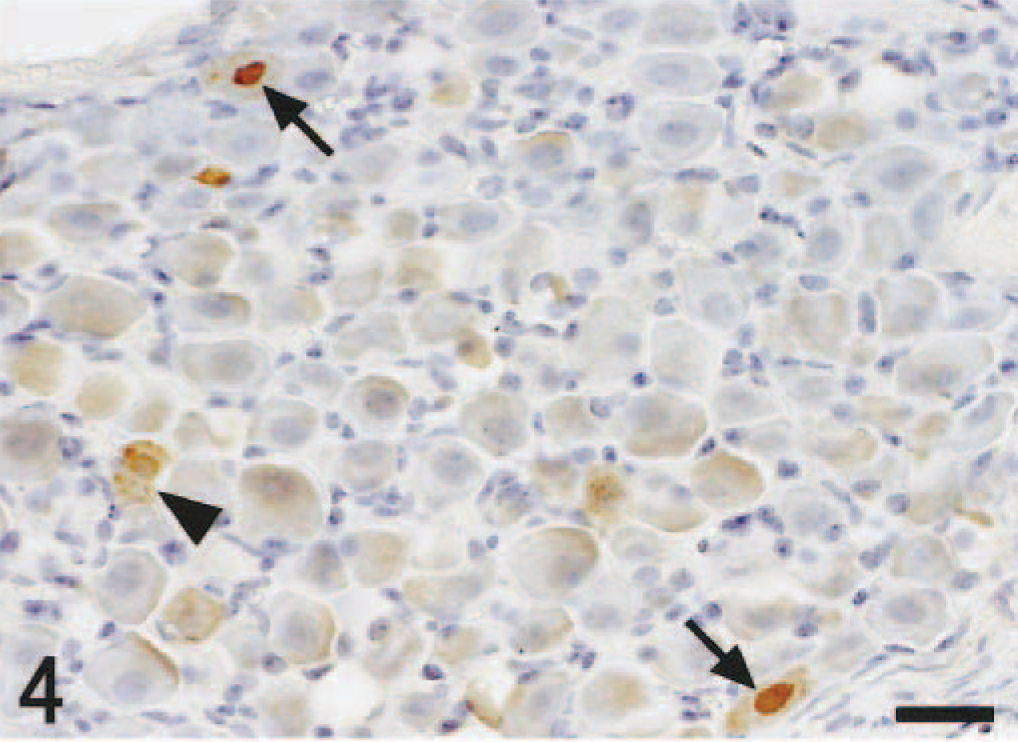
Vagal (nodose) ganglion of nonvagectomized side; mouse, unilaterally vagectomized and infected with influenza A virus strain 24a5b, day 3 pi. Viral antigens in the nuclei (arrows) and cytoplasm (arrowhead) of gangliocytes. Immunohistochemistry by the streptavidin–biotin–immunoperoxidase complex method. Mayer's hematoxylin counterstain. Bar = 25 µm.
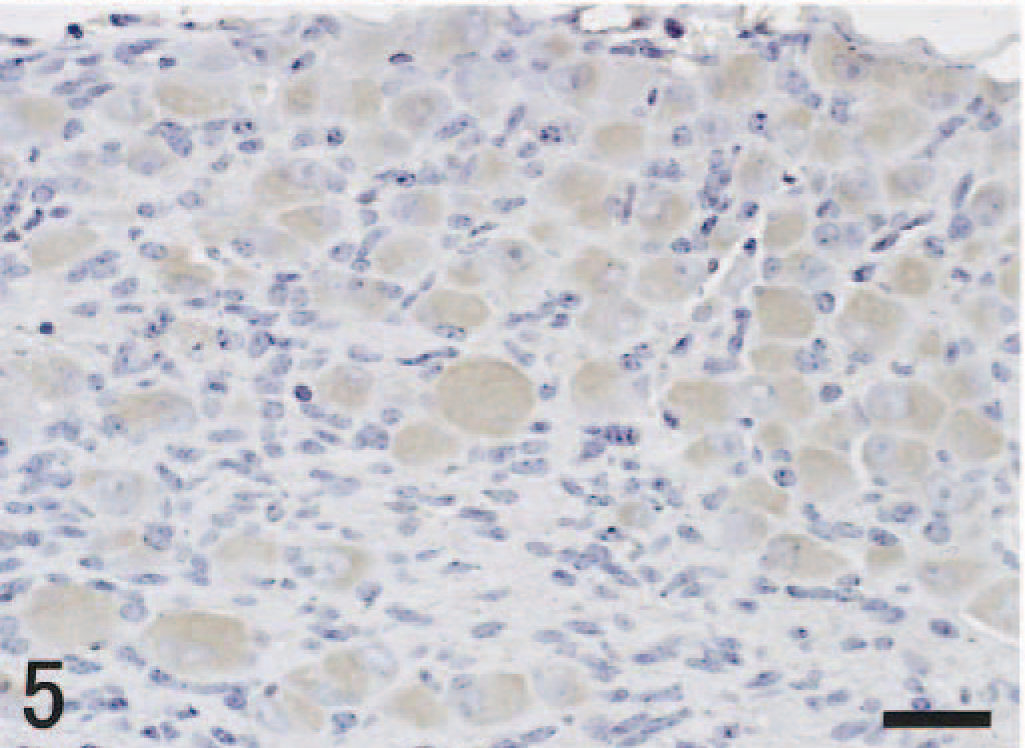
Vagal (nodose) ganglion of vagectomized side; mouse, unilaterally vagectomized and infected with influenza A virus strain 24a5b, day 3 pi. There are no antigen-positive cells. Immunohistochemistry by the streptavidin–biotin–immunoperoxidase complex method. Mayer's hematoxylin counterstain. Bar = 25 µm.
In situ hybridization
Hybridization signals were detected in the NTS and thoracic spinal cord as seen with immunohistochemistry (Fig. 6). Hybridization signal was located within the nuclei of neurons and glial cells.
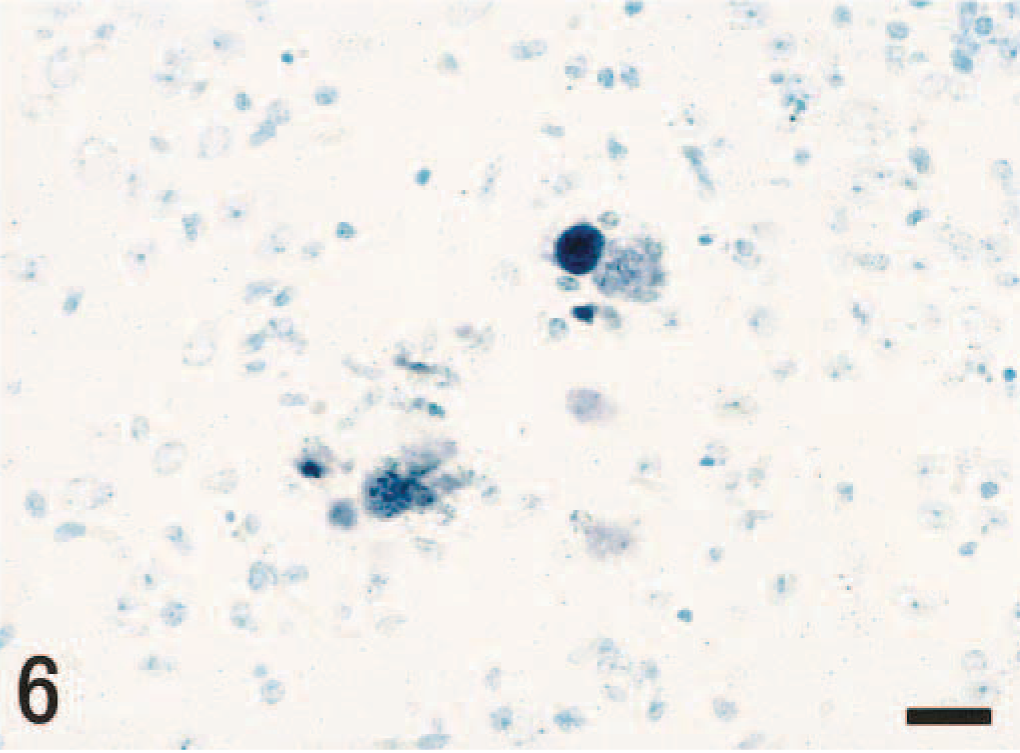
NTS, brain stem; mouse, unilaterally vagectomized and infected with influenza A virus strain 24a5b, day 5 pi. The same region depicted in Fig. 2. Viral RNA is detected in the nuclei of neurons and glial cells. In situ hybridization. Methyl green counterstain. Bar = 25 µm.
Discussion
The distribution of virus in control mice (Table 1) was consistent with those reported in the studies by Shinya et al. (strain 24a5b) 34 and Park et al. (strain HK483), 26 in which transneural invasion of the virus into the CNS was postulated. The viral distribution in the CNS of the vagectomized mice (Table 2) was similar to that in nonvagectomized control mice (Table 1), but infections appeared earlier and more frequently, probably because of stress caused by the surgical treatment. In unilaterally vagectomized mice, the most prominent asymmetric distribution of the viral antigens was observed in vagal (nodose) ganglia. In the left vagal ganglion on the intact side, viral antigens were detected from day 3 pi, whereas viral antigens appeared in the right ganglion from day 5 pi. The most probable explanation for this difference is that, on days 3 and 4 pi, virus was unable to spread to the vagal ganglion of the neurectomized side from the respiratory mucosa because of a discontinuity of the neural pathway. This indicates that the virus spreads from the respiratory tract to the vagal ganglia through the vagus nerves.
The main regions where the viral antigens were detected in the brain stem were the NTS and NA, which along with the dorsal motor nucleus of the vagus nerve (DMV) comprise the vagal medullary centers. The NTS is innervated by the afferent processes of sensory neurons in the vagal ganglion and receives sensory information from respiratory mechano- and chemoreceptors. 12,16,27 The efferent fibers arising from motoneurons in the NA and DMV run with the vagal sensory nerve fibers and provide innervation to airway smooth muscle, glands, and blood vessels. Interconnections between the vagal medullary centers and a single decussation of the vagus nerves inside the thorax have been demonstrated. 27 Considering these neuroanatomic interconnections, virus replicated in the lungs and lower respiratory airways could have spread through intact vagal nerves to the NTS via the vagal ganglion and to the NA directly (Fig. 7, bold lines). Interconnections between the NTS and NA of both sides might have provided another route for viral spread, thus resulting in bilateral effects on the nuclei (Fig. 7, dotted lines). It took 2 days for the virus to reach the vagal ganglion of the vagectomized side from the NTS of the opposite, nonvagectomized side (Fig. 7, broken lines). Pterygopalatine ganglia connected with the salivary and lachrymal glands by parasympathetic nerves and trigeminal ganglia connected with nasal mucosa by sensory nerves were also infected, but their nuclei in the brain were not usually affected. These results indicate that the trigeminal and facial nerves are also possible routes for viral transport to the CNS but are not preferentially used. The spread of the influenza virus to the thoracic spinal cord via sympathetic trunk nerves 26 was also confirmed in the present experiment; sympathetic transport occurred later than the vagal transport.
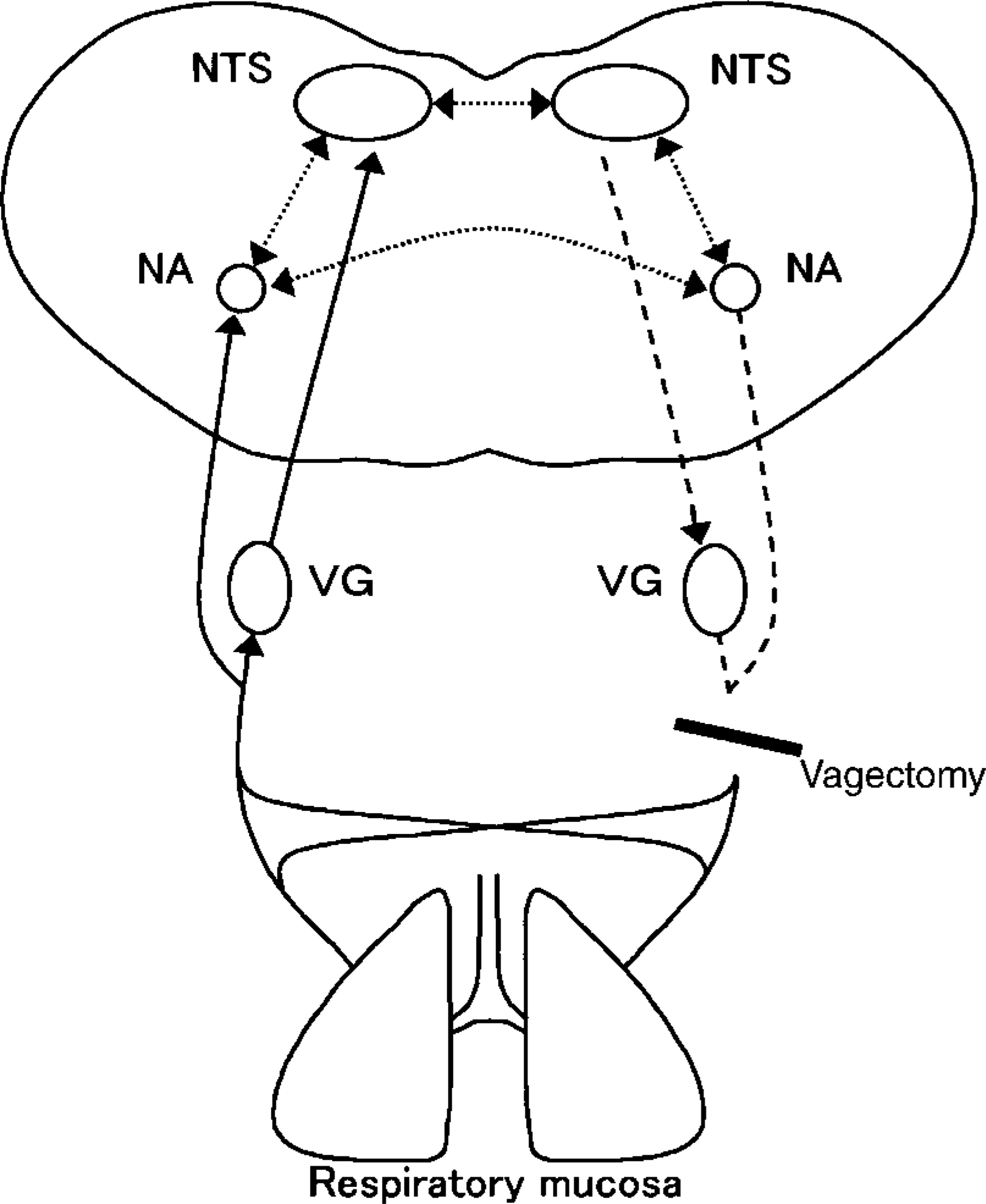
Diagram of possible viral transmission from the respiratory mucosa to the brain stem through the vagus nerves. Bold, dotted, and broken lines indicate afferent pathways to VG, NTS, and NA; pathways between vagal medullary centers; and an efferent pathway to VG of the vagectomized side, respectively. VG, vagal (nodose) ganglion; NTS, nucleus of the solitary tract; NA, nucleus ambiguus.
Neurotropic influenza virus strain 24a5b used in this study and in the report by Shinya et al. 34 was obtained from water fowl and was serially passaged in the air sac and brain of chicks, 15,35,36 whereas strain HK483 was isolated from the throat of a human patient with fatal influenza infection in Hong Kong in 1997. 26 These two different H5 neurotropic influenza viruses showed similar transneural spread from the respiratory mucosa to the CNS. Transvagal transport has also been reported for certain neurotropic enteroviruses, 23,41 reovirus serotype 3, 23,24 pseudorabies virus, 3 and hemagglutinating encephalomyelitis virus, 1 but the details of viral transport through vagas nerve are not known. We introduced the same virus strain used in this study into several peripheral sites in BALB/c mice including anterior chamber of the eye, brachial plexus, articular cavity of the knee joint, sciatic nerve, and hindlimb footpad, but none of the inoculations resulted in a transneural spread of virus to the CNS (K. Matsuda et al., unpublished). The results contrast markedly with those for pseudorabies virus, in which the virus spreads through both somatic and autonomic nerves. 8
The spread of neurotropic viruses from the periphery to the CNS depends on microtubule-associated fast axonal transport in a retrograde or anterograde direction (or both). 20,25,37,38,41 The molecular mechanisms for the transport have been clarified in some viruses: rabies virus and certain adenoviruses can be transported using dynein, a microtubule-associated motor protein, 17,28 and reovirus can bind directly to isolated microtubules. 2 Schwann cells have been recently demonstrated to be another pathway for neural spread in herpes simplex virus infections. 33 In myelinated nerve fibers, the myelin sheath surrounding the axon has been reported to act as a physical barrier preventing the passage of virus from the surrounding endoneurial tissue into the axon. 14 Somatic nerves are myelinated, and autonomic nerves have unmyelinated axons. The absence of a myelin sheath in autonomic nerves helps virus to penetrate the axon, which may account for the preferential transmission of neurotropic influenza virus through autonomic nerves (vagas and sympathetic nerves) in this study.
Clinical features of patients in the H5N1 influenza epidemic in Hong Kong in 1997 included gastrointestinal manifestations (abdominal pain, vomiting, and diarrhea), in addition to respiratory disease, without definitive evidence of viral replication in organs other than the respiratory tract. 43 In this study and in previous H5 influenza studies in mice 26,34 and ferrets, 44 some infected animals showed gastrointestinal manifestations such as abdominal distention, due to retention of gas in the alimentary tract, and diarrhea. Therefore, clinical signs in the alimentary tracts may be due to abnormal peristalsis caused by influenza virus infection of intra- and extramural autonomic nerves and their centers in the brain stem and thoracic cord.
