Abstract
This report describes 2 genetically related paint mares, case Nos. 1 and 2, presented to the Oklahoma State University Boren Veterinary Medical Teaching Hospital for chronic weight loss and abnormal gait, respectively. Notable findings in both cases included marked persistent eosinophilia and multiple intramuscular lateral thoracic masses. Histologic examination of masses revealed eosinophilic, centrally necrotic granulomas and marked eosinophilic myositis. Granulomas in case No. 1 also contained intralesional Sarcocystis sp material, and adjacent muscle fibers contained intact protozoal cysts. Case No. 1 developed severe refractory muscle pain and recurrent esophageal dysphagia. At necropsy, disseminated, grossly visible granulomas were present throughout all examined striated muscles. Nested polymerase chain reaction of the 18S rRNA gene revealed >99% homology with Sarcocystis fayeri. Sarcocystis spp are apicomplexan protozoa that infect striated muscle of many omnivorous species, typically without inciting clinical disease. Sarcocystosis should be considered a rare cause of granulomatous eosinophilic myositis and choke in horses.
Sarcocystis spp are highly prevalent apicomplexan protozoa that infect the striated muscle of many domestic animal species, typically without significant clinical disease. The prevalence of Sarcocystis sp infection in equids has a wide range, from 0.5% to 21% in 1 US study 3 and 15% to 23% in German studies. 6 A prospective study performed at the Oklahoma Animal Disease Diagnostic Laboratory determined that 11 of 22 (50%) pasture-housed horses aged >1 year of age that presented for necropsy examination between February and June 2014 contained Sarcocystis sp cysts within the tongue and/or esophagus, without associated clinical or microscopic disease (H. R. Herd, unpublished data).The usually nonpathogenic Sarcocystis fayeri is the only reported intramuscular Sarcocystis sp in North American equids; Sarcocystis bertrami is the reported species infecting striated muscle of horses and donkeys in Europe. 8 Equids are the intermediate host for S. fayeri and S. bertrami (Sarcocystis equicanis). Canids are considered the definitive host for both. 2 Infected horses typically do not exhibit any clinical signs; however, rarely reported signs are limited to mild acute fever, mild to severe muscle soreness, dysphagia, and weight loss. 1 Clinicopathologic findings range from acute mild anemia to chronic mild to severe anemia. 1 This report describes the investigation of disseminated eosinophilic and granulomatous myositis caused by S. fayeri infection in 2 genetically related horses.
Case No. 1, a 3-year-old paint mare, presented to the Oklahoma State University Boren Veterinary Medical Teaching Hospital with a 3- to 4-month history of weight loss. Physical examination revealed tachycardia (average, 60 beats per minute; reference, 28–44 beats per minute); multiple immobile, firm, irregularly shaped intramuscular masses on the lateral thorax; and intermittent muscle fasciculations. The most significant finding from serial routine blood work was a marked, persistent eosinophilia (2210–4950 µl; reference interval, 0–1000 µl). Muscle biopsies of thoracic masses revealed multiple linear, poorly defined granulomas, surrounded by eosinophilic to lymphoplasmacytic myositis, edema, and muscle fiber degeneration and loss (Fig. 1). Granulomas rarely contained intralesional pale eosinophilic outlines of zoites and cyst wall–like material (Fig. 2). Within less severely affected regions, muscle fibers occasionally contained intact immature, protozoal cysts consistent with Sarcocystis sp. Initial differentials for eosinophilic myositis included multisystemic epitheliotropic eosinophilic syndrome, aberrant parasite migration of a larval nematode, and sarcocystosis. Case No. 1 was treated with anthelmintics (moxidectin/praziquantel, 0.4 mg/kg, oral) and antiprotozoals (sulfamethoxazole and trimethoprim, 20 mg/kg, oral, every 24 hours), without incident. Aggressive anti-inflammatory and pain management was administered over a 7-month period without resolution of clinical signs of generalized pain (muscle fasciculations, tachypnea, and tachycardia). The horse then developed recurrent unmanageable esophageal dysphagia and spontaneous reflux and was euthanized at the owner’s request.
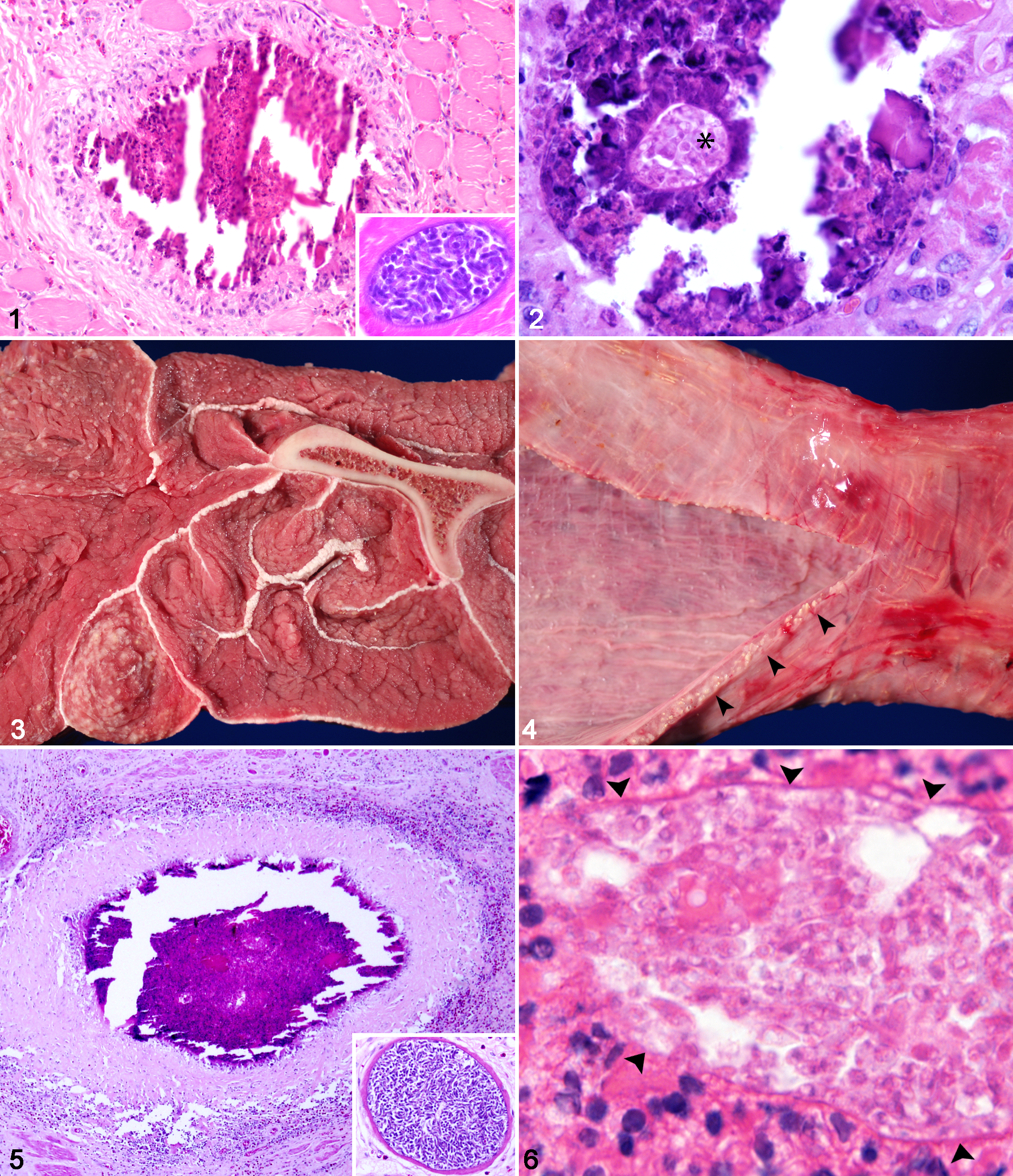
Horse, Sarcocystis fayeri infection.
A complete necropsy with histopathologic examination was performed on case No. 1. All examined striated muscles contained innumerable, disseminated, pale tan, rod-shaped, firm nodules (0.7 × 0.1 cm). Particularly affected were the muscles of the esophagus and diaphragm, large shoulder and thigh muscles, and intercostal muscles (Fig. 3). Severely affected muscles were atrophied and mottled by the pallor of fibrosis. The esophagus was intermittently dilated and flaccid and regionally contracted by pale tan fibrous connective tissue. The mucosal and adventitial surfaces of the esophagus were mottled by hemorrhage and innumerable pale tan foci (Fig. 4). Tissue samples obtained at necropsy were fixed in 10% neutral buffered formalin, paraffin embedded, sectioned at 5 µm, and routinely stained with hematoxylin and eosin. The grossly visible pale tan foci were confirmed as eosinophilic granulomas with central necrosis, similar to, but more chronic than, those seen in the antemortem biopsies, as evidenced by the thick band of surrounding fibrous connective tissue (Fig. 5). Granulomas were composed of a core of hypereosinophilic amorphous material admixed with pyknotic and karyorrhectic cellular debris, deeply basophilic mineral, and, occasionally, intralesional pale eosinophilic outlines of zoites and cyst wall–like material (Fig. 6). Eosinophilic cores were surrounded by macrophages, multinucleate giant cells, and eosinophils and were further surrounded by thick bands of fibrous connective tissue. Surrounding myocytes were variably separated, surrounded, and replaced by abundant expansion of perimysial fibrous connective tissue and infiltrating eosinophils. Remaining myofibers were irregularly atrophied and hypereosinophilic and contained fragmented sarcoplasm and pyknotic nuclei. Randomly scattered within individual myofibers were numerous mature protozoal cysts (100- to 200-µm diameter) that contained abundant crescent-shaped bradyzoites (4–6 µm), consistent with Sarcocystis sp.
Immunohistochemistry was performed on skeletal muscle from case No. 1 for Sarcocystis neurona—the causative agent of equine protozoal myeloencephalitis, to rule out an atypical manifestation—and was negative. Immunohistochemistry for smooth muscle actin confirmed that intact sarcocysts were present within skeletal muscle but not smooth muscle of the distal esophagus.
Approximately 1 year after the index case, case No. 2, a 5-year-old paint mare from the same farm and sired by the same stallion presented to the same teaching hospital with several years’ history of abnormal gait. Similar to case No. 1, serial blood work revealed a marked persistent eosinophilia (2907 to 5350 µl, reference interval: 0–1000 µl), and physical examination identified multiple immobile, firm thoracic masses, which largely mirrored the biopsy findings of case No. 1; a notable exception was lack of intact S. fayeri cysts. Case No. 2 was treated with an antiprotozoal (ponazuril, 10 mg/kg, oral, every 24 hours) for 28 days and responded well with improvement of clinical signs; however, marked eosinophilia and abnormal gait persisted for more than 10 months.
Polymerase chain reaction was performed on tissue from both cases and from 1 control horse, and it included sections of tongue and esophagus obtained from case No. 1 at necropsy, a wedge muscle biopsy from the lateral thorax of case No. 2, and tongue obtained from an unrelated control horse that was presented for necropsy with incidental microscopically visible, intact sarcocysts. DNA extraction and nested polymerase chain reaction to amplify 18S rRNA gene were performed as previously described. 7 Sequencing yielded a 532–base pair segment of the 18S rRNA gene from case Nos. 1 and 2 and the unrelated control horse. These sequences were >99% identical to one another and shared >99% similarity with a S. fayeri (AB661438) sequence obtained from a horse in Japan. Sarcocystis hirsuta (KC209741) was the next-highest match, at a 93% homology.
Intact Sarcocystis sp cysts within biopsy specimens obtained early in the disease course of case No. 1 contained immature sarcocysts lined by ill-defined metrocytes, in contrast to mature Sarcocystis sp cysts in later specimens obtained at necropsy that were characterized by well-defined, crescent-shaped bradyzoites. Similarly, granulomas from the biopsy of case No. 1 were smaller and appeared less chronic than those identified in necropsy specimens, as evidenced by the lack of a thick band of fibrous connective tissue. This apparent synchronous aging of the cysts and granulomas suggests that the formation of granulomas and development of persistent eosinophilia were in response to the sarcocysts. The presence of degenerate sarcocyst material within multiple granulomas and polymerase chain reaction confirmation support a primary causative role of S. fayeri in these 2 cases; however, absolute certainty cannot be ensured. To our knowledge, only 1 case of multifocal myositis associated with and suggested to be caused by S. fayeri in a horse has been previously reported. 9 However, in that case report, discrete granulomas and eosinophilia were not reported.
A similar disease, known as eosinophilic myositis, is described in cattle and sheep and is usually found incidentally at carcass inspection. 8 Eosinophilic myositis is classically described as well-demarcated areas of gray to green discoloration within skeletal muscle; however, rare cases are granulomatous, characterized by single or multiple pale tan discrete granulomas within skeletal muscle. Sarcocystis cruzi is a probable cause of eosinophilic myositis in cattle. The rarity of clinical disease in equids is mirrored in cattle, in which 100% of animals are infected by Sarcocystis sp, and yet only 5% have grossly evident eosinophilic myositis at slaughter. 4
A type 1 hypersensitivity is thought to play a role in cattle with S. cruzi–associated eosinophilic myositis based on increased IgE serum levels in affected animals; however, elevated parasite-specific IgE levels are also found in parasitized animals without myositis. 5 Additional theories for potential pathogenesis, which have been considered, include ingestion of large doses of sporozoites or infection by a more pathogenic strain of Sarcocystis sp. The familial relationship of the currently reported cases may suggest a genetic basis. S. fayeri should be included as a differential for eosinophilic and granulomatous myositis in horses with muscle pain and esophageal dysphagia.
Footnotes
Acknowledgements
We thank the Oklahoma State University Department of Veterinary Pathobiology for funding this work, the Krull-Ewing endowment and the National Center for Veterinary Parasitology (![]() ) for supporting molecular identification of the organisms, and the staff and pathologists of Oklahoma Animal Disease Diagnostic Laboratory for their assistance in collecting and processing samples.
) for supporting molecular identification of the organisms, and the staff and pathologists of Oklahoma Animal Disease Diagnostic Laboratory for their assistance in collecting and processing samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
