Abstract
This study defined the best site for quantifying osteoclasts in male Lewis rats with mycobacteria-induced adjuvant arthritis. Hind paw sections of normal and arthritic rats (
Bone growth, maintenance, and repair during normal remodeling and skeletal disease are mediated by the reciprocal activities of osteoblasts and osteoclasts. Osteoblasts, which originate from mesenchymal stem cells in bone marrow, produce bone matrix and express receptors for most bone-regulating molecules—regardless of whether the predominant action is to drive bone formation or bone resorption. 13 Osteoclasts are derived from phagocytic precursors of the monocyte/macrophage lineage and degrade bone matrix. Normal remodeling is tightly controlled by way of cell-to-cell communication between osteoclasts and osteoblasts, with osteoblasts both supporting and regulating osteoclast activity. 7 However, in pathologic states “activated” cells (e.g., infiltrating leukocytes and synovial fibroblasts) contribute other molecules that shift the equilibrium between osteoblastic and osteoclastic activities. 8,10,14 These molecules act by way of a common pathway to enhance osteoclast differentiation, activity, and survival (reviewed in Bolon et al. 2 ), thus contributing significantly to bone loss in such human conditions as osteoporosis and rheumatoid arthritis.
Animal models of many skeletal diseases also are characterized by increased numbers of osteoclasts. Therefore, assessment of osteoclast populations in animal bones is an important step in understanding the pathogenesis of skeletal diseases and in developing new bone-sparing therapies. For some purposes, an exact count of osteoclasts is made using labor- and time-intensive histomorphometric methods. 4,9,11 In other instances, intralesional osteoclasts may be assessed using simple, rapid, semiquantitative scoring systems. 1,3 The objective of this retrospective study was to define the optimal site for rapid quantification of intralesional osteoclasts in the hind paws of arthritic rats. For this purpose, we used rats in which adjuvant-induced arthritis (AdA) was initiated with an inoculation of mycobacteria. This model yields a severe immune-mediated, jointeroding disease that resembles rheumatoid arthritis.
Materials and Methods
Experimental design
This study was performed with archival tissues from two control groups from an AdA study with rats. We selected this model because animals develop extensive inflammation and localized bone erosions in conjunction with profound osteoclast production within 2 weeks of adjuvant inoculation. Rats from one cohort were normal (no arthritis), whereas rats from the second had arthritis but were not treated. The extent and severity of lesions were assessed independently in the same sections using both semiquantitative and quantitative methods.
Arthritis model
Young adult, male Lewis rats (Charles River, Wilmington, MA) weighing 180–200 g had been acclimated for 1 week and then randomly assigned to either the normal control or the untreated arthritic control group (
Tissue acquisition and processing
At necropsy, all animals had been anesthetized with carbon dioxide, and hind paws had been removed and fixed for 2 days by immersion in zinc formalin (Z-fix; Anatech Ltd., Battle Creek, MI). Paws subsequently were decalcified in eight serial changes of a 1 : 1 mixture of 8 N formic acid and 1 N sodium formate, divided longitudinally along the median axis, and processed into paraffin.
A 4-µm-thick section was stained to demonstrate osteoclasts using an indirect immunoperoxidase method and a proprietary rabbit anti-human monoclonal antibody (Amgen, Thousand Oaks, CA) directed against the osteoclast marker cathepsin K, which is an osteoclast protease that bears primary responsibility for resorption of bone. The procedure was performed at room temperature on an automated tissue stainer (Model Mark 5; DPC, Flanders, NJ). When needed, each section was washed in phosphate-buffered saline (1 ml). Deparaffinized sections were pretreated for 15 minutes with trypsin (0.1%, in 1% calcium chloride; Sigma, St. Louis, MO) and then covered for 10 minutes with CAS Block (Zymed Laboratories, San Francisco, CA). Sections were incubated with the primary antibody (1 µg/ml) for 60 minutes and with the biotinylated goat anti-rabbit polyclonal secondary antibody (Vector, Burlingame, CA; used at 1 : 200) for 25 minutes. Next, peroxidase-blocking solution (DAKO Corporation, Carpenteria, CA) was applied for 25 minutes, after which the indirect immunoperoxidase bridge was developed for 25 minutes using a commercial kit incorporating avidin–biotin complex and peroxidase (ABC Elite kit; Vector). Reaction sites were observed using diaminobenzidine (DAB+ Substrate/Chromagen System; DAKO) for 3 minutes and counterstained for 1 minute each with hematoxylin and eosin (HE).
Histopathologic and histomorphometric assessments
Paw sections first were evaluated using tiered, semiquantitative scoring criteria (Table 1) to grade bone erosions and intralesional osteoclasts (visible as cathepsin K–labeled [brown], multinucleated cells) in bones throughout the tibiotarsal and intertarsal joints (Fig. 1). Subsequently, joint integrity was evaluated in a quantitative fashion by measuring bone area (an index of erosion), osteoclasts, and osteoblasts (Table 2) at three specific sites: distal tibia (Fig. 2), navicular tarsal (Fig. 3), and calcaneus (Fig. 4). These three bones were selected because they are affected most severely in rats with AdA when joint structure is examined 7 days after the onset of clinical disease. Quantitative assessments were made using the 20× objective of a Nikon Eclipse E400 microscope and commercial bone histomorphometry software (Osteomeasure, version 4.0; Osteometrics, Decatur, GA). In the tibia (Fig. 2) and calcaneus (Fig. 4), the analysis was performed in two 0.36-mm2 (0.6 × 0.6 mm) fields oriented parallel to but not encompassing the growth plate cartilage. In the navicular tarsal (Fig. 3), the analysis was conducted by imaging a 1.08-mm2 (0.6 × 1.8 mm) rectangle (comprised of three 0.36-mm2 fields) that included 80–90% of the total tarsal marrow area. Osteoclasts (i.e., cathepsin K–positive cells) were enumerated in two ways, those in contact with bone and those in marrow. Total osteoclast numbers were calculated as the sum of the bone and marrow subpopulations.
Histopathologic criteria for arthritis lesion scores.∗

∗ Criteria published previously in Bolon et al. 1
Definitions for quantitative counts of joint integrity.
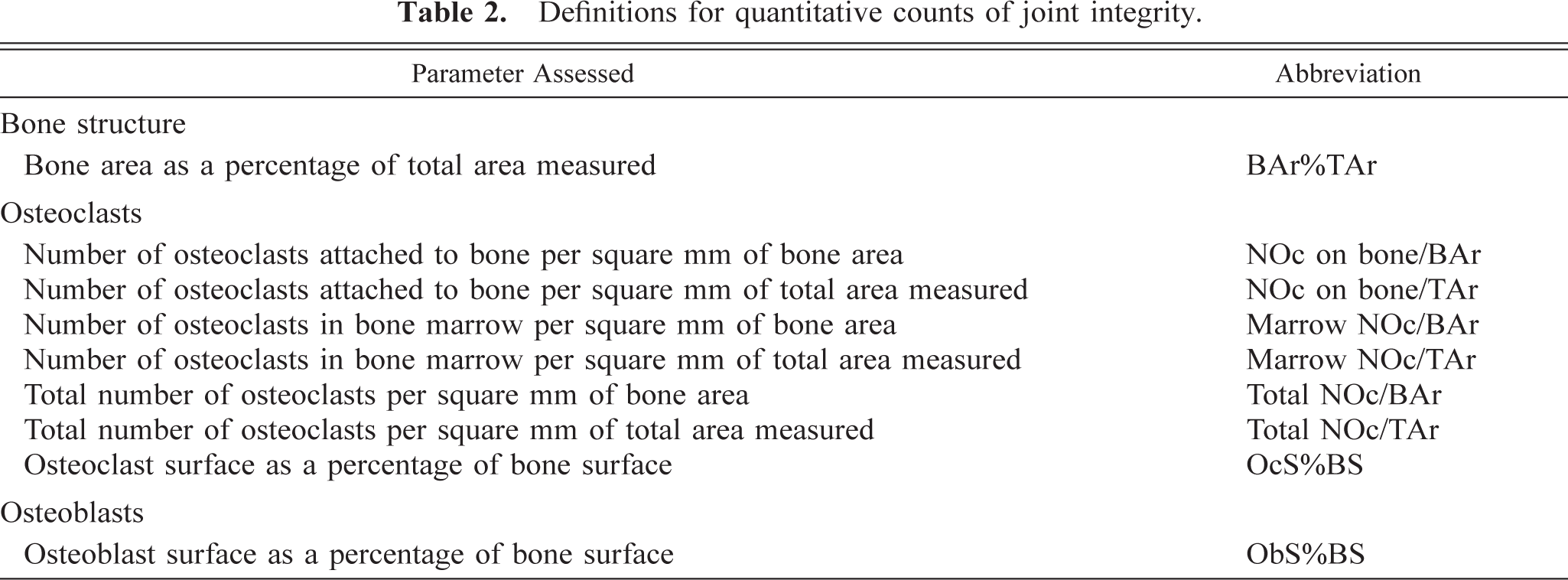
Tibiotarsal region of the hind paw (midaxial orientation); normal adult rat. Schematic diagram showing major anatomic features, including the three bones in which the quantitative parameters were measured: calcaneus (C), navicular tarsal (N), and tibia (T). The semiquantitative scores for erosions and osteoclasts represent composite values obtained for these three bones and another tarsal, the talus (Ta). Bar = 1 mm.
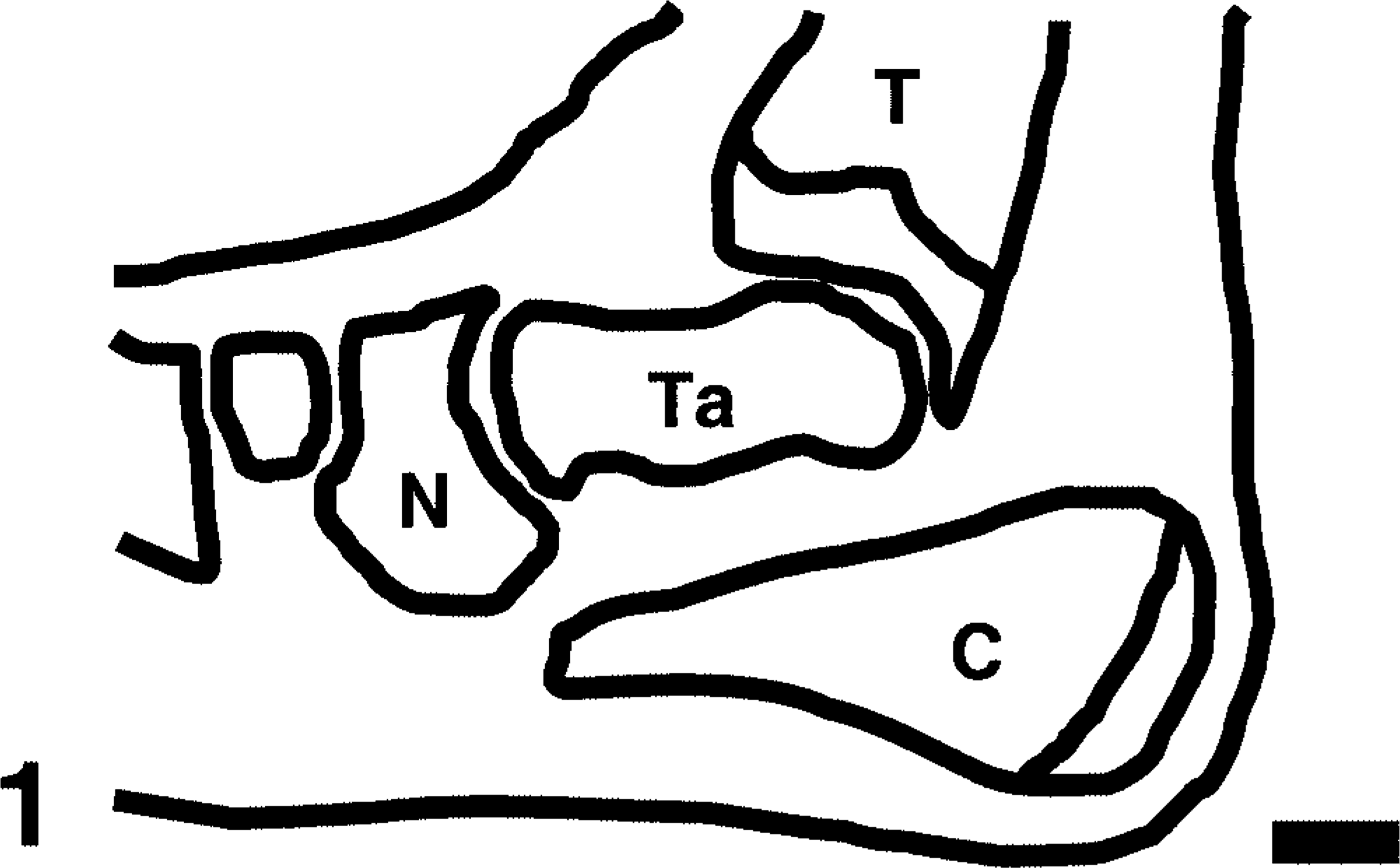
Distal tibia; control rat. The normal distal tibia features a prominent growth plate (G) and epiphysis (E), cortical bone (B) with smooth periosteal and endosteal bone surfaces, a few osteoclasts associated with the primary spongiosa (PS), and fat-filled marrow cavity (M). The synovium (S) of the tibiotarsal joint (J) is thin. Black squares (side = 0.6 mm) define fields in which quantitative parameters were measured. Ta = [proximal] talus. Anti–human cathepsin K immunohistochemical method to detect osteoclasts, with HE counterstains. Bar = 0.5 mm.
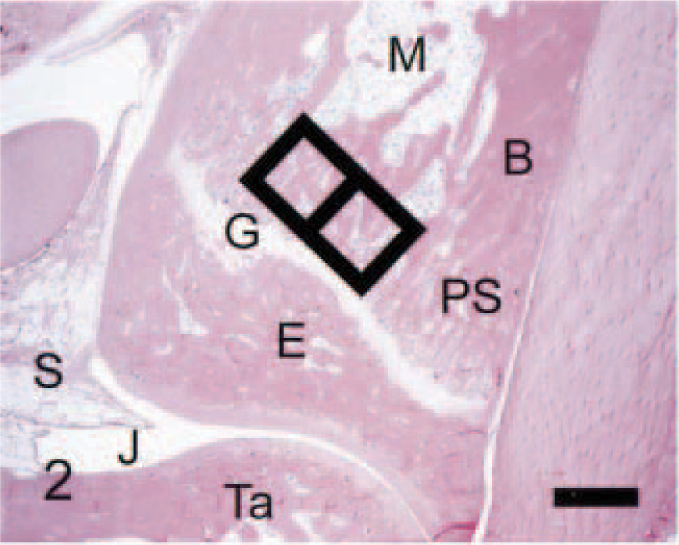
Navicular tarsal; control rat. In normal animals, this bone is characterized by cortical bone (B) with smooth periosteal and endosteal bone surfaces, intact joint cartilages (C) resting on subchondral bone plates (P), essentially no osteoclasts, and fat-filled marrow cavity. Black squares (side = 0.6 mm) delineate fields in which quantitative parameters were measured. Ta = [distal] talus. Anti–human cathepsin K immunohistochemical method to detect osteoclasts, with HE counterstains. Bar = 0.5 mm.
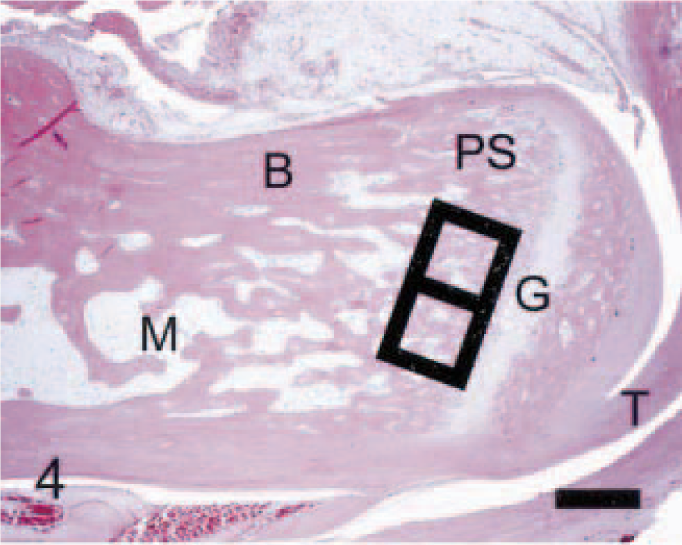
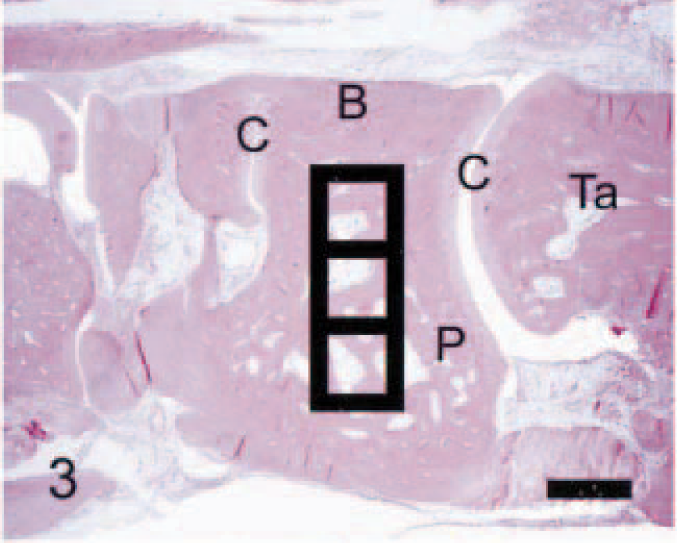
Calcaneus; control rat. Normally, the calcaneus is distinguished by a distinct growth plate (G) and attached gastrocnemius tendon (T), cortical bone (B) with smooth periosteal and endosteal bone surfaces, scattered osteoclasts associated with the primary spongiosa (PS), and fat-filled marrow cavity (M). Black squares (side = 0.6 mm) define fields in which quantitative parameters were measured. Anti–human cathepsin K immunohistochemical method to detect osteoclasts, with HE counterstains. Bar = 0.5 mm.
Statistical analyses
The histopathologic lesion scores (ordinal variables) were analyzed using the chi-square test, whereas the histomorphometric data (continuous variables) were assessed using analysis of variance followed by Dunnett's
Results
Semiquantitative lesion scores
As expected, the arthritic animals had significantly higher (
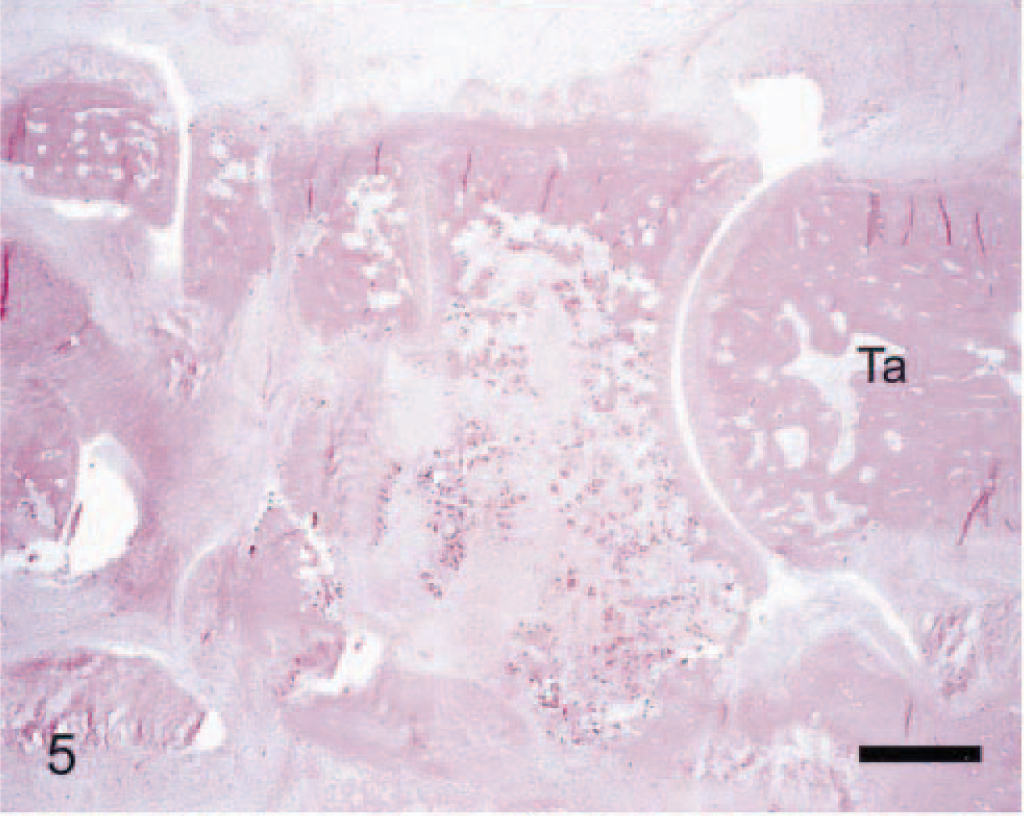
Navicular tarsal bone; arthritic rat. Compared with its normal anatomy (Fig. 3), this severe lesion in the navicular tarsal bone features marked leukocyte infiltration (in both adjacent soft tissues and bone marrow) with extensive erosion of both cortical and trabecular bones. Myriad osteoclasts are strewn within the marrow, particularly near eroding (scalloped) trabeculae. Anti–human cathepsin K immunohistochemical method to detect osteoclasts, with HE counterstains. Bar = 0.5 mm.
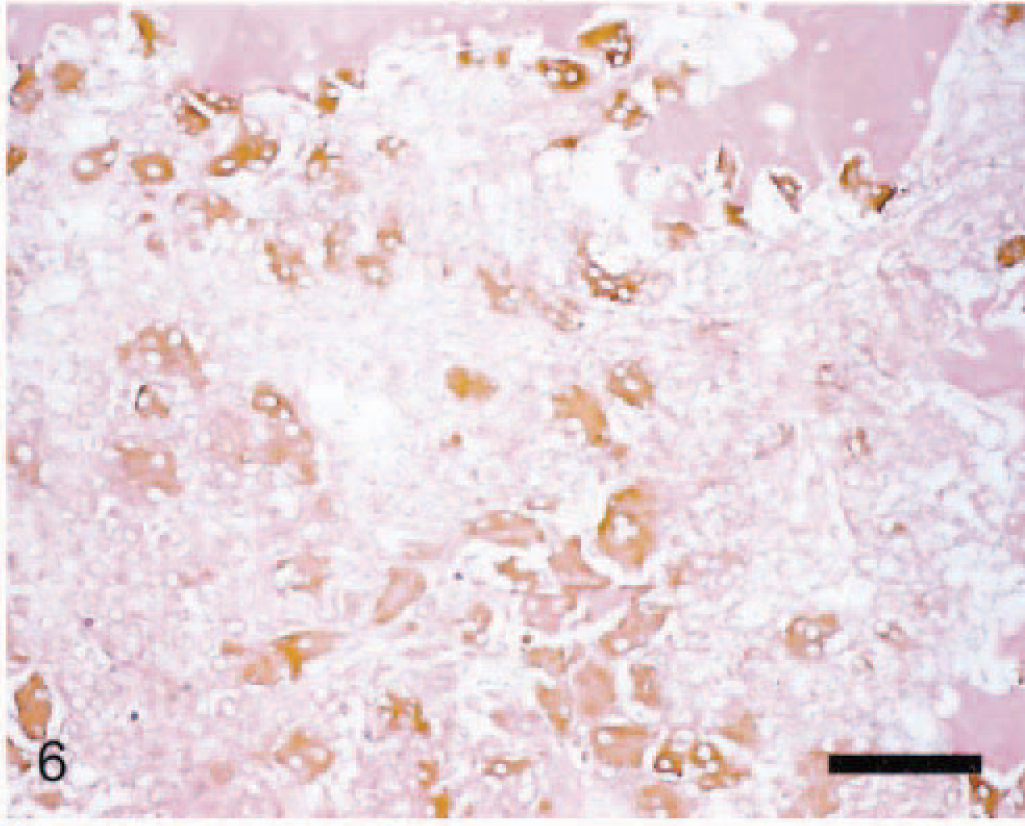
Navicular tarsal bone; arthritic rat. This marrow field is subjacent to the proximal cartilage. Numerous activated osteoclasts (large, multinucleated cells with brown cytoplasm) line eroding (scalloped) endosteal surfaces and are spread throughout the marrow. Necrotic inflammatory cells efface most of the marrow. Anti–human cathepsin K immunohistochemical method to detect osteoclasts, with HE counterstain. Bar = 50 µm.
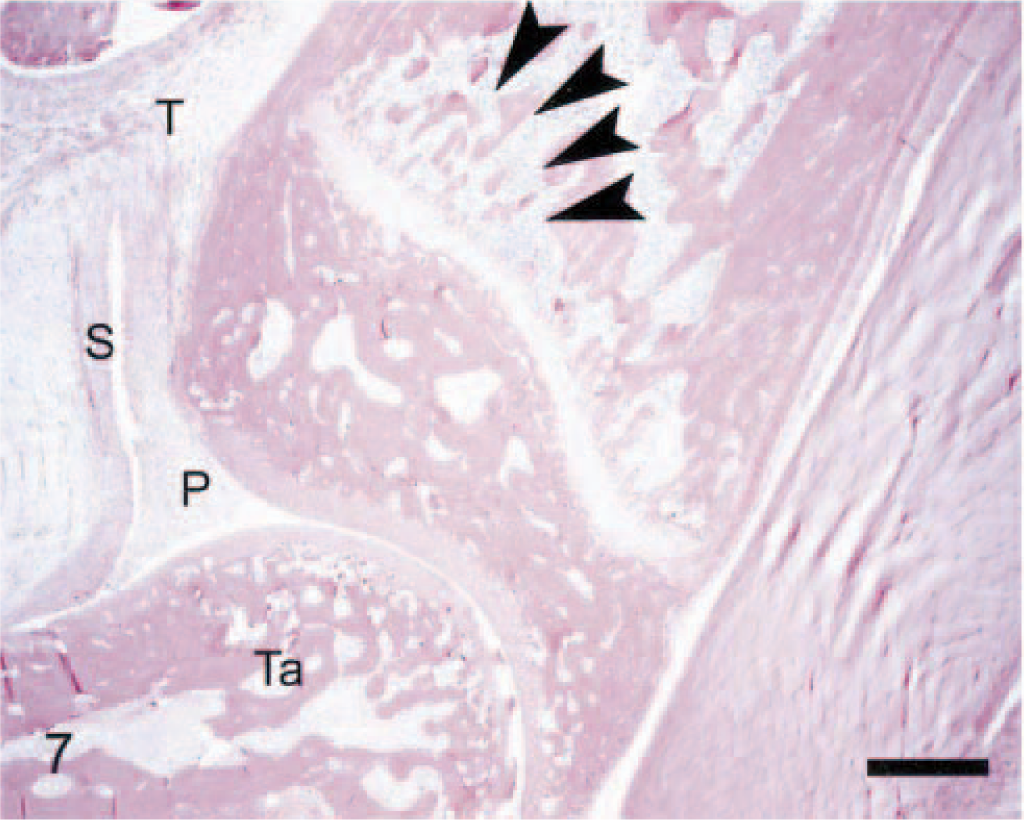
Distal tibia; arthritic rat. Compared with its normal architecture (Fig. 2), this bone is only modestly affected. Marked arthritis is evident as indicated by a frond of pannus (P) extending into the tibiotarsal joint, marked thickening of the synovium (S), and a diffuse influx of leukocytes into the marrow and periarticular soft tissues (T). Early erosion in the primary spongiosa is revealed by the decreased thickness and connectivity of the bone trabeculae near the growth plate (arrowheads). Anti–human cathepsin K immunohistochemical method to detect osteoclasts, with HE counterstains. Bar = 0.5 mm.
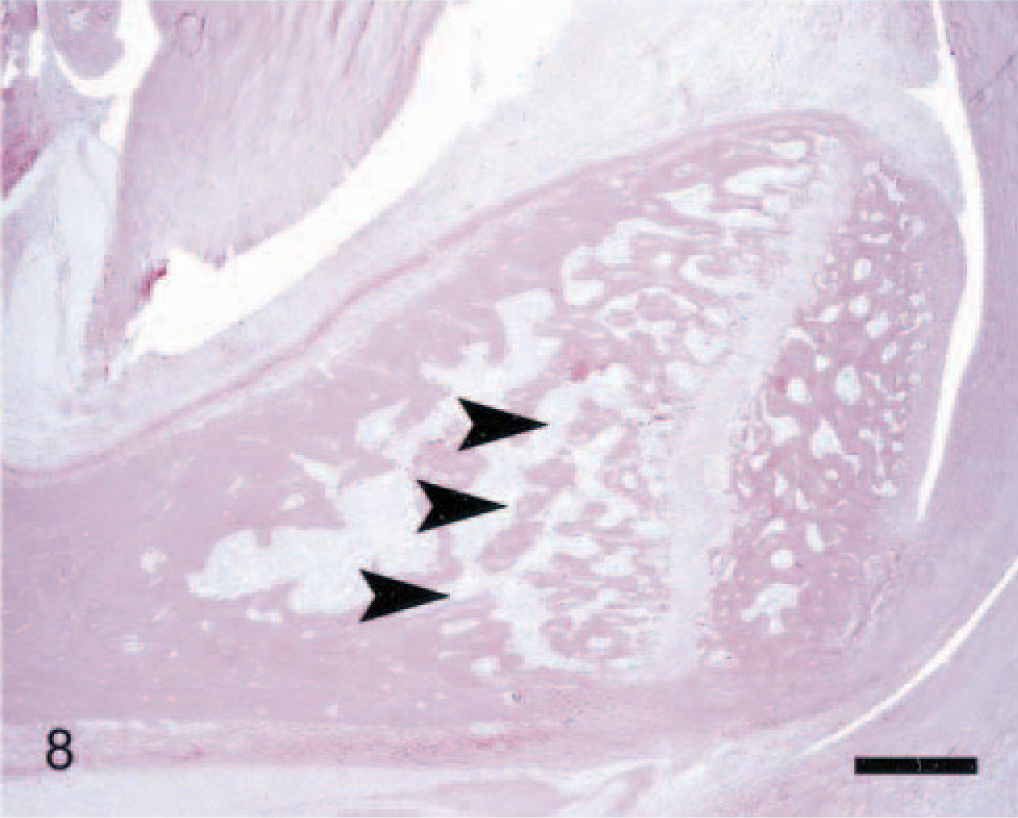
Calcaneus; arthritic rat. Compared with its normal structure (Fig. 4), this bone is relatively unaffected. The only prominent feature of arthritis is diffuse leukocyte infiltration into the adjacent soft tissues and, to a minor extent, marrow. Minimal erosion of bone is demonstrated by thinning of the trabeculae adjacent to the growth plate (arrowheads). Anti–human cathepsin K immunohistochemical method to detect osteoclasts, with HE counterstains. Bar = 0.5 mm.
Quantitative data: bone area and cell counts
The quantitative analysis agreed well with data acquired from this qualitative assessment (Table 3). Osteoclasts were greatly increased in arthritic rats at all three sites, although significant increases in osteoclasts (
Quantitative measurements of joint integrity.∗
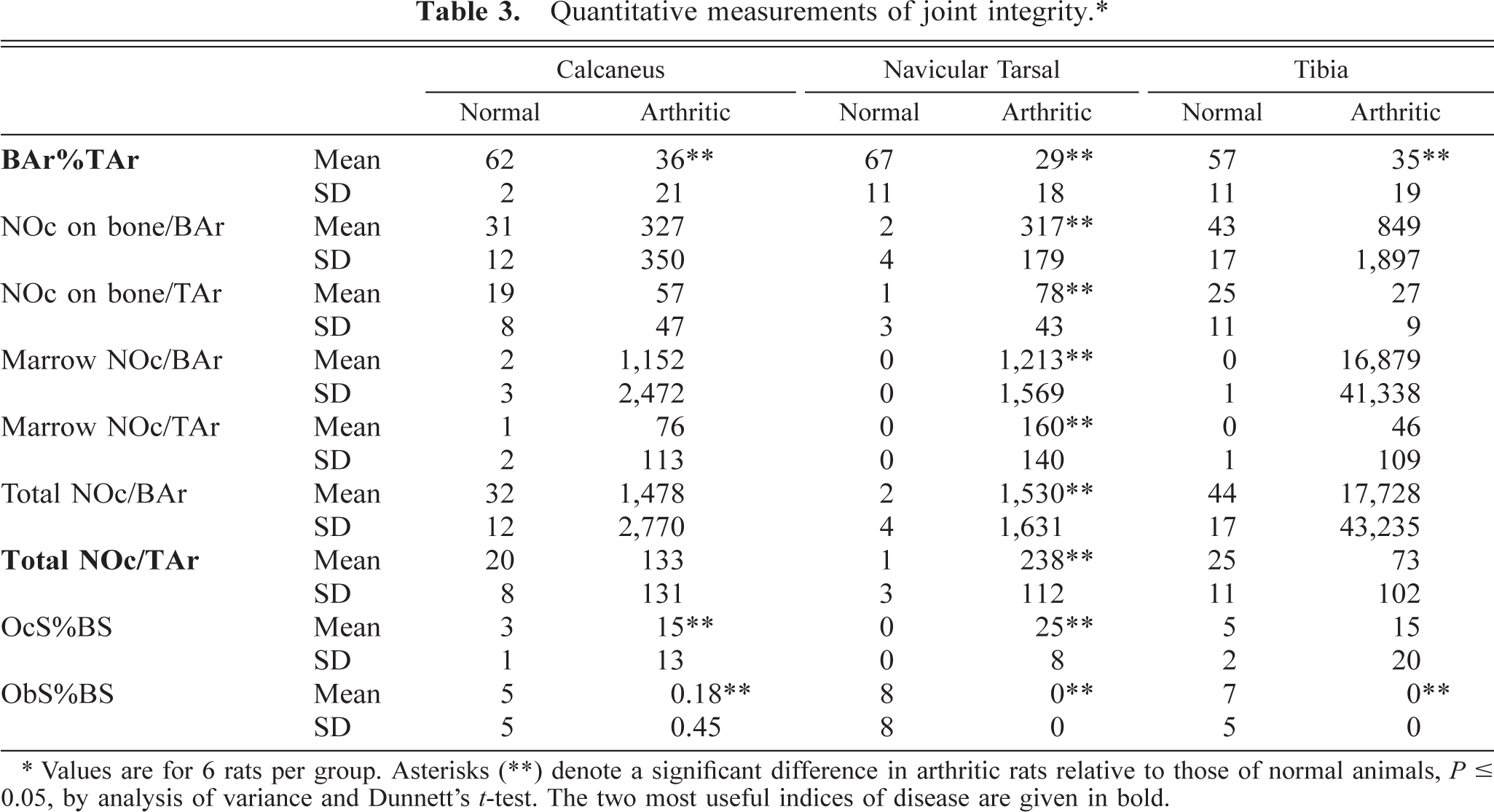
∗ Values are for 6 rats per group. Asterisks (∗∗) denote a significant difference in arthritic rats relative to those of normal animals,
Correlation between semiquantitative scores and quantitative data
The correlation between semiquantitative findings and the quantitative indices of joint destruction in rats with AdA was dependent on the site at which the quantitative measurements were taken. The best agreement occurred in the tarsal, where good correlations existed between erosion score and bone area (BAr%TAr,
Discussion
The results of this retrospective study indicate that a rapid histologic exam using semiquantitative scoring criteria can discriminate the severity of joint destruction in rats with AdA to a degree consistent with that afforded by more time-consuming quantitative assessments. However, in those instances where accurate enumeration is required, our data define preferred parameters to measure as well as the optimal site for rapidly quantifying bone destruction and intralesional osteoclasts. As shown in Table 3, the most relevant quantifiable parameters for measuring the progression of arthritis were bone area as a percentage of the total area measured (BAr%TAr, an index of bone integrity [erosions]) and the total number of osteoclasts per millimeter of total area measured (Total NOc/TAr, an assessment of osteoclasts associated with bone removal). The BAr%TAr was significantly reduced (
The anti–cathepsin K method is the primary means of detecting osteoclasts in our laboratory because of its sensitivity and specificity when applied to sections of rat bone. Although the antibody we use is proprietary, similar results might be obtained using commercial antibodies. Several firms manufacture anti–human cathepsin K antibodies. 12 Although their use would have to be verified empirically in sections of rat bone, we anticipate that one or more of these molecules would function in a manner equivalent to our proprietary anti-human antibody. Should such attempts prove futile, several other means of osteoclast detection exist in the public domain. For example, the enzyme histochemical procedure to demonstrate tartrate-resistant acid phosphatase (TRAP) is a standard technique for identifying osteoclasts in many species. Indeed, our laboratory routinely used a commercial TRAP kit (No. 387-A; Sigma Diagnostics, St. Louis, MO) to detect osteoclasts in rodent bone before switching to the anti–cathepsin K immunohistochemical method, a change driven by the tedious nature of the TRAP protocol and the similar results obtained using the two techniques (D. Duryea, unpublished). Antibodies against two other osteoclast markers—calcitonin receptor and vitronectin receptor 6,15 —are available from several vendors; 12 again, the capacity of these anti-human reagents to function in animal bones has yet to be reported. Molecular probes suitable for in situ hybridization could be designed for osteoclast markers in the event that commercial anti-human antibodies do not work in animal sections. For example, we have labeled osteoclasts in rat tissues with reagents directed against the known gene sequences of mouse calcitonin receptor (GenBank [Gb]: U18542, nucleotides [nts] 1,478–1,807), mouse cathepsin K (Gb: x94444, nts 450–933), and rat vitronectin receptor αVβ3 integrin (Gb: S58528, nts 359–716). The mouse Gb entries were deemed suitable for probing rat specimens because mouse and rat genes exhibit ∼ 95% homology over the specified sequences (S. Scully, personal communication). Finally, osteoclast profiles can be enhanced in HE sections by altering the eosin composition to include two additional dyes (final working solution: eosin Y, 238 ml; phloxine B, 6 ml; orange G, 6 ml). An eosin formulation that includes phloxine B and orange G imparts a vibrant rose pink hue to osteoclast cytoplasm that exceeds the contrast afforded by eosin alone.
In conclusion, the navicular tarsal bone is the most sensitive site for assessing bone destruction and intra-lesional osteoclasts in the hind paws of male Lewis rats with mycobacteria-induced adjuvant arthritis. The two most useful gauges of joint integrity were bone area as a percentage of total area measured (an erosion index) and total number of osteoclasts per millimeter of total area measured (which measures all intralesional osteoclasts). If arthritis quantification was limited to this site and these two indices, lesion severity in each arthritic paw could be enumerated in 20–30 minutes, which represents an 80% reduction in quantification time relative to the full counting method.
Footnotes
Acknowledgements
We thank Diane Duryea, Efrain Pacheco, Sheila Scully, and Gwyneth Van for technical assistance.
