Abstract
Using an immunohistochemical method, we attempted to detect the transmission of abnormal prion protein (PrPsc) to the enterocytes of the small intestine of neonatal mice by oral exposure with sheep brain affected by scrapie. Five 1-day-old neonatal mice were exposed by oral inoculation to the homogenized brain of a scrapie-affected sheep. In the small intestine of all mice 1 hour after inoculation, immunoreactivity with antinormal prion protein (PrPc) antibody was seen in the cytoplasm of villus enterocytes. This finding suggests transmission of abnormal PrPsc into the cytoplasm of enterocytes. In control mice treated with normal sheep brain, no PrPc signal was seen in enterocytes of the small intestine. Immunopositivity for neurofilament protein and glial fibrillary acidic protein was seen in the cytoplasm of enterocytes of mice inoculated with scrapie and normal sheep brain. This suggests that the enterocytes of neonatal mice can absorb PrPsc and other macromolecular proteins of the sheep brain affected by scrapie and may be more important than previously thought as a pathway for PrPsc transmission in neonatal animals.
Animal-transmissible spongiform encephalopathies or prion diseases, including ovine scrapie and bovine spongiform encephalopathy (BSE), are caused by acquisition of abnormal prion protein (PrPsc). Replication of the PrPsc depends critically on the host normal prion protein (PrPc), which develops into an abnormal, detergent-insoluble, proteinase-resistant isoform, PrPsc, in affected tissues such as the central nervous system (CNS) and lymphoid tissues. 3 However, how and when animals acquire PrPsc in the course of natural scrapie infection is not known. In experimental studies involving intragastric or oral inoculation of rodents with PrPsc from scrapie brain, PrPsc was detected in Payer's patches, gut-associated lymphoid tissues, 7,9,10 and ganglia of the enteric nervous systems before its detection in the CNS. 2,14 After the ingestion of PrPsc, uptake by the small intestine may follow either M-cell dependent or M-cell independent routes. 5 However, little is known about the transport mechanisms by which PrPsc reaches the germinal centers of lymphoid tissues from the gut lumen.
In neonatal animals, histologically, distended vacuoles containing colostral protein are frequently found in the cytoplasm of enterocytes lining villi of the small intestine, suggesting intestinal macromolecular uptake by pinocytosis during the first weeks after birth. 1,15 In this study, using immunohistochemical methods, we attempted to detect the entry of PrPsc into the enterocytes of the small intestine of neonatal mice following oral exposure to sheep brain containing abnormal prion of scrapie.
The first occurrence of ovine scrapie in Japan was reported in 1984. 13 Mice inoculated with fresh brain homogenates of an affected sheep showed the clinical signs and histopathologic lesions of scrapie after a long incubation period. 11
Five 1-day-old neonatal mice were exposed by oral inoculation to sheep brain containing PrPsc. The sheep brain used for this study had been fixed in 10% formalin solution for 7 days and embedded in paraffin for 20 yrs. 13 Deparaffinized tissues (100 mg) of the medulla oblongata of the sheep were homogenized in 1 ml phosphate-buffered saline (PBS) with a polytron for 3 minutes, and 20 ml of homogenized brain was used for each peroral inoculation. For immunohistochemical controls, tissue sections were stained by procedures that omitted only the primary antibody. For negative controls, three neonatal mice were exposed by oral inoculation to normal sheep brain fixed in 10% formalin, embedded in paraffin, and extracted by identical procedures. All inoculated mice were sacrificed under anesthesia 1 hour after inoculation. Duodenum and jejunum were fixed in 4% paraformaldehyde in PBS for 24 hours and embedded in paraffin. Thin paraffin sections (4 mm) of the small intestine of mice and the medulla oblongata of the sheep brain affected by scrapie were deparaffinized and stained with hematoxylin and eosin (HE).
Histologic sections of sheep brain and mouse small intestine were stained immunohistochemically by the avidin-biotin-peroxidase complex (ABC) procedure (ABC-peroxidase staining kit (Elite; Vector Laboratories, Burlingame, CA). The specific antibody used in the present study was an affinity-purified polyclonal rabbit anti-PrPc antibody described in previous reports. 6,12 The synthetic peptide used as immunogen was B-103, corresponding to bovine PrP codons 103–121. This polyclonal antibody reacted strongly with PrPc-enriched fractions of brain tissues of cattle, sheep, and mouse on Western blots. 6
Deparaffinized tissue sections for PrP immunostaining were autoclaved at 121 C for 30 minutes after immersion in 98% formic acid for 30 minutes and in 60 µg/ml proteinase K (Wako Pure Chemical Industries, Tokyo, Japan) in 0.1 mol/liter phosphate buffer saline for 5 minutes. Sections were incubated with primary antibodies at room temperature for 30 minutes. After washing with distilled water, sections were incubated with 0.5% biotinylated goat antirabbit antibody for 30 minutes at room temperature and with ABC conjugated peroxidase for 30 minutes. Color was developed in a 0.05% 3,3′-diaminobenzidine solution. Tissue sections without pretreatment were incubated with rabbit antibody to bovine glial fibrillary acidic protein (GFAP) (Dako, Co., Carpinteria, CA) and a rabbit antibody cocktail to human neurofilament (NF) (Affinity Research Products Limited, Mamhead, UK); these proteins served as markers of nervous tissue.
In the brain of the sheep with scrapie, cytoplasmic vacuolation of neurons and spongiform change of the gray matter were identified in the HE sections of the medulla oblongata. 13 Immunoreactivity for PrPc was found in the neuropil of the medulla oblongate (Fig. 1).
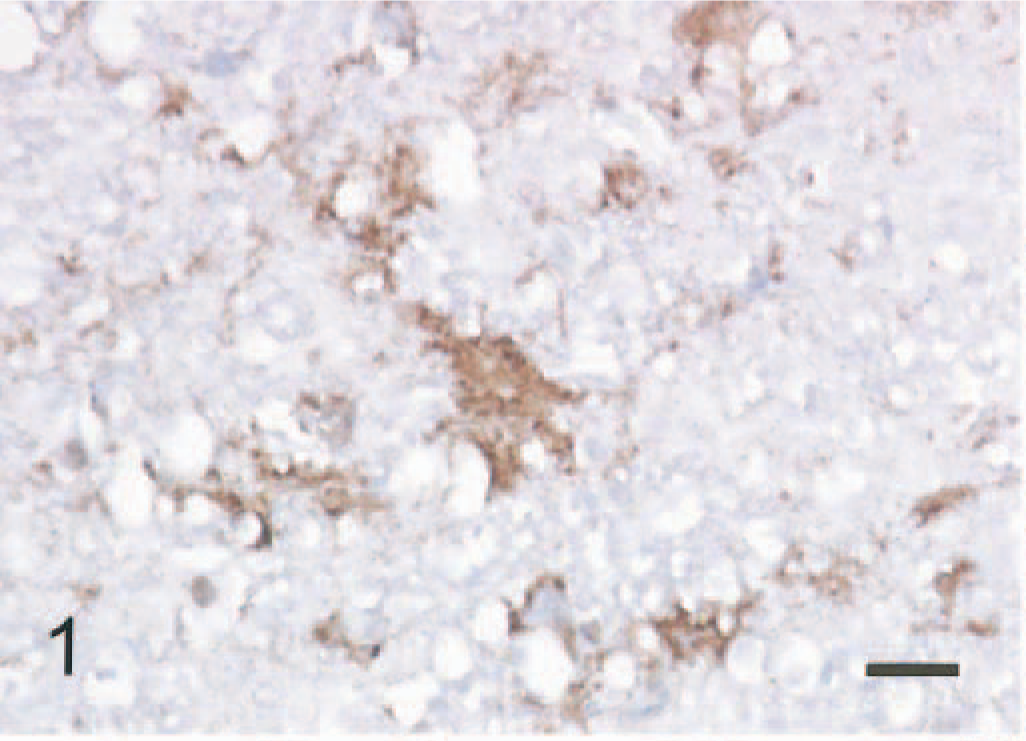
Medulla oblongata; scrapie infected sheep, positive PrPc signal is seen in neuropil with spongiform change. ABC method, Mayer's hematoxylin counterstain. Bar = 20 mm.
In the small intestine of all five mice given scrapie-affected brain 1 hour prior to being killed, sheep brain tissue was present in the gut lumen. Immunopositive signals for PrPc were found in the intralumenal sheep brain tissue (Fig. 2) and the apical and basilar cytoplasm of villus and crypt enterocytes (Fig. 3) in the small intestine of all mice treated with scrapie brain. When PrPc antibody was omitted, no positive signals for PrPc were found in enterocytes (Fig. 4). PrPc-positive cells ranged from 1 to 2 per high-power field (Fig. 5). Positive signals for GFAP and NF were found in the medulla oblongata of the sheep brain with scrapie and in enterocytes of the small intestine of all mice inoculated with scrapie brain (Figs. 6 and 7).
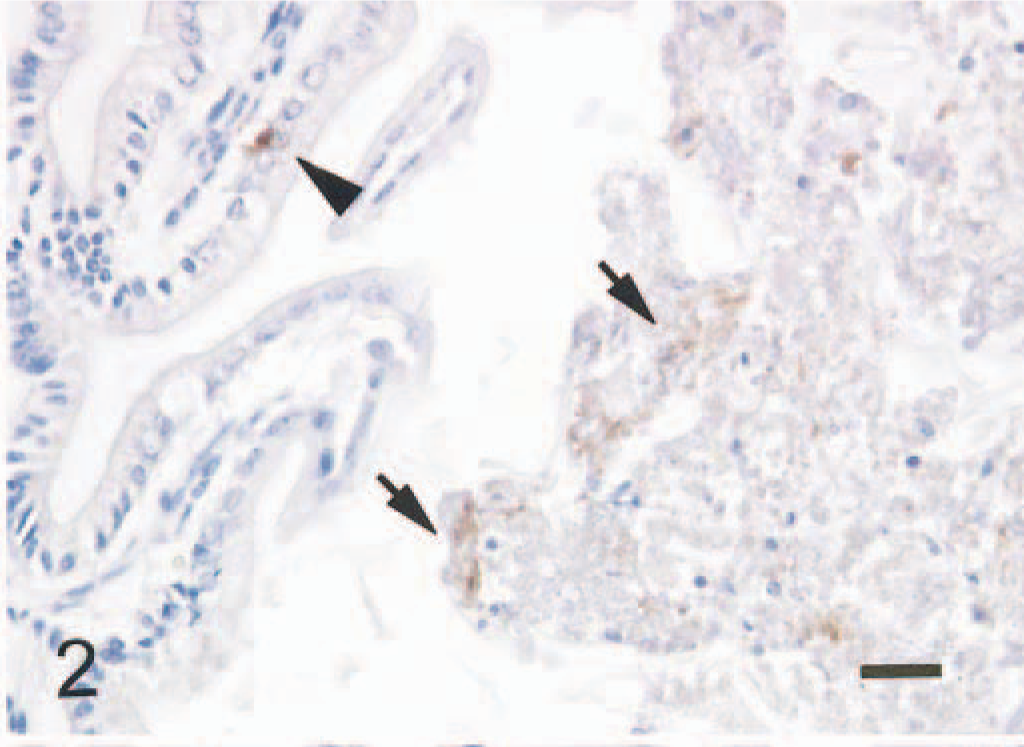
Small intestine; mouse, 1 hour after inoculation with homogenized sheep brain with scrapie. Positive signal for PrPc is present in the intralumeral sheep brain tissue (arrows) and in an enterocyte (arrowhead). ABC method, Mayer's hematoxylin counterstain. Bar = 30 mm.
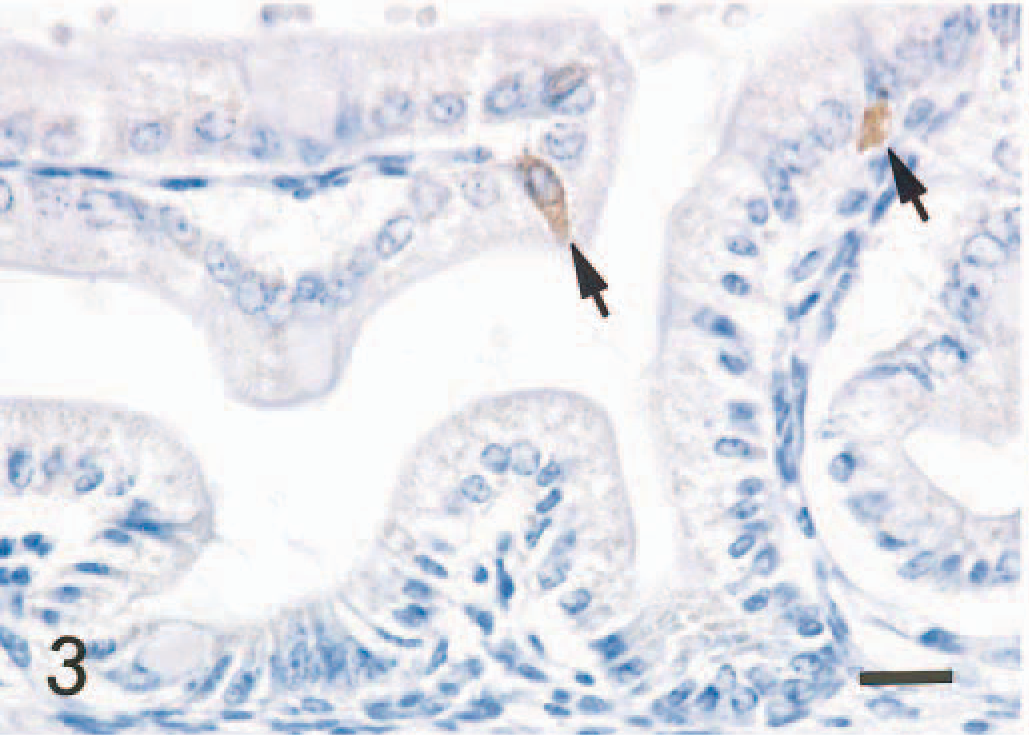
Small intestine; mouse, 1 hour after inoculation with homogenized sheep brain with scrapie. Positive signal for PrPc is present in the apical and basilar cytoplasm of villus enterocytes (arrows). ABC method, Mayer's hematoxylin counterstain. Bar = 20 mm.
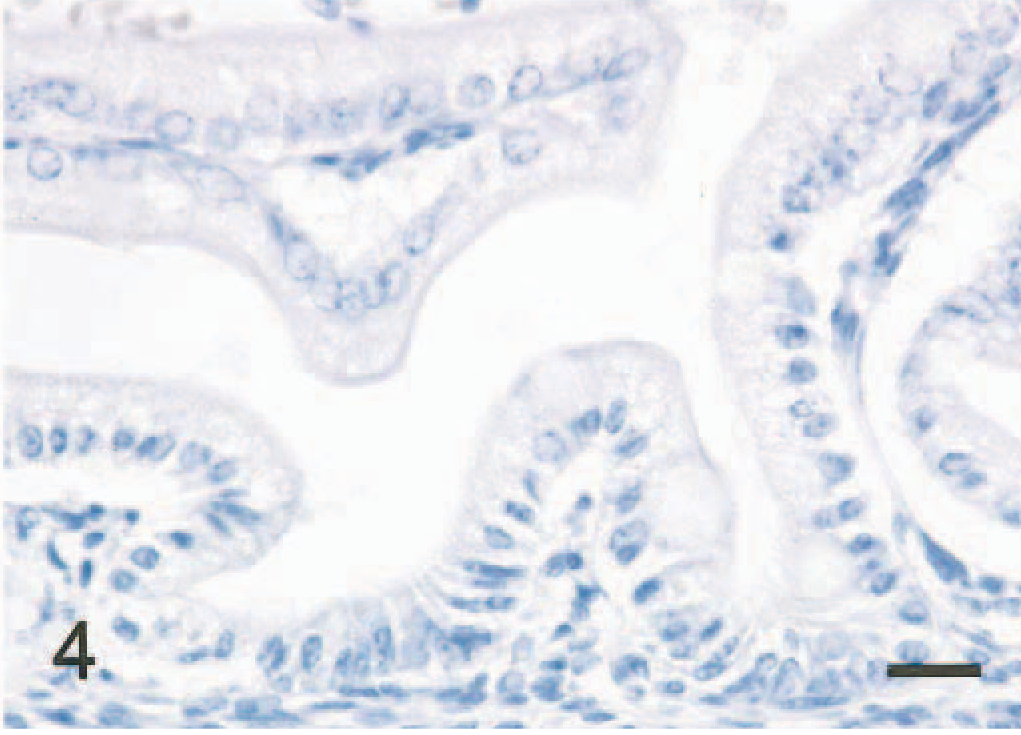
Small intestine; mouse, 1 hour after inoculation with homogenized sheep brain with scrapie. Immunostain omitting PrPc antibody shows no PrPc signal in enterocytes. ABC method, Mayer's hematoxylin counterstain. Bar = 20 mm.
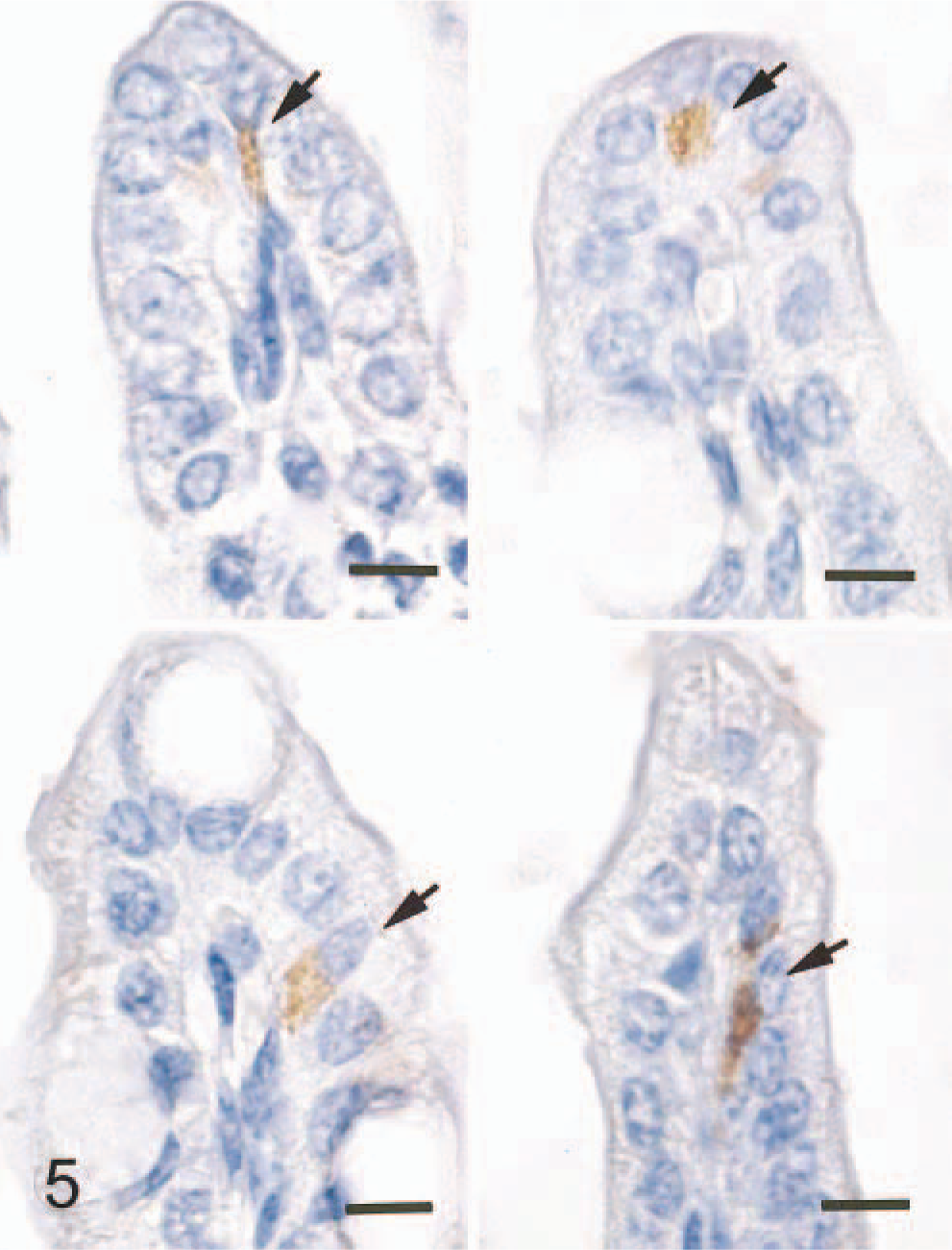
Small intestine; mouse, 1 hour after inoculation with homogenized sheep brain with scrapie. There is PrPc signal mainly in the basilar cytoplasm of enterocytes at tip of the villus adjacent to the nucleus (arrows). ABC method, Mayer's hematoxylin counterstain. Bar = 10 mm.
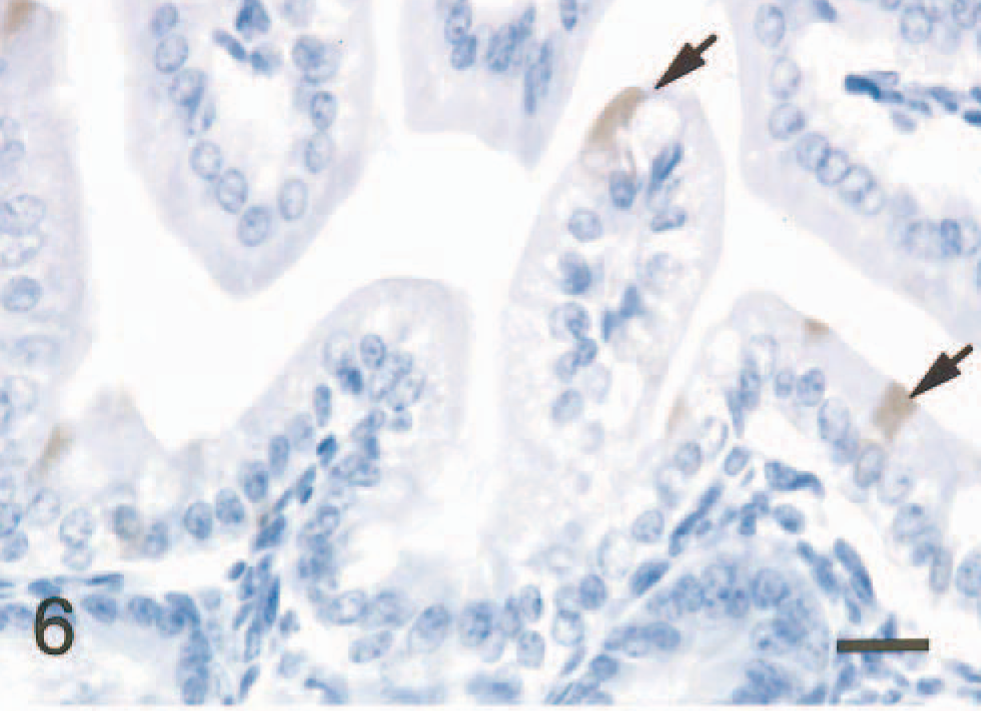
Small intestine; mouse, 1 hour after inoculation with homogenized sheep brain with scrapie. GFAP signal is seen in the apical cytoplasm of enterocytes (arrows). ABC method, Mayer's hematoxylin counterstain. Bar = 20 mm.
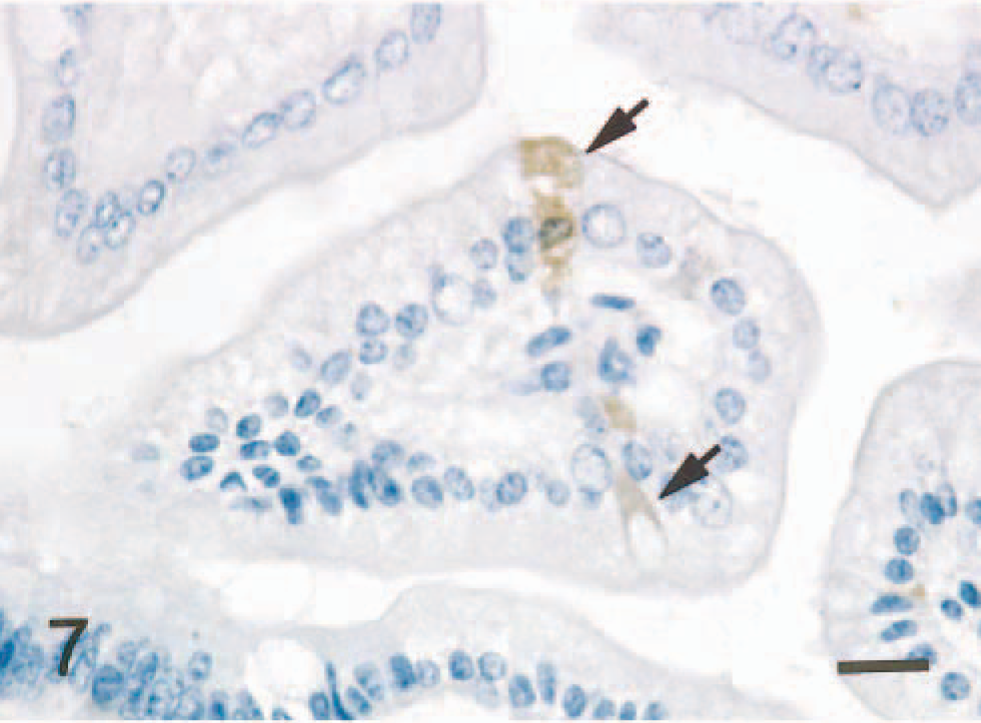
Small intestine; mouse, 1 hour after inoculation with homogenized sheep brain with scrapie. NF signal is observed in the apical and basilar cytoplasm of enterocytes (arrows). ABC method, Mayer's hematoxylin counterstain. Bar = 20 mm.
In all mice given normal brain, no PrPc was seen in the intralumenal brain tissue or enterocytes of the small intestine (Fig. 8), but signal for GFAP- and NF-positive signals was found in the enterocytes.
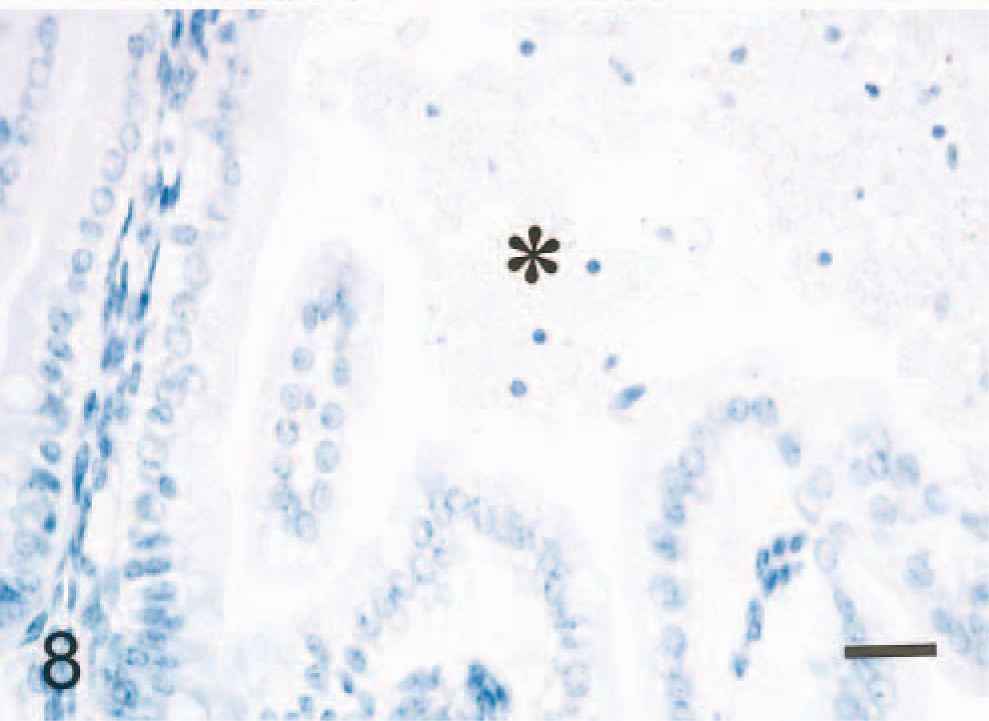
Small intestine; mouse, 1 hour after inoculation with homogenized brain of normal sheep (∗). No PrPc signal is seen in the cytoplasm of enterocytes. Tissues were subjected to multiple pretreatments. ABC method, Mayer's hematoxylin counterstain. Bar = 50 mm.
For immunohistochemical detection of abnormal PrPsc in the brain, several pretreatments have been employed, including picric acid, formic acid, steam autoclaving, and microwave treatment, because epitope retrieval after formalin fixation and paraffin embedding of the brain tissue is very difficult. 4 Without pretreatment there is a high probability of false-negative immunostaining. 4 Therefore, single or multiple pretreatments are necessary for immunohistochemical detection of the PrPsc. In the present study, we used multiple pretreatments. Immunopositivity for PrPc was detected in the sheep brain with scrapie and in enterocytes of mice inoculated with sheep brain containing the abnormal scrapie prion. The immunohistochemical results suggest entry of PrPsc into the cytoplasm of enterocytes. However, absorption of sheep brain containing PrPsc occurred only in enterocytes in the duodenum and jejunum in this study. Intralumenal sheep brain was observed only as far distally as the jejunum. This material could not be observed in the ileum in mice at only 1 hour after ingestion. Observation of absorbed PrPsc in the ileum would require being killed later. We have demonstrated absorption of PrPsc in the jejunum and ileum of adult mice in another study (M. Okamoto, T. Noguchi, and H. Tamiyama, unpublished data). It is not clear from our studies whether different segments of the small intestine differ in their ability to absorb the macromolecular proteins of sheep brain.
In Germany, milk substitutes made from bone meal and fat of diseased animals are believed to play an important role in the transmission of BSE in cattle. 8 In the present study, immunohistochemistry suggests that PrPsc of sheep brain affected by scrapie enters neonatal murine enterocytes by pinocytosis. Thus, the results support the possibility that PrPsc included in milk substitutes may be absorbed by enterocytes of the small intestine of the neonate and may thus be an important pathway for neonatal PrPsc transmission in sheep, cattle, and mice. The mechanism of PrPsc transmission from enterocytes to the germinal centers of lymphoid tissues or peripheral nerve systems remains unclear.
Footnotes
Acknowledgements
The present study was supported in part by a grant from Gakujutsu-Frontier cooperative research in Rakuno Gakuen University.
