Abstract
The Cd40l–/– mouse is a well-established model of X-linked hyper-immunoglobulin M (IgM) syndrome, an immunodeficiency disorder of human beings characterized by the lack of expression of the CD40 ligand (CD40L) on activated T-cells, predisposing to infections with opportunistic pathogens like Pneumocystis jirovecii. The aim of our study was to describe the pulmonary lesions in Cd40l–/– mice experimentally infected with Pneumocystis murina, in comparison with naturally infected severe combined immunodeficient (SCID) mice. Formalin-fixed paraffin-embedded lungs from 26 Cd40l–/–, 11 SCID, and 5 uninfected Cd40l–/– mice were examined by histology and immunohistochemistry for the presence of the pathogen and for leukocyte populations (CD3, CD4, CD45R/B220, CD8a, Iba-1, Ly-6G, CD206, MHC II, and NKp46/NCR1). Infection was confirmed by immunohistochemistry in 18/26 (69%) Cd40l–/– mice and in 11/11 (100%) SCID mice. Fourteen out of 26 (54%) Cd40l–/– mice had interstitial pneumonia. Twenty-three out of 26 (88%) Cd40l–/– mice had peribronchiolar/perivascular lymphoplasmacytic infiltrates, rich in B-cells and Mott cells. Acidophilic macrophage pneumonia was additionally found in 20/26 (77%) Cd40l–/– mice. Only 4/11 (36%) SCID mice had interstitial pneumonia, but no peribronchiolar/perivascular infiltrates or acidophilic macrophage pneumonia were observed in this strain. This study represents the first description of pulmonary histopathological lesions in Cd40l–/– mice infected with P. murina. We speculate that the singular characteristics of the inflammatory infiltrates observed in Cd40l–/– mice could be explained by the specific immune phenotype of the model.
Pneumocystis spp. are fungal pulmonary opportunistic pathogens found in a wide range of mammalian species, including humans and mice. In humans, Pneumocystis jirovecii infection is particularly relevant in immunocompromised patients, causing a severe interstitial pneumonia called P. jirovecii pneumonia. 8 Spontaneous infection with the host-specific P. murina species is relatively common in immunodeficient strains of mice and has similar pathological features. 2
X-linked hyper-immunoglobulin M (IgM) syndrome (HIGM1) is one of the several immunodeficiency disorders in which affected patients become susceptible to opportunistic pathogens like P. jirovecii.12,24 Hyper-immunoglobulin M (IgM) syndrome is a primary immunodeficiency characterized by inactivating mutations of CD40LG gene that encodes CD40 ligand (CD40L). 42 CD40L is commonly expressed on activated CD4+ T-cells, but certain levels of expression were reported on other immune cell types including B-cells, natural killer (NK) cells, monocytes/macrophages, and dendritic cells. 41 The role of CD40L is particularly crucial on activated CD4+ T-cells because it is responsible for engaging CD40 receptor on B-cells triggering their proliferation, maturation, and immunoglobulin class-switching.41,42 For this reason, HIGM1 patients have impaired production of switched Ig isotypes, with normal or elevated serum levels of IgM. 42 The Cd40l–/– mouse is a well-established model of human HIGM1, showing the same disease phenotype as the human counterpart, although the immune defect is less severe.34,37
Pneumocystis spp. infection has been described in several immunodeficient murine models, like CB-17 severe combined immunodeficiency (SCID), Rag-1 and -2-deficient, CD4+ T-cell-depleted BALB/c, and nude mice, resulting in different pulmonary lesions depending on the type and level of immunodeficiency.3,4,5,7,10,15,20,21,35,36,38,43–46 In Rag-1 and -2-deficient mice, no pulmonary inflammation is observed, while in nude mice, inflammation is inconspicuous.15,20,44,45 The SCID mouse is one of the first and most studied models of Pneumocystis spp. infection.6,7,10,17,26,35,36,43,44,46–48 The histological hallmarks of Pneumocystis spp. infection are eosinophilic foamy material in the alveolar spaces, thickening of alveolar septa by mononuclear inflammatory cells, and peribronchiolar and perivascular lymphocytic infiltrates.10,35,36,43,44 In the SCID model, however, cases with no peribronchiolar/perivascular inflammation rather than no inflammation at all are also reported.35,46 In a previous study performed in Cd40l–/–mice experimentally infected with P. murina, no inflammatory lesions or upregulation of immune response-related genes were observed, and no influx of immune cells in the lung was found by immunohistochemistry. 18
The aim of this study was to characterize the pulmonary lesions and the immune cells infiltrate in Cd40l–/– mice experimentally infected with P. murina, in comparison to pulmonary lesions induced in SCID mice spontaneously infected with the same opportunistic pathogen.
Materials and Methods
Case Selection
Formalin-fixed paraffin-embedded lungs from 26 C57BL/6J Cd40l–/– mice (B6.129S2-Cd40lgtm1Imx/J, 8 males and 18 females) experimentally infected with P. murina through intranasal inoculation were examined. Cd40l–/– mice were purchased from The Jackson Laboratory. The mice were infected between 2 and 8 months of age and sacrificed between 9 and 12 months of age when clinical sings of the disease appeared. The presence of the organism in the lung was confirmed by molecular analysis with polymerase chain reaction (PCR). Details on experimental infection and PCR analysis are described elsewhere. 42 All the animal procedures were designed and performed with the approval of the Institutional Animal Care and Use Committee of the San Raffaele Hospital (IACUC #749, #818) and approved by the Ministry of Health and local authorities according to Italian law. Lungs from 5 age-matched uninfected Cd40l–/–mice (2 males and 3 females) were included as controls, and lungs from 11, 2- to 8-month-old CB-17 SCID mice (CB17/lcr-Prkdcscid/lcrlcoCrl) spontaneously infected with P. murina were retrieved from the archive of the Mouse and Animal Pathology Laboratory (MAPLab, Milan, Italy). Severe combined immunodeficient mice were purchased from Charles River Laboratory.
For both strains, pulmonary co-morbidities at the time of infection were excluded by periodic health monitoring, in accordance with the Federation of European Laboratory Animal Science Associations (FELASA) recommendations. 31 This included testing for Sendai virus, pneumonia virus of mice, Mycoplasma spp., and Corynebacterium kutscheri.
Histology and Immunohistochemistry
Four micrometer thick paraffin sections were stained with hematoxylin and eosin for histopathological examination and immunohistochemistry (IHC) was carried out to evaluate the presence of P. murina and characterize the immune cell infiltrates. Immunohistochemistry was performed automatically using the Thermo Scientific Autostainer 480S System (Thermo Fisher Scientific, Fremont, CA, USA). Dewaxing of tissue sections and heat-induced epitope retrieval were simultaneously performed using the Dewax and HIER Buffer H pH 9 (Thermo Fisher Scientific, Fremont, CA, USA) at boiling temperature for 40 minutes. Endogenous peroxidase was blocked with 3% H2O2 for 10 min at room temperature. Nonspecific protein binding was prevented with 10% normal goat or rabbit serum for 30 minutes at room temperature. Sections were incubated for 1 hour at room temperature with the primary antibodies listed in Table 1. Sections were incubated with biotinylated goat anti-rabbit, rabbit anti-rat, or rabbit anti-goat IgG (Vector Laboratories, Burlingame, CA, USA) diluted 1:200. Biotinylated goat anti-mouse IgM 1:1000 (Vector Laboratories, Burlingame, CA, USA) was used for direct identification of IgM-secreting plasma cells. Labeling was performed with Vectastain Elite ABC-Peroxidase kit (Vector Laboratories, Burlingame, CA, USA) diluted 1:150, and the reaction was visualized with Peroxidase ImmPACT DAB Substrate (Vector Laboratories, Burlingame, CA, USA). Sections were counterstained with Mayer’s hematoxylin and mounted with Micromount (Diapath, Martinengo, Italy). For immune cells markers, sections of murine healthy lymphoid organs were included in the immunolabeling runs as positive controls. For P. murina, internal controls of known positive mice were used. Negative controls were the same sections used as positive controls but without primary antibody.
List of primary antibodies used for immunohistochemistry.
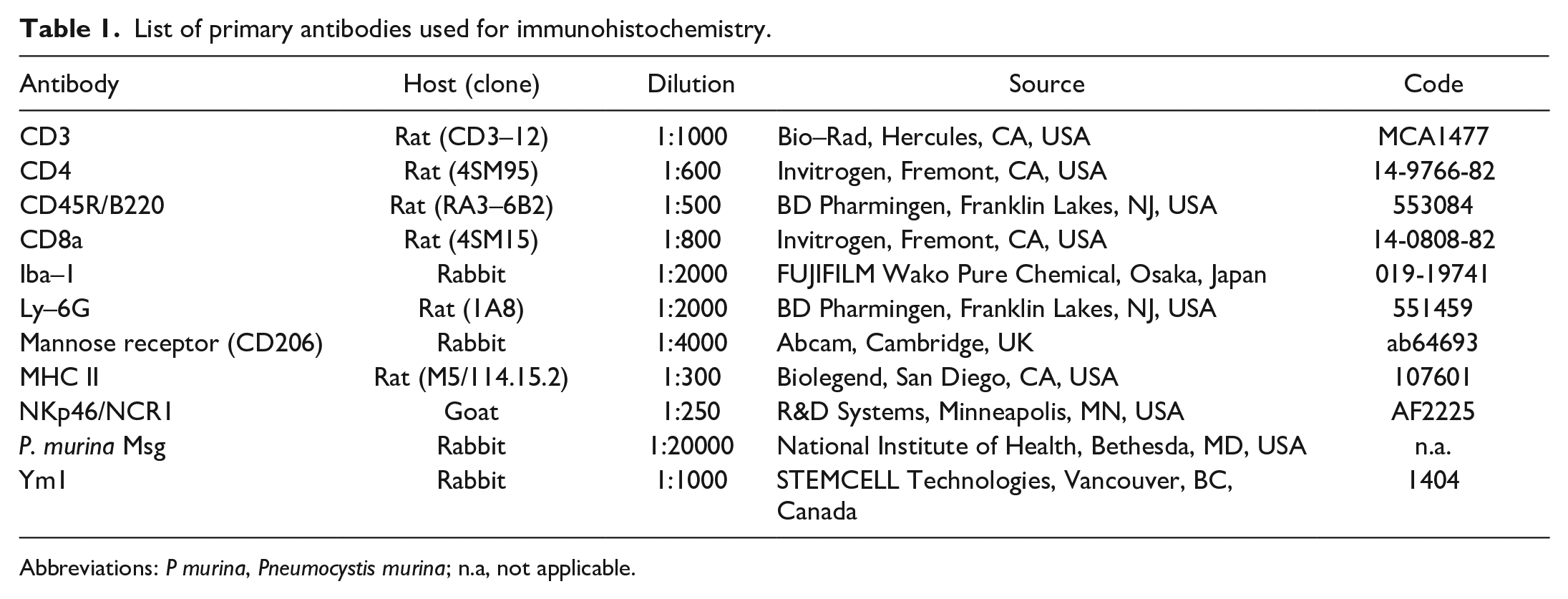
Abbreviations: P murina, Pneumocystis murina; n.a, not applicable.
Slides were digitalized using the NanoZoomer-XR Digital Slide Scanner C12000 (Hamamatsu Photonics K.K., Hamamatsu, Japan) and visualized using NDP.view2 Image viewing software (Hamamatsu Photonics K.K., Hamamatsu, Japan). In hematoxylin and eosin-stained sections, the presence of intra-alveolar eosinophilic foamy material (morphologically consistent with P. murina) was evaluated according to the following semiquantitative scoring system: 0 = absent; 1 = occasional organisms in <10% of alveoli; 2 = moderate numbers of organisms in 10% to 50% of alveoli; 3 = numerous organisms in >50% of alveoli. Peribronchiolar and perivascular lymphoplasmacytic infiltrates were evaluated according to the following semiquantitative scoring system, as previously described: 5 0 = absent; 1 = mild infiltrate around few bronchioles and vessels; 2 = moderate infiltrate around many bronchioles and vessels; 3 = heavy infiltrate around most bronchioles and vessels. Acidophilic macrophage pneumonia (AMP) was evaluated according to the following semiquantitative scoring system: 0 = absent; 1 = occasional cells in <10% of alveoli; 2 = moderate numbers of cells in 10% to 50% of alveoli; 3 = numerous cells and multinucleated giant cells in >50% of alveoli. Interstitial pneumonia was evaluated according to the following semiquantitative scoring system: 0 = absent; 1 = slight thickening of alveolar septa with occasional mononuclear cells; 2 = moderate thickening of alveolar septa with moderate numbers of mononuclear cells; 3 = severe thickening of alveolar septa with numerous mononuclear cells. Slides immunolabeled with anti-P. murina Msg antibody were scored as follows: 0 = no organisms; 1 = occasional organisms (organisms in less than 3 fields of 0.406 mm2 each); 2 = moderate numbers of organisms (organisms in 3-5 fields of 0.406 mm2 each); 3 = numerous organisms (organisms in more than 5 fields of 0.406 mm2 each). Slides stained with anti-CD45R/B220, -CD3, -CD4, -CD8a, -Iba-1, -MHC II, -CD206, -NKp46/NCR1, and -Ly-6G antibodies were evaluated for quantification of immune cells in interstitial pneumonia and peribronchiolar/perivascular infiltrates and scored as follows: 0 = no positive cells; 1 = occasional positive cells (positive cells in less than 3 fields of 0.406 mm2 each); 2 = moderate numbers of positive cells (positive cells in 3-5 fields of 0.406 mm2 each); 3 = numerous positive cells (positive cells in more than 5 fields of 0.406 mm2 each).
Statistical Analysis
Statistical analysis was performed with GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA, version 8.0). The frequency of pulmonary lesions in Cd40l–/– and SCID mice was compared with Fisher’s exact test. The correlation between P. murina burden and histopathological findings was studied with Spearman’s correlation. P < .05 was considered statistically significant.
Results
Neither P. murina nor pulmonary lesions were detected in uninfected Cd40l–/– control mice (Fig. 1a). P. murina infection was detected in hematoxylin and eosin-stained sections and confirmed by IHC in 18/26 (69%) Cd40l–/–mice (Table 2; Supplemental Table S1). Fourteen out of 26 (54%) Cd40l–/– mice had interstitial pneumonia, characterized by variably sized areas of pulmonary parenchymal consolidation, variable amounts of intra-alveolar organisms associated with foamy macrophages and multinucleated giant cells containing the organisms, and thickening of alveolar septa by mixed inflammatory cell infiltrates composed of mononuclear cells and occasional granulocytes (Fig. 1b). Infection with P. murina was detected in hematoxylin and eosin-stained sections and confirmed by IHC in all the 11 (100%) SCID mice. Interstitial pneumonia was found in 4/11 (36%) SCID mice, only when large numbers (score 3) of intra-alveolar organisms were detected (Fig. 1c). In both strains, only occasional CD45R/B220+ B-cells (Figs. 2a, b) and CD3+ T-cells (Figs. 2c, d) were present in the interstitial pneumonia, whereas a predominance of Iba-1+ macrophages was observed (Figs. 3a, b). Ly-6G+ neutrophils were infrequent in the interstitial pneumonia in Cd40l–/– mice, whereas they were more abundant in SCID mice (Figs. 3c, d). NKp46/NCR1+ NK cells were rare in both strains (Supplemental Figure S1). A statistically significant correlation was found in both Cd40l–/– and SCID mice between P. murina burden and the severity of interstitial pneumonia (P < .0001 and P < .001, respectively).
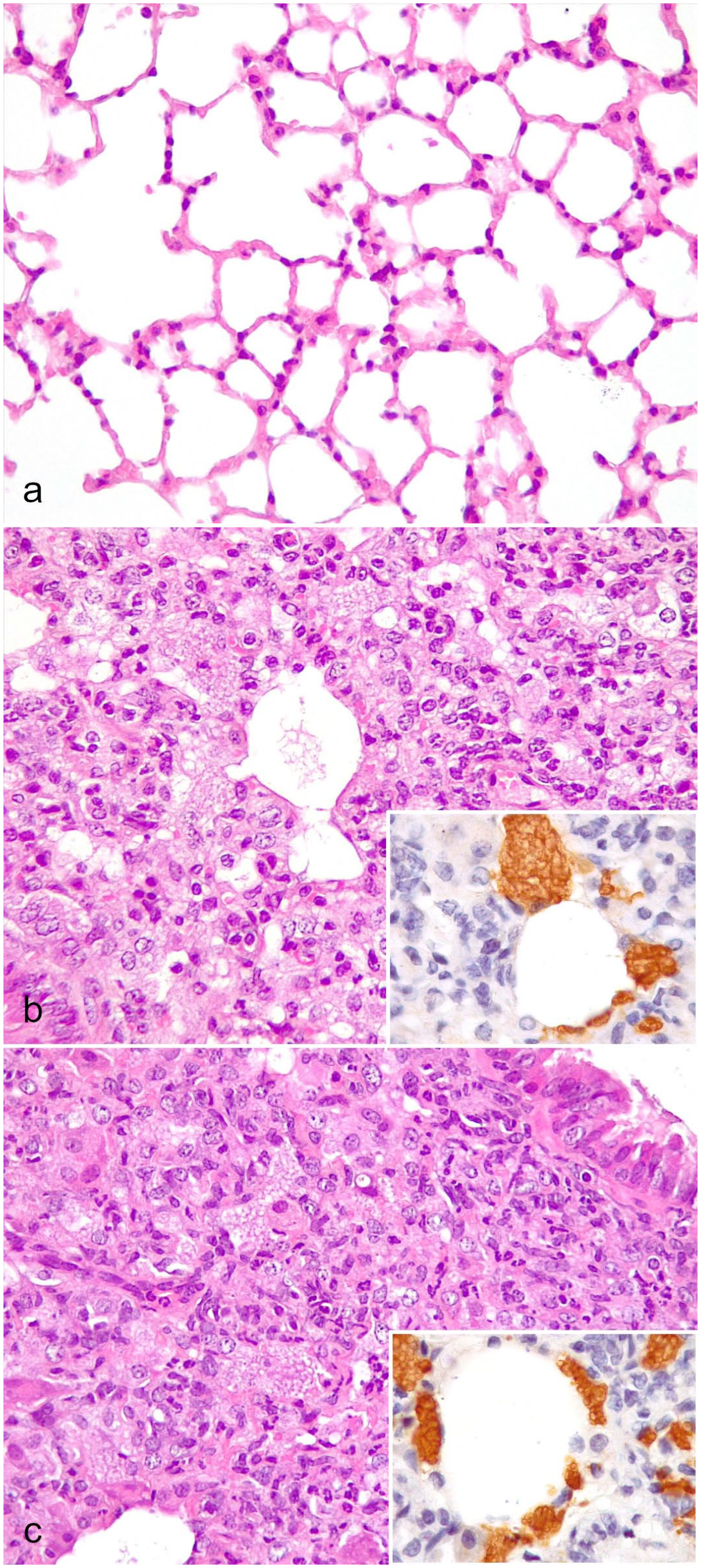
Pneumocystis murina interstitial pneumonia, lung. (a) Uninfected healthy Cd40l–/– mouse. Hematoxylin and eosin (HE). (b) Experimentally infected Cd40l–/– mouse. HE. Inset: numerous immunohistochemically labeled P. murina organisms. Pneumocystis murina Msg immunohistochemistry (IHC). (c) Naturally infected SCID mouse. HE. Inset: numerous immunohistochemically labeled P. murina organisms. Pneumocystis murina Msg IHC.
Frequency of pulmonary findings in Cd40l–/– and SCID mice infected with P. murina. Results are reported as number of affected mice (%).
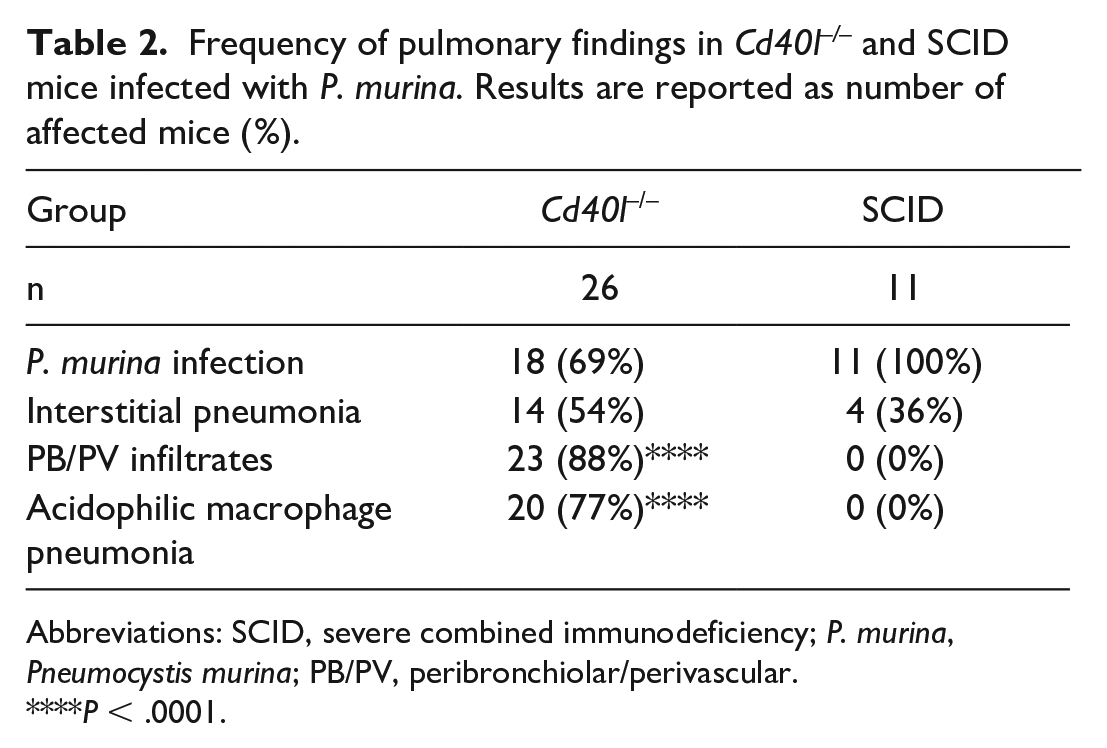
Abbreviations: SCID, severe combined immunodeficiency; P. murina, Pneumocystis murina; PB/PV, peribronchiolar/perivascular.
*P < .0001.
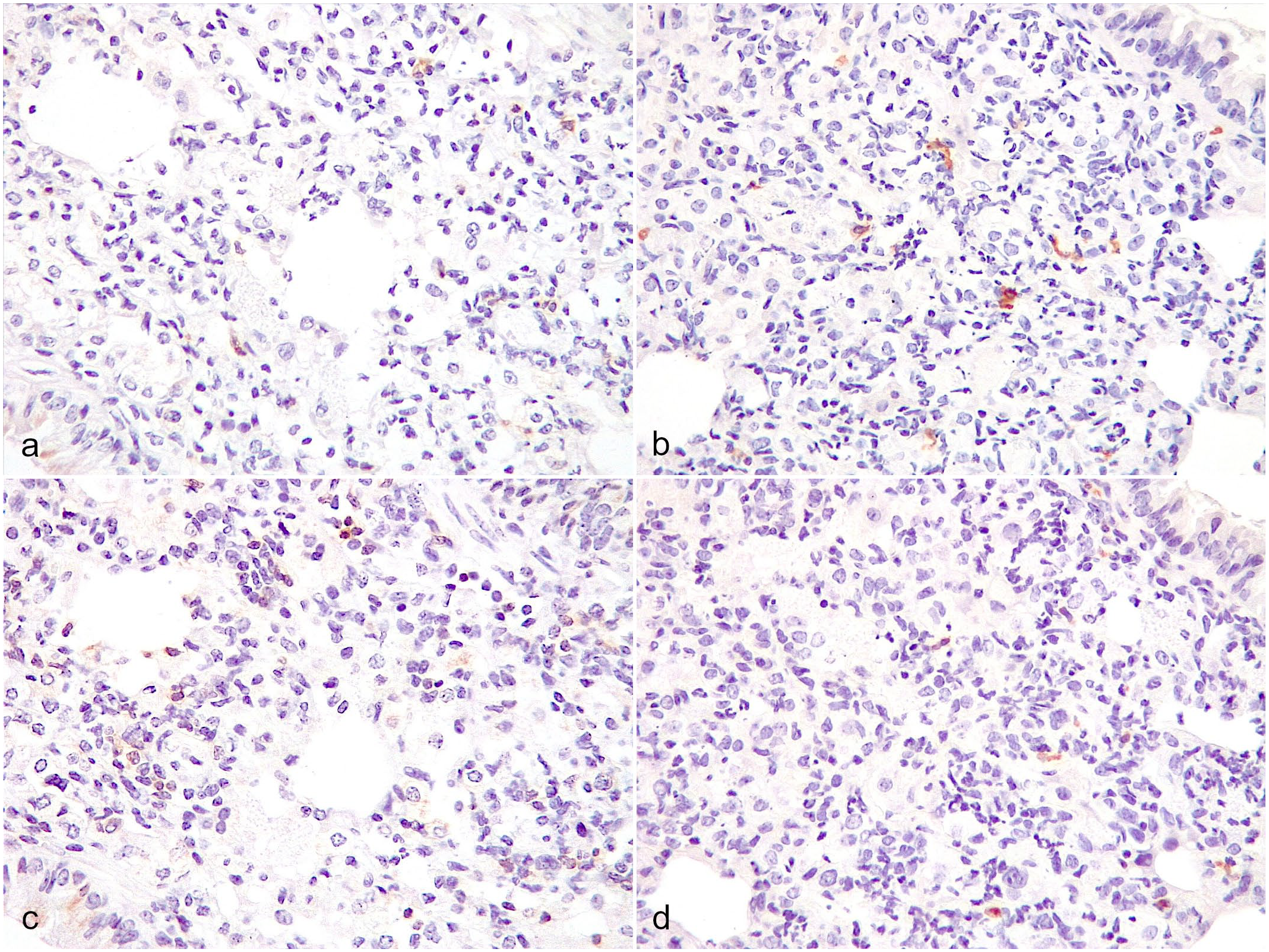
Pneumocystis murina interstitial pneumonia, lung. (a, c) Cd40l–/–mouse and (b, d) SCID mouse. (a, b) Few scattered B-cells with membranous immunolabeling for CD45R/B220. CD45R/B220 immunohistochemistry (IHC). (c, d) Few scattered T-cells with membranous immunolabeling for CD3. CD3 IHC.
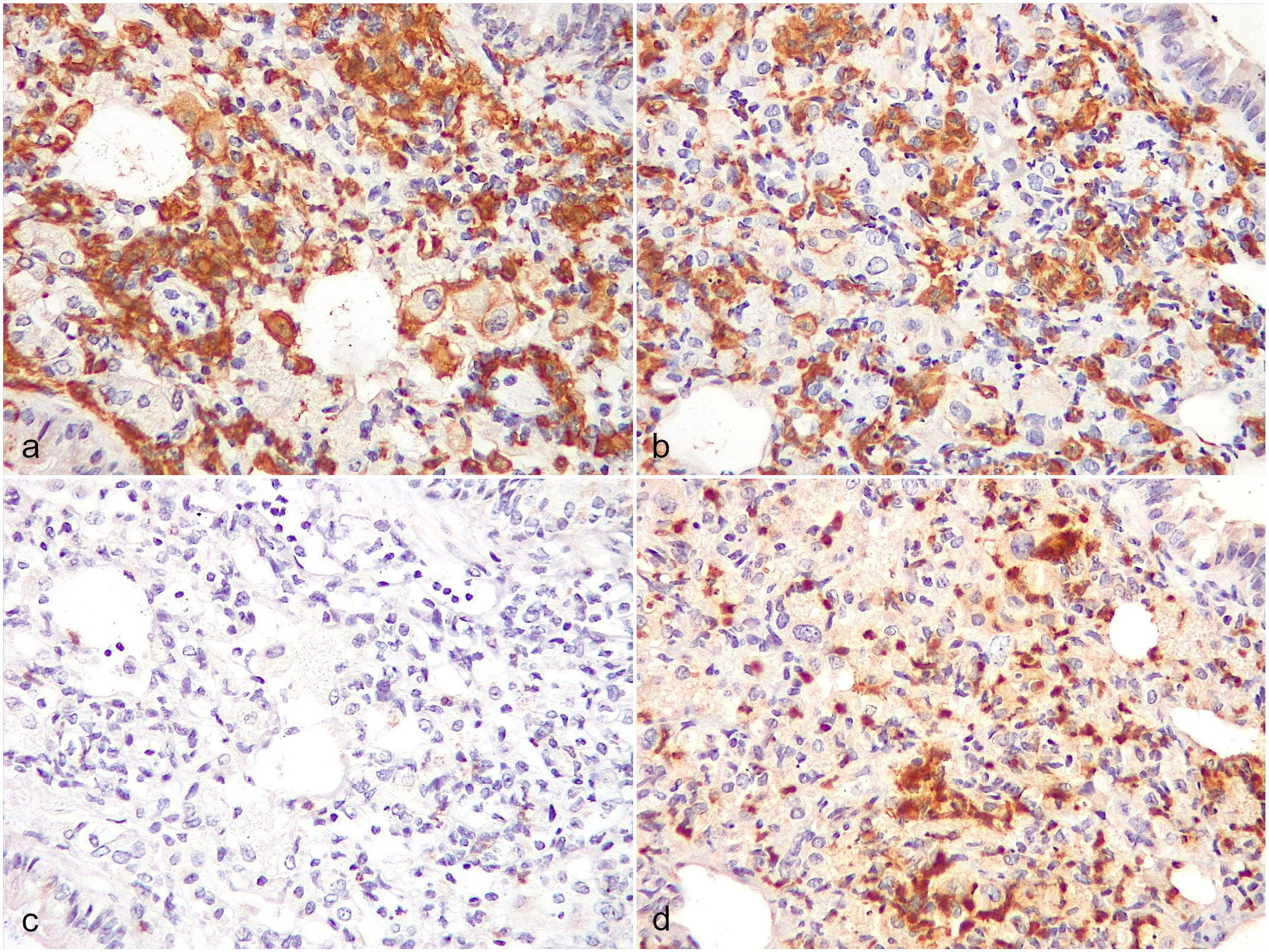
Pneumocystis murina interstitial pneumonia, lung. (a, c) Cd40l–/–mouse and (b, d) SCID mouse. (a, b) Numerous macrophages with cytoplasmic immunolabeling for Iba-1. Iba-1 immunohistochemistry (IHC). (c, d) Few scattered neutrophils with membranous immunolabeling for Ly-6G in the (c) Cd40l–/– mouse, compared to more numerous neutrophils immunolabeled in the (d) SCID mouse. Ly-6G IHC.
No peribronchiolar/perivascular lymphoplasmacytic infiltrates were observed in uninfected Cd40l–/–control mice and SCID mice (Figs. 4a, b). In 23/26 (88%) Cd40l–/–mice, including 7 mice that were negative for P. murina by IHC, multifocal peribronchiolar/perivascular infiltrates composed of lymphocytes and plasma cells, with variable numbers of Mott cells were present (Fig. 4c). In 10 mice, the infiltrates were also organized in follicle-like structures (Fig. 4d). CD45R/B220+ B-cells were prevalent in the peribronchiolar/perivascular infiltrates (Figs. 4e, f). Plasma cells were immunopositive for IgM. A statistically significant correlation was found in Cd40l–/– mice between P. murina burden and peribronchiolar/perivascular lymphoplasmacytic infiltrates (P < .05). In 2/26 (8%) Cd40l–/– mice, peribronchiolar and perivascular sheets of large, atypical CD3+ lymphocytes were found, suggestive of T-cell lymphoma. Both mice were positive for P. murina at IHC.
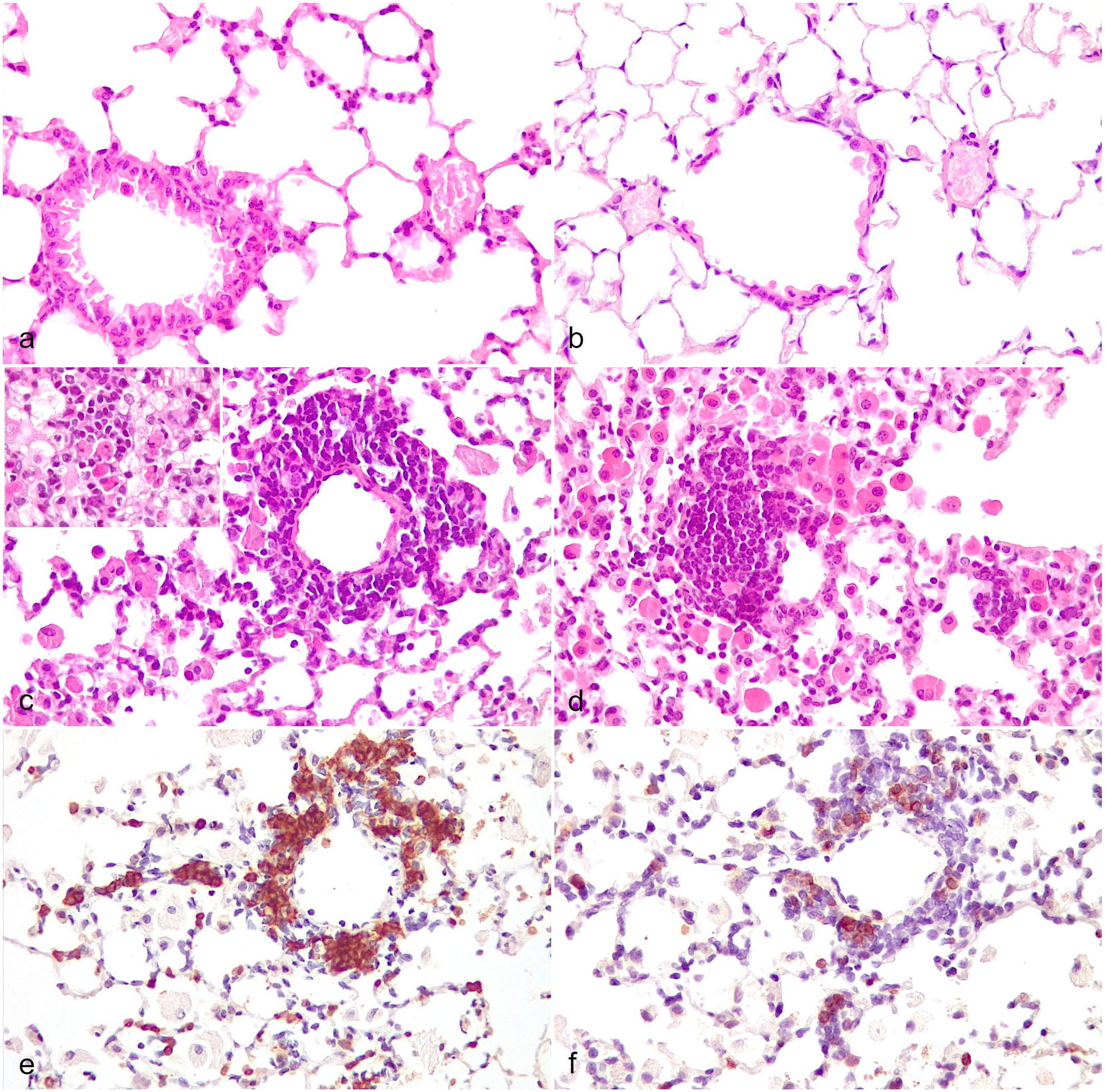
Peribronchiolar and perivascular lymphoplasmacytic infiltrates, lung. (a) Uninfected healthy Cd40l–/– mouse. Hematoxylin and eosin (HE). (b) Naturally infected SCID mouse. Note the absence of peribronchiolar/perivascular infiltrates. HE. (c) Experimentally infected Cd40l–/– mouse. HE. Inset: numerous Mott cells in the infiltrate. (d) Perivascular lymphoplasmacytic inflammatory infiltrate organized in a follicle-like structure in a Cd40l–/– mouse. Note the numerous intra-alveolar macrophages laden with bright eosinophilic crystals, characteristic of acidophilic macrophage pneumonia. HE. (e-f) Perivascular lymphoplasmacytic inflammatory infiltrate in a Cd40l–/– mouse. The infiltrate is almost exclusively composed of (e) B-cells, with a minority of (f) T-cells. (e) CD45R/B220 and (f) CD3 immunohistochemistry.
In 20/26 (77%) Cd40l–/– mice, intra-alveolar macrophages and multinucleated giant cells laden with bright eosinophilic crystals were found (Fig. 5a). Occasional crystals were also detected free in the alveoli or in the lumen of bronchioles. The lesion was consistent with AMP. Macrophages of the AMP lesions were immunopositive for Ym1 (Fig. 5b). No statistically significant correlation was found between P. murina burden and AMP. No AMP was found in uninfected Cd40l–/–control mice and SCID mice.
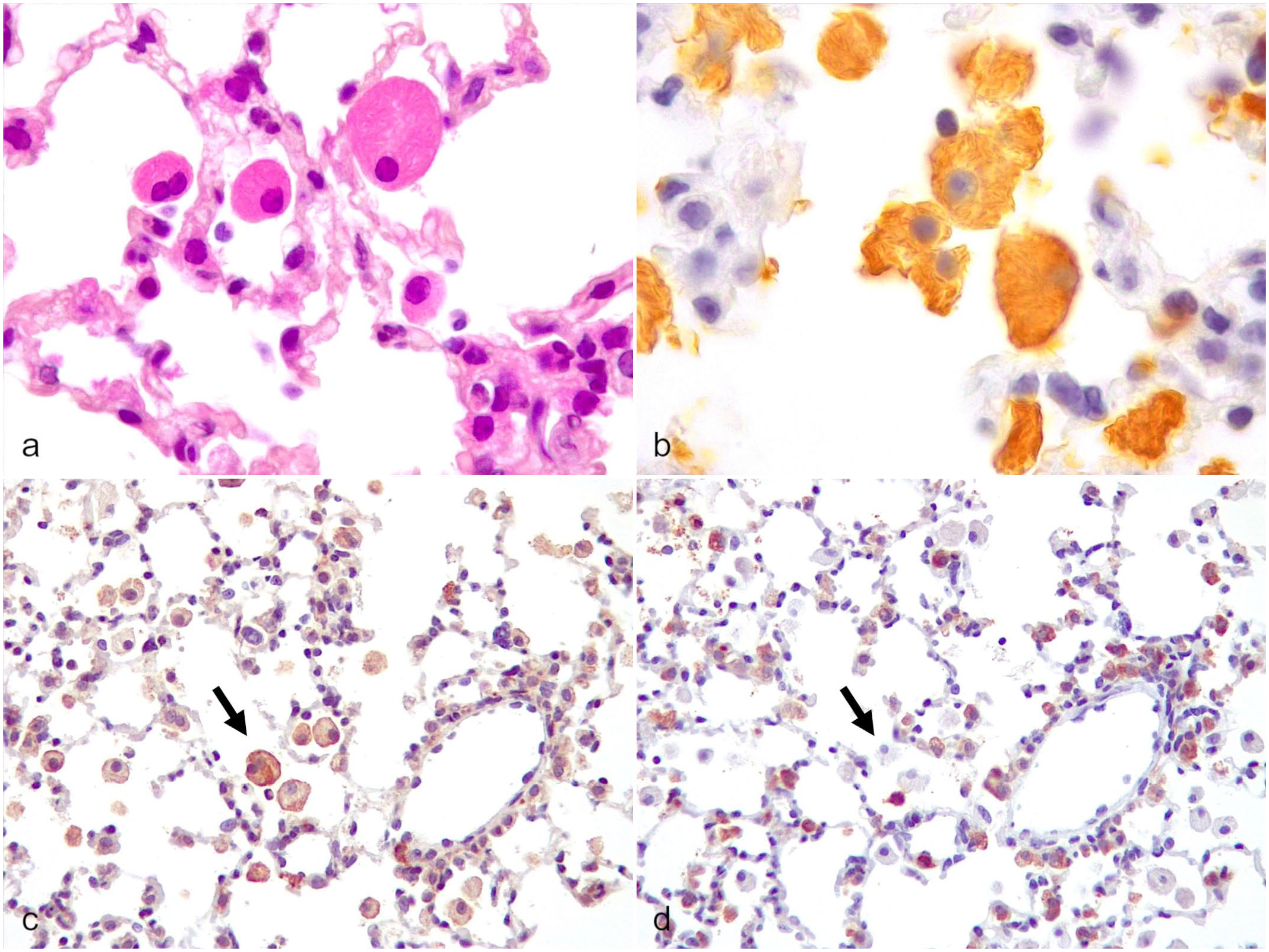
Acidophilic macrophage pneumonia (AMP), lung, Cd40l–/–mouse. (a) Intra-alveolar macrophages laden with bright eosinophilic crystals. Hematoxylin and eosin. (b) AMP macrophages display diffuse cytoplasmic immunolabeling for Ym1. Ym1 immunohistochemistry (IHC). (c) AMP macrophages display strong membranous and weaker cytoplasmic immunolabeling for CD206, indicating alternative activation/M2 polarization (arrow), which is required for clearing the Pneumocystis spp. infection. CD206 IHC. (d) AMP macrophages are negative to MHC II, indicating lack of classical activation/M1 polarization (arrow). MHC II IHC.
To further characterize the subtypes of CD3+ T-cells and macrophages (M1/M2 polarization) in P. murina infected mice, additional IHC for CD4, CD8a, mannose receptor (CD206), and MHC II were performed on 6 Cd40l–/–mice and the 4 affected SCID mice with different severity of interstitial pneumonia (Supplemental Table S1). In Cd40l–/– mice, there was a higher prevalence of CD8+ than CD4+ T-cells and MHC II+ than CD206+ macrophages in the interstitial pneumonia (Supplemental Figures S2a, c). In SCID mice, there were occasional CD4+ and CD8+ T-cells in the interstitial pneumonia, whereas macrophages were mainly MHC II+, with only few CD206+ macrophages (Supplemental Figures S2b, d).
In the peribronchiolar/perivascular lymphoplasmacytic infiltrates, CD4+ T-cells were predominant in Cd40l–/– mice with higher burdens of the pathogen and/or with more severe interstitial pneumonia, whereas CD8+ T-cells were prevalent in cases of less severe infection. In the peribronchiolar/perivascular infiltrates, MHC II+ macrophages were prevalent, and only occasional CD206+ macrophages were present. Macrophages of the AMP lesions were CD206+ and MHC II–, indicating an M2 phenotype (Figs. 5c, d).
Discussion
In this study, we described pulmonary lesions caused by P. murina after experimental intranasal infection in Cd40l–/–mice, an animal model that recapitulates the HIGM1 of humans, and we compared the results with spontaneously infected SCID mice. Despite the limitation of comparing experimental infection versus natural infection (i.e. standardized vs uncontrolled dose of infection), the aim of our study was to describe P. murina-induced pulmonary lesions in a still poorly characterized model of opportunistic infection like the Cd40l–/– mouse, in comparison to those occurring in a well-characterized immunodeficient model like the SCID mouse.
Interstitial pneumonia was identified in both SCID and Cd40l–/– mice and was always associated with presence of intra-alveolar organisms. The prevalence of interstitial pneumonia was higher in Cd40l–/– mice than in SCID mice (54% vs 36%, respectively). Moreover, in SCID mice, interstitial pneumonia was present only when large numbers of intra-alveolar organisms were identified.
In contrast to SCID mice, peribronchiolar/perivascular lymphoplasmacytic infiltrates were consistently found in Cd40l–/– mice. Peribronchiolar/perivascular lymphoplasmacytic infiltrates are common findings in mice exposed to a variety of antigenic stimuli, including infectious agents, toxic compounds, and tumors. 33 In the context of our study, these were considered specific for P. murina infection, since no lesions were identified in the lungs of uninfected Cd40l–/– mice. In some cases, the infiltrates were organized into B-cell rich follicle-like structures, suggesting a possible early attempt toward the formation of organized tertiary lymphoid structures. 32 In support of this potential, inducible bronchus-associated lymphoid tissue formation was recently demonstrated in C57BL/6 mice infected with P. murina. 13 However, no germinal center formation was observed in the follicle-like structures, thus confirming the immunological impairment of the Cd40l–/– mice. 34
Our results are in contrast with those reported in the only study previously performed in Cd40l–/– mice, where no pulmonary lesions due to Pneumocystis spp. infection were reported. 18 However, in that study, the infection was transmitted by co-housing study animals with seeder infected mice, 18 while in our case, intranasal infection was used. The substantially larger organism load delivered by intranasal inoculation likely accounts for the different quality and magnitude of immune response in our study.18,27
In line with other studies, 46 we observed a low prevalence of interstitial pneumonia in SCID mice. The low prevalence of an inflammatory response is most likely due to the constitutive lack of both B- and T-cells, the latter of which are particularly required for the activation of macrophages via interferon-gamma production.28,48,47 When an inflammatory response to Pneumocystis spp. was reported in SCID mice, the formation of peribronchiolar and perivascular lymphoplasmacytic infiltrates was observed only in experimentally infected mice,7,10,35,36 confirming that the conditions of infection and the organism burden are major factors involved in the outcome of the infection.18,27
We found higher numbers of neutrophils in the interstitial pneumonia of SCID mice compared to Cd40l–/–. The influx of neutrophils was previously reported in P. carinii-infected SCID mice and was thought to be due to a CD4+ T-lymphocyte-independent aberrant inflammatory response that is incapable of clearing the infection. 46 In support of this hypothesis, an interesting model of host response during Pneumocystis spp. infection was recently published. 9 Our results further demonstrate that the Cd40l–/–mouse is not deficient in CD4+ T-cells; instead its defect being in the function of CD4+ T-cells (due to the lack of CD40L), rather than in their number. 34
In our study, P. murina was not detected by IHC in 8/26 Cd40l–/–mice in which the infection was previously confirmed by PCR. This could be explained by a low sensitivity of IHC as compared to molecular methods.
Iba-1+ macrophages represented the main population of inflammatory cells in interstitial pneumonia of both Cd40l–/– and SCID mice. The major role of macrophages in the elimination of Pneumocystis spp. is well known.9,31 We suppose that, despite the immunological defect of the Cd40l–/–mouse, an alternative mechanism of T-cell-mediated macrophagic activation by means of interferon-gamma production could be preserved in this mouse. This was also demonstrated in HIGM1 patients, whom CD40L-deficient T-cells showed little if any impairment in the ability to produce cytokines.25,40
The peribronchiolar and perivascular lymphoplasmacytic infiltrates in Cd40l–/– mice were almost exclusively composed of CD45R/B220+ B-cells. This might represent a typical feature of this model. We hypothesize that the lack of an effective activation of B-cells and production of IgGs might result in accumulation of B-cells and plasma cells with aberrant production of IgMs that are not sufficient to clear the infection. This feature of an “overwhelmed” immune response is supported by the notable number of Mott cells in the infiltrates. By definition, Mott cells are IgM-producing plasma cells defective in immunoglobulin secretion, 1 and their presence is thus perfectly fitting with the immunological status of the Cd40l–/–mouse model.
Regarding mice with pulmonary lymphoma, organs other than lungs were not available for examination; thus, we cannot exclude that these were part of a “spontaneous” multicentric neoplasia. This is supported by a high incidence of leukocytic tumors in the C57BL/6 strain. 23 Although the incidence of lymphoma is reported to be increased in some immunodeficient strains of mice,11,22,39 to date, there is no evidence that the Cd40l–/– mouse is predisposed to developing lymphoproliferative disorders.
Acidophilic macrophage pneumonia was another common finding in Cd40l–/–mice in our study. Acidophilic macrophage pneumonia is considered an idiopathic pulmonary disease of laboratory mice, more frequently occurring in C57BL/6 and 129Sv strains.14,19,29 Moreover, it was reported to be enhanced in P. carinii infection.14,19,29 This is supported by the fact that AMP macrophages are alveolar macrophages, the first line of defense against Pneumocystis spp.16,30 In line with this observation, we demonstrated that AMP macrophages have an M2 phenotype, which is the one required for clearing the Pneumocystis spp. infection effectively;9,30 therefore, suggesting that AMP could arise (or be worsened) as an attempt to clear the pathogen.
This study represents the first description of pulmonary histopathological lesions in Cd40l–/– mice infected with P. murina. We demonstrated that Cd40l–/– mice are susceptible to infection with P. murina and mount an inflammatory response in the attempt to clear the infection, recapitulating the susceptibility of HIGM1 patients to similar opportunistic pathogens. Notably, findings like the B-cell- and Mott cell-rich peribronchiolar and perivascular infiltrates in the lungs of our mice seem to be specific features of the model. We believe that our data could contribute to a better understanding of the inflammatory response patterns in the Cd40l–/– mouse, with possible useful implications for translational research.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241252409 – Supplemental material for Pneumocystis murina lesions in lungs of experimentally infected Cd40l–/– mice
Supplemental material, sj-pdf-1-vet-10.1177_03009858241252409 for Pneumocystis murina lesions in lungs of experimentally infected Cd40l–/– mice by Andrea Cappelleri, Simone Canesi, Luca Bertola, Valentina Capo, Alessandra Zecchillo, Luisa Albano, Anna Villa, Eugenio Scanziani and Camilla Recordati in Veterinary Pathology
Supplemental Material
sj-xlsx-2-vet-10.1177_03009858241252409 – Supplemental material for Pneumocystis murina lesions in lungs of experimentally infected Cd40l–/– mice
Supplemental material, sj-xlsx-2-vet-10.1177_03009858241252409 for Pneumocystis murina lesions in lungs of experimentally infected Cd40l–/– mice by Andrea Cappelleri, Simone Canesi, Luca Bertola, Valentina Capo, Alessandra Zecchillo, Luisa Albano, Anna Villa, Eugenio Scanziani and Camilla Recordati in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank JA Kovacs and LR Bishop (NIH Clinical Center, National Institutes of Health, Bethesda, MD, USA) for providing the antibody against P. murina.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Italian Ministry for Universities and Research (grant no. PRIN 2017 Prot. 20175XHBPN).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
