Abstract
Mice are commonly used as an experimental model to investigate the Equid herpesvirus 1 (EHV-1) infection. This model easily reproduces the disease, and the clinical signs are more or less similar to those observed in the horse, the natural host. During natural infection, the acute course of respiratory infection is mandatory for the development of adaptive immune response. Since interactions between EHV-1 and anesthetics are possible, the study investigated whether the early events of murine pulmonary immune response could be affected by different anesthetics. Therefore, mice were experimentally infected with a unique EHV-1 strain under the effects of ether, ketamine/xylazine, or isoflurane. Clinical signs and histopathological lesions in the lungs were described, and the cell death and proliferation rates of sham-inoculated or infected animals were quantified using immunohistochemistry. Clinical signs were more severe in animals anesthetized with ether. Qualitative differences in the recruited inflammatory cells were observed following application of anesthesia. The level of infection between the infected groups was not statistically significant. However, lungs from ketamine/xylazine-anesthetized animals showed the highest cell death rates, whereas those from isoflurane-anesthetized animals showed the highest proliferation rates. It has been emphasized that anesthetics alone or their interactions with EHV-1 modify the response against the infection. An appropriate selection of the anesthetic during experimental studies is relevant to minimize wrong conclusions.
The family Herpesviridae includes a large number of viruses that infect a wide range of hosts. Equid herpesvirus (EHV-1) is a varicellovirus belonging to the subfamily Alphaherpesvirinae and causes a worldwide epidemic of abortion, perinatal mortality, respiratory disease, and neurological disorders in horses. 19 EHV-1 enters the host through the respiratory airways and causes respiratory disease. The virus has the ability to remain latent in lymphoid cells and neurons of the trigeminal ganglia. 17,23 Reactivation frequently occurs under immune depression. 10 A leukocyte-associated viremia allows the virus to reach the uterus and the central nervous system, causing reproductive and neurological disorders. 30 Primary and reactivated herpesvirus infections can be asymptomatic or result in diseases of variable severity. 2
Awan et al 4 used a murine model to induce EHV-1 infection, allowing a better understanding of different virological, histopathological, and immunological aspects of the disease. 28 In this study, animals were intranasally inoculated with infected cell culture medium under a light anesthesia. The anesthesia is used to reduce the animal’s suffering and the possible expulsion of the inoculum that may reduce the effectiveness of the infection. In the model proposed by Awan et al, 4 ether was used extensively in many experiences. However, because of the irritant properties to mucous membranes, especially those of the respiratory tract, and the significant risk of fire or explosion, this popular anesthetic was replaced by ketamine/xylazine 13 or by isoflurane. 14 Several studies have suggested that anesthetics may alter tissue homeostasis. 5,9,25 So far, the possible effects of such anesthetics on EHV-1 infection have not been investigated.
Therefore, the aim of this work was to study possible differences in clinical signs, histopathological lesions, cell proliferation, and death rates in sham-inoculated and infected mice under the effects of 3 anesthetics.
Material and Methods
Experimental Design
To explore the effects of ether in the murine model, sections of paraffin-embedded lungs from a previous experiment were studied. One of the researchers of our group actively participated in the experimental proceeding of that assay (C.M.G.). In that experiment, 10 specific pathogen–free BALB/c male mice that were 6 weeks old were anesthetized with ether and intranasally inoculated with EHV-1 (AR4 strain) or sham-inoculated. All the experimental conditions were rigorously registered.
Two groups of 10 mice each were anesthetized with ketamine/xylazine and isoflurane, respectively; then 5 animals of each of these groups were intranasally inoculated with AR4 EHV-1 strain and the other 5 were sham-inoculated. All the animals shared the same age, strain, and gender and had the same microbiological status. Two days post inoculation, mice were anesthetized with the same anesthetics selected for the inoculation and then were killed by exsanguination. Volatile compounds including ether (Merck, Darmstadt, Germany) and isoflurane (Baxter Co., Deerfield, IL) were dispensed in a closed chamber until animal were unconscious. Ketamine (Ketamina, Laboratorios Holiday, Buenos Aires, Argentina) (drug dose 50 mg/kg) and xylazine (Xilacina clorhidrato; Laboratorios Ritchmond, Buenos Aires, Argentina) (drug dose 10 mg/kg) were intraperitoneally administered.
Virus Strain and Intranasal Inoculation
The abortigenic EHV-1 strain (AR4) was isolated in Argentina from a foal that died 24 hours after birth. 20 After 4 passages in RK13 cells (rabbit kidney), a viral stock was prepared, titrated by standard Reed and Muench method, and stored at –70°C in liquid nitrogen. Aliquots of this viral strain stock were propagated in RK13 cells and used in both assays. Cell cultures were allowed to grow with a minimal essential medium supplemented with 10% fetal calf serum and titrated as previously described. 12 Fifty microliters of supernatant (105.75 CCID50/50 μl) or 50 μl of virus-free cell culture was intranasally inoculated in the animals of both experiments. 12
Clinical Assessment
Mice were examined at the beginning and at the end of the experiment. Clinical signs were recorded as highly severe, severe, or mild by the same operator in both experiments (C.M.G.). The current proceedings were revised and approved by the Institutional Animal Care and Use Committee of the Faculty of Veterinary Sciences of La Plata National University and were performed in accordance with the Guide for the Care and Use of Laboratory Animals of the National Research Council.
Histopathological Analysis
Tissue processing and histopathological analysis
Lungs were fixed in 10% buffered formaldehyde (pH 7.4) for 12 hours and then dehydrated, clarified, and embedded in paraffin wax. Paraffin-embedded sections (3 μm thick) were stained with hematoxylin and eosin (HE). Morphological descriptions were conducted by 2 independent researchers (M.L.E., C.N.Z.).
Immunohistochemistry
The magnitude of the infection and the proliferation and death rates in the sites of infection by EHV-1 were studied using immunohistochemistry to detect EHV-1 antigen proliferating cell nuclear antigen (PCNA) and caspase 3 antigen, respectively.
Sections (3 μm thick) were mounted on positively charged slides (Biotraza microscope slides, Cat. #HDAS001A, Huida Medical Instruments Co., Jiangsu, China), deparaffinized, dehydrated, incubated with 3% H2O2 in methanol for 30 minutes at room temperature, and rinsed in phosphate-buffered saline (PBS, pH 7.4). Antigen retrieval was applied using 750 W of microwave irradiation twice for 5 minutes in citrate buffer (pH 6.0). Nonspecific binding sites were blocked with 2% bovine serum albumin (BSA) + 5% skim milk (for EHV-1 antigen) or 1% BSA (for PCNA and caspase 3 antigens) for 30 minutes in a humid chamber at 4°C, followed by the primary antibody incubation for 1 hour at 37°C. Liquid 3,3-diaminobenzidine tetrahydrochloride (DAB) was used as chromogen (DakoCytomation, Glostrup, Denmark), and Hill’s hematoxylin was used for counterstaining. 33
Detection of EHV-1 antigen
A polyclonal anti-EHV-1 rabbit serum produced in our laboratory was used as primary antibody (1/1500 dilution). Slides were incubated for 1 hour at 37°C, rinsed in PBS 3 times for 5 minutes each, and incubated with anti-rabbit EnVision detection system+HRP (DakoCytomation) for 30 minutes at room temperature. Then sections were rinsed in PBS, revealed, and counterstained. In each slide, a section of a positive infected sample was included. As a negative control, the primary antibody was replaced with a normal rabbit serum. 33
Proliferation assessment
Mouse monoclonal anti-PCNA was used as a primary antibody (clone PC 10, ascites fluid, Sigma Chemical Co., St. Louis, MO, EE.UU, 1/3000 dilution). The primary antibody was pretreated with Dako ARK (Animal Research Kit) Peroxidase System (code K3954) to reduce the possible reactivity of the detection system with endogenous immunoglobulin. Nonspecific binding sites of endogenous biotin were blocked with an avidin-biotin blocking kit (Cat. #HK102-5K, Biogenex, Fremont, CA). The antibody-Dako ARK polylinker complex was then incubated for 1 hour at 37°C, rinsed in PBS 3 times for 5 minutes each, revealed, and counterstained. 33
Apoptosis assessment
The sections were incubated with rabbit anti-caspase 3 (active form) polyclonal antibody (1/50 dilution) (Cat. #AB3623 Chemicon International, Inc., Temecula, CA) overnight at 4°C. Anti-rabbit EnVision detection system+HRP (DakoCytomation) was then used for 30 minutes at room temperature. Sections were then rinsed in PBS twice for 5 minutes, treated with DAB, and counterstained.
Infection, Cell Proliferation, and Cell Death Quantification
The quantification of these parameters was performed by digital image analysis with the program Image-Pro Plus 6.0 (MediaCybernetics, Bethesda, MD). A total of 20 digital images (40× magnification) for each animal were captured. The areas of bronchus/bronchiole were manually selected, and the ratio of immunostained tissue for EHV-1 antigen versus total selected area was measured. PCNA and caspase 3 immunostained cells were counted. A set of pixels were defined as immunostained tissue, and the clusters of pixels, whose size was in accordance with nucleus (PCNA) or cell size (caspase 3), were counted.
Antigen stability was tested comparing the staining intensity of tissue samples (archival and current samples) with the same positive control.
Statistical Analysis
To compare mean values in the infection ratio, 1-way analysis of variance (ANOVA) was performed followed by multiple comparisons Tukey test to compare mean values of infection. When proliferation rates were compared, interactions between anesthetic and infection were studied using 2-way ANOVA and Tukey test, after logarithmic data transformation. Homoscedasticity was evaluated using the Levene test. Because of the nonhomoscedastic nature of caspase 3 data distributions, nonparametric tests were performed. Mann-Whitney test was performed to compare the mean value of sham-inoculated groups versus infected groups for each anesthetized animal. Kruskal-Wallis test was used to compare the anesthetics effects within sham-inoculated and infected groups. The validation of antigen stability was analyzed using Dunnett multiple comparisons test.
Results
Clinical Signs
Three sham-inoculated animals anesthetized with ether showed mild or severe dyspnea 24 hours post inoculation, whereas no clinical signs were observed in the other 2 sham-inoculated animals. All infected mice showed depression, dyspnea, tachypnea, ruffled fur, and hunched posture. These signs were mild in the groups anesthetized with ketamine/xylazine or isoflurane, and they were highly severe in the infected mice anesthetized with ether.
Histopathological Findings
No histological evidence of infection was observed in the lungs of sham-inoculated mice anesthetized with ketamine/xylazine (Fig. 1) or isoflurane, although the lungs from the latter showed slightly thickened interalveolar walls (Fig. 2). Mild inflammatory reactions were found in the lungs from sham-inoculated mice anesthetized with ether. In such animals, lymphocytic and neutrophilic infiltration was observed around bronchi, bronchioles, and blood vessels. Furthermore, interalveolar walls appeared thickened, and erosion foci were present in bronchial and bronchiolar epithelia (Fig. 3).
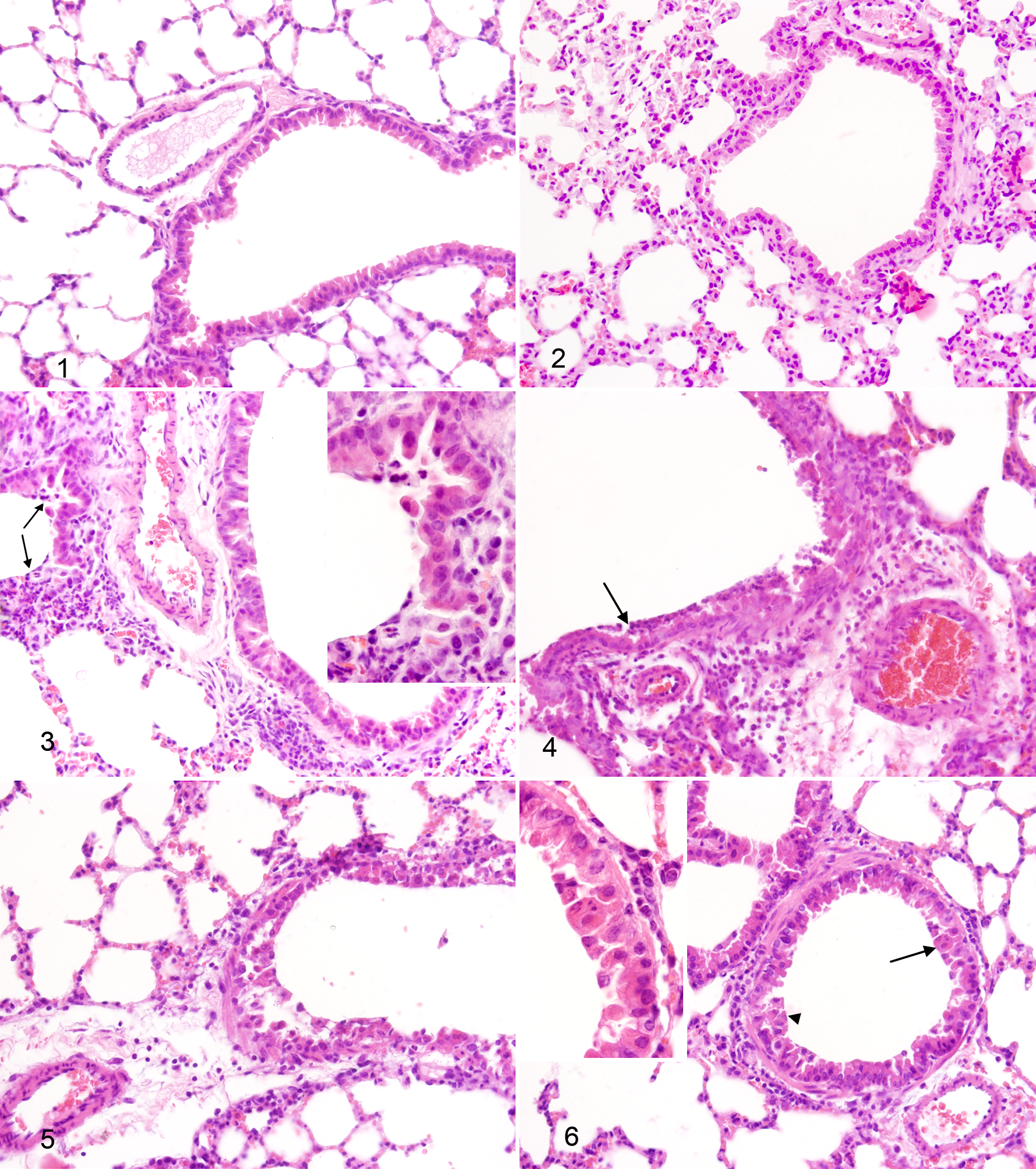
Lung, mice. Sham-inoculated (control) animal. No histopathological lesions were found in lungs from sham-inoculated animals anesthetized with ketamine/xylazine. HE.
Lungs of all infected mice showed typical foci of EHV-1 infection in bronchial and bronchiolar epithelia, including desquamation, syncytia, and epithelial cells with eosinophilic intranuclear inclusion bodies. Although the morphological changes induced by EHV-1 infection were similar in all groups, recruited inflammatory cells were qualitatively varied. Lungs of infected mice anesthetized with ether showed severe neutrophilic infiltration and, to a lesser extent, monocytes in the bronchiolar lumen and lymphocytes surrounding bronchioles and blood vessels (Fig. 4). In the lungs of infected mice anesthetized with ketamine/xylazine (Fig. 5) or isoflurane (Fig. 6), there was thickening of alveolar walls and lymphocytic infiltration surrounding respiratory airways and blood vessels.
Immunohistochemistry
No statistically significant difference was found in the intensity of EHV-1, PCNA, or caspase 3 immunostaining in all infected animals.
EHV-1 Antigen
EHV-1 immune reactivity was found in bronchial and bronchiolar epithelia and in some alveolar cells in lungs of infected mice (Fig. 7). The level of infection between the infected groups was not statistically significant (P > .09; ANOVA) (Fig. 10).
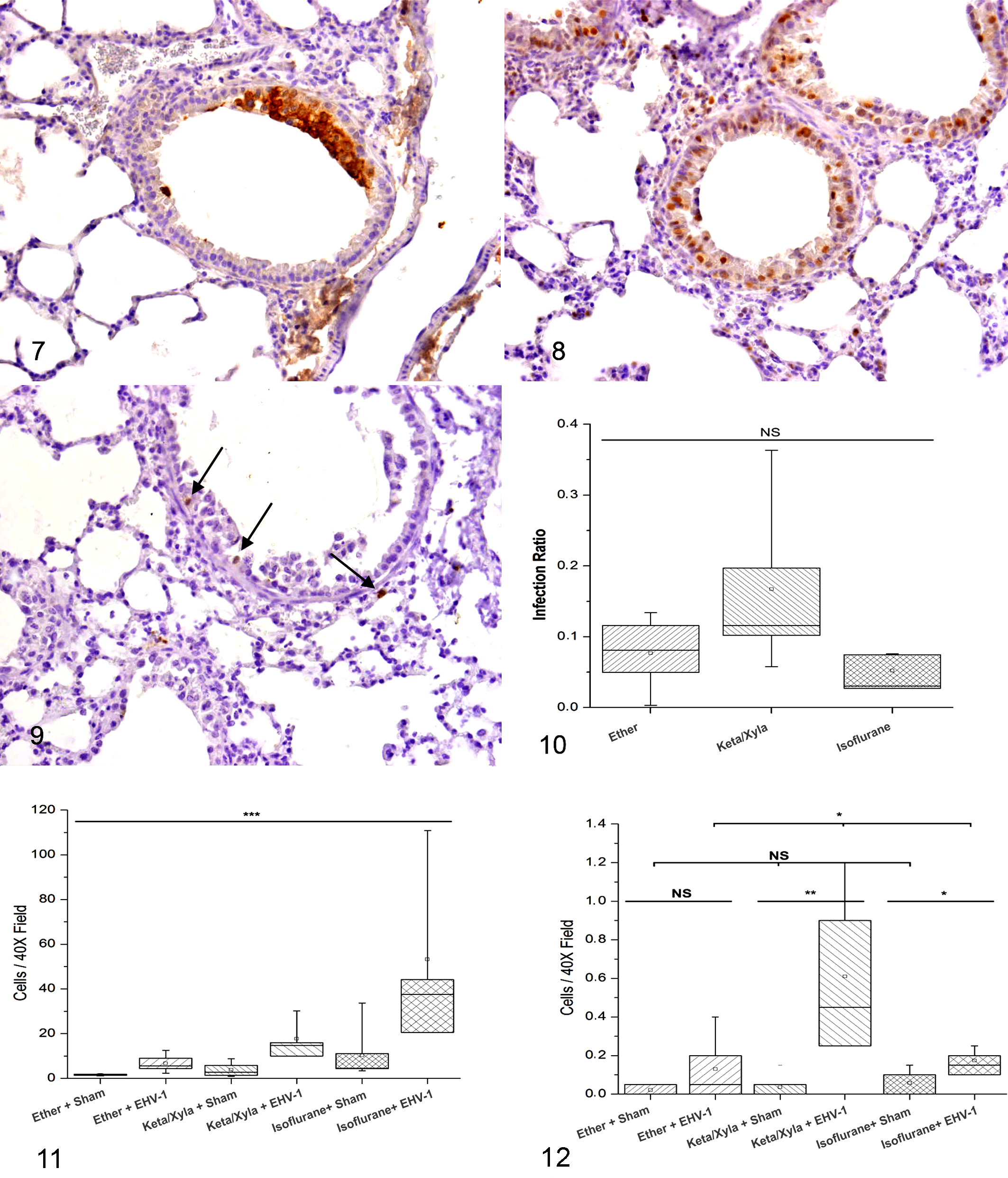
Lung, mice. Immunochemical labeling for EHV-1 antigen at the bronchiolar epithelial cells from an infected animal anesthetized with ketamine/xylazine. Diaminobenzidine chromogen, hematoxylin counterstain.
Cell proliferation
Lungs from all animals showed the same antigenic distribution pattern. The PCNA-stained nuclei were predominantly found in the epithelium, in some instances, and in some detached cells of the bronchial lumen. Furthermore, some alveolar infected cells were positively stained (Fig. 8). The number of PCNA-positive cells increased in all the infected groups with respect to sham-inoculated groups. Between the infected groups, the number of PCNA-positive cells significantly increased in mice anesthetized with isoflurane, whereas no significant difference was found between sham-inoculated groups (Fig. 11).
Cell death
The number of caspase 3–positive cells in the lungs of infected animals increased with respect to sham-inoculated animals for all the anesthetics, although this increase was not significant for ether. The highest number of immunostained cells was observed in the lungs of infected animals anesthetized with ketamine/xylazine (P < .05, Kruskal-Wallis test; Fig. 12). The distribution pattern of caspase 3–positive immunostained cells was found surrounding the infection foci and not within them (Fig. 9).
Discussion
In this work we used a murine model of EHV-1 infection under the effect of 3 different anesthetics to study the clinical signs, histopathological lesions, and cell proliferation and death rates in the lungs. Infected animals anesthetized with ether showed the most severe clinical signs and histopathological lesions. However, no relationship was observed between the intensity of the histopathological lesions and the level of infection determined by immunohistochemistry, since sham-inoculated animals anesthetized with ether also showed dyspnea after inoculation. The infected animals anesthetized with ketamine/xylazine or isoflurane showed mild clinical signs, less intense histopathological lesions, and a higher infection level (although it was not statistically significant). The dyspnea found in animals anesthetized with ether may be more related to the impairment in ventilation caused by the inflammatory changes and the increased thickness of interalveolar walls than to the epithelial loss itself. Other researchers reported that mice infected with influenza virus showed extended pneumonia after ether anesthesia in comparison with other anesthetics. 16 The great number of neutrophils found in lungs of animals anesthetized with ether disagrees with the fact that glycoprotein G (gG) of EHV-1 has the ability to impair neutrophil and monocyte recruitment to the infection foci. 26 Moreover, gG binds to different chemokines, like murine CCL3, 6 and the interaction between these chemokines and gG is mandatory during the primary EHV-1 infection. 27 These findings were obtained using isoflurane in vitro and in vivo. 26 Despite the fact that EHV-1 is able to inhibit neutrophil chemotaxis, we suggest that ether may block or overtake the effect of gG, since in sham-inoculated and infected animals there was a great neutrophilic infiltration.
Ether increases vascular permeability and produces necrosis and irritation of the respiratory airways. 15 As a result, certain molecules are released and activate Toll like-receptor (TLR) 2 and 4. 11 EHV-1 could also activate TLR 2, 3, 7, and 9. 22,31 The pattern of TLR activation is critical to define the innate and adaptive immune response profile. 1 It could be suggested that activation of TLRs by damage associated molecular pattern molecules and by EHV-1 might explain the discrepancies found in mice anesthetized with ether. In addition, histopathological changes described in the lungs of such animals might be related to nonspecific acute inflammatory response rather than the well-known specific pulmonary reaction described in the natural host. 3
Despite similar histopathological changes, lungs from infected animals anesthetized with isoflurane and ketamine/xylazine showed different proliferation and cell death rates, thus suggesting temporal variations in the resolution of the infection reaction. Many studies have reported that ketamine possesses anti-inflammatory properties. 18,25 Moreover, ketamine impairs the release of interleukins (1b, 6, and 8) and tumor necrosis factor alfa (TNF-α) in response to different stimuli 7,8,24,29 and inhibits the maturation of bone marrow-derived dendritic cells and T-helper-1 type immune response priming. 21 In a murine model of burn injury, it was shown that ketamine increases the apoptotic rates in the intestinal epithelium. 32 In addition, the positive immunoreactivity for caspase 3 antigen in lungs of infected animals increased under ketamine anesthesia. This might be explained by the proapoptotic effect of ketamine, although the interaction between the infection and the anesthetics could not be tested. The present study was performed at a particular time of the infection (2 days post inoculation) during which changes under isoflurane anesthesia may occur sooner than those under ketamine/xylazine. As a result, the lower cell death level and the higher proliferation rate found in infected animals anesthetized with isoflurane may indicate the beginning of the regeneration and recovery phase.
The present work emphasizes the relevance of an appropriate selection of the anesthetic during experimental studies, particularly to study EHV-1 infection. Although many advances in the immune response, pathogenesis, or immunogenic capacity of EHV-1 strains have been obtained in experimental assays using different anesthetics, such as ether and ketamine/xylazine, it is clear that undesirable effects caused by the anesthesia or a possible interaction between anesthetics and EHV-1 might force a modified immune response against the natural infection or experimental inoculation. These effects might also induce different changes from those specific to the virus, and the elicited response may lead to wrong outcomes.
In conclusion, the diversity of compounds used as anesthetics might affect the extrapolation and comparison of data between different experimental assays used to reproduce the EHV-1 infection in mice. Therefore, isoflurane could be considered a good anesthetic for such investigations since it produces few known side effects. Besides, animals suffer little distress and show a better postanesthesia recovery.
Footnotes
Acknowledgments
We thank Lic Jimena Barbeito Andrés for her helpful advice with the statistical methods, Dr Enrique L. Portiansky (FCV-UNLP, CCT-CONICET La Plata, Argentina) for his technical assistance in image processing and analysis, and Sr Juan L. Piccioni and Sr Claudio A. Leguizamón for their technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by grants from FONCyT (PICT 2011-1163), CIC PBA, the Department for Science and Technology of the National University of La Plata (SeCyt), and the National Academy of Agronomy and Veterinary (ANAV).
