Abstract
Left ventricular (LV) outflow tract-right atrial (RA) communication associated with bacterial endocarditis is described in a 6-year-old intact male Great Pyrenees dog with a 4- to 5-day history of fever, lethargy, weight loss, severe regenerative anemia, and asplenia. Typical vegetative mural endocardial lesions were observed grossly. Histologic evaluation revealed small gram-negative coccobacilli that were consistent with Bordetella avium-like organisms. These bacteria were associated with severe endocardial inflammation characterized by neutrophilic infiltration, extensive necrosis of endocardium, and fibrin deposition. LV-RA shunt (Gerbode defect) is a rare cardiac defect in humans that can be either congenital or, more rarely, secondary to septic endocarditis, valve replacement procedures, or thoracic trauma. B. avium-like organisms causing septicemia and endocarditis in immunocompromised and asplenic human patients have been described. To our knowledge, no previous descriptions of Gerbode defect associated with bacterial endocarditis in domestic animals have been reported in veterinary literature.
Keywords
Left ventricular (LV) outflow tract-right atrial (RA) communications are rare types of ventricular septal defects (VSDs), known collectively as Gerbode defects. 8 In humans, these defects are usually congenital, 1,8,11,15 but a few cases secondary to bacterial endocarditis have been described. 20,22
Gerbode defects may be divided into two main groups, depending on whether the defect is above or below the insertion of the tricuspid valve in the membranous interventricular septum (MIS). The MIS is divided by the septal leaflet of the tricuspid valve into atrioventricular and interventricular portions (Fig. 1: center). Infravalvular-type defects are considered to be congenital and are always associated with malformation of the tricuspid valve. This Gerbode defect consists of a defect in the interventricular portion of the MIS that is associated with anomalies in the septal leaflet of the tricuspid valve, such as perforations or clefts, fenestrations, aneurysmal transformations, or adhesions of the valvular leaflets to the margins of the defect, that allow LV blood to enter the RA and right ventricle (RV). The second group of Gerbode defects are supravalvular and occur in the atrioventricular portion of the MIS, resulting in a direct LV-RA communication (Fig. 1: center). 1,8,11,15,22 Occasionally, both supravalvular and infravalvular areas are affected. 15 An LV-RA shunt occurs through the communication because of the greater systolic pressure in the LV than in the RA. During diastole, the RA pressure is greater than that in the LV, resulting in blood flow to both ventricles, causing biventricular volume overload, four-chamber enlargement, 1,22 and decreased arterial oxygen saturation. 11 This anomaly is clinically recognized in the neonatal period or during life by the presence of a murmur. 15 Clinical signs in congenital or long-term heart disease are those of congestive heart failure and poor tissue perfusion, including respiratory difficulty, exercise intolerance, depression, weakness, and syncope. 13
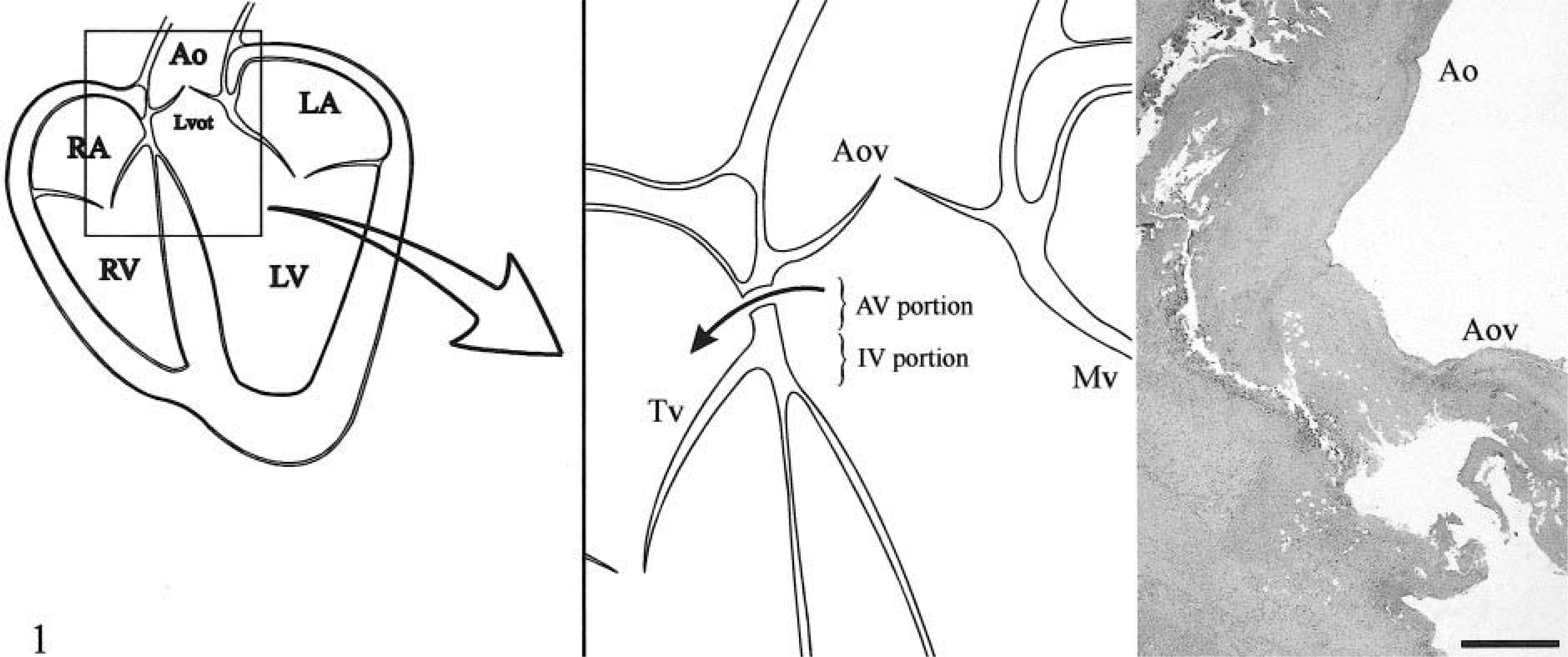
Diagram showing normal anatomic relationships of the interventricular septum to the LV outflow tract, aortic valve, and tricuspid valve.
This report describes the clinical and pathologic features of subacute bacterial mural endocarditis associated with perforation of the interventricular septum in a dog. The perforation resulted in direct communication between the LV outflow tract and the RA.
A 6-year-old intact male Great Pyrenees dog with a previous history of fever, lethargy, depression, anorexia, and weight loss for 4–5 days was submitted for necropsy. Severe regenerative anemia and the presence of Ehrlichia spp. had been demonstrated by hematology. Asplenia was observed by echography; there was no history of previous splenectomy.
The dog was presented in good body condition and had generalized lymphadenomegaly. At necropsy, a 1-cm-diameter fistula through the MIS, between the LV and RA, was observed. From the left heart, the defect was in the LV outflow tract, just below the aortic valve (Fig. 2). From the right heart, the defect was just above the septal leaflet of the tricuspid valve (Fig. 3). There were multiple irregular, rough, yellow-red, friable vegetative lesions in and around the fistula on both sides. The inflammation associated with these vegetations extended to the adjacent aortic valve ring. The aortic valve was slightly thickened and opaque, but there were no vegetations on it. The mitral valve and its chordae tendinae were slightly thickened. The tricuspid valve and cardiac chambers contained no abnormalities. Multiple white-yellow, elevated, soft, wedge-shaped areas consistent with embolic infarcts were detected in both kidneys. The liver was dark red and enlarged, with an acinar pattern, consistent with passive congestion. The small intestine contained blood in the lumen, with multifocal hemorrhages on its mucosal and serosal surfaces. The spleen was absent, and no evidence of laparotomy or splenectomy, such as sutures, scars, or adhesions, was observed. Tissues from the heart and cervical lymph nodes were collected and cultured for bacteria.
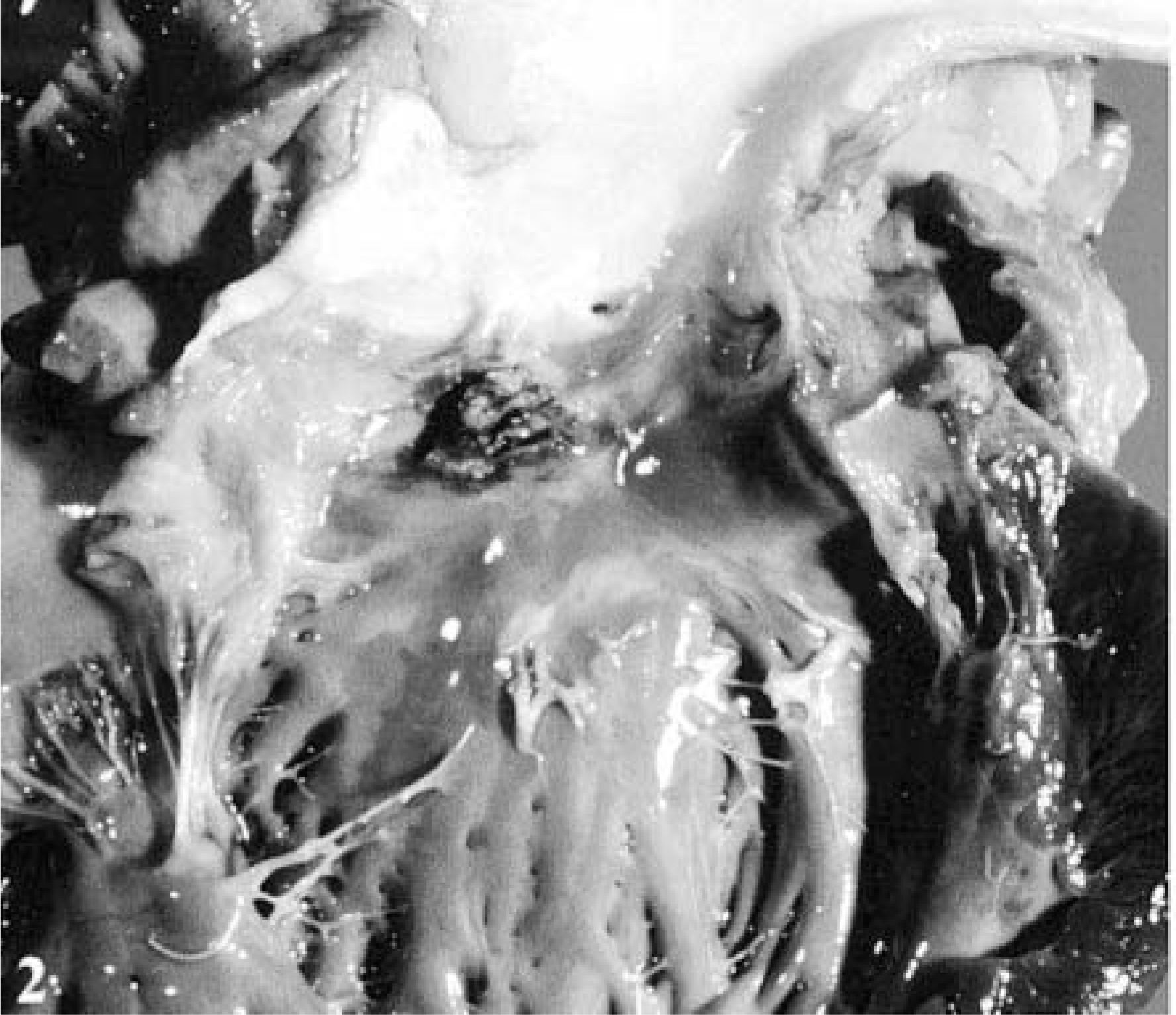
Heart; dog. LV aspect of perforation located in the LV outflow tract, just below the aortic valve. Note the multiple irregular, rough, and friable prominent vegetations.
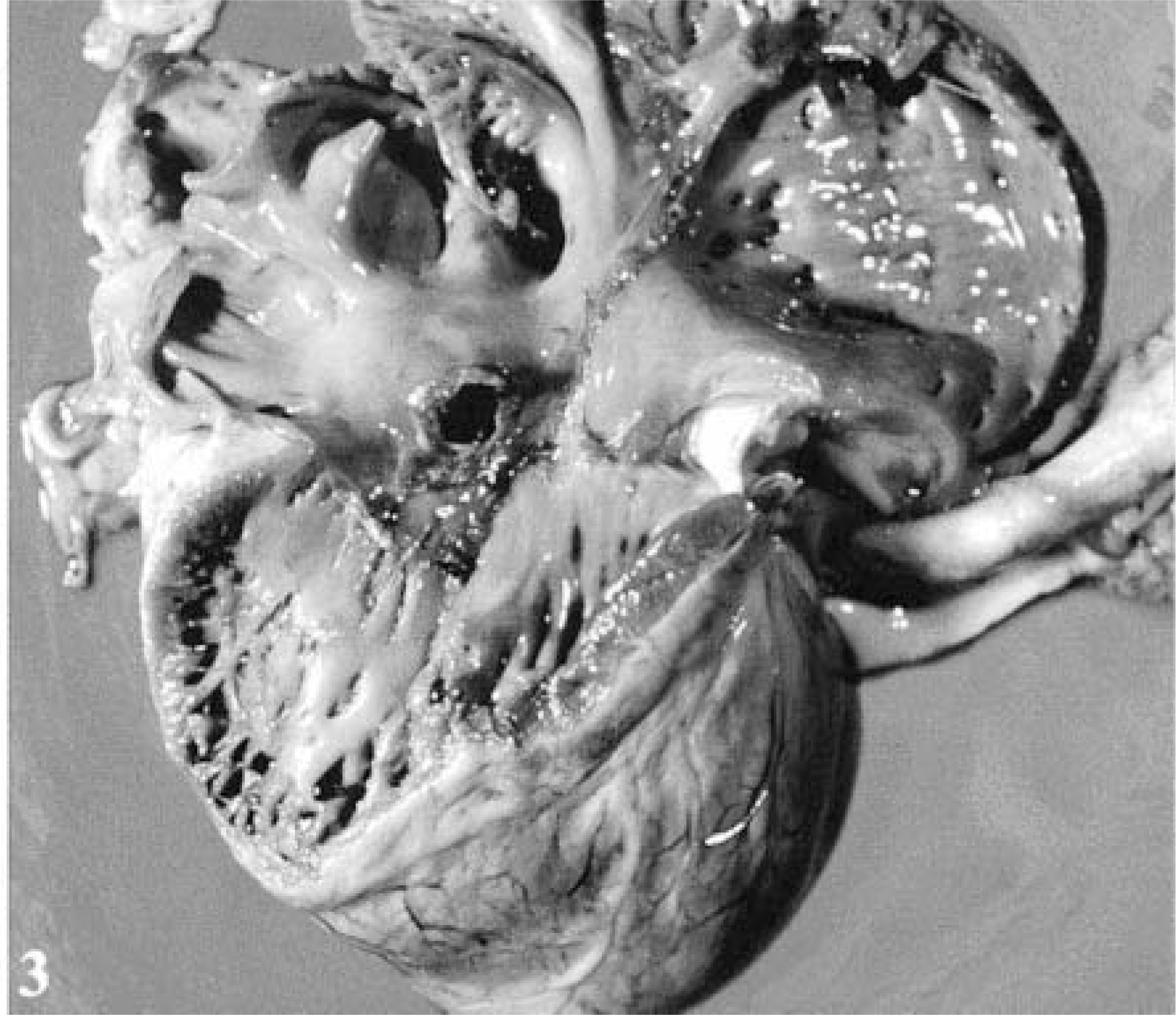
Heart; dog. Right heart view showing the perforation situated above the insertion of the septal leaflet of the tricuspid valve with associated vegetations.
Tissue samples were fixed in 10% neutral buffered formalin, trimmed, processed routinely, embedded in paraffin wax, sectioned at 5–6 µm, and stained with hematoxylin and eosin and with Gram's stain (Brown and Brenn modified method). Histologic evaluation of sections from the heart revealed severe necrotizing endocarditis characterized by infiltration of neutrophils and macrophages (Fig. 1: right). This inflammatory exudate extended to the adjacent aortic valve ring and root of the aortic valve. The inflammation also extended into the adjacent myocardium, where fibroplasia without collagen deposition was prominent. Numerous small gram-negative coccobacilli were seen within these lesions. Cultures of heart and cervical lymph node yielded, on the basis of their biochemical profile, Bordetella avium–like bacteria. The renal cortex contained large areas of ischemic necrosis, inflammation, and bacteria, consistent with septic infarcts. The liver contained dilated sinusoids, with thrombi and atrophy of the centrolobular hepatocytes. Lymph nodes showed diffuse lymphoid hyperplasia. Bacteria identical to those in the heart were observed in subcapsular sinuses of cervical lymph nodes. Intravascular thrombi containing neutrophils and bacteria were present in multiple organs.
On the basis of the gross and histologic features and isolation of B. avium–like organisms, a diagnosis of supravalvular LV-RA communication (Gerbode-type defect) associated with bacterial endocarditis was made.
LV-RA communication is an extremely rare lesion in humans. 1,3,11,15,20,22 We have not been able to find previous descriptions of this lesion in domestic animals. The pathogenesis of the LV-RA communication in this animal is uncertain because it is unclear whether the endocarditis was primary or secondary; Gerbode defects can be either congenital 1,8,11,15 or acquired. 6,11,20,22 In human medicine, congenital Gerbode defects are more frequent than acquired ones and represent about 0.08% of all congenital cardiac lesions. 3,15,22 Congenital LV-RA communication with secondary endocarditis was considered in this animal because congenital heart defects are frequently associated with gastrointestinal, genitourinary, neuromuscular, and, as in this case, lymphoreticular growth anomalies such as asplenia. 14,17 However, this was considered unlikely because there were no previous clinical signs of heart disease and no secondary lesions of heart disease were observed. This defect often causes more marked signs than a conventional VSD because of the greater pressure difference between LV and RA than between the two ventricles and hence an increased flow across the VSD. 3 Human patients with LV-RA shunts have consistently marked enlargement of the RA and RV. Defects of long duration usually result in biventricular enlargement. 1,22 Extracardiac lesions expected with right-side overload include enlargement of the pulmonary artery, congestion of the pulmonary vessels, and chronic passive hepatic congestion. 8 None of these lesions were observed in this animal. Also, Gerbode defects have not been described in conjunction with asplenia. Although congenital asplenia without cardiac defects is a rare phenomenon, it has been described as an isolated defect as a result of a specific defect of spleen development. 9,17
Because the dog in this case had been asymptomatic until 4–5 days before death, had never been found to have a heart murmur, and had had no compensatory changes associated with chronic heart disease, the endocarditis was considered acute and fulminant. The endocarditis may have developed in a membranous defect that was congenital in origin and had closed years earlier by any of the several mechanisms suggested by Anderson et al. (1983). 1 The mechanism most often implicated is redundant tricuspid valve tissue closure of the defect created when a septal aneurysm forms. The residual septal aneurysm could then perforate secondarily to bacterial invasion, giving rise to the acute, fatal condition described. Damaged or denuded vascular endothelium readily promotes the development of platelet-fibrin aggregates and provides a nidus for bacterial invasion and colonization. 13 It is possible that bacterial endocarditis in a previously damaged septum (closed congenital septal defect) resulted in a septal fistula. The second possibility is an acquired septal defect as a consequence of bacterial endocarditis. Acquired Gerbode defects arise from chest trauma, 6 as a complication in aortic 11,22 or mitral valve replacement, 11 and from interventricular septal perforation secondary to infectious endocarditis. 18,20,22 Supravalvular Gerbode defects have been indicated as being more likely acquired than congenital, 22 and bacterial invasion resulting in acute endocarditis in an intact septum is also possible. 18,20 Acute endocarditis is described as a destructive, tumultuous infection by a highly virulent organism, frequently in a previously normal heart. 18 Perforation of the MIS also has been described as a complication of rupture of aortic valve ring abscesses. 7,18 In the animal presented here, the septic, necrotizing endocarditis involved the aortic ring and root of the aortic valve. Because the relatively avascular anulus offers little resistance to infection, extension of an aortic root abscess into contiguous structures is abetted by the high velocity and turbulence of blood flow across the aortic valve. 7 In our opinion, the acute and fulminating endocarditis in the supravalvular septum of this dog with absence of previous clinical signs and heart-related lesions could have resulted from either of these mechanisms, and none of them can be ruled out on the basis of the morphologic findings.
In dogs, the pathogens most frequently found in endocarditis include strains of Streptococcus spp., Escherichia coli, and Erysipelothrix rhusiopathiae. 16 B. avium–like organisms are confirmed pathogens mainly associated with septicemia and endocarditis in humans and respiratory disease in poultry. 5,12,19 These bacteria may be carried normally in the oral or respiratory tract of humans or animals, including dogs, from where they may reach the bloodstream. 12,19 Although the portal of entry was not identified in this case, large and numerous bacterial colonies were present in lymph nodes of the cervical area. Asplenic or immunocompromised patients are at risk for infection with these organisms. 12,19 In general, congenital or acquired asplenia carries an increased risk of life-threatening infections, especially those caused by gram-negative or encapsulated bacteria, 4,9,21,23 which tend to be fulminant in nature. 4,23 Ehrlichiosis is more severe in asplenic individuals as well 4 and per se induces bone marrow suppression. 10 All these factors could have influenced the “easy and rapid” dissemination of these opportunistic bacteria to produce acute endocarditis and death of this animal.
This report represents, as far as we have been able to find, the first case of LV-RA communication associated with bacterial endocarditis in dogs or other domestic animals. B. avium–like bacteria are agents to be suspected as pathogens in immunocompromised animals.
