Abstract
A 9-year-old, spayed, female Maremmano shepherd had a bilateral mastectomy for multiple mammary adenocarcinomas 2 years previous and was referred to the Cardiology Service of the School of Veterinary Medicine of Milan after an acute episode of cardiogenic collapse. Because of severe cardiovascular symptoms and poor prognosis, the dog was euthanized. Necropsy examination revealed the presence of multiple firm grayish neoplastic nodules in the myocardium of the left ventricle and scattered in the pulmonary parenchyma. Neoplastic nodules were also detected in the spleen, pancreas, liver, kidneys, and omentum. Histological examination revealed the coexistence of tubular adenocarcinoma and an undifferentiated sarcoma in the myocardium. Immunohistochemical staining of the sarcoma cells was negative for cytokeratin, desmin, and smooth muscle myosin, thus excluding their epithelial or myoepithelial origin, as well as an origin from smooth muscles cells. These findings, together with the coexpression of vimentin and α-smooth muscle actin, suggested that the sarcoma was derived from myofibroblasts. To the authors' knowledge, this is the first report describing cardiac sarcoma of presumptive myofibroblastic origin in a dog with simultaneous occurrence of cardiac metastasis of mammary gland adenocarcinoma.
Primary and metastatic tumors involving the heart are relatively uncommon in humans and in canine species. 6,24 Primary tumors of the heart occur with the same frequency in the dog and in humans and represent 0.69% and 0.1–0.5% of cardiac tumors, respectively. 15,24 In humans, metastatic tumors are reported to be 20–40 times more frequent than primary ones. 15 In canines, the majority of studies report a high prevalence of primary tumors, 13,24 although in a recent study, a large number of cardiac metastases, mostly carcinomas, have been reported. 2 In dogs, hemangiosarcoma arising in the right atrium is the most common primary cardiac tumor. 13,14 Other cardiac tumors of mesenchymal origin reported in the dog are rhabdomyosarcoma, leiomyosarcoma, fibrosarcoma, and, recently, a case of malignant mixed mesenchymal tumor. 13,14 As far as metastatic tumors in dogs are concerned, cardiac metastases of mammary and pulmonary carcinoma, lymphoma, and malignant melanoma have been most frequently reported. 2,23 This report describes the simultaneous occurrence of 2 different tumors—1 of mesenchymal and the other of epithelial origin—in the myocardium of a dog.
A 9-year-old, 35-kg, spayed, female Maremmano shepherd was referred to the Cardiology Service of the School of Veterinary Medicine of Milan after an acute collapse. Two years earlier, the dog had bilateral mastectomy for multiple neoplastic nodules that were composed of neoplastic cells arranged in tubules and resembling luminal epithelial cells of a mammary gland. These features allowed the diagnosis of multiple mammary simple tubular adenocarcinomas (Fig. 1). At clinical examination, the dog had pale mucous membranes, increased refill time (>2 sec), and weak, arrhythmic, and rapid arterial pulses. On auscultation of the thorax, the heart sounds appeared arrhythmic and rapid; no cardiac murmur was detected. Pulmonary auscultation was unremarkable. The maximum systolic blood pressure, measured by the Doppler method, was 110 mm Hg. A 12-lead electrocardiogram revealed the presence of sustained ventricular tachycardia with a heart rate of about 300 beats per minute. Thoracic radiographs performed in both right lateral and ventrodorsal planes revealed the presence of multiple lung masses ranging from a few millimeters to 10 cm. The largest mass appeared in the cranial right pulmonary lobe and partially covered the heart silhouette. Hematology was within reference limits; serum biochemistry was normal with the exception of the serum cardiac troponin I (cTnI) level, which was 15.88 ng/ml (normal range <0.03–0.07 ng/ml). 20 Because of the poor prognosis, euthanasia was done at request of the owner.
Gross findings observed at necropsy included generalized severe hyperemia of all organs and a small amount of pale reddish fluid in both the abdominal and thoracic cavities. In the abdomen, multiple firm grayish neoplastic nodules, ranging from 1–4 cm in diameter, were in the spleen, pancreas, liver, kidneys, and omentum. In the thoracic cavity, similar nodules were scattered in the lungs. The cranial right pulmonary lobe was replaced by a large (10-cm diameter) mass that extended and adhered to the pericardium of the base of the heart. Four small nodules from 3 to 10 mm in diameter were identified in the myocardium of the left ventricle. On cut surface, all neoplastic nodules were tan, soft, and friable and showed focal hemorrhages. Samples of all neoplastic tissues were fixed in 10% buffered formalin and routinely processed for histological examination.
Immunohistochemical staining was performed on deparaffinized sections with the avidin-biotin-peroxidase complex (ABC) a technique 10 (Table 1). DAB (3,3′-diaminobenzidine) a was used as the chromogen. Sections were counterstained with Mayer hematoxylin. Negative controls were performed under identical conditions, incubating the sections with normal horse serum. A section of the primary mammary adenocarcinoma was used as a positive control for cytokeratin. Interstitial cells and vascular walls present in the cardiac sections close to the sarcoma were considered as internal positive control for vimentin and muscular markers (actin, myosin, and desmin), respectively.
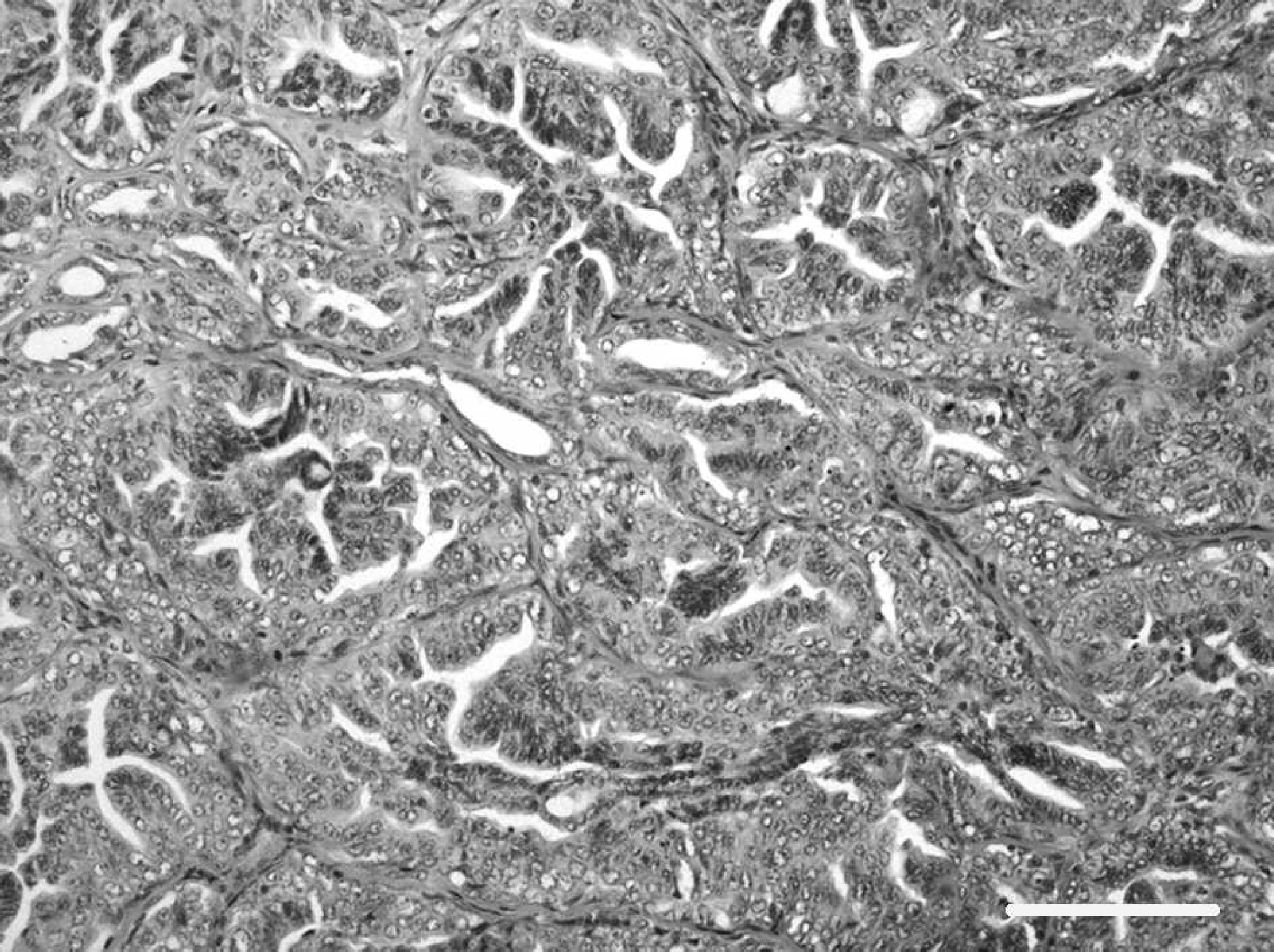
Primary mammary simple tubular adenocarcinoma. Hematoxylin and eosin. Bar = 40 μm. 100X.
Histological examination revealed that, in spite of similar gross appearance, neoplastic nodules belonged to 2 different tumors. All the pulmonary neoplastic lesions and 2 of the 4 nodules observed in the left ventricle were composed of irregularly shaped tubular structures, lined by 1–3 layers of cuboidal epithelial cells, immersed in an abundant and dense fibrous stroma. Focal hemorrhages and scattered areas of coagulative necrosis were also observed. Immunohistochemical staining gave specific labeling with low background with all the antibodies employed. Cells were negative for vimentin, desmin, smooth muscle myosin, and α-smooth muscle actin. They were immunohistochemically positive for cytokeratins (Fig. 2), demonstrating their epithelial origin. Neoplastic nodules of epithelial origin shared morphology with the mammary adenocarcinoma diagnosed 2 years earlier and were thus interpreted as metastases of mammary origin.
On the other hand, omental, hepatic, and renal nodules, together with the other 2 ventricular myocardial small nodules, were composed of haphazardly arranged, pleomorphic, spindle to polygonal, 20–80-μm-wide neoplastic cells with faintly eosinophilic cytoplasm and large oval nuclei with dispersed chromatin and 1 prominent eosinophilic nucleolus. They had marked atypia, anisocytosis, and anisokaryosis. Mitotic figures, mostly atypical, ranged from 0–4 per high-power field (HPF; 400X). These cells were negatively immunolabeled for cytokeratin, excluding their origin from an epithelial or myoepithelial component of a mammary gland neoplasm. The positive reaction of neoplastic cells for vimentin suggested the diagnosis of sarcoma. The negative reaction for both smooth muscle myosin and desmin suggested the sarcoma cells did not originate from smooth muscle cells. 4,21 On the other hand, their positive reaction for vimentin (Fig. 3A) and α-smooth muscle actin (Fig. 3B) suggested that the sarcoma cells were mostly derived from myofibroblasts.
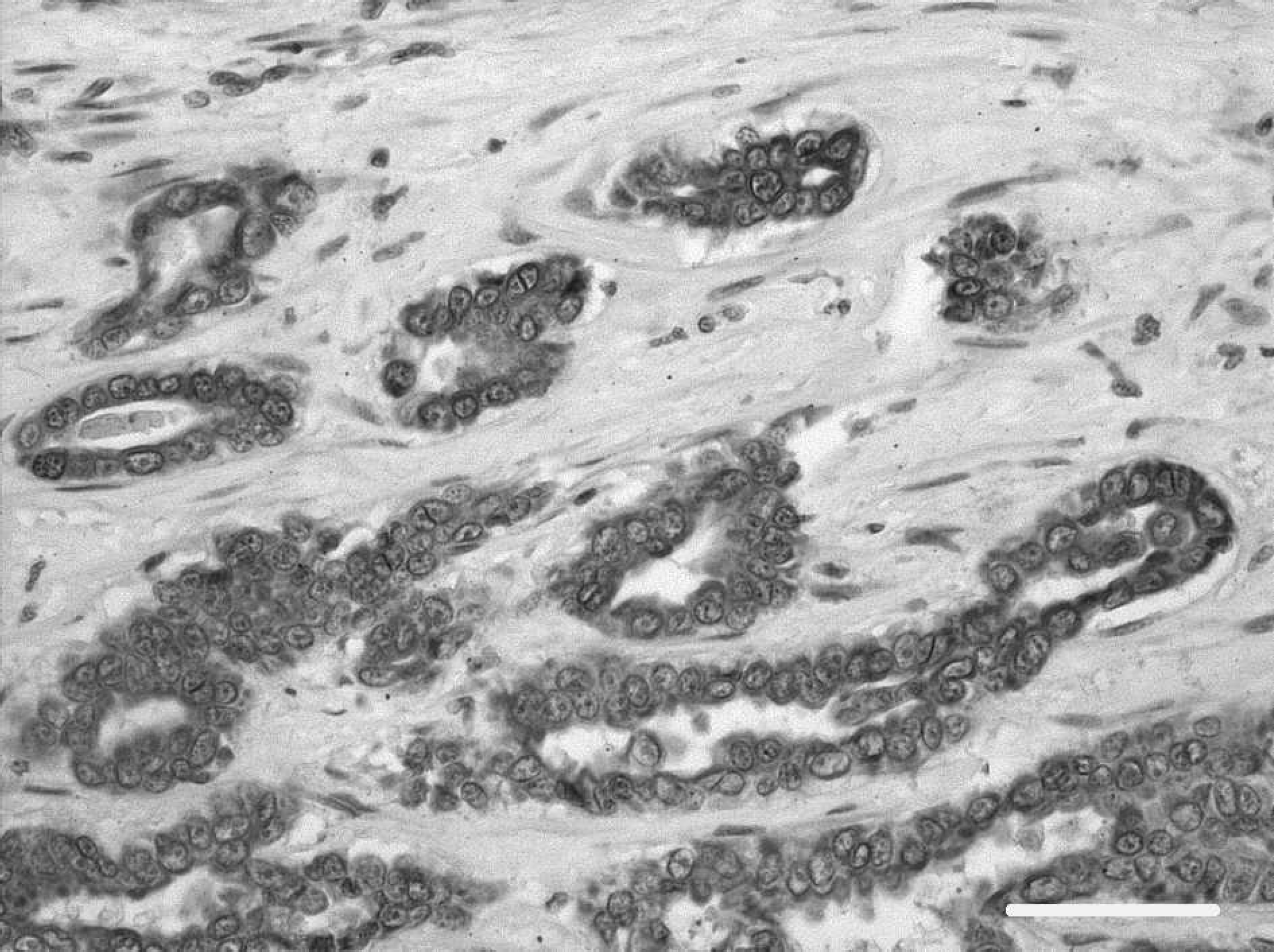
Myocardial metastasis of mammary tubular adenocarcinomas. Neoplastic epithelial cells are strongly positive for pan-cytokeratin marker. Avidin-biotin immunoperoxidase complex method, monoclonal antibody CK AE1/AE3, hematoxylin counterstain. Bar = 80 μm. 200X.
Myofibroblasts are a differentiated fibroblast cell type characterized by increased contractile capacity and elevated production of extracellular matrix. 9,17 These cells, sharing morphologic and functional features with fibroblast and smooth muscle cells, were initially described in granulation tissue, granulomas, and other reactive conditions. 8 In the heart, spindle cells with myofibroblastic differentiation are reported in humans as well as in canines and are implicated in fibrosis associated with pressure-overload hypertrophy and postinfarctive tissue remodeling. 5,7,17 Malignant mesenchymal tumors composed of neoplastic cells with myofibroblastic characteristics are uncommon in humans and canines. Myofibroblast has been frequently used to describe fibroblasts that proliferate in granulation tissue and contain both vimentin and smooth muscle actin but not desmin or myoglobin. Ultrastructurally, myofibroblasts are differentiated from quiescent fibroblasts in tissues by intracytoplasmic bundles of contractile microfilaments, extensive cell-to-matrix attachment sites, and intercellular adherens and gap junctions. 9 In humans, myofibroblastic sarcomas are considered to have a wide-ranging anatomic distribution with a predilection for the head and the neck, whereas cardiac occurrence is rare. 11,16 In a dog, a case of sarcoma partially composed of myofibroblasts was observed in the neck as a reaction to microchip implantation, 22 whereas, in the heart, no sarcomas with myofibroblastic component or true myofibroblastic sarcomas have been previously reported. To the authors' knowledge, this is the first description of a tumor presumptively totally composed of myofibroblasts in the heart of a dog. Unfortunately, because all the sarcomatous nodules had similar size and because they all exhibited necrotic foci and hemorrhage, it was impossible to determine whether the cardiac nodules were primary or metastatic. However, it could be hypothesized that, in this case, myofibroblastic sarcoma originated primarily in the heart from tissue remodeling after the spread of mammary tumor metastases. In fact, metastatic emboli induce small infarcts leading to tissue remodeling. In the heart, it is well known that fibroblasts account for up to two thirds of the total cells and are responsible for its structural integrity through controlled proliferation and extracellular matrix turnover. 18 In the myocardium during the postinfarction remodeling process, 19 intracardiac fibroblasts, ordinarily quiescent, transform into a proliferative and invasive myofibroblast phenotype and initiate structural changes to cardiac interstitium. In the case presented here, myocardial injury secondary to mammary tumor metastasis might be considered the cause of myofibroblastic activation and proliferation. During this latter process, neoplastic transformation could occur.
Monoclonal antibodies employed in the immunohistochemical assays.

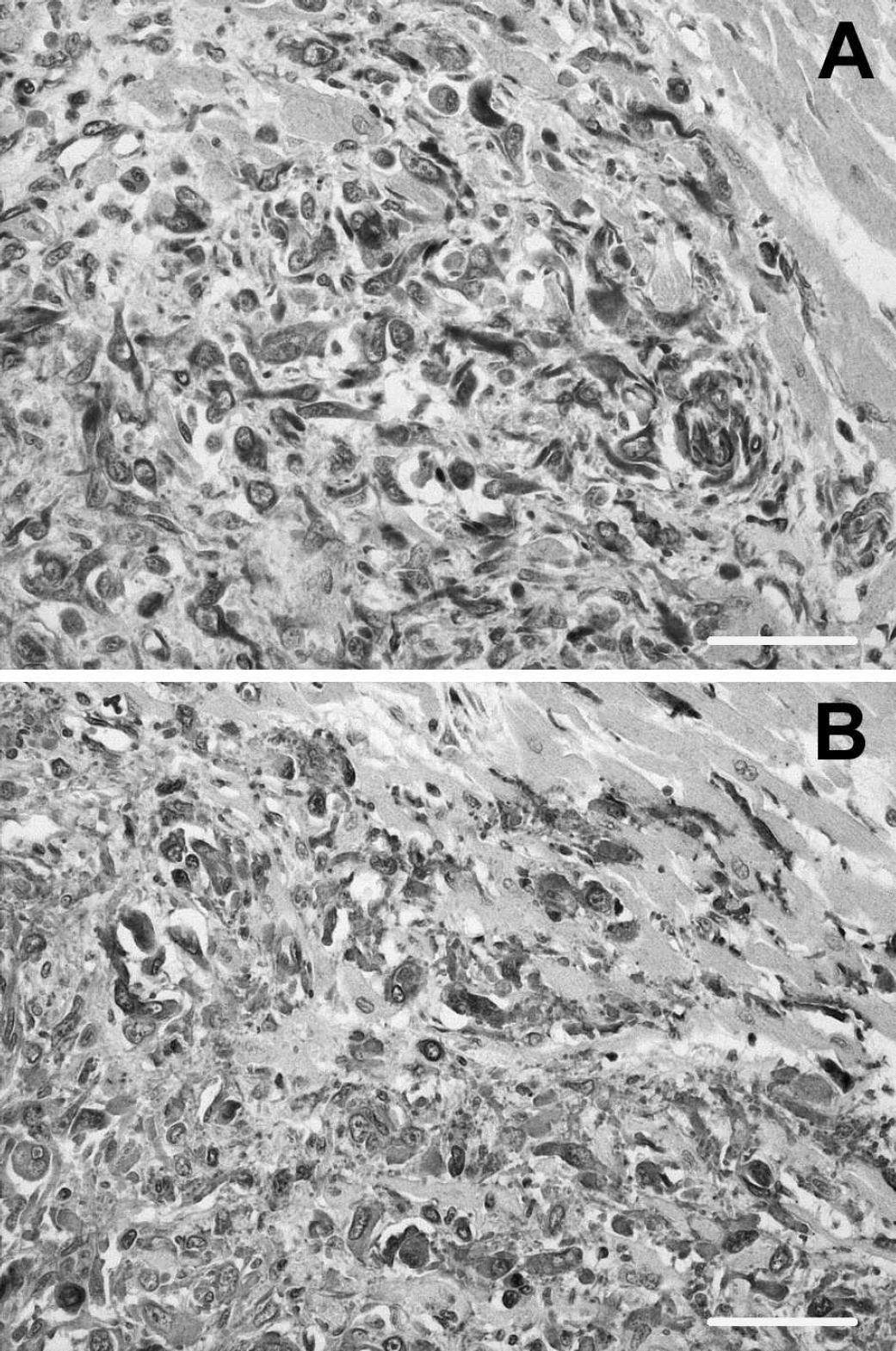
Myocardial metastasis of the myofibroblastic sarcoma. Neoplastic spindle cells arranged in short bundles are strongly positive for both
Mammary tumors and related metastasis are not infrequent in female dogs; in a recent study, 6 out of 12 dogs with malignant mammary tumors presented with cardiac metastasis, mainly in the left ventricle. 2 Mammary metastatic nodules were observed in the myocardium of the left ventricle in this case as well. In the aforementioned study, only a few dogs with heart tumors had cardiac symptoms. 2 Cardiac symptoms are generally related to size and location of the tumor, and electrocardiographic abnormalities seem to be secondary to infiltration of the myocardium by neoplastic cells associated with damage of the conduction system. 6,12 In the case presented here, the dog was referred for cardiac symptoms, and electrocardiogram revealed ventricular tachycardia and an elevated serum cTnI level. Because neoplastic nodules of mesenchymal and epithelial neoplasia were both localized in the left ventricle, it was impossible to determine the exact origin of ventricular tachycardia. The elevation of cTnI suggested myocardial damage. Because serum cTnI is 100% specific for the heart, it is a biochemical test for myocardial injury, 1,3 as in this case with tumors (or expansile tumors) in the myocardium.
This paper describes a rare case of double tumors in the heart of a dog: 1 of epithelial and 1 of mesenchymal origin. Immunohistochemistry suggested that the latter was presumptively totally composed by myofibroblasts. To the authors' knowledge, this is the first report describing a cardiac sarcoma, most likely derived from myofibroblasts in a dog, occurring concurrently with cardiac metastasis of mammary gland adenocarcinoma.
Acknowledgements. The authors thank Mr. Marco Colombo for photographic support.
Footnotes
a.
Vector Laboratories, Burlingame, CA.
b.
Zymed Laboratories, San Francisco, CA.
c.
Dako North America, Inc., Carpinteria, CA.
d.
Novocastra Laboratories, Newcastle upon Tyne, UK.
e.
Sigma-Aldrich, St. Louis, MO.
